Research Administrators Forum May 8th, Marcia Smith Associate Vice Chancellor for Research
|
|
|
- Juliet McCormick
- 5 years ago
- Views:
Transcription
1 Research Administrators Forum May 8th, 2014 Marcia Smith Associate Vice Chancellor for Research
2 Agenda Welcome and Announcements Marcia Smith, Office of Research Administration OHRPP Retention of Research Records Marcia Malmet, Office of the Human Research Protection Program OCGA Updates Patti Manheim, Office of Contracts and Grants Administration Operational Management Challenges EPASS Changes Master Training Calendar EFM Updates Kevin Cook, Interim Director, Extramural Fund Management Organizational Updates Pool to Subaccount Close-out Timeline PAMS Questions and Discussion
3 Retention of Research Records FDA Investigational Drug Studies: Clinical investigators must retain study records for a period of two years following the date a marketing application is approved for the drug/indication for which it is being investigated or If no application is to be filed or if the application is not approved for such indication, until two years after the investigation is discontinued and FDA is notified. Review sponsor contract! 1
4 Retention of Research Records FDA Investigational Device Studies: Clinical investigators must retain study records for a period of two years after the latter of the following two dates: The date on which the investigation is terminated or completed, or The date that the records are no longer required for purposes of supporting a premarket approval application or a notice of completion of a product development protocol. NIH Studies three years after close of study or six years if PHI involved. 2
5 Patti Manheim Director May 8, 2014
6 Today s Topics Operational Management Challenges OCGA Master Training Calendar Revised EPASS Introduction of New Staff
7 Operational Management Challenges Background - Leadership Bodies RAPID Results, Updates, Accomplishments, and Initiatives RAPID Steering Committee Convened by EVC Scott Waugh Membership includes Vice Chancellors, Deans, Senior Administrators AVC Marcia Smith reports to the Steering Committee on ORA operations and the direction of RAPID initiatives RAPID Faculty Advisory Committee reports to the Steering Committee and advises on RAPID priorities
8 Operational Management Challenges Background Presented Challenges in OCGA Operations Management to Steering Committee in March Stunned/Astounded by ramifications and complexities of OCGA day-to-day operations Provost invitation to present to Dean s Council
9 Operational Management Challenges Background Dean s Council Similarly Stunned/Astounded by ramifications and complexities Provost summarizes risks to the institution Deans agree Provost solicits Deans analysis and recommendations
10 The Issues By the Numbers FY13 Proposals Submitted 4,674 Received 2 days (16 working hours) 1,899 42% or less in advance of sponsor deadline Received 4 days (32 working hours) or less in advance of sponsor deadline 3,468 74% Larger component of our portfolio is complex 20% decrease in federal dollars 56% increase in state, foundation, other dollars
11 Impact of Late Proposals OCGA accepts ALL proposals regardless of: How late it is received: 5 minutes prior to deadline? How complete: highly variable quality and completeness highly variable preparer expertise How inflexible the deadline and the electronic system Process to accommodate a reasonable number of late proposals/real emergencies
12 Delays in Award Set-Up $21M of awards (174) currently on hold waiting for Department or PI response FY14 median award processing time: 9 days -- complete proposal record 13 days -- PI/Department information missing 46% of all awards in FY13 required additional information from the Department or PI before set-up 10% of awards ($88M) in FY13 received for proposals submitted without institutional approval (No department, DRA or OCGA approval)
13 Increasing Complexity of OCGA Transactions Increasing percentage of non-routine one-of-akind or few-of-a-kind transactions Complex transactions require coordination with campus experts: Intellectual Property Development Risk Management UCOP General Counsel Humans/Animals Export Control Conflict of Interest PI/Department
14 Complex, Coordinated Collaboration CTAO PI Campus Counsel Cap Progs OHRPP Chairs RPC Purch. OARO OCGA Sponsor VCs Other UCs ISR Dvlpmt Deans EFM Risk Mgmt UCOP Tax Svs OIP
15 What are Other UC Campuses Doing? Berkeley Hard Line Must be complete (including internal forms and all signatures) or returned Draft and placeholder sections are not acceptable Except scope/research plan Five days prior to the deadline Including pre-proposals, white papers, LOI s Incomplete returned and considered late Exceptions require written request for VCR approval can be denied
16 What are Other UC Campuses Doing? Berkeley Hard Line SPO s office hours: 8:00 am to 5:00 pm (M F) SPO reserves the right to process noncompliant proposals only after on-time proposals for day are submitted Unreasonable and unfair to expect on-time proposals to be delayed or receive lesser review due to late proposals
17 What are Other UC Campuses Doing? Irvine Hard Line Must be complete (including internal forms and all signatures) or returned Draft and placeholder sections are not acceptable Except scope/research plan Standard Proposals: 5 days prior to the deadline Non-standard Proposals: 10 days prior to the deadline Sponsor is not Government, UC, or Domestic Higher Ed. Proposal is a contract Proposal includes Award Terms and Conditions Proposal includes SubAwards Limited Submissions Reduced F&A
18 What are Other UC Campuses Doing? Irvine Hard Line Applies to all extramural proposals even if NO signature or institutional concurrence Final scope/research plan no later than 8 business hours before deadline time First-In/First-Out no preference for late proposals Exception to 5/10 days require written request for VCR approval can be denied
19 What are Other UC Campuses Doing? Will provide data/feedback to Provost and Deans Council Currently reviewing standard/mandatory expectations for proposal completeness Currently reviewing OCGA proposal review processes and expectations based on when received Finalizing proposal receipt process (via Proposal Intake Team)
20 Revised EPASS Based on campus and ORA feedback Updated Form and Instructions available on the OCGA Forms site
21 Revised EPASS Section 2: Department or ORU Added two ORUs to the pick-list: California Center for Population Research (CCPR) Grand Challenges
22 Revised EPASS Section 5: Sponsor Information Reorganized fields and provided more information to clarify direct versus prime sponsor Left side: Sponsor Information(Entity which will provide funding directly to UCLA) Right Side: Prime Sponsor Information (Complete this section when UCLA is a subrecipient)
23 Revised EPASS Section 6: Proposal Checklist Added Cost Sharing Amount to cost sharing question(s): Human and Animal Subjects; If yes, indicate Pending or IRB #:
24 Revised EPASS Section 7: Additional Forms Required Added clarifying language to 700-U Required for Research Hover over help for types of Research
25 OCGA Master Training Calendar Beginning September 2014 First Thursday of each month Kinross minute sessions on a focused topic Presented by subject matter experts
26 OCGA Master Training Calendar Click on the topic for details
27 OCGA Master Training Calendar Topic: Filling out the EPASS: How and Why Date: Thursday, September 4, Kinross Ave, Room 210 9:30AM-11:00AM Presenter: Summary: RSVP: Patti Manheim This session will address the background and purpose of the EPASS. We will review and discuss each section of the EPASS with specific examples of questions from users. This session is appropriate for anyone with responsibility for completing, reviewing or processing EPASS forms. Click here to RSVP
28 New OCGA Staff Sam Perez, Award Intake/Grant Analyst Experience Nine years in OCGA as a Contract and Grant Specialist Comprehensive understanding of OCGA processes and procedures, technical expertise with ORA/OCGA systems
29 New OCGA Staff Rae Anne Robinett, Senior Grant Analyst Previous experience: UCLA Office of Intellectual Property and Industry Sponsored Research Eight years of in-depth and comprehensive expertise in reviewing proposals, negotiating complex agreements.
30 OCGA New Staff Shauna Huntsman, Senior Grant Analyst Effective June 2, 2014 Experience: Senior Research Administrator at UCSF Grant and contract proposal preparation and award negotiation UC expertise Solid background in federal and non-profit sponsors
31 OCGA Staff Open Positions Contract and Grant Officer Contract and Grant Specialist Assistant to the Director
32
33 EFM Updates Kevin Cook Interim Director, EFM
34 Agenda Organization Updates DHHS Pooled to Subaccounting Transition Closeout Timeline PAMS Update
35 Organization Updates
36 Organization Updates Four Open Positions 3 Accountant I s 1 Accountant II Accountant I s Verbal offers accepted Anticipated to start 5/19 Accountant II Finalists identified Goal is to minimize changes to department contacts as we assimilate the new hires
37 Pooled to Subaccounting Transition
38 Pooled to Subaccounting Transition Change in cash draw in PMS Reimbursements for DHHS expenditures are drawn from the Federal Payment Management System (PMS) EFM currently draws cash from a pooled account EFM must draw cash from subaccounts for new and continuing awards as NIH establishes them in the PMS subaccounts in FY14 and FY15 respectively. How will UCLA address the change? Goal: To minimize additional burden for departments while being in compliance with the new requirement Proposal: Treat SNAP and Non-SNAP differently to isolate population where change is necessary to accommodate the subaccounting transition
39 Pooled Account vs. Subaccounts Pooled Account: Total Draw: $20 million Total Awards: 1,000 Subaccount: Total Draw: $20 million Total Awards: 1,000 $20 million draw by UCLA Draws submitted in aggregate Request made for all award under DHHS/ NIH Award Expense Draw Amount A B C D E F G Draw requested by award
40 SNAP Example Project Period (Original competitive segment) January 2010 December 2015 Budget Period End Dates (New smaller competitive segments) December 2014 and December 2015 FFR Due Dates March 2015 and March 2016 New Fund? No EFM/Department Interaction for FFR Due March 2015? No Report off Ledger as of December 2014, this FFR is administrative only January 2010 December 2014 December 2015 March 2015 (FFR Due) March 2016 (FFR Due) Pooled Account (Same Fund) Sub Account (Same Fund)
41 Non-SNAP Example Project Period (Original competitive segment) January 2010 December 2015 Budget Period End Dates (New smaller competitive segments) December 2014 and December 2015 FFR Due Dates March 2015 and March 2016 New Fund? Yes ORA will create the new Funds as RAS Funds in advance of the transition FM/Department Interaction for FFR Due March 2015? Yes We will be requesting a Closeout Packet for both Funds Overlap January 2010 December 2014 December 2015 March 2015 (FFR Due) March 2016 (FFR Due) Pooled Account (Old Fund) Sub Account (New Fund)
42 Summary Restricted Carryforward? Type SNAP Non-SNAP No Yes New Fund? No Yes Confirmation w/ Dept of Interim FFR? No NIH Notice for award payment transition to subaccounts: NIH FAQs for award payment transition to subaccounts: Yes
43 Closeout Timeline
44 Closeout Phases Phase 1 Notification and Preparation 90/30/0 Days Before RAPID Closeout Notification to Department/PI Department/PI Review Fund Fund Ends Phase 2 Reconciliation and Report 45 Days Before Department finalizes review of Fund 30 Days Before Department will provide Fund with Closeout Packet FFR is Due
45 Drivers for Closeout Phases Final financial reports/invoices are currently due 90 days after the Budget End Date for almost all direct federal awards Final financial reports/invoices are currently due 60 days after the Budget End Date for almost all pass-through federal awards
46 Drivers for Closeout Phases (cont d) Effective under the new subaccounting process, no reimbursements will be made on requests for payments more than 90 days after Budget End Date Impact: All expenditures must post in a timely manner to meet the above referenced deadlines for direct and pass-through federal awards in order to be reimbursed.
47 Example of FFR Due 90 Days After
48 Example of Invoice Due 60 Days After
49 Avoiding Late Transactions Monthly expenditure reconciliation while a Fund is active is the best practice for avoiding out of period transactions This allows the PI and Department to ensure that: Expenses that should have hit are accurately reflected Expenses that hit erroneously are moved in a timely manner F&A expenses have been incurred correctly It is the responsibility of the PI and Department to ensure that expenses on a Fund match those reported to EFM and the FFR/invoice Phase 2, representing the day period after the Fund ends, is the appropriate time to make sure that any final expenses are applied to a Fund, and any reconciling or correcting entries are executed
50 Currently Under Consideration EFM forming a working group, which will include department representatives, to discuss impact of hard deadlines on closeout process If Closeout Packet is not received by the due date, then UCLA is considering reporting off the ledger after EFM does a cursory review of the expenses If expenses are deemed questionable by EFM and appropriate and timely justification is not received, then the department will be expected to cover those expenses If expenses are posted or received after the contractual deadline for the final report/invoice, then the department or vendor will be expected to cover those expenses
51 PAMS Updates
52 PAMS Daily Usage PAMS Daily Usage All Users
53 System Availability Current Availability: 7:30 AM 6:00 PM, M-F System Uptime
54 Highlights Successfully processed 4 Closeout Packets! Bugs and Enhancements Submit to PAMS Help Fixed 75 bugs since go-live Current priority: Assignments and TIF PAMS Help PAMSHelp@research.ucla.edu (310) Quick guides:
Please fill out the survey forms 2/9/2012. Upcoming ORA Training Opportunities. Agenda
 Agenda Welcome and Announcements Marcia Smith Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Data Management Tool Sharon Farb, Associate University Librarian,
Agenda Welcome and Announcements Marcia Smith Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Data Management Tool Sharon Farb, Associate University Librarian,
Research Administrators Forum August 14th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum August 14th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Associate Vice Chancellor, Office of Research Administration
Research Administrators Forum August 14th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Associate Vice Chancellor, Office of Research Administration
Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith Data Management Tool Sharon Farb, Associate University Librarian,
Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith Data Management Tool Sharon Farb, Associate University Librarian,
FEDERAL FUND CLOSEOUT. RAF August 13, 2015 Yoon Lee
 FEDERAL FUND CLOSEOUT RAF August 13, 2015 Yoon Lee Agenda Communication update Applicability and population Next steps Resources 2 Federal Fund Closeout Procedure: Communication Update April 2015 Presented
FEDERAL FUND CLOSEOUT RAF August 13, 2015 Yoon Lee Agenda Communication update Applicability and population Next steps Resources 2 Federal Fund Closeout Procedure: Communication Update April 2015 Presented
Research Administration Forum March 9, Marcia Smith Associate Vice Chancellor for Research
 Research Administration Forum March 9, 2017 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements - Marcia Smith Immigration Update and Q & A Sam Nahidi, Assistant Director,
Research Administration Forum March 9, 2017 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements - Marcia Smith Immigration Update and Q & A Sam Nahidi, Assistant Director,
Patti Manheim, Director, OCGA
 Research Administrators Forum Patti Manheim, Director, OCGA Agenda Welcome - Patti Manheim OCGA Updates Patti Manheim * Consortium/contractual arrangements versus purchase of services * Master Calendar
Research Administrators Forum Patti Manheim, Director, OCGA Agenda Welcome - Patti Manheim OCGA Updates Patti Manheim * Consortium/contractual arrangements versus purchase of services * Master Calendar
Outgoing Subaward Basics - Subawards Issued by UCLA under Extramurally Funded Grants/Cooperative Agreements
 Outgoing Subaward Basics - Subawards Issued by UCLA under Extramurally Funded Grants/Cooperative Agreements PREVIEW: Master Training (Mary Haskins, OCGA Subaward Officer) When: Wednesday, April 18, 2018
Outgoing Subaward Basics - Subawards Issued by UCLA under Extramurally Funded Grants/Cooperative Agreements PREVIEW: Master Training (Mary Haskins, OCGA Subaward Officer) When: Wednesday, April 18, 2018
Updates on American Recovery and Reinvestment Act (ARRA) Funding. Virginia Anders, Acting Director, OCGA Evelyn Balabis, Director, EFM
 Updates on American Recovery and Reinvestment Act (ARRA) Funding Virginia Anders, Acting Director, OCGA Evelyn Balabis, Director, EFM 15 OCGA ARRA Help Desk 10:00 a.m. 5:00 p.m., Monday Friday 310-794-0548
Updates on American Recovery and Reinvestment Act (ARRA) Funding Virginia Anders, Acting Director, OCGA Evelyn Balabis, Director, EFM 15 OCGA ARRA Help Desk 10:00 a.m. 5:00 p.m., Monday Friday 310-794-0548
Research Administrators Forum September 13, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum September 13, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith OCGA Initiatives Patti Manheim, Kim Duiker Proposal Intake
Research Administrators Forum September 13, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith OCGA Initiatives Patti Manheim, Kim Duiker Proposal Intake
Sponsored by Office of Research Administration. Marcia Smith Associate Vice Chancellor Research Administration. July 9, 2009
 Research Administrators Forum Sponsored by Office of Research Administration Marcia Smith Associate Vice Chancellor Research Administration July 9, 2009 Agenda 2 Updates from the Animal Research Committee
Research Administrators Forum Sponsored by Office of Research Administration Marcia Smith Associate Vice Chancellor Research Administration July 9, 2009 Agenda 2 Updates from the Animal Research Committee
Patti Manheim - Director February 13, 2014
 Patti Manheim - Director February 13, 2014 Today s Topics NIH Fiscal Operations for FY 2014 Updates from the January 2014 FDP Meeting Other OCGA Updates NIH Fiscal Operations for FY2014 NIH Budget $30.15
Patti Manheim - Director February 13, 2014 Today s Topics NIH Fiscal Operations for FY 2014 Updates from the January 2014 FDP Meeting Other OCGA Updates NIH Fiscal Operations for FY2014 NIH Budget $30.15
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018
 CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
FEDERAL FUND CLOSEOUT. RAF November 12, 2015 Yoon Lee
 FEDERAL FUND CLOSEOUT RAF November 12, 2015 Yoon Lee Agenda Key Steps in the Procedure September and October FFR Submission Frequently Asked Questions December Deadlines 2 Federal Fund Closeout Key Steps
FEDERAL FUND CLOSEOUT RAF November 12, 2015 Yoon Lee Agenda Key Steps in the Procedure September and October FFR Submission Frequently Asked Questions December Deadlines 2 Federal Fund Closeout Key Steps
Research Administrators Forum October 10th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum October 10th, 2013 Marcia Smith Associate Vice Chancellor for Research Agenda Meeting Agenda Welcome and Announcements Marcia Smith, Office of Research Administration Contract
Research Administrators Forum October 10th, 2013 Marcia Smith Associate Vice Chancellor for Research Agenda Meeting Agenda Welcome and Announcements Marcia Smith, Office of Research Administration Contract
Research Administrators Forum November 8, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum November 8, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith RAPID Update PAMS Jessica Carney, Allison Philabaum Post
Research Administrators Forum November 8, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith RAPID Update PAMS Jessica Carney, Allison Philabaum Post
VCR Five Day Proposal Submission Policy Implementation Guidance RAC Forum March 16, 2016
 UC BERKELEY VCR Five Day Proposal Submission Policy Implementation Guidance RAC Forum March 16, 2016 Why did Berkeley need a new policy? Some PIs were using late proposals as SOP SUN faculty members noted
UC BERKELEY VCR Five Day Proposal Submission Policy Implementation Guidance RAC Forum March 16, 2016 Why did Berkeley need a new policy? Some PIs were using late proposals as SOP SUN faculty members noted
SJSU Research Foundation
 SJSU Research Foundation The Nuts & Bolts of Proposal Preparation and Award Management Sponsored Programs Workshop February 16, 2017 Agenda Discussion of Common Issues and Solutions Terms and Conditions
SJSU Research Foundation The Nuts & Bolts of Proposal Preparation and Award Management Sponsored Programs Workshop February 16, 2017 Agenda Discussion of Common Issues and Solutions Terms and Conditions
What You Need to Know When Submitting an NIH SF424 R&R Grant Application Through the UCLA Office of Contract and Grant Administration (OCGA)
 What You Need to Know When Submitting an NIH SF424 R&R Grant Application Through the UCLA Office of Contract and Grant Administration (OCGA) Kim Duiker, Assistant Director Mary Haskins, Contract & Grant
What You Need to Know When Submitting an NIH SF424 R&R Grant Application Through the UCLA Office of Contract and Grant Administration (OCGA) Kim Duiker, Assistant Director Mary Haskins, Contract & Grant
Presentation 2/26/2013. Director, TitleOPERA
 Presentation Title Michelle Presented G. Bulls By: Name Director, TitleOPERA Office OER AASCU Washington Conference February 21, 2013 2 NIH is operating under a continuing resolution. See NOT-OD- 13-018
Presentation Title Michelle Presented G. Bulls By: Name Director, TitleOPERA Office OER AASCU Washington Conference February 21, 2013 2 NIH is operating under a continuing resolution. See NOT-OD- 13-018
Patti Manheim, Director April 11, 2013
 Patti Manheim, Director April 11, 2013 Today s Topics NIH Research Performance Project Report (RPPR) Update Patti Manheim NIH Public Access Policy Reporting Requirements Patti Manheim and Lisa Federer,
Patti Manheim, Director April 11, 2013 Today s Topics NIH Research Performance Project Report (RPPR) Update Patti Manheim NIH Public Access Policy Reporting Requirements Patti Manheim and Lisa Federer,
UNIVERSITY RESEARCH ADMINISTRATION FINANCIAL ROLES AND RESPONSIBILITIES MATRIX - WORK IN PROGRESS 10/03/2013 Roles.
 UNIVERSITY RESEARCH ADMINISTRATION Roles Business Internal Controller's Clinical Responsibilities PI Office Chair Dean Audit Office OCR GCFA GCA PROVOST Trials Office I. GENERAL RESEARCH ADMINISTRATION
UNIVERSITY RESEARCH ADMINISTRATION Roles Business Internal Controller's Clinical Responsibilities PI Office Chair Dean Audit Office OCR GCFA GCA PROVOST Trials Office I. GENERAL RESEARCH ADMINISTRATION
SUMMARY. Extramural Funds Accounting effectively conducts post award financial management:
 SUMMARY Extramural Funds Accounting assists campus faculty and staff in meeting their teaching, research, and operational needs by providing quality financial services for externally funded projects. Protecting
SUMMARY Extramural Funds Accounting assists campus faculty and staff in meeting their teaching, research, and operational needs by providing quality financial services for externally funded projects. Protecting
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP)
 Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Complex Award Management Program Projects/Multi-Fund Awards- Post Award NUMBER: RAS SOP 2001 VERSION: 4.0
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Complex Award Management Program Projects/Multi-Fund Awards- Post Award NUMBER: RAS SOP 2001 VERSION: 4.0
ORA Quarterly Meeting April
 ORA Quarterly Meeting April 19 2017 Agenda 1. Workday Grants Implementation Items General Information Blackout Period Sub K s - Set-up and Invoicing Advance Accounts 2. Jackson/ UM Partnership Master Agreement
ORA Quarterly Meeting April 19 2017 Agenda 1. Workday Grants Implementation Items General Information Blackout Period Sub K s - Set-up and Invoicing Advance Accounts 2. Jackson/ UM Partnership Master Agreement
University of Colorado Denver
 University of Colorado Denver Campus Guidelines Title:, 4-13 Source: Prepared by: Approved by: Office of Grants and Contracts Director, Office of Grants and Contracts Vice Chancellor for Research Effective
University of Colorado Denver Campus Guidelines Title:, 4-13 Source: Prepared by: Approved by: Office of Grants and Contracts Director, Office of Grants and Contracts Vice Chancellor for Research Effective
Subrecipient Monitoring Procedures
 Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015
 OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015 INTRODUCTION The University of Wisconsin-Milwaukee (UWM) is the prime recipient on a wide range of sponsored awards
OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015 INTRODUCTION The University of Wisconsin-Milwaukee (UWM) is the prime recipient on a wide range of sponsored awards
Subaward Policies and Procedures Manual
 JOHNS HOPKINS UNIVERISTY Krieger School of Arts and Sciences Business and Research Administration Subaward Policies and Procedures Manual 2016 Contents Introduction...3 Differentiating types of relationships...3
JOHNS HOPKINS UNIVERISTY Krieger School of Arts and Sciences Business and Research Administration Subaward Policies and Procedures Manual 2016 Contents Introduction...3 Differentiating types of relationships...3
University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures
 Summary 1. Subaward Definitions A. Subaward B. Subrecipient University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures C. Office of Contracts and Grants (OCG) 2. Distinguishing
Summary 1. Subaward Definitions A. Subaward B. Subrecipient University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures C. Office of Contracts and Grants (OCG) 2. Distinguishing
Sponsored by Office of Research Administration
 Research Administrators Forum Sponsored by Office of Research Administration Marcia Smith Associate Vice Chancellor Research Administration January 14, 2010 Meeting Agenda Meeting Agenda Welcome and Announcements
Research Administrators Forum Sponsored by Office of Research Administration Marcia Smith Associate Vice Chancellor Research Administration January 14, 2010 Meeting Agenda Meeting Agenda Welcome and Announcements
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM. December 2017
 Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Outgoing Subaward Guide and F-A-Qs
 1. What is a subaward? Outgoing Subaward Guide and F-A-Qs 2. After an award is made to Rutgers, how is a subaward established? 3. What is the outgoing subaward process? 4. How can a subaward be changed/amended?
1. What is a subaward? Outgoing Subaward Guide and F-A-Qs 2. After an award is made to Rutgers, how is a subaward established? 3. What is the outgoing subaward process? 4. How can a subaward be changed/amended?
Cost Share In SAP. Presented by Paige Brown Sponsored Projects Accounting. Cost Share in IRIS
 Cost Share In SAP Presented by Paige Brown paige.brown@uky.edu 257-1750 Sponsored Projects Accounting Cost Share in IRIS Define Cost Share When and How to use Cost Share Display postings How to fund the
Cost Share In SAP Presented by Paige Brown paige.brown@uky.edu 257-1750 Sponsored Projects Accounting Cost Share in IRIS Define Cost Share When and How to use Cost Share Display postings How to fund the
Ramp up and Wrap up projects. Riding the Wave to Close Out
 Ramp up and Wrap up projects Riding the Wave to Close Out To maximize our time together today: Please turn off or silence your cell phones Limit side chatter so everyone can hear Best of all. Stay for
Ramp up and Wrap up projects Riding the Wave to Close Out To maximize our time together today: Please turn off or silence your cell phones Limit side chatter so everyone can hear Best of all. Stay for
Kathy Hancock, Assistant Grants Compliance Officer, DGCO, OPERA, Office of Extramural Research, NIH, HHS
 Division of Grants Compliance and Oversight (DGCO) Office of Policy for Extramural Research Administration (OPERA), OER National Institutes of Health (NIH), DHHS 19 th Annual SBIR/STTR Conference Milwaukee,
Division of Grants Compliance and Oversight (DGCO) Office of Policy for Extramural Research Administration (OPERA), OER National Institutes of Health (NIH), DHHS 19 th Annual SBIR/STTR Conference Milwaukee,
After the Award is Made THEN WHAT?
 After the Award is Made THEN WHAT? George Tucker Chief Grants Management Officer National Institute of Environmental Health Sciences October 2016 TOPICS OF DISCUSSION The ins & outs of the NoA Accessing
After the Award is Made THEN WHAT? George Tucker Chief Grants Management Officer National Institute of Environmental Health Sciences October 2016 TOPICS OF DISCUSSION The ins & outs of the NoA Accessing
Agenda. NIH Update. Other Updates. Proposal & Progress Report Statistics Research Administration Training Topic:
 Agenda NIH Update Salary Cap Guidance NOT OD 17 037: NIH Implementation of the Interim RPPR While a Renewal Application is Under Consideration Prior Approval for No Cost Extensions via the era Commons
Agenda NIH Update Salary Cap Guidance NOT OD 17 037: NIH Implementation of the Interim RPPR While a Renewal Application is Under Consideration Prior Approval for No Cost Extensions via the era Commons
Emory Research A to Z ERAZ
 Emory Research A to Z ERAZ July 17, 2014 Whitehead Auditorium G01 Whitehead Building Agenda Controlled Substances Sub Award Processing RAS Updates SAM Kiosk Federal Updates OSP/OGCA Training Updates Christine
Emory Research A to Z ERAZ July 17, 2014 Whitehead Auditorium G01 Whitehead Building Agenda Controlled Substances Sub Award Processing RAS Updates SAM Kiosk Federal Updates OSP/OGCA Training Updates Christine
AURA Meeting. July 23, 2014
 AURA Meeting July 23, 2014 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored
AURA Meeting July 23, 2014 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored
Research Administrators Forum August 8th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum August 8th, 2013 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Office of Research Administration Conflicts of Interest
Research Administrators Forum August 8th, 2013 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Office of Research Administration Conflicts of Interest
University of Pittsburgh SPONSORED PROJECT FINANCIAL GUIDELINE Subject: SUBRECIPIENT MONITORING
 I. Scope Subrecipient monitoring is the process by which the University selects, monitors, controls, and accounts for University subcontractors or subrecipients utilized on sponsored projects in accordance
I. Scope Subrecipient monitoring is the process by which the University selects, monitors, controls, and accounts for University subcontractors or subrecipients utilized on sponsored projects in accordance
Sponsored Projects: Curriculum Plan Johns Hopkins University Panel Members:
 Sponsored Projects: Curriculum Plan Johns Hopkins University Panel Members: Mike Alexander, University Administration Jim Aumiller, University Administration Holly Benze, Engineering Don Cornely, University
Sponsored Projects: Curriculum Plan Johns Hopkins University Panel Members: Mike Alexander, University Administration Jim Aumiller, University Administration Holly Benze, Engineering Don Cornely, University
Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS
 Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar Chicago, IL October 2016 Diane Dean, Director
Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar Chicago, IL October 2016 Diane Dean, Director
Grant/Sponsor Related Systems. Department and OSP Perspectives on ERA
 Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
Contract & Users Group
 Contract & Users Group September 12, 2016 10:00 a.m.- 11:30 a.m. CHASS INT North 1020. Business & Financial Services A Division of Business & Administration Services (BAS) BAS Agenda Payroll Certification
Contract & Users Group September 12, 2016 10:00 a.m.- 11:30 a.m. CHASS INT North 1020. Business & Financial Services A Division of Business & Administration Services (BAS) BAS Agenda Payroll Certification
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Preparing campus for what is changing and what is not What is Grants Reform and when is it effective? What
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Preparing campus for what is changing and what is not What is Grants Reform and when is it effective? What
Research Administration Services Roles & Responsibilities For Grants and Contracts (Excludes Clinical Trials) Version 3.1
 Research Administration Services Roles & Responsibilities For Grants and Contracts (Excludes Clinical Trials) Version 3.1 Category PI/PI Staff Department* RAS Unit School/Unit ORA Offices Relevant SOP
Research Administration Services Roles & Responsibilities For Grants and Contracts (Excludes Clinical Trials) Version 3.1 Category PI/PI Staff Department* RAS Unit School/Unit ORA Offices Relevant SOP
Grants Management Workshop. Proposal and Award Management Support
 Grants Management Workshop Proposal and Award Management Support Agenda Sponsored Project Administration UVM and RSENR Support Systems Provide an overview of the grant life cycle For each stage of the
Grants Management Workshop Proposal and Award Management Support Agenda Sponsored Project Administration UVM and RSENR Support Systems Provide an overview of the grant life cycle For each stage of the
Research Administration at Emory Foundation Program DAY TWO: The Financial Administration of Sponsored Programs
 Revised 05/2018 Research Administration at Emory Foundation Program DAY TWO: The Financial Administration of Sponsored Programs Presenter: Finance: Grants and Contracts (FGC) Jumionne Tiako AD Post Award
Revised 05/2018 Research Administration at Emory Foundation Program DAY TWO: The Financial Administration of Sponsored Programs Presenter: Finance: Grants and Contracts (FGC) Jumionne Tiako AD Post Award
How to Manage Externally Funded Grants PROJECTS - FUND CODE 501 or 502
 How to Manage Externally Funded Grants PROJECTS - FUND CODE 501 or 502 What is a Grant? Funding that is coming from a source external to Wesleyan Project conceived by the Investigator Response to a solicitation
How to Manage Externally Funded Grants PROJECTS - FUND CODE 501 or 502 What is a Grant? Funding that is coming from a source external to Wesleyan Project conceived by the Investigator Response to a solicitation
SERVICE LEVEL AGREEMENT: University of Arkansas for Medical Sciences OFFICE OF SPONSORED PROGRAMS ADMINISTRATIVE NETWORK (OSPAN)
 SERVICE LEVEL AGREEMENT: University of Arkansas for Medical Sciences OFFICE OF SPONSORED PROGRAMS ADMINISTRATIVE NETWORK (OSPAN) Table of Contents Complete later Section I General Information and Objectives
SERVICE LEVEL AGREEMENT: University of Arkansas for Medical Sciences OFFICE OF SPONSORED PROGRAMS ADMINISTRATIVE NETWORK (OSPAN) Table of Contents Complete later Section I General Information and Objectives
Sponsored Projects Services Post Award. Arizona s First University.
 Sponsored Projects Services Post Award Arizona s First University. AGENDA An overview of Postaward Administration and the resources we use Today s discussion will include: The Award Documents Types of
Sponsored Projects Services Post Award Arizona s First University. AGENDA An overview of Postaward Administration and the resources we use Today s discussion will include: The Award Documents Types of
Cost Sharing. Policy Statement and Purpose
 Cost Sharing Policy Type: Administrative Responsible Office: Office of Sponsored Programs (proposal and award), Office of Research and Innovation, and Grants and Contracts Accounting (fiscal and accounting),
Cost Sharing Policy Type: Administrative Responsible Office: Office of Sponsored Programs (proposal and award), Office of Research and Innovation, and Grants and Contracts Accounting (fiscal and accounting),
Sponsored Programs Roles & Responsibilities
 The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
UC BERKELEY. The Seven Deadly Sins. Proposal Development. October 16, 2014
 UC BERKELEY The Seven Deadly Sins Proposal Development October 16, 2014 After all What Can Go Wrong? X-PI Status Cost Sharing Administrative Salaries F&A Sub-awards Collaborations Problematic Terms Topic:
UC BERKELEY The Seven Deadly Sins Proposal Development October 16, 2014 After all What Can Go Wrong? X-PI Status Cost Sharing Administrative Salaries F&A Sub-awards Collaborations Problematic Terms Topic:
ORA Closeout Process for NIH Awards
 Office of Research Administration ORA Closeout Process for NIH Awards ORA CLOSEOUT GUIDELINES ORA is responsible for making sure that necessary closeout documents are submitted to NIH within 90 days of
Office of Research Administration ORA Closeout Process for NIH Awards ORA CLOSEOUT GUIDELINES ORA is responsible for making sure that necessary closeout documents are submitted to NIH within 90 days of
Diane Dean, Director Kathy Hancock, Assistant Grants Compliance Officer Joel Snyderman, Assistant Grants Compliance Officer
 Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar San Diego, CA October 2015 Diane Dean,
Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar San Diego, CA October 2015 Diane Dean,
Sponsored Programs Roles & Responsibilities
 The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6
 Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6 KC-PRE28: Prepare Summary Proposal KC-AWD22: Research Gifts November 13, 2013 Goals and Outcomes Goals Obtain feedback on the proposed
Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6 KC-PRE28: Prepare Summary Proposal KC-AWD22: Research Gifts November 13, 2013 Goals and Outcomes Goals Obtain feedback on the proposed
The Lifecycle of a Sponsored Project
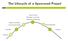 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
Title: OFFICE OF SPONSORED PROGRAMS/IRB/ORIP COORDINATION
 SYRACUSE UNIVERSITY HUMAN RESEARCH PROTECTION PROGRAM STANDARD OPERATING PROCEDURES TITLE: OFFICE OF SPONSORED PROGRAMS/IRB/ORIP COORDINATION DOCUMENT NUMBER: 033 REVISION NUMBER REVISION DATE (SUPERSEDES
SYRACUSE UNIVERSITY HUMAN RESEARCH PROTECTION PROGRAM STANDARD OPERATING PROCEDURES TITLE: OFFICE OF SPONSORED PROGRAMS/IRB/ORIP COORDINATION DOCUMENT NUMBER: 033 REVISION NUMBER REVISION DATE (SUPERSEDES
Financial Oversight of Sponsored Projects Principal Investigator and Department Administrator Responsibilities
 Principal Investigator and Department Administrator Responsibilities Boston College Office for Sponsored Programs Office for Research Compliance and Intellectual Property March 2004 Introduction This guide
Principal Investigator and Department Administrator Responsibilities Boston College Office for Sponsored Programs Office for Research Compliance and Intellectual Property March 2004 Introduction This guide
UC Davis Policy and Procedure Manual
 UC Davis Policy and Procedure Manual Chapter 230, Sponsored Programs Section 07, Public Health Service Regulations on Objectivity in Research Date: Supersedes: 8/24/12 Responsible Department: Office of
UC Davis Policy and Procedure Manual Chapter 230, Sponsored Programs Section 07, Public Health Service Regulations on Objectivity in Research Date: Supersedes: 8/24/12 Responsible Department: Office of
FAQ S FOR UNIFORM GUIDANCE
 FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
NOVA SOUTHEASTERN UNIVERSITY
 NOVA SOUTHEASTERN UNIVERSITY DIVISION OF RESPONSIBILITIES FOR RESEARCH AND SPONSORED PROGRAMS Vice President of Research & Technology Transfer: The responsibilities of the Vice President of Research &
NOVA SOUTHEASTERN UNIVERSITY DIVISION OF RESPONSIBILITIES FOR RESEARCH AND SPONSORED PROGRAMS Vice President of Research & Technology Transfer: The responsibilities of the Vice President of Research &
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates!
 FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
Successful Stewardship: The Effective Management of Sponsored Projects. Closeout of a Sponsored Project Presented by Tim Patterson February 23, 2004
 Huron Consulting Group. All rights reserved. Successful Stewardship: The Effective Management of Sponsored Projects Closeout of a Sponsored Project Presented by Tim Patterson February 23, 2004 Experience.Redefined.
Huron Consulting Group. All rights reserved. Successful Stewardship: The Effective Management of Sponsored Projects Closeout of a Sponsored Project Presented by Tim Patterson February 23, 2004 Experience.Redefined.
Designing Internal Controls for Federal Grant Programs. April 9, 2015
 Designing Internal Controls for Federal Grant Programs April 9, 2015 Overview Grants Management FAMU Policies and Procedures Grants Management Roles and Responsibilities OMB Uniform Guidance (Super Circular)
Designing Internal Controls for Federal Grant Programs April 9, 2015 Overview Grants Management FAMU Policies and Procedures Grants Management Roles and Responsibilities OMB Uniform Guidance (Super Circular)
UC San Diego Policy & Procedure Manual
 UC San Diego Policy & Procedure Manual Search A Z Index Numerical Index Classification Guide What s New CONTRACTS AND GRANTS (RESEARCH) Section: 150-45 Effective: 08/01/2001 Supersedes: 05/26/1999 Review
UC San Diego Policy & Procedure Manual Search A Z Index Numerical Index Classification Guide What s New CONTRACTS AND GRANTS (RESEARCH) Section: 150-45 Effective: 08/01/2001 Supersedes: 05/26/1999 Review
Instructions for Completion of EXTRAMURAL PROPOSAL APPROVAL AND SUBMISSION SUMMARY "EPASS"
 Instructions for Completion of EXTRAMURAL PROPOSAL APPROVAL AND SUBMISSION SUMMARY "EPASS" This form is used for the review and approval of applications or proposals to be submitted to extramural funding
Instructions for Completion of EXTRAMURAL PROPOSAL APPROVAL AND SUBMISSION SUMMARY "EPASS" This form is used for the review and approval of applications or proposals to be submitted to extramural funding
The purpose of this survey
 The purpose of this survey Dear Colleague: As part of our ongoing effort to strengthen grants administration across Boston University, we have created this survey to gain a better understanding of your
The purpose of this survey Dear Colleague: As part of our ongoing effort to strengthen grants administration across Boston University, we have created this survey to gain a better understanding of your
PI Department. 12. Project Period Start 07/01/15 End 06/30/ Budget Period Start 07/01/15 End 06/30/ Award Type. 17.
 See PADR-at-a-Glance for how to use your PADR. 1. Account 3048119999 Awarded 2. MIS # 201509281969 3. Research Administrator RA name p - 257-9420 f - 323-1060 RA email 6. Sponsor 1001001332 Sponsor Name
See PADR-at-a-Glance for how to use your PADR. 1. Account 3048119999 Awarded 2. MIS # 201509281969 3. Research Administrator RA name p - 257-9420 f - 323-1060 RA email 6. Sponsor 1001001332 Sponsor Name
Contract & Grant User s Group
 Contract & Grant User s Group November 14, 2017 1:15 p.m. 3:15 p.m. Genomics Auditorium Business & Financial Services A Division of Business & Administration Services (BAS) BAS Agenda Overview of Temporary
Contract & Grant User s Group November 14, 2017 1:15 p.m. 3:15 p.m. Genomics Auditorium Business & Financial Services A Division of Business & Administration Services (BAS) BAS Agenda Overview of Temporary
Today s Topics. OCGA Quick Updates Kim Duiker and Heather Winters Updates from NIH Regional Seminar Kim Duiker S2S Grants Updates Cindy Gilbert
 August 8, 2013 Today s Topics OCGA Quick Updates Kim Duiker and Heather Winters Updates from NIH Regional Seminar Kim Duiker S2S Grants Updates Cindy Gilbert Quick Updates OCGA Staffing Update Three retirements
August 8, 2013 Today s Topics OCGA Quick Updates Kim Duiker and Heather Winters Updates from NIH Regional Seminar Kim Duiker S2S Grants Updates Cindy Gilbert Quick Updates OCGA Staffing Update Three retirements
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP)
 Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Research Proposal Application Process NUMBER: RAS SOP 1002 VERSION: 4.0 LAST REVISED: PREPARED BY: Office
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Research Proposal Application Process NUMBER: RAS SOP 1002 VERSION: 4.0 LAST REVISED: PREPARED BY: Office
IRES Proposal Tracking (PT) Presented by: Kathi Goodfriend Office of Sponsored Projects Revised 03/15/2018 PRN: 5/14/ :19 PM
 IRES Proposal Tracking (PT) Presented by: Kathi Goodfriend Office of Sponsored Projects Revised 03/15/2018 PRN: 5/14/2018 12:19 PM Topics Covered Class Objective:. To become more familiar with the IRES
IRES Proposal Tracking (PT) Presented by: Kathi Goodfriend Office of Sponsored Projects Revised 03/15/2018 PRN: 5/14/2018 12:19 PM Topics Covered Class Objective:. To become more familiar with the IRES
Dept/ College. Dept/ College. Dept/ College
 Office of Research Administration Matrix Revision date -2.10.17 Submitting proposals, executing awards, conducting research, and administrating sponsored projects involves many different people and units
Office of Research Administration Matrix Revision date -2.10.17 Submitting proposals, executing awards, conducting research, and administrating sponsored projects involves many different people and units
SJSU Research Foundation
 SJSU Research Foundation Grant Proposal Development Taking a Bite Out of Forms Sponsored Programs Workshop November 19, 2015 Overview of Workshop Why We Have Forms Forms from OSP Forms from Human Resources
SJSU Research Foundation Grant Proposal Development Taking a Bite Out of Forms Sponsored Programs Workshop November 19, 2015 Overview of Workshop Why We Have Forms Forms from OSP Forms from Human Resources
UNIFORM GUIDANCE IMPLEMENTATION
 UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
FINANCE-315 7/1/2017 SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a proposal to UACES. It provides a checklist of documents and certifications required by sponsors, as well as an
SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a proposal to UACES. It provides a checklist of documents and certifications required by sponsors, as well as an
OSR Monthly Meeting. August 16, 2018 (Chicago) August 21, 2018 (Evanston)
 OSR Monthly Meeting August 16, 2018 (Chicago) August 21, 2018 (Evanston) Agenda OSR Announcements/Staffing Updates ESPR Replacement Project Update Year-end Procedures NSF Updates NIH Biosketch Reminder
OSR Monthly Meeting August 16, 2018 (Chicago) August 21, 2018 (Evanston) Agenda OSR Announcements/Staffing Updates ESPR Replacement Project Update Year-end Procedures NSF Updates NIH Biosketch Reminder
UC San Francisco Supplier Diversity Basics. Module 2: Campus Responsibilities
 UC San Francisco Supplier Diversity Basics Module 2: Campus Responsibilities Supplier Diversity Basics Curriculum For UC San Francisco Employees What you need to know: Module 1: Policy and Regulatory Requirements
UC San Francisco Supplier Diversity Basics Module 2: Campus Responsibilities Supplier Diversity Basics Curriculum For UC San Francisco Employees What you need to know: Module 1: Policy and Regulatory Requirements
SPONSORED PROGRAMS AWARDS, EXPENDITURES, AND ALLOWABILTY APRIL 2015 Policy
 SPONSORED PROGRAMS AWARDS, EXPENDITURES, AND ALLOWABILTY APRIL 2015 Policy 2.2.01 Purpose: This policy was developed to ensure consistent compliance with 2 CFR part 200 Uniform Guidance and the Cost Accounting
SPONSORED PROGRAMS AWARDS, EXPENDITURES, AND ALLOWABILTY APRIL 2015 Policy 2.2.01 Purpose: This policy was developed to ensure consistent compliance with 2 CFR part 200 Uniform Guidance and the Cost Accounting
Grants Financial Procedures (Post-Award) v. 2.0
 Grants Financial Procedures (Post-Award) v. 2.0 1 Grants Financial Procedures (Post Award) Version Number: 2.0 Procedures Identifier: Superseded Procedure(s): BU-PR0001 N/A Date Approved: 9/1/2013 Effective
Grants Financial Procedures (Post-Award) v. 2.0 1 Grants Financial Procedures (Post Award) Version Number: 2.0 Procedures Identifier: Superseded Procedure(s): BU-PR0001 N/A Date Approved: 9/1/2013 Effective
Research Administration Frequently Asked Questions
 Research Administration Frequently Asked Questions I am submitting a proposal to NIH, but I can t download the grants.gov package, how do I submit? A. Use an old grants.gov package from a previous submission
Research Administration Frequently Asked Questions I am submitting a proposal to NIH, but I can t download the grants.gov package, how do I submit? A. Use an old grants.gov package from a previous submission
Proposal Preparation and Submission February 2018
 Proposal Preparation and Submission February 2018 Introductions We are. Chris Dye-Hixenbaugh, C&G Officer, Proposals Brooke Herevia, C&G Analyst, Proposals Shanna Nation Jose, C&G Analyst, Proposals Kassie
Proposal Preparation and Submission February 2018 Introductions We are. Chris Dye-Hixenbaugh, C&G Officer, Proposals Brooke Herevia, C&G Analyst, Proposals Shanna Nation Jose, C&G Analyst, Proposals Kassie
December 2015 Research Administration Working Group WELCOME OFFICE OF THE VICE PRESIDENT FOR RESEARCH
 December 2015 Research Administration Working Group WELCOME Open Mike Fringe Rates FY17 proposed Budget v. Charging Distribution of Indirects PI award letter out & accounts funded Departments- this month
December 2015 Research Administration Working Group WELCOME Open Mike Fringe Rates FY17 proposed Budget v. Charging Distribution of Indirects PI award letter out & accounts funded Departments- this month
Roles & Responsibilities University of Rochester Department Administrative Staff
 Roles and Responsibilities - UR Department Administrative Staff Roles & Responsibilities University of Rochester Department Administrative Staff Administrative Staff at the departmental, center or unit
Roles and Responsibilities - UR Department Administrative Staff Roles & Responsibilities University of Rochester Department Administrative Staff Administrative Staff at the departmental, center or unit
PERALTA COMMUNITY COLLEGE DISTRICT SINGLE AUDIT REPORT JUNE 30, 2010
 PERALTA COMMUNITY COLLEGE DISTRICT SINGLE AUDIT REPORT JUNE 30, 2010 TABLE OF CONTENTS JUNE 30, 2010 Independent Auditors' Report on Internal Control Over Financial Reporting and on Compliance and Other
PERALTA COMMUNITY COLLEGE DISTRICT SINGLE AUDIT REPORT JUNE 30, 2010 TABLE OF CONTENTS JUNE 30, 2010 Independent Auditors' Report on Internal Control Over Financial Reporting and on Compliance and Other
Roles & Responsibilities UR Office of Research and Project Administration
 Roles and Responsibilities - UR ORPA Roles & Responsibilities UR Office of Research and Project Administration The Office of Research and Projects Administration (ORPA) is the administrative office legally
Roles and Responsibilities - UR ORPA Roles & Responsibilities UR Office of Research and Project Administration The Office of Research and Projects Administration (ORPA) is the administrative office legally
Hello. National Grants Management Association Monthly Training November 16, Eric J. Russell, CIA, CGAP, CGMS, MPA Crowe Horwath LLP
 Hello. National Grants Management Association Monthly Training November 16, 2016 Eric J. Russell, CIA, CGAP, CGMS, MPA Crowe Horwath LLP 2016 Crowe 2016 Crowe Horwath Horwath LLP LLP Agenda Third Party
Hello. National Grants Management Association Monthly Training November 16, 2016 Eric J. Russell, CIA, CGAP, CGMS, MPA Crowe Horwath LLP 2016 Crowe 2016 Crowe Horwath Horwath LLP LLP Agenda Third Party
UNC Lineberger Developmental Funding Program. Proposal Due Dates: 5:00pm March 15 and September 15
 Proposal Due Dates: 5:00pm March 15 and September 15 Letter of Intent for Tier 3: Multi-Project Awards due three weeks before due date The UNC Lineberger Developmental Funding Program is intended to support
Proposal Due Dates: 5:00pm March 15 and September 15 Letter of Intent for Tier 3: Multi-Project Awards due three weeks before due date The UNC Lineberger Developmental Funding Program is intended to support
2017 UC Multicampus Research Funding Opportunities
 2017 UC Multicampus Research Funding Opportunities The UC Multicampus Research Programs and Initiatives The President s Research Catalyst Award REQUEST FOR PROPOSALS v. March 9, 2016 (draft; proposals
2017 UC Multicampus Research Funding Opportunities The UC Multicampus Research Programs and Initiatives The President s Research Catalyst Award REQUEST FOR PROPOSALS v. March 9, 2016 (draft; proposals
Uniform Guidance. Overview and Implementation Plan. November 21, 2014
 Uniform Guidance Overview and Implementation Plan November 21, 2014 Uniform Guidance. Change is coming! College or Department name here 1 Overarching goal of the reform is to: streamline the rules and
Uniform Guidance Overview and Implementation Plan November 21, 2014 Uniform Guidance. Change is coming! College or Department name here 1 Overarching goal of the reform is to: streamline the rules and
Objectives for Financial Control over Grant Programs
 Objectives for Financial Control over Grant Programs I. Cash management of grant funds is monitored for appropriate timing of receipts and disbursements of grant funds. (Cash Management) II. Procedures
Objectives for Financial Control over Grant Programs I. Cash management of grant funds is monitored for appropriate timing of receipts and disbursements of grant funds. (Cash Management) II. Procedures
Subrecipient Risk Assessment and Monitoring of Northeastern University Issued Subawards
 Subrecipient Risk Assessment and Monitoring of Northeastern University Issued Subawards What is a Subaward? A Subaward is a contractual agreement between Northeastern University and a third party organization
Subrecipient Risk Assessment and Monitoring of Northeastern University Issued Subawards What is a Subaward? A Subaward is a contractual agreement between Northeastern University and a third party organization
N a t i o n a l C o u n c i l o f U n i v e r s i t y R e s e a r c h A d m i n i s t r a t o r s NCURA PEER REVIEW FINAL REPORT JUNE 27, 2012
 N a t i o n a l C o u n c i l o f U n i v e r s i t y R e s e a r c h A d m i n i s t r a t o r s UNIVERSITY OF CALIFORNIA MERCED NCURA PEER REVIEW FINAL REPORT JUNE 27, 2012 Site Visit April 9-11, 2012
N a t i o n a l C o u n c i l o f U n i v e r s i t y R e s e a r c h A d m i n i s t r a t o r s UNIVERSITY OF CALIFORNIA MERCED NCURA PEER REVIEW FINAL REPORT JUNE 27, 2012 Site Visit April 9-11, 2012
SPONSORED PROJECT CLOSE-OUT/PAYMENT COLLECTION GUIDELINE
 SPONSORED PROJECT CLOSE-OUT/PAYMENT COLLECTION GUIDELINE Purpose: To provide guidance to assist Principal Investigators (PI s), Award Managers, UAB research/fiscal administrators in close-out, invoicing,
SPONSORED PROJECT CLOSE-OUT/PAYMENT COLLECTION GUIDELINE Purpose: To provide guidance to assist Principal Investigators (PI s), Award Managers, UAB research/fiscal administrators in close-out, invoicing,
PEER (PAPERLESS ENVIRONMENT FOR ELECTRONIC REVIEW)
 PEER (PAPERLESS ENVIRONMENT FOR ELECTRONIC REVIEW) 1 What is PEER? PEER is a web portal maintained by Vanderbilt University which handles internal processing of grant related requests and contract submission
PEER (PAPERLESS ENVIRONMENT FOR ELECTRONIC REVIEW) 1 What is PEER? PEER is a web portal maintained by Vanderbilt University which handles internal processing of grant related requests and contract submission
Financial Research Compliance. April 2013
 Financial Research Compliance April 2013 Overview I. What is a Sponsored Award? II. Sponsored Awards at WFU III. Compliance IV. Hot Topics V. Audits VI. WFU Updates VII. Questions VIII. Contacts What is
Financial Research Compliance April 2013 Overview I. What is a Sponsored Award? II. Sponsored Awards at WFU III. Compliance IV. Hot Topics V. Audits VI. WFU Updates VII. Questions VIII. Contacts What is
