AURA Meeting. July 23, 2014
|
|
|
- Thomasine Carroll
- 6 years ago
- Views:
Transcription
1 AURA Meeting July 23, 2014
2 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration
3 Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored Projects Administration go.uth.edu\spa
4 Assigning a Delegate Sometimes, faculty members are unavailable. Delegates can be assigned to act in their stead. Delegates can see and edit proposals the faculty member can see or edit and will get the same notifications as the faculty member does, both and in the system. This includes the ability to route the application. Information on Assigning a Delegate can be found in three locations: Systems & Reporting -> Cayuses424 ->Helpful Information Table of Contents-> Assigning a Delegate Table of Contents-> Cayuse424-> Helpful Information
5 SPA Twitter Keith Comfort Financial Analyst, Post Award Finance Sponsored Projects Administration
6 SPA Twitter Home Page
7 Organizations on Twitter
8 Sample Lists of Health Tweeters
9 Sample NIH Tweets
10 Medical & Health Tweets
11 Tweets by Federal Health Agencies
12 OSP Name Change Karen Niemeier Contracts Director Sponsored Projects Administration
13 OSP Name Change
14 Research Conflicts of Interest Revised Certification and Disclosure Forms Loretta Davis, MPA Manager, Conflicts of Interest Program Office of Institutional Compliance (713) UCT 1510
15 Research COI Requirements - Overview Covered Individuals must complete the following requirements: 1. Complete the required RCOI education. (Employees fulfill this requirement through UTHealth s Mandatory Compliance Training. Others may take the RCOI training module available on the CITI site if they have not taken a comparable course at their own institution or company.) 2. Submit a "Research COI Certification Form" for each proposed research project: to SPA; to CPHS; upon request by the AWC; to the department for startup funds, gifts, endowments, etc. designated for their research; and to the program or official making the funding decision for internal awards. 2A. If there is a related Significant Financial Interest for a research project, submit a "Research COI Disclosure Form. 3. Submit an annual Financial Disclosure Statement.
16 Research Conflict of Interest Certification Form Effective July, 2014, a revised Research COI Certification Form is available. As always, the 1-page form is required of all Covered Individuals on proposed research grants, contracts, and protocols. The form is located at: and is also embedded within the OSP Review & Approval Form, and within the iris submission form. New: Two questions have been added, for a total of seven Yes/No questions:
17 Research Conflict of Interest Certification Form 1. Do you or a family member participate in an outside activity with the research sponsor that would be defined as a Significant Financial Interest? 2. Do you or a family member own stock, stock options, business ownership, or rights to such interests in the research sponsor, that would be defined as a Significant Financial Interest? 3. Do you or a family member personally own rights to the technology that will be studied or validated in the proposed research? (Does not include intellectual property owned by UTHealth.) 4. Do you or a family member have a Significant Financial Interest in an entity that owns or licenses the technology that will be studied or validated in the proposed research? 5. Do you or a family member have any other personal relationship or financial interest that could appear to affect, or be affected by, the proposed research? 6. New: Do you have an existing UTHealth Research Conflict of Interest Management Plan for this sponsor, or for the technology that will be studied or validated in the proposed research? 7. New: Are you aware of any Significant Financial Interest held by your supervisor (including a department chair, or a graduate or postdoctoral advisor) with the sponsor, or in the technology that will be studied or validated in the proposed research?
18 Research Conflict of Interest Disclosure Form Effective July, 2014, a revised Research COI Disclosure Form will be available. As always, the 3-page form is required only when a Covered Individual has certified a Significant Financial Interest with a proposed research project (as reported on his/her RCOI Certification Form). The Form is located at: New: The form is no longer required to be submitted with the proposal. New: Once the Research COI Office initiates a research conflict of interest review, the researcher will be contacted and the form will be requested at that time. New: Forms will be submitted electronically to Research_COI@uth.tmc.edu. (They may also be mailed to the RCOI Program Office, UCT 1510; however, incomplete forms will not be accepted.)
19 Research Conflict of Interest Disclosure Form The RCOI Disclosure Form obtains information about the investigator s related Significant Financial Interests, and the research, so that the RCOI Committee may conduct its review and make its recommendations to the EVPARA. 1. Financial interests and relationships; 2. Other financial interests (of supervisors, UTHealth, etc.); 3. Intellectual property; 4. Manufacture or commercialization of research-related items; 5. Use of experimental animals; 6. Human subjects research; 7. Research performance sites; 8. Description of the research; 9. Role(s) in the research; and 10. Whether an existing Management Plan (if one has been implemented) provides sufficient management strategies.
20 Financial Conflicts of Interest in Research From the Research Conflicts of Interest Policy (HOOP 94), Section III F: Research may be allowed in the face of a conflict depending upon: 1) the nature of the science; 2) the specific financial interest; 3) the magnitude of the interest and the degree to which it is related to the research; and 4) the extent to which the interest is amenable to effective oversight and management. Neither the institution nor a Covered Individual may expend research funds unless the EVPARA has determined that no financial conflict of interest exists or that any financial conflict of interest is manageable in accordance with the terms of a management plan that has been adopted and implemented.
21 SPA Training Program Amaris Ogu, MBA Clinical Research Financial Analyst Sponsored Projects Administration
22 SPA Training Program SPA Training Program is a series of training resources developed by Sponsored Projects Administration (SPA) to cover basic and advanced topics in research administration relevant to UTHealth policies and procedures.
23 Training Resources SPA Training Course o 3 day training course o Basic to intermediate topics o SPA and guest presenters o Hands on experience o First Sessions October 2014 & April 2015 Online Modules o Advanced Topics o Available September 2014 Website o Internal Training Opportunities o External Training Opportunities o Tips and Tools o go.uth.edu/ SPATraining
24 01 SPA Overview Purpose SPA Breakdown o Grants, Contracts, PAF, Systems & Reporting, Clinical Research Finance o Contact/Specialist List Signature Authority Hoop 124 o Delegation of Authority o SPA signature matrix Institutional Information o Institutional Data Sheet o Tax Status o UT Foundation Website Resources (News Feed, Training section: checklist & index) AURA Internal Routing/Review Process o Life Cycle of a Sponsored Project o Internal review process o Internal Deadlines Finding Funding/PI Initiates Proposal o SciVal o Other Mechanism to Find Funding Proposal Development A&F Packet Investigator/Department Responsibility R&A Forms (includes filling out) o R&A Form o Non monetary R&A o R&A Addendum o Additional Personnel Addendum o Signature Page Addendum RCOI o Conflict of Interest Disclosure Form o Annual Financial Disclosure o Define Covered Individual Agency Instructions & Guidelines o Uniform Circular Subcontracts (In/Out o Scope of Work o Letter of Intent/Face Page o IDC rate agreement o PHS FCOI Regulation/ Sub recipient COI form/fdp Institutional Clearinghouse o Budget/Budget Justification Exception Letters o Indirect Cost Waiver o Cost Sharing o Appointment Letters o Limited Nomination Assurances o Types Grant Application o Components/Parts of Application o Personnel Types o Multi PI Plan Budget & Budget Justification o Agency Guidelines o Effort Minimum Effort (HOOP 93) o Modular vs. Non Modular o Allowable Cost/ OMB A 21 o Indirect Cost o Exclusions Clinical Trial Budgets o Research Cost/Standard of Care Cost 02 Contracts Definition of a contract ocontract vs. Grant Internal Review Process o Submit R&A: Non monetary vs Monetary o Assurances Needed Types o Authorized Officials Negotiation Process o Negotiation Points Confidential Disclosure Agreements o OTM s involvement Material Transfer Agreements o UBMTA o AUTM Clinical Trial /Sponsored Research Agreements Subcontracts In/Out o FDP o A 133 Compliance Validation o Sub recipient vs. Vendor Data Use Agreements In/Out Salary Reimbursement Agreements Collaboration Agreements Proposal Submission/Tracking Tracking for Federal Proposal After the Fact Proposals Withdrawing a Proposal JIT o What is a JIT? o Internal review process o Timeline for submission o Required documents Post Submission Materials Award Receipt/NoA Notice of Award o Restrictions Project Set Up Process Guarantee Accounts Split Account Request X TRAIN appointment Award Management Transfer In/Out Budget Modifications No Cost Extensions Account Deficits Buy Cards Cost Transfers Personnel Actions o Retro PA s Prior Approval Request o Change in Scope o Change in Personnel o Change in Effort Account Reconciliation Financial & Progress Reporting Progress Report/RPPR/Continuations Financial Status Reports 03 o Carry Forward Request Award Close out Relinquishing Letters Final Progress Report PAF Project Account Close Out Process FSR & FIS Effort Effort/Effort Certification at UTHEALTH o What is/not Effort? o What is ecrt o Why is Effort Certification Important? o Effort Reporting Lifecycle & Calendar o Reporting Roles & Responsibilities o What is cost sharing? o Salary Cap Calculations o Clinical Trials o Forms o Certifying Effort Frequently asked questions Effort Guidelines & Policies o Federal Requirements o Minimum & Maximum Levels o Risk of Noncompliance o Reporting Schedule o Travel Restrictions Procedure Frequently asked questions with Effort Systems & Reporting Cayuse o Creating an account o Administrator Access o Login Instructions o How to create a new proposal o Routing chain creations o Non federal proposal upload o Subcontract upload o Transform and existing proposal o Permissions era Commons & Other Systems o ID Requests o Help Desk Issues SAM Registration
25 SPA Training Contact Information For more information please us at
26 CRF Policies/Training Heather Cody, MHA Supervisor, Clinical Research Finance Sponsored Projects Administration
27 CRF Policies/Training Effective 9/1 Clinical Research Finance will launch 2 new procedures designed to ensure the accuracy of clinical research billing. Needed to meet new regulation Ensures accurate billing of research costs
28 CRF Policies/Training Coverage Analysis Evaluates whether clinical services/items in a research study should be billed to the sponsor or as standard of care to a 3 rd party payor Justifies procedures that are deemed standard of care Registration/Scheduling Outlines a procedure to identify study patients in GE Centricity Designates how the study patient will be scheduled Allows for markers to identify claims that should receive evaluation prior to submission to 3 rd party payors
29 CRF Policies/Training Training for new policies Overview trainings for Coverage Analysis and Registration/Scheduling (3 available times) Trainings will cover a completed coverage analysis example and a high overview of the clinical research billing process. Advanced Training (2 available times) You should attend the overview 1 st A coverage analysis will be completed during training
30 CRF Team Heather Cody, MHA - Supervisor Heather.cody@uth.tmc.edu Amaris Ogu, MBA Financial Analyst Amaris.e.ogu@uth.tmc.edu Trae Rohan, BA, CHRC - Regulatory Specialist Trae.Rohan@uth.tmc.edu CRF Group - grp-crf@uthouston.edu
31 Effort Reporting William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration
32 Effort Reporting Certification Period is now open July 21, 2014 through August 21, 2014 Travel restrictions effective August 25, 2014 Submit any retroactive PAs Place cards on hold if changes pending Contact the Effort Reporting Team or
33 Subaccount Transition Update Victoria Briscoe Assistant Director, Post Award Finance Sponsored Projects Administration
34 Subaccount Transition Update NIH Notice: NOT-OD
35 Facilities & Administrative Cost Rate Agreement Jodi S. Ogden, MBA, CRA Associate Vice President Sponsored Projects Administration
36 F&A Rate Agreement Institutional Information: go.uth.edu/instinfo
37 F&A Rate Agreement Off campus definition: For all activities performed in facilities not owned by the institution and to which rent is directly allocated to the project(s) the off-campus rate will apply. Grants or contracts will not be subject to more than one F&A cost rate. If more than 50% of a project is performed offcampus, the off-campus rate will apply to the entire project.
38 HOOP 69
39 HOOP 69 Changes B. Non-Federal Sponsored Projects Applications for non-federal sponsored projects shall include a minimum of 30 percent of total direct costs as indirect costs in the budget, unless: The funding agency has a written policy that it pays a different fixed indirect cost rate The request for proposal or funding opportunity announcement details a different indirect cost rate The agency provides a letter signed by a representative on official letterhead that it will pay a different rate on the specific award
40 Uniform Guidance Jodi S. Ogden, MBA, CRA Associate Vice President Sponsored Projects Administration
41 Uniform Guidance OMB is streamlining the Federal government's guidance on Administrative Requirements, Cost Principles, and Audit Requirements for Federal awards. These modifications are a key component of a larger Federal effort to more effectively focus Federal resources on improving performance and outcomes while ensuring the financial integrity of taxpayer dollars in partnership with non-federal stakeholders.
42 Uniform Guidance or A-81 This final guidance supersedes and streamlines requirements from OMB Circulars: A-21 A-87 A-110 A-122 A-89 A-102 A-133 A-50
43 Uniform Guidance or A-81 SPA Leadership team is reviewing the following: Cost Accounting Standards & DS2 Internal Controls Procurement Standard Terms and Conditions Closeout Subawards & Subrecipient Monitoring Conflict of Interest Compensation & Effort Reporting Compensation & Fringe Benefits Performance Measurement Direct Costs Administrative Salaries Prior Approval Requirements for Pass-Through Entities Indirect Costs Travel Costs Exchange Rates Fixed Amount Awards Equipment Program Income
44 Uniform Guidance or A-81 Implementation date is December 27, 2014
45 Next Meeting: September 24, 2014 Presentations & Schedule Posted at: go.uth.edu/aura
RESEARCH CONFLICT OF INTEREST. Vyju Ram, MD Conflict of Interest Program
 RESEARCH CONFLICT OF INTEREST Vyju Ram, MD Conflict of Interest Program Research Conflict of Interest (RCOI) Policy Federal policy (42 CFR Part 50, Subpart F)- purpose is to promote objectivity in research
RESEARCH CONFLICT OF INTEREST Vyju Ram, MD Conflict of Interest Program Research Conflict of Interest (RCOI) Policy Federal policy (42 CFR Part 50, Subpart F)- purpose is to promote objectivity in research
An Overview of the Pre- Award Grant Process
 An Overview of the Pre- Award Grant Process Tiffany Sagers Institutional Wide Grant II Direct: 713-500-3492 Main: 713-500-3999 Email: Tiffany.Sagers@uth.tmc.edu Sponsored Projects Administration (SPA)
An Overview of the Pre- Award Grant Process Tiffany Sagers Institutional Wide Grant II Direct: 713-500-3492 Main: 713-500-3999 Email: Tiffany.Sagers@uth.tmc.edu Sponsored Projects Administration (SPA)
OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015
 OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015 INTRODUCTION The University of Wisconsin-Milwaukee (UWM) is the prime recipient on a wide range of sponsored awards
OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015 INTRODUCTION The University of Wisconsin-Milwaukee (UWM) is the prime recipient on a wide range of sponsored awards
Outgoing Subagreements: Subawards and Subcontracts
 Effective Date: March 1, 2014 Version: 1 Page: 1 of 6 Overview Sponsored programs may include partnerships with other organizations or institutions whereby a portion of the project is conducted by investigators
Effective Date: March 1, 2014 Version: 1 Page: 1 of 6 Overview Sponsored programs may include partnerships with other organizations or institutions whereby a portion of the project is conducted by investigators
An Overview of the Pre- Award Grant Process
 An Overview of the Pre- Award Grant Process Tiffany Sagers Institutional Wide Grant II Direct: 713-500-3492 Main: 713-500-3999 Email: Tiffany.Sagers@uth.tmc.edu Sponsored Projects Administration (SPA)
An Overview of the Pre- Award Grant Process Tiffany Sagers Institutional Wide Grant II Direct: 713-500-3492 Main: 713-500-3999 Email: Tiffany.Sagers@uth.tmc.edu Sponsored Projects Administration (SPA)
Subrecipient Monitoring Procedures
 Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
Sponsored Programs Roles & Responsibilities
 The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
Pre Award Table of Contents
 Pre Award Table of Contents ORA Internal Procedures for Account Establishment 2 Conflict of Interest: Thomas Jefferson University Conflict of Interest (COI) Policy 4 Cost Sharing and Matching Funds 6 Developing
Pre Award Table of Contents ORA Internal Procedures for Account Establishment 2 Conflict of Interest: Thomas Jefferson University Conflict of Interest (COI) Policy 4 Cost Sharing and Matching Funds 6 Developing
Sponsored Programs Roles & Responsibilities
 The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
Subawards and Subrecipient Monitoring
 Subawards and Subrecipient Monitoring H O L LY S O M M E R S D I R E C T O R, O S P G R A N T S J O S H R O S E N B E R G A C T I N G D I R E C T O R, O G C A Objectives 2 Distinguish Between: Procurement
Subawards and Subrecipient Monitoring H O L LY S O M M E R S D I R E C T O R, O S P G R A N T S J O S H R O S E N B E R G A C T I N G D I R E C T O R, O G C A Objectives 2 Distinguish Between: Procurement
Principal Investigator
 Appendix A: Roles and Responsibilities Details Principal Investigator 1. Proposal Preparation 1.1 Has an overall responsibility to assemble a submission ready proposal meaning a complete, accurate proposal
Appendix A: Roles and Responsibilities Details Principal Investigator 1. Proposal Preparation 1.1 Has an overall responsibility to assemble a submission ready proposal meaning a complete, accurate proposal
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP)
 Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Research Proposal Application Process NUMBER: RAS SOP 1002 VERSION: 4.0 LAST REVISED: PREPARED BY: Office
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Research Proposal Application Process NUMBER: RAS SOP 1002 VERSION: 4.0 LAST REVISED: PREPARED BY: Office
UNIVERSITY RESEARCH ADMINISTRATION FINANCIAL ROLES AND RESPONSIBILITIES MATRIX - WORK IN PROGRESS 10/03/2013 Roles.
 UNIVERSITY RESEARCH ADMINISTRATION Roles Business Internal Controller's Clinical Responsibilities PI Office Chair Dean Audit Office OCR GCFA GCA PROVOST Trials Office I. GENERAL RESEARCH ADMINISTRATION
UNIVERSITY RESEARCH ADMINISTRATION Roles Business Internal Controller's Clinical Responsibilities PI Office Chair Dean Audit Office OCR GCFA GCA PROVOST Trials Office I. GENERAL RESEARCH ADMINISTRATION
University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures
 Summary 1. Subaward Definitions A. Subaward B. Subrecipient University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures C. Office of Contracts and Grants (OCG) 2. Distinguishing
Summary 1. Subaward Definitions A. Subaward B. Subrecipient University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures C. Office of Contracts and Grants (OCG) 2. Distinguishing
Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed:
 COEUS NIH-GRANTS.GOV PROPOSAL REVIEW CHECKLIST REVIEWER: Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed: Has the Program Announcement been reviewed? What is
COEUS NIH-GRANTS.GOV PROPOSAL REVIEW CHECKLIST REVIEWER: Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed: Has the Program Announcement been reviewed? What is
The Lifecycle of a Sponsored Project
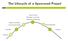 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
Designing Internal Controls for Federal Grant Programs. April 9, 2015
 Designing Internal Controls for Federal Grant Programs April 9, 2015 Overview Grants Management FAMU Policies and Procedures Grants Management Roles and Responsibilities OMB Uniform Guidance (Super Circular)
Designing Internal Controls for Federal Grant Programs April 9, 2015 Overview Grants Management FAMU Policies and Procedures Grants Management Roles and Responsibilities OMB Uniform Guidance (Super Circular)
INDIRECT COST POLICY
 UNIVERSITY OF LOUISIANA AT LAFAYETTE OFFICE OF THE VICE PRESIDENT FOR RESEARCH INDIRECT COST POLICY Revision Date: 8/11/2014 Original Effective Date: 11/08/2006 Responsible Office: Reference: Vice President
UNIVERSITY OF LOUISIANA AT LAFAYETTE OFFICE OF THE VICE PRESIDENT FOR RESEARCH INDIRECT COST POLICY Revision Date: 8/11/2014 Original Effective Date: 11/08/2006 Responsible Office: Reference: Vice President
Proposal Preparation and Submission February 2018
 Proposal Preparation and Submission February 2018 Introductions We are. Chris Dye-Hixenbaugh, C&G Officer, Proposals Brooke Herevia, C&G Analyst, Proposals Shanna Nation Jose, C&G Analyst, Proposals Kassie
Proposal Preparation and Submission February 2018 Introductions We are. Chris Dye-Hixenbaugh, C&G Officer, Proposals Brooke Herevia, C&G Analyst, Proposals Shanna Nation Jose, C&G Analyst, Proposals Kassie
Finding Funding, Budget Preparation, and Proposal Submission for Sponsored Research
 Finding Funding, Budget Preparation, and Proposal Submission for Sponsored Research Laura Johnston, Office of Sponsored Programs, Asst. Director Craig O Neill, Office of Sponsored Programs, Manager February
Finding Funding, Budget Preparation, and Proposal Submission for Sponsored Research Laura Johnston, Office of Sponsored Programs, Asst. Director Craig O Neill, Office of Sponsored Programs, Manager February
Grant Administration Glossary of Commonly-Used Terms in Sponsored Programs
 Page 1 of 6 Grant Administration Allowability: The determination of whether or not costs can be charged to a sponsored project as a direct or indirect cost. Allocability: A cost is allocable to a particular
Page 1 of 6 Grant Administration Allowability: The determination of whether or not costs can be charged to a sponsored project as a direct or indirect cost. Allocability: A cost is allocable to a particular
SJSU Research Foundation Cost Share Policy
 SJSU Research Foundation Cost Share Policy Office of Sponsored Programs Policy No.: Effective Date: Supersedes: n/a Publication Date: OSP. 03-04-001 Rev. A 05/01/2017 6/29/2017 1.0 Purpose The Cost Share
SJSU Research Foundation Cost Share Policy Office of Sponsored Programs Policy No.: Effective Date: Supersedes: n/a Publication Date: OSP. 03-04-001 Rev. A 05/01/2017 6/29/2017 1.0 Purpose The Cost Share
University of Pittsburgh SPONSORED PROJECT FINANCIAL GUIDELINE Subject: SUBRECIPIENT MONITORING
 I. Scope Subrecipient monitoring is the process by which the University selects, monitors, controls, and accounts for University subcontractors or subrecipients utilized on sponsored projects in accordance
I. Scope Subrecipient monitoring is the process by which the University selects, monitors, controls, and accounts for University subcontractors or subrecipients utilized on sponsored projects in accordance
Subrecipient vs. Contractor: Guidance on Appropriate Classification of Legal Relationship
 HARVARD UNIVERSITY Responsible Office: Office for Sponsored Programs Date First Effective: October 18, 2011 Revision Date: December 26, 2014 Subrecipient vs. Contractor: Guidance on Appropriate Classification
HARVARD UNIVERSITY Responsible Office: Office for Sponsored Programs Date First Effective: October 18, 2011 Revision Date: December 26, 2014 Subrecipient vs. Contractor: Guidance on Appropriate Classification
Grant/Sponsor Related Systems. Department and OSP Perspectives on ERA
 Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
Award Transfer Guidelines
 Award Transfer Guidelines The process of transferring awards from one institution to another is not a simple task and requires negotiation between the sponsoring agency, the old institution and the new
Award Transfer Guidelines The process of transferring awards from one institution to another is not a simple task and requires negotiation between the sponsoring agency, the old institution and the new
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP)
 Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Complex Award Management Program Projects/Multi-Fund Awards- Post Award NUMBER: RAS SOP 2001 VERSION: 4.0
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Complex Award Management Program Projects/Multi-Fund Awards- Post Award NUMBER: RAS SOP 2001 VERSION: 4.0
THE DRA S GUIDE TO ERA
 THE DRA S GUIDE TO ERA Updated: 11/8/2017 Contents I. Getting Started 2 How do I log in to era?... 2 How do I find and open proposal records?... 2 II. Proposals 4 PT Navigation... 4 How do I find the status
THE DRA S GUIDE TO ERA Updated: 11/8/2017 Contents I. Getting Started 2 How do I log in to era?... 2 How do I find and open proposal records?... 2 II. Proposals 4 PT Navigation... 4 How do I find the status
Outgoing Subaward Guide and F-A-Qs
 1. What is a subaward? Outgoing Subaward Guide and F-A-Qs 2. After an award is made to Rutgers, how is a subaward established? 3. What is the outgoing subaward process? 4. How can a subaward be changed/amended?
1. What is a subaward? Outgoing Subaward Guide and F-A-Qs 2. After an award is made to Rutgers, how is a subaward established? 3. What is the outgoing subaward process? 4. How can a subaward be changed/amended?
Policy on Cost Allocation, Cost Recovery, and Cost Sharing
 President Page 1 of 11 PURPOSE: Provide guidance and structure when allocating and documenting costs (direct and indirect) for extramurally funded awards. Serves to provide direction for budgeting, allocating
President Page 1 of 11 PURPOSE: Provide guidance and structure when allocating and documenting costs (direct and indirect) for extramurally funded awards. Serves to provide direction for budgeting, allocating
Research Administration at Emory Foundation Program DAY TWO: The Financial Administration of Sponsored Programs
 Revised 05/2018 Research Administration at Emory Foundation Program DAY TWO: The Financial Administration of Sponsored Programs Presenter: Finance: Grants and Contracts (FGC) Jumionne Tiako AD Post Award
Revised 05/2018 Research Administration at Emory Foundation Program DAY TWO: The Financial Administration of Sponsored Programs Presenter: Finance: Grants and Contracts (FGC) Jumionne Tiako AD Post Award
Emory Research A to Z ERAZ
 Emory Research A to Z ERAZ July 17, 2014 Whitehead Auditorium G01 Whitehead Building Agenda Controlled Substances Sub Award Processing RAS Updates SAM Kiosk Federal Updates OSP/OGCA Training Updates Christine
Emory Research A to Z ERAZ July 17, 2014 Whitehead Auditorium G01 Whitehead Building Agenda Controlled Substances Sub Award Processing RAS Updates SAM Kiosk Federal Updates OSP/OGCA Training Updates Christine
OMB Uniform Guidance ( UG ) Briefing. ASRSP & OSR Brown Bag Tuesday, January 27 th
 OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
Guidelines for the Acceptance and Use of Externally Funded Grants and Contracts
 Guidelines for the Acceptance and Use of Externally Funded Grants and Contracts http://www.una.edu/sponsored-programs/ Tanja F. Blackstone, PhD Director, Sponsored Programs tfblackstone@una.edu University
Guidelines for the Acceptance and Use of Externally Funded Grants and Contracts http://www.una.edu/sponsored-programs/ Tanja F. Blackstone, PhD Director, Sponsored Programs tfblackstone@una.edu University
University of Kansas Medical Center Research Institute. What Every Administrator Ought to Know About Grant Management
 University of Kansas Medical Center Research Institute What Every Administrator Ought to Know About Grant Management Who is the RI? The University of Kansas Medical Center Research Institute, Inc. is a
University of Kansas Medical Center Research Institute What Every Administrator Ought to Know About Grant Management Who is the RI? The University of Kansas Medical Center Research Institute, Inc. is a
SYSTEM REQUIREMENTS AND USEFUL INFORMATION LOGGING INTO THE PERIS PORTAL
 SYSTEM REQUIREMENTS AND USEFUL INFORMATION ------------------------------------------------- LOGGING INTO THE PERIS PORTAL -------------------------------------------------------------------------- CREATING
SYSTEM REQUIREMENTS AND USEFUL INFORMATION ------------------------------------------------- LOGGING INTO THE PERIS PORTAL -------------------------------------------------------------------------- CREATING
East Carolina University Sponsored Projects Administration Roles and Responsibilities Matrix By Responsibility
 (See legend at end of matrix.) East Carolina University Sponsored Projects Administration Roles and Responsibilities Matrix By Responsibility This matrix is designed to provide general guidance on administrative
(See legend at end of matrix.) East Carolina University Sponsored Projects Administration Roles and Responsibilities Matrix By Responsibility This matrix is designed to provide general guidance on administrative
Subaward Policies and Procedures Manual
 JOHNS HOPKINS UNIVERISTY Krieger School of Arts and Sciences Business and Research Administration Subaward Policies and Procedures Manual 2016 Contents Introduction...3 Differentiating types of relationships...3
JOHNS HOPKINS UNIVERISTY Krieger School of Arts and Sciences Business and Research Administration Subaward Policies and Procedures Manual 2016 Contents Introduction...3 Differentiating types of relationships...3
Sponsored Programs and Research Compliance SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a subaward proposal to University of North Carolina Wilmington (UNCW). Please complete this form and send all required
SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a subaward proposal to University of North Carolina Wilmington (UNCW). Please complete this form and send all required
Finding Funding, Budget Preparation, and Proposal Submission for Sponsored Research
 Finding Funding, Budget Preparation, and Proposal Submission for Sponsored Research Laura Johnston, Office of Sponsored Programs, Asst. Director February 24, 2017 1 Agenda A Walkthrough of MSU s Proposal
Finding Funding, Budget Preparation, and Proposal Submission for Sponsored Research Laura Johnston, Office of Sponsored Programs, Asst. Director February 24, 2017 1 Agenda A Walkthrough of MSU s Proposal
Roles & Responsibilities UR Office of Research and Project Administration
 Roles and Responsibilities - UR ORPA Roles & Responsibilities UR Office of Research and Project Administration The Office of Research and Projects Administration (ORPA) is the administrative office legally
Roles and Responsibilities - UR ORPA Roles & Responsibilities UR Office of Research and Project Administration The Office of Research and Projects Administration (ORPA) is the administrative office legally
Research Administration Services Roles & Responsibilities For Grants and Contracts (Excludes Clinical Trials) Version 3.1
 Research Administration Services Roles & Responsibilities For Grants and Contracts (Excludes Clinical Trials) Version 3.1 Category PI/PI Staff Department* RAS Unit School/Unit ORA Offices Relevant SOP
Research Administration Services Roles & Responsibilities For Grants and Contracts (Excludes Clinical Trials) Version 3.1 Category PI/PI Staff Department* RAS Unit School/Unit ORA Offices Relevant SOP
Research Administration Frequently Asked Questions
 Research Administration Frequently Asked Questions I am submitting a proposal to NIH, but I can t download the grants.gov package, how do I submit? A. Use an old grants.gov package from a previous submission
Research Administration Frequently Asked Questions I am submitting a proposal to NIH, but I can t download the grants.gov package, how do I submit? A. Use an old grants.gov package from a previous submission
STATEMENT OF POLICY PURPOSE
 STATEMENT OF POLICY PURPOSE INDIRECT COST POLICY Winston-Salem State University receives reimbursement of Facilities and Administrative (F&A) costs related to grants and contracts and will allocate these
STATEMENT OF POLICY PURPOSE INDIRECT COST POLICY Winston-Salem State University receives reimbursement of Facilities and Administrative (F&A) costs related to grants and contracts and will allocate these
2017 POST AWARD FREQUENTLY ASKED QUESTIONS (FAQS) Post Award FAQS
 2017 POST AWARD FREQUENTLY ASKED QUESTIONS (FAQS) Post Award FAQS 1. What is Pre-Award and where is it located? Pre-Award is under the purview of the Office of Sponsored Programs (OSP). They assists in
2017 POST AWARD FREQUENTLY ASKED QUESTIONS (FAQS) Post Award FAQS 1. What is Pre-Award and where is it located? Pre-Award is under the purview of the Office of Sponsored Programs (OSP). They assists in
4.12 Effort Certification
 4.112 Grants and Cooperative Agreements 4.1121 The Public Health Service ("PHS") and the National Science Foundation ("NSF") have regulations promoting objectivity in research by requiring that a university
4.112 Grants and Cooperative Agreements 4.1121 The Public Health Service ("PHS") and the National Science Foundation ("NSF") have regulations promoting objectivity in research by requiring that a university
Research Administrators Forum September 13, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum September 13, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith OCGA Initiatives Patti Manheim, Kim Duiker Proposal Intake
Research Administrators Forum September 13, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith OCGA Initiatives Patti Manheim, Kim Duiker Proposal Intake
FINANCE-315 7/1/2017 SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a proposal to UACES. It provides a checklist of documents and certifications required by sponsors, as well as an
SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a proposal to UACES. It provides a checklist of documents and certifications required by sponsors, as well as an
Research Administration Updates. May 16, :00 am IOP Auditorium
 Research Administration Updates May 16, 2018 11:00 am IOP Auditorium Topics Covered Using ClinCard (Greenphire) for Federally Sponsored Clinical Research Studies Ryan Mulligan, SCTR Institute OCR & ORSP
Research Administration Updates May 16, 2018 11:00 am IOP Auditorium Topics Covered Using ClinCard (Greenphire) for Federally Sponsored Clinical Research Studies Ryan Mulligan, SCTR Institute OCR & ORSP
SJSU Research Foundation
 SJSU Research Foundation Grant Proposal Development Taking a Bite Out of Forms Sponsored Programs Workshop November 19, 2015 Overview of Workshop Why We Have Forms Forms from OSP Forms from Human Resources
SJSU Research Foundation Grant Proposal Development Taking a Bite Out of Forms Sponsored Programs Workshop November 19, 2015 Overview of Workshop Why We Have Forms Forms from OSP Forms from Human Resources
Agenda. NIH Update. Other Updates. Proposal & Progress Report Statistics Research Administration Training Topic:
 Agenda NIH Update Salary Cap Guidance NOT OD 17 037: NIH Implementation of the Interim RPPR While a Renewal Application is Under Consideration Prior Approval for No Cost Extensions via the era Commons
Agenda NIH Update Salary Cap Guidance NOT OD 17 037: NIH Implementation of the Interim RPPR While a Renewal Application is Under Consideration Prior Approval for No Cost Extensions via the era Commons
Proposal Submission Guidelines
 March 28, 2016 Proposal Submission Guidelines Elliott School faculty have led a rapid increase in sponsored research over the past few years and the Dean s Office has increased its administrative capacity
March 28, 2016 Proposal Submission Guidelines Elliott School faculty have led a rapid increase in sponsored research over the past few years and the Dean s Office has increased its administrative capacity
ORA Quarterly Meeting April
 ORA Quarterly Meeting April 19 2017 Agenda 1. Workday Grants Implementation Items General Information Blackout Period Sub K s - Set-up and Invoicing Advance Accounts 2. Jackson/ UM Partnership Master Agreement
ORA Quarterly Meeting April 19 2017 Agenda 1. Workday Grants Implementation Items General Information Blackout Period Sub K s - Set-up and Invoicing Advance Accounts 2. Jackson/ UM Partnership Master Agreement
Financial Conflict of Interest Policy
 Financial Conflict of Interest Policy Office of Sponsored Programs Policy No.: OSP.1-03 Effective: 8/24/12 Supersedes: OSP.n-03 1.0 Purpose The San Jose State University Research Foundation (SJSURF) Financial
Financial Conflict of Interest Policy Office of Sponsored Programs Policy No.: OSP.1-03 Effective: 8/24/12 Supersedes: OSP.n-03 1.0 Purpose The San Jose State University Research Foundation (SJSURF) Financial
Standard Operating Procedures for P209: Investigator Conflict of Interest Policy
 Standard Operating Procedures for P209: Investigator Conflict of Interest Policy Table of Contents Applicability... 4 Institutional Roles... 5 Conflict of Interest (COI) Committee... 5 Designated Institutional
Standard Operating Procedures for P209: Investigator Conflict of Interest Policy Table of Contents Applicability... 4 Institutional Roles... 5 Conflict of Interest (COI) Committee... 5 Designated Institutional
Title: OFFICE OF SPONSORED PROGRAMS/IRB/ORIP COORDINATION
 SYRACUSE UNIVERSITY HUMAN RESEARCH PROTECTION PROGRAM STANDARD OPERATING PROCEDURES TITLE: OFFICE OF SPONSORED PROGRAMS/IRB/ORIP COORDINATION DOCUMENT NUMBER: 033 REVISION NUMBER REVISION DATE (SUPERSEDES
SYRACUSE UNIVERSITY HUMAN RESEARCH PROTECTION PROGRAM STANDARD OPERATING PROCEDURES TITLE: OFFICE OF SPONSORED PROGRAMS/IRB/ORIP COORDINATION DOCUMENT NUMBER: 033 REVISION NUMBER REVISION DATE (SUPERSEDES
Research Administrators Forum May 8th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum May 8th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Office of Research Administration OHRPP Retention of Research
Research Administrators Forum May 8th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Office of Research Administration OHRPP Retention of Research
University of Pittsburgh
 I. Scope This guideline establishes the requirements for recording direct and indirect costs on the financial accounting records of the University in accordance with Federal regulations and generally accepted
I. Scope This guideline establishes the requirements for recording direct and indirect costs on the financial accounting records of the University in accordance with Federal regulations and generally accepted
Financial Conflict of Interest: Investigator Procedures. Office of Research, Innovation, and Economic Development Research Integrity and Compliance
 Financial Conflict of Interest: Investigator Procedures Office of Research, Innovation, and Economic Development Research Integrity and Compliance June 2018 2 Table of Contents Introduction... 3 Private,
Financial Conflict of Interest: Investigator Procedures Office of Research, Innovation, and Economic Development Research Integrity and Compliance June 2018 2 Table of Contents Introduction... 3 Private,
MASSACHUSETTS INSTITUTE OF TECHNOLOGY. Policy for Cost Sharing and Matching Funds on Sponsored Projects Effective July 1, 1998
 INTRODUCTION This policy rescinds and supersedes the MIT Guidelines for Cost Sharing and Matching Funds on Sponsored Projects dated June 25, 1997. PURPOSE AND SCOPE The Institute must ensure that cost
INTRODUCTION This policy rescinds and supersedes the MIT Guidelines for Cost Sharing and Matching Funds on Sponsored Projects dated June 25, 1997. PURPOSE AND SCOPE The Institute must ensure that cost
GUIDELINES FOR GRADUATE STUDENTS
 GUIDELINES FOR GRADUATE STUDENTS IMPORTANT INFORMATION WHEN APPLYING FOR EXTERNAL FUNDING Applying for and receiving external funding for student research and creative projects may finance some or all
GUIDELINES FOR GRADUATE STUDENTS IMPORTANT INFORMATION WHEN APPLYING FOR EXTERNAL FUNDING Applying for and receiving external funding for student research and creative projects may finance some or all
Level of Effort Reporting
 Level of Effort Reporting Introduction Federal agencies provide funding to Eastern Washington University for a variety of research, scholarship and service activities. Appropriately certified effort reports
Level of Effort Reporting Introduction Federal agencies provide funding to Eastern Washington University for a variety of research, scholarship and service activities. Appropriately certified effort reports
Rebecca Trahan. Office of Sponsored Programs December 9, ORED Limited Submission Update
 Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
IRES Proposal Tracking (PT) Presented by: Kathi Goodfriend Office of Sponsored Projects Revised 03/15/2018 PRN: 5/14/ :19 PM
 IRES Proposal Tracking (PT) Presented by: Kathi Goodfriend Office of Sponsored Projects Revised 03/15/2018 PRN: 5/14/2018 12:19 PM Topics Covered Class Objective:. To become more familiar with the IRES
IRES Proposal Tracking (PT) Presented by: Kathi Goodfriend Office of Sponsored Projects Revised 03/15/2018 PRN: 5/14/2018 12:19 PM Topics Covered Class Objective:. To become more familiar with the IRES
Guidance on Grant Award Transfers
 University of the Pacific Office of Sponsored Programs Guidance on Grant Award Transfers It is not unusual for funded Principal Investigators (PIs) to move from one institution to another. However, The
University of the Pacific Office of Sponsored Programs Guidance on Grant Award Transfers It is not unusual for funded Principal Investigators (PIs) to move from one institution to another. However, The
Electronic Research Administration (era) March 8, 2018
 Electronic Research Administration (era) March 8, 2018 Introductions We are. Terry Duperron, era Analyst Marlene Mooshian, Proposal Analyst Kassie Obelleiro, Training Officer Jinger Snyder, Award Analyst
Electronic Research Administration (era) March 8, 2018 Introductions We are. Terry Duperron, era Analyst Marlene Mooshian, Proposal Analyst Kassie Obelleiro, Training Officer Jinger Snyder, Award Analyst
Quickguide to Creating a Proposal in Kuali Coeus
 Proposal Tab v.1 Revised 7/18/2014 Panel Field Action Notes Proposal Development Guide Reference Page(s) Enter a description of the proposal in this format: Document Overview Description Due Date xx/xx/xx_pi
Proposal Tab v.1 Revised 7/18/2014 Panel Field Action Notes Proposal Development Guide Reference Page(s) Enter a description of the proposal in this format: Document Overview Description Due Date xx/xx/xx_pi
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM. December 2017
 Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Outgoing Subaward Basics - Subawards Issued by UCLA under Extramurally Funded Grants/Cooperative Agreements
 Outgoing Subaward Basics - Subawards Issued by UCLA under Extramurally Funded Grants/Cooperative Agreements PREVIEW: Master Training (Mary Haskins, OCGA Subaward Officer) When: Wednesday, April 18, 2018
Outgoing Subaward Basics - Subawards Issued by UCLA under Extramurally Funded Grants/Cooperative Agreements PREVIEW: Master Training (Mary Haskins, OCGA Subaward Officer) When: Wednesday, April 18, 2018
TEXAS TECH UNIVERSITY HEALTH SCIENCES CENTER EL PASO
 TEXAS TECH UNIVERSITY HEALTH SCIENCES CENTER EL PASO Operating Policy and Procedure HSCEP OP: PURPOSE: REVIEW: 65.07, Effort Reporting: Certifying Time and Effort on Sponsored Projects The purpose of this
TEXAS TECH UNIVERSITY HEALTH SCIENCES CENTER EL PASO Operating Policy and Procedure HSCEP OP: PURPOSE: REVIEW: 65.07, Effort Reporting: Certifying Time and Effort on Sponsored Projects The purpose of this
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018
 CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
OMB Uniform Guidance: Cost Principles, Audit, and Administrative Requirements for Federal Awards
 OMB Uniform Guidance: Cost Principles, Audit, and Administrative Requirements for Federal Awards Chad Person May 1, 2013 Presented By: Devesh Kamal, CPA Shareholder deveshk@cshco.com Jesse Young, CPA Principal
OMB Uniform Guidance: Cost Principles, Audit, and Administrative Requirements for Federal Awards Chad Person May 1, 2013 Presented By: Devesh Kamal, CPA Shareholder deveshk@cshco.com Jesse Young, CPA Principal
Updates on American Recovery and Reinvestment Act (ARRA) Funding. Virginia Anders, Acting Director, OCGA Evelyn Balabis, Director, EFM
 Updates on American Recovery and Reinvestment Act (ARRA) Funding Virginia Anders, Acting Director, OCGA Evelyn Balabis, Director, EFM 15 OCGA ARRA Help Desk 10:00 a.m. 5:00 p.m., Monday Friday 310-794-0548
Updates on American Recovery and Reinvestment Act (ARRA) Funding Virginia Anders, Acting Director, OCGA Evelyn Balabis, Director, EFM 15 OCGA ARRA Help Desk 10:00 a.m. 5:00 p.m., Monday Friday 310-794-0548
SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients must complete this form when submitting a proposal to KSU, along with documents and certifications required by sponsors. This form must be endorsed by the
SUBRECIPIENT COMMITMENT FORM All subrecipients must complete this form when submitting a proposal to KSU, along with documents and certifications required by sponsors. This form must be endorsed by the
UNIFORM ADMINISTRATIVE REQUIREMENTS, COST PRINCIPLES, AND AUDIT REQUIREMENTS FOR FEDERAL AWARDS - UPDATE FEBRUARY 2015
 UNIFORM ADMINISTRATIVE REQUIREMENTS, COST PRINCIPLES, AND AUDIT REQUIREMENTS FOR FEDERAL AWARDS - UPDATE FEBRUARY 2015 AOA Conference Pasadena, CA February 9, 2015 Agenda 1. Introduction / Disclaimer 2.
UNIFORM ADMINISTRATIVE REQUIREMENTS, COST PRINCIPLES, AND AUDIT REQUIREMENTS FOR FEDERAL AWARDS - UPDATE FEBRUARY 2015 AOA Conference Pasadena, CA February 9, 2015 Agenda 1. Introduction / Disclaimer 2.
Emory University Research Administration Services (RAS) Standard Operating Procedure
 Emory University Research Administration Services (RAS) Standard Operating Procedure TITLE: NUMBER: RAS SOP 2016 VERSION: 4.1 LAST REVISED: PREPARED BY: Office of Research Administration, Research Administration
Emory University Research Administration Services (RAS) Standard Operating Procedure TITLE: NUMBER: RAS SOP 2016 VERSION: 4.1 LAST REVISED: PREPARED BY: Office of Research Administration, Research Administration
XAVIER UNIVERSITY. Financial Conflict of Interest Policy-Federal Grant Proposals
 Effective Date: XAVIER UNIVERSITY Financial Conflict of Interest Policy-Federal Grant Proposals Last Updated: May 2013 Responsible University Office: Office of Grant Services Responsible Executive: Associate
Effective Date: XAVIER UNIVERSITY Financial Conflict of Interest Policy-Federal Grant Proposals Last Updated: May 2013 Responsible University Office: Office of Grant Services Responsible Executive: Associate
Office of Sponsored Programs and Contracts
 Office of Sponsored Programs and Contracts Proposal Process (Part A) Charles E. Patterson, Ph.D. Assistant Director Baylor University Office of Sponsored Programs 4 th Floor, Pat Neff Hall (254) 710-3817
Office of Sponsored Programs and Contracts Proposal Process (Part A) Charles E. Patterson, Ph.D. Assistant Director Baylor University Office of Sponsored Programs 4 th Floor, Pat Neff Hall (254) 710-3817
ROLES AND RESPONSIBILITIES* FOR EXTERNAL AWARDS MADE TO DAVIDSON COLLEGE
 ROLES AND RESPONSIBILITIES* FOR EXTERNAL AWARDS MADE TO DAVIDSON COLLEGE Responsible (individual ultimately responsible for getting the work done) Accountable (signatory authority; expected to justify
ROLES AND RESPONSIBILITIES* FOR EXTERNAL AWARDS MADE TO DAVIDSON COLLEGE Responsible (individual ultimately responsible for getting the work done) Accountable (signatory authority; expected to justify
Page 1 of 10 1/31/2008
 Page 1 of 10 1/31/2008 Step by Step Tips to Avoid Errors and Warnings o Full instructions can be found at: http://grants1.nih.gov/grants/funding/424/index.htm. o This document is intended to assist you
Page 1 of 10 1/31/2008 Step by Step Tips to Avoid Errors and Warnings o Full instructions can be found at: http://grants1.nih.gov/grants/funding/424/index.htm. o This document is intended to assist you
RESEARCH ADMINISTRATION SERIES: SESSION 2 BUDGET PREPARATION WORKSHOP Amber Jensen Sarah Marcotte November 17 th, 2016
 RESEARCH ADMINISTRATION SERIES: SESSION 2 BUDGET PREPARATION WORKSHOP Amber Jensen Sarah Marcotte November 17 th, 2016 What we are covering today: Proposal Announcement Proposal Docs Budget & Budget Justification
RESEARCH ADMINISTRATION SERIES: SESSION 2 BUDGET PREPARATION WORKSHOP Amber Jensen Sarah Marcotte November 17 th, 2016 What we are covering today: Proposal Announcement Proposal Docs Budget & Budget Justification
Table of Contents. Introduction 2 How to Use This Guide 2 Best Practices for Budget Preparation 2
 Prepared by Kathy Thatcher, Liberal Arts Grants Services Revised: May 15, 2015 Table of Contents Introduction 2 How to Use This Guide 2 Best Practices for Budget Preparation 2 Direct Costs 3 Salaries 4
Prepared by Kathy Thatcher, Liberal Arts Grants Services Revised: May 15, 2015 Table of Contents Introduction 2 How to Use This Guide 2 Best Practices for Budget Preparation 2 Direct Costs 3 Salaries 4
SERVICE LEVEL AGREEMENT: University of Arkansas for Medical Sciences OFFICE OF SPONSORED PROGRAMS ADMINISTRATIVE NETWORK (OSPAN)
 SERVICE LEVEL AGREEMENT: University of Arkansas for Medical Sciences OFFICE OF SPONSORED PROGRAMS ADMINISTRATIVE NETWORK (OSPAN) Table of Contents Complete later Section I General Information and Objectives
SERVICE LEVEL AGREEMENT: University of Arkansas for Medical Sciences OFFICE OF SPONSORED PROGRAMS ADMINISTRATIVE NETWORK (OSPAN) Table of Contents Complete later Section I General Information and Objectives
Effort Reporting at UNR
 Effort Reporting at UNR M I C H E L E D O N D A N V I L L E R E S E A R C H C O M P L I A N C E P R O G R A M M A N A G E R 775-784-6360 M D O N D A N V I L L E @ U N R. E D U The Goals of Effort Reporting
Effort Reporting at UNR M I C H E L E D O N D A N V I L L E R E S E A R C H C O M P L I A N C E P R O G R A M M A N A G E R 775-784-6360 M D O N D A N V I L L E @ U N R. E D U The Goals of Effort Reporting
RESTRICTED PROJECTS: ROLES AND RESPONSIBILITIES May 2009
 RESTRICTED PROJECTS: ROLES AND RESPONSIBILITIES May 2009 Training and Education Review, interpret, and disseminate policies from sponsors Review, interpret, and disseminate policies from research compliance
RESTRICTED PROJECTS: ROLES AND RESPONSIBILITIES May 2009 Training and Education Review, interpret, and disseminate policies from sponsors Review, interpret, and disseminate policies from research compliance
Grant Review and Submission Standard Operating Procedure
 Grant Review and Submission Standard Operating Procedure Stephen Hunt, MPA Assistant Director, Policy, Compliance & Reporting Standard Operating Procedures GRANT LIFECYCLE PRIMARY BUSINESS FUNCTIONS Application
Grant Review and Submission Standard Operating Procedure Stephen Hunt, MPA Assistant Director, Policy, Compliance & Reporting Standard Operating Procedures GRANT LIFECYCLE PRIMARY BUSINESS FUNCTIONS Application
An Exercise in Effort
 3 rd Annual Symposium for Research Administrators An Exercise in Effort Brian Bertlshofer, Director, Cost Analysis and Compliance bertlsbj@email.unc.edu Aja Saylor, Central Effort Coordinator ajasaylor@unc.edu
3 rd Annual Symposium for Research Administrators An Exercise in Effort Brian Bertlshofer, Director, Cost Analysis and Compliance bertlsbj@email.unc.edu Aja Saylor, Central Effort Coordinator ajasaylor@unc.edu
Sponsored Program Administration Policy Approved by Academic Senate on 4/4/06
 Sponsored Program Administration Policy Approved by Academic Senate on 4/4/06 Page 1 of 10 Article 1. Definitions 1.1. Auxiliary means an Auxiliary Organization as defined in Executive Order No. 698. 1.2.
Sponsored Program Administration Policy Approved by Academic Senate on 4/4/06 Page 1 of 10 Article 1. Definitions 1.1. Auxiliary means an Auxiliary Organization as defined in Executive Order No. 698. 1.2.
Subrecipient Profile Questionnaire
 Subrecipient Profile Questionnaire How to use: The questionnaire is used to help determine a subrecipient organization s financial and management strength, which helps assess risk and dictates the monitoring
Subrecipient Profile Questionnaire How to use: The questionnaire is used to help determine a subrecipient organization s financial and management strength, which helps assess risk and dictates the monitoring
What You Need to Know When Submitting an NIH SF424 R&R Grant Application Through the UCLA Office of Contract and Grant Administration (OCGA)
 What You Need to Know When Submitting an NIH SF424 R&R Grant Application Through the UCLA Office of Contract and Grant Administration (OCGA) Kim Duiker, Assistant Director Mary Haskins, Contract & Grant
What You Need to Know When Submitting an NIH SF424 R&R Grant Application Through the UCLA Office of Contract and Grant Administration (OCGA) Kim Duiker, Assistant Director Mary Haskins, Contract & Grant
University of Central Florida
 Guidance & Directive No: ORC-01 University of Central Florida Guidance & Directive Date of Adoption/Revision: February 2013 Subject 1.0 Statement and Purpose Federal and state agencies, private foundations,
Guidance & Directive No: ORC-01 University of Central Florida Guidance & Directive Date of Adoption/Revision: February 2013 Subject 1.0 Statement and Purpose Federal and state agencies, private foundations,
Second Chance Act Grant Recipients Bureau of Justice Assistance (BJA) Compliance with Federal Grant Management Frequently Asked Questions (FAQs)
 Second Chance Act Grant Recipients Bureau of Justice Assistance (BJA) Compliance with Federal Grant Management Frequently Asked Questions (FAQs) 1. When does the grant term begin? Generally, for most grantees,
Second Chance Act Grant Recipients Bureau of Justice Assistance (BJA) Compliance with Federal Grant Management Frequently Asked Questions (FAQs) 1. When does the grant term begin? Generally, for most grantees,
Grants Management Workshop. Proposal and Award Management Support
 Grants Management Workshop Proposal and Award Management Support Agenda Sponsored Project Administration UVM and RSENR Support Systems Provide an overview of the grant life cycle For each stage of the
Grants Management Workshop Proposal and Award Management Support Agenda Sponsored Project Administration UVM and RSENR Support Systems Provide an overview of the grant life cycle For each stage of the
The Uniform Guidance 2 CFR 200 A Guide to Risk-Based Grants Management
 This image cannot currently be displayed. The Uniform Guidance 2 CFR 200 A Guide to Risk-Based Grants Management 2015 This image cannot currently be displayed. Increase in Federal Grants Activity The Catalog
This image cannot currently be displayed. The Uniform Guidance 2 CFR 200 A Guide to Risk-Based Grants Management 2015 This image cannot currently be displayed. Increase in Federal Grants Activity The Catalog
Introduction to Cayuse424
 Introduction to Cayuse424 In this training you will learn: How to: Navigate Cayuse424 Personalize your password Create Professional Profiles Create Budgets Create subcontract proposal, budget Initiate
Introduction to Cayuse424 In this training you will learn: How to: Navigate Cayuse424 Personalize your password Create Professional Profiles Create Budgets Create subcontract proposal, budget Initiate
Narration: Welcome to the Anatomy of an Administrative Shell mini course.
 Welcome to the Anatomy of an Administrative Shell mini course. 1 If you have previously joined us for other sponsored project mini courses, you will be familiar with the Sponsored Project Life Cycle. In
Welcome to the Anatomy of an Administrative Shell mini course. 1 If you have previously joined us for other sponsored project mini courses, you will be familiar with the Sponsored Project Life Cycle. In
CAYUSE Research Suite
 CAYUSE Research Suite Internal Clearance & Submission Process Principal Investigator/Program Director Cayuse SP is used by ORSP to process Internal Clearance and when applicable to submit a proposal for
CAYUSE Research Suite Internal Clearance & Submission Process Principal Investigator/Program Director Cayuse SP is used by ORSP to process Internal Clearance and when applicable to submit a proposal for
PEER (PAPERLESS ENVIRONMENT FOR ELECTRONIC REVIEW)
 PEER (PAPERLESS ENVIRONMENT FOR ELECTRONIC REVIEW) 1 What is PEER? PEER is a web portal maintained by Vanderbilt University which handles internal processing of grant related requests and contract submission
PEER (PAPERLESS ENVIRONMENT FOR ELECTRONIC REVIEW) 1 What is PEER? PEER is a web portal maintained by Vanderbilt University which handles internal processing of grant related requests and contract submission
Developing Proposal Budgets
 Grant Writing & Developing Proposal Budgets Sandra H. Harpole Associate VP for Research Sandy Williamson ORED Fiscal Officer January 8, 2008 Idea First or Opportunity First? Idea Have an idea for a project
Grant Writing & Developing Proposal Budgets Sandra H. Harpole Associate VP for Research Sandy Williamson ORED Fiscal Officer January 8, 2008 Idea First or Opportunity First? Idea Have an idea for a project
Are You Ready for This? The New Uniform Grant Guidance 2 CFR 200
 Are You Ready for This? The New Uniform Grant Guidance 2 CFR 200 Increase in Federal Grants Activity The Catalog of Federal Domestic Assistance lists over 2,000 Federal grant programs $600B $200B $7B $24B
Are You Ready for This? The New Uniform Grant Guidance 2 CFR 200 Increase in Federal Grants Activity The Catalog of Federal Domestic Assistance lists over 2,000 Federal grant programs $600B $200B $7B $24B
