SERVICE LEVEL AGREEMENT: University of Arkansas for Medical Sciences OFFICE OF SPONSORED PROGRAMS ADMINISTRATIVE NETWORK (OSPAN)
|
|
|
- Toby Eaton
- 5 years ago
- Views:
Transcription
1 SERVICE LEVEL AGREEMENT: University of Arkansas for Medical Sciences OFFICE OF SPONSORED PROGRAMS ADMINISTRATIVE NETWORK (OSPAN) Table of Contents Complete later Section I General Information and Objectives SLA Purpose and Expectations The purpose of this Service Level Agreement (SLA) is to establish the foundation for a cooperative partnership between the OFFICE OF SPONSORED PROGRAMS ADMINISTRATIVE NETWORK (OSPAN) and other stakeholders. This SLA will outline: The services that OSPAN offers and the working relationship between OSPAN and all stakeholders. The key performance indicators used to measure the quality of services provided. OSPAN Mission OSPAN's mission is to provide sponsored programs administration to all stakeholders of the University of Arkansas for Medical Sciences. OSPAN will be staffed by experts in sponsored program administrative services. OSPAN Vision Conducting the administrative business of sponsored awards so stakeholders can perform the sponsored program objectives. OSPAN Guiding Principles 1. Service: Excellent service is at the core of the Center. 2. Communication: OSPAN staff will provide clear and transparent communications to stakeholders. 3. Integrity and Respect: Mutual respect among all stakeholders will be upheld and all transactions will be performed with utmost integrity. OSPAN Goals 1. Provide expert and professional support for all UAMS stakeholders 2. Provide clear roles and responsibilities for OSPAN staff members and the stakeholders whom they serve. 3. Provide timely, streamlined and standardized processes that support stakeholders 4. Provide solutions to sponsored awards administration questions and issues. OSPAN Structure Each administrative unit will have a designated OSPAN staff member to serve sponsored program administration needs. OSPAN has the flexibility to bring multiple 1
2 staff together when needed for more complex grant submissions, and to bring together specialists from other teams to meet the needs of the stakeholder. If your OSPAN staff member is unavailable, designated back-up individuals will be available to meet the stakeholder s needs. The current list of OSPAN teams and the stakeholders they serve can be found on the University of Arkansas for Medical Sciences OSPAN website (include hyperlink). The Director of OSPAN reports to the Vice Chancellor for Research, with a secondary reporting relationship to the OSPAN Governance Advisory Board (GAB). Section II OSPAN Service Components OSPAN Locations The OSPAN teams and the administrative units they serve are listed on the OSPAN Website (insert hyperlink). OSPAN Services High level roles and responsibilities are outlined below. Pre-Award Process OSPAN Responsibilities Stakeholders/Administrative Units Responsibilities Identifying Funding Opportunities Collects and distributes targeted funding opportunities from all sources to stakeholders Proposal Development Manage TRACKs entry Budget Development Review and complete administrative components of the proposal Contact the subcontractors to gather required sub-awardee forms Contact editors as requested or as needed. Proposal Review and Submission Completes the final proposal package, reviews, and alerts STAKEHOLDER to review and submit the TRACKs 2 Stakeholders, in partnership with their Administrative Unit, makes the decision on whether or not to pursue the opportunity Stakeholder alerts OSPAN team of their intent to submit a proposal by following the timeline located in the appendix. Stakeholder completes scientific components of the proposal and assists in budget/budget justification development. Dept. Chair and administrative unit review and approve submission of the proposal.
3 Just in Time (JIT)Submissions Edit Manuscripts funding proposal. Prepare all administrative components of Just in Time or other pre-award requests from the sponsor Assist in obtaining regulatory approvals required for Just in Time submissions Submit JIT materials to ORSP Editors will identify relevant writing guidelines, instructions, and ethical standards (e.g., journal instructions for authors, regulatory requirements) and ensure the manuscript follows these. Editors will ensure the manuscript is grammatically correct and punctuated and formatted according to the journal s style guide. Editors will ensure that the required information for each section is present. prepares all scientific components of JIT submissions can delegate the assigned OSPAN Administrator to assist with a JIT in era Commons, if desired Stakeholder will contact OSPAN team member to schedule editing time no later than two weeks before manuscript is due. Stakeholder/author will submit to editor a fully written manuscript or a manuscript that is as complete as possible. Award Phase Process OSPAN Responsibilities Award Acceptance Reviews award document and determines if revised budget is required If revised budget is required, will collaborate with administrative unit. Assist in obtaining regulatory approvals required to accept award Stakeholders/Administrative Units Responsibilities approves revised budget, if necessary review award terms and conditions Administrative Unit will collaborate with OSPAN team on required changes. Stakeholders/Administrative Units approves TRACKS (for advance accounts, pre-award costs, and cost sharing) 3
4 Award Modifications Identifies need for award modification that requires internal or Sponsor approval In collaboration with STAKEHOLDER, drafts letter of request to the Sponsor (if applicable). Forwards requested forms to internal administrative unit or ORSP for review and submission, if applicable. Progress Report Submissions Beginning 30 days prior to the due date of a progress report prepares all administrative components for Progress Report submissions Assist in obtaining regulatory approvals required for Progress Report submissions. Submits all materials to ORSP Alerts OSPAN team of significant events that may require modification to the award. prepares all scientific components of Progress Report submissions (including ensuring the proper publications are linked in accordance with the NIH Public Access Policy) can delegate the assigned OSPAN team member to assist with a Progress Report in era Commons if desired Post-Award Process OSPAN Responsibilities STAKEHOLDER/ Administrative Unit Responsibilities Monthly Budget Reconciliation Monthly Administrative Unit Review Monitors spending on sponsored accounts. Prepares monthly financial reports to include budget, actual, commitments, projection and balance after the monthly financial close. Will meet with administrative unit leaders for summary review of all sponsored programs 4 meets with OSPAN team member on a monthly basis (at a minimum) to review grant accounts. Administrative Unit may attend monthly meetings if desired Administrative Unit leaders meets with OSPAN team member on a monthly basis (at a minimum) to review sponsored programs
5 Interim Financial Reporting Payment of Subcontract Invoices Reviews financial report with grants accounting for accuracy. Verifies with grants accounting carryforward amount. Reviews all invoices received from subcontractors. Follows-up with subcontractors who are not submitting invoices in a timely manner Routes to STAKEHOLDER for approval Routes to accounts payable for payment Closeout Reconcile all sponsored accounts in preparation for closeout Process CSRs as required. Notify STAKEHOLDER and administrative units that grant is ending at 90, 60 and 30 days prior to expiration. Ensure final invoices are processed. N/A approves subcontract invoices for payment notifies OSPAN team member should there be an issue with the work being completed by a subcontractor review and sign CSR completes all final technical reports required by Sponsor At least 90 days prior to closeout, Administrative Unit reviews and initiates separation procedures for employees who will be terminated upon grant close out. Research Contracts and Clinical Trials Process OSPAN Responsibilities STAKEHOLDER/ Department Responsibilities Pre-Award Clinical Trials Management Contract Development and Execution Review and negotiate budget with sponsor and stakeholder. Maintain CLARA record for routing and approval Input, as needed, for Research Contract negotiations 5 Review and sign the negotiated CDA Approve negotiated budget Input, as needed, for negotiations
6 Coverage Analysis Input, as needed Input, as needed, for development Final review and approval Budgets Input, as needed, for budget development Invoicing/AR Management Review study calendars and milestones Generate invoice and submit to sponsor Receive payment and ensure it is applied to appropriate clinical trial account Monitor accounts receivable and escalate risky accounts receivable (accounts receivable not paid within 120 days of invoicing) for potential bad debt write off Notifies Research Contracts and STAKEHOLDER if contract should be amended due to accrual/financial implications Closeout Reconcile account financials Confirm contract terms for closure General final invoice for unbilled activity Receive final payment Close account Input, as needed, for budget development Final budget approval Enter patient data in tracking system Confirm milestones have been reached and OSPAN can invoice sponsor Confirm payment received is tied to specific protocol Confirm study outcomes Conduct site close-out activities Draft final letter/communication to sponsor Approve study for closing Section III OSPAN Performance Metrics Tracking Effectiveness Timely, efficient, and accurate service and accountability are critical components of OSPAN. To ensure that those goals are met, key performance indicators will be tracked and monitored. Quarterly reports will be sent to the GAB and published on the OSPAN website. 6
7 Key Performance Indicators Pre-award Services Level Measures Number of proposals submitted per month. Number of proposal submitted early (successful submission 2 day before deadline.). Time in State (itemize the states). Total Proposal Days (administrative Items; OSPAN tracking) Percentage of proposals that do not require the administrative unit to return them to OSPAN for revisions prior to submission to sponsor Percentage of proposals that do not require ORSP to return them to OSPAN for revisions prior to submission to funding agency. Number of proposals using editor services. Number of scientific publications using editor services. Post-Award Compliance Risk and Service Level Measures Number of Advance Accounts open greater than 90 days without cause Number of Accounts where the project period has ended and the account deficit or surplus 5 %. Number of cost summary reports that are submitted past the due date. Number of cost summary reports returned for revisions. Number of transactions that posted after the grant/budget end date Number of cost transfers greater than 90 days from the original transaction date. Number of Effort reports submitted after institutional due date Number of days award is under department review. Number of days award is under grants accounting review. Number of days from notice of award to activation. Number of monthly reports reviewed. Research Contracts and Clinical Trials Management Service Level Measure Number of invoices submitted within 30 days of receiving notice from clinical coordinators that the milestone has been met. Accounts Receivable by aging category (> 30 days, days, > 90 days): OSPAN will escalate payments outstanding over 90 days. Number of accounts with no billing activity over 90 days without cause. Number of open trials with a project end date > 90 days Number of outgoing subcontracts submitted within 14 days of receiving notice from administrative unit. Faculty/Department Service Related Metrics Service Level Measure 7
8 STAKEHOLDER and administrative unit satisfaction with OSPAN support (survey) Escalation Procedures: OSPAN Failure to Meet Performance Expectations In the event that OSPAN fails to meet the expectations outlined in this document, a STAKEHOLDER or administrative unit can take the following steps: 1. Communicate the problem directly to your OSPAN team leader Communication is the key to the success of the partnership between the Stakeholder, administrative unit and OSPAN. Problems should be clearly identified and possible resolutions discussed with your OSPAN team leader. 2. Contact OSPAN Director If discussions with the appropriate OSPAN Team Leader fail to resolve the problem, please contact the OSPAN Director to discuss the problem and resolution. Escalation Procedures: Stakeholders Failure to Meet Expectations In the event that STAKEHOLDER fails to meet the expectations outlined in this document, a OSPAN or administrative unit can take the following steps: 1. Communicate the problem directly to the OSPAN Team leader and STAKEHOLDER. Communication is the key to the success of the partnership between the Stakeholder, administrative unit and OSPAN. Problems should be clearly identified and possible resolutions discussed with the OSPAN team leader and STAKEHOLDER. 2. Contact OSPAN Director If discussions with the appropriate OSPAN Team Leader and STAKEHOLDER fail to resolve the problem, please contact the OSPAN Director to discuss the problem and resolution. If failure to resolve the problem, the director will discuss problem with GAB and VCR. Section IV SLA Maintenance Conditions Periodic Review of SLA The Vice Chancellor for Research, OSPAN Director, and Governance Advisory Board (GAB) will meet annually to review the SLA to determine whether the services and 8
9 service level commitment are meeting the needs of the departments served. During the early stages of OSPAN implementation, more frequent review may be necessary to ensure that the SLA accurately reflects the STAKEHOLDERs and administrative units needs and the capacity of OSPAN to meet them. Substantial changes in the University, such as a significant increase in sponsored programs, should also prompt a review of the SLA. SLA Modification Review The SLA will be reviewed for proposed changes/revisions and to consider any outstanding resource needs. The reviewers include the OSPAN Director, Vice Chancellor for Research, and GAB. The Vice Chancellor for Research, OSPAN Director, GAB, stakeholders and administrative units may submit requested updates and recommendations, with a brief rationale for each recommendation. At the direction of the review committee, the OSPAN Director will make all approved changes to the SLA document. The SLA will be posted on the OSPAN website to promote accountability. The Vice Chancellor for Research and the OSPAN Director will be responsible for implementing changes to the SLA and communicating SLA changes to their stakeholder group. Appendix Include Acronyms, Definitions, work flow chart and organizational chart. 9
SERVICE LEVEL AGREEMENT: THOMAS JEFFERSON UNIVERSITY OFFICE OF RESEARCH ADMINISTRATION (ORA) JANUARY, 2018
 SERVICE LEVEL AGREEMENT: THOMAS JEFFERSON UNIVERSITY OFFICE OF RESEARCH ADMINISTRATION (ORA) JANUARY, 2018 SLA Updated 2/1/2015, 1/4/2016, 9/1/2016, 1/4/2017, 7/1/2017,1/22/2018 21 Table of Contents Acronyms
SERVICE LEVEL AGREEMENT: THOMAS JEFFERSON UNIVERSITY OFFICE OF RESEARCH ADMINISTRATION (ORA) JANUARY, 2018 SLA Updated 2/1/2015, 1/4/2016, 9/1/2016, 1/4/2017, 7/1/2017,1/22/2018 21 Table of Contents Acronyms
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP)
 Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Complex Award Management Program Projects/Multi-Fund Awards- Post Award NUMBER: RAS SOP 2001 VERSION: 4.0
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Complex Award Management Program Projects/Multi-Fund Awards- Post Award NUMBER: RAS SOP 2001 VERSION: 4.0
Thomas Jefferson University
 Thomas Jefferson University Offices of Research Support Services April 2017 *For Discussion Purposes Only* Agenda Topic Presenter Email Phone Duration Offices of Research Support Services: Introduction
Thomas Jefferson University Offices of Research Support Services April 2017 *For Discussion Purposes Only* Agenda Topic Presenter Email Phone Duration Offices of Research Support Services: Introduction
ROLES AND RESPONSIBILITIES* FOR EXTERNAL AWARDS MADE TO DAVIDSON COLLEGE
 ROLES AND RESPONSIBILITIES* FOR EXTERNAL AWARDS MADE TO DAVIDSON COLLEGE Responsible (individual ultimately responsible for getting the work done) Accountable (signatory authority; expected to justify
ROLES AND RESPONSIBILITIES* FOR EXTERNAL AWARDS MADE TO DAVIDSON COLLEGE Responsible (individual ultimately responsible for getting the work done) Accountable (signatory authority; expected to justify
Research Administration Services Roles & Responsibilities For Grants and Contracts (Excludes Clinical Trials) Version 3.1
 Research Administration Services Roles & Responsibilities For Grants and Contracts (Excludes Clinical Trials) Version 3.1 Category PI/PI Staff Department* RAS Unit School/Unit ORA Offices Relevant SOP
Research Administration Services Roles & Responsibilities For Grants and Contracts (Excludes Clinical Trials) Version 3.1 Category PI/PI Staff Department* RAS Unit School/Unit ORA Offices Relevant SOP
OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015
 OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015 INTRODUCTION The University of Wisconsin-Milwaukee (UWM) is the prime recipient on a wide range of sponsored awards
OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015 INTRODUCTION The University of Wisconsin-Milwaukee (UWM) is the prime recipient on a wide range of sponsored awards
Roles & Responsibilities University of Rochester Department Administrative Staff
 Roles and Responsibilities - UR Department Administrative Staff Roles & Responsibilities University of Rochester Department Administrative Staff Administrative Staff at the departmental, center or unit
Roles and Responsibilities - UR Department Administrative Staff Roles & Responsibilities University of Rochester Department Administrative Staff Administrative Staff at the departmental, center or unit
SPONSORED PROJECT CLOSE-OUT/PAYMENT COLLECTION GUIDELINE
 SPONSORED PROJECT CLOSE-OUT/PAYMENT COLLECTION GUIDELINE Purpose: To provide guidance to assist Principal Investigators (PI s), Award Managers, UAB research/fiscal administrators in close-out, invoicing,
SPONSORED PROJECT CLOSE-OUT/PAYMENT COLLECTION GUIDELINE Purpose: To provide guidance to assist Principal Investigators (PI s), Award Managers, UAB research/fiscal administrators in close-out, invoicing,
Vertex Investigator-Initiated Studies Program Overview
 Vertex Investigator-Initiated Studies Program Overview Our Goal Our Investigator Initiated Study grants support independent, investigator-initiated research designed to advance scientific knowledge of
Vertex Investigator-Initiated Studies Program Overview Our Goal Our Investigator Initiated Study grants support independent, investigator-initiated research designed to advance scientific knowledge of
Dept/ College. Dept/ College. Dept/ College
 Office of Research Administration Matrix Revision date -2.10.17 Submitting proposals, executing awards, conducting research, and administrating sponsored projects involves many different people and units
Office of Research Administration Matrix Revision date -2.10.17 Submitting proposals, executing awards, conducting research, and administrating sponsored projects involves many different people and units
RESTRICTED PROJECTS: ROLES AND RESPONSIBILITIES May 2009
 RESTRICTED PROJECTS: ROLES AND RESPONSIBILITIES May 2009 Training and Education Review, interpret, and disseminate policies from sponsors Review, interpret, and disseminate policies from research compliance
RESTRICTED PROJECTS: ROLES AND RESPONSIBILITIES May 2009 Training and Education Review, interpret, and disseminate policies from sponsors Review, interpret, and disseminate policies from research compliance
n e m e rch Research Administration Process l A ity lia ral t / C ffic DCG SPA Identification of Funding Opportunities Proposal Preparation
 Research Administration rocess Identification of Funding Opportunities Seek funding opportunities for individual proposals S S S Seek funding opportunities for interdisciplinary proposals S S Distribute
Research Administration rocess Identification of Funding Opportunities Seek funding opportunities for individual proposals S S S Seek funding opportunities for interdisciplinary proposals S S Distribute
UNIVERSITY RESEARCH ADMINISTRATION FINANCIAL ROLES AND RESPONSIBILITIES MATRIX - WORK IN PROGRESS 10/03/2013 Roles.
 UNIVERSITY RESEARCH ADMINISTRATION Roles Business Internal Controller's Clinical Responsibilities PI Office Chair Dean Audit Office OCR GCFA GCA PROVOST Trials Office I. GENERAL RESEARCH ADMINISTRATION
UNIVERSITY RESEARCH ADMINISTRATION Roles Business Internal Controller's Clinical Responsibilities PI Office Chair Dean Audit Office OCR GCFA GCA PROVOST Trials Office I. GENERAL RESEARCH ADMINISTRATION
Sponsored Programs Roles & Responsibilities
 The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
Subrecipient Monitoring Procedures
 Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
The Lifecycle of a Sponsored Project
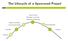 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP)
 Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: ions and Forecasting NUMBER: RAS SOP 2007 VERSION: 4.0 LAST REVISED: PREPARED BY: Office of Research Administration,
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: ions and Forecasting NUMBER: RAS SOP 2007 VERSION: 4.0 LAST REVISED: PREPARED BY: Office of Research Administration,
City of Fernley GRANTS MANAGEMENT POLICIES AND PROCEDURES
 1 of 12 I. PURPOSE The purpose of this policy is to set forth an overall framework for guiding the City s use and management of grant resources. II ` GENERAL POLICY Grant revenues are an important part
1 of 12 I. PURPOSE The purpose of this policy is to set forth an overall framework for guiding the City s use and management of grant resources. II ` GENERAL POLICY Grant revenues are an important part
Sponsored Programs Roles & Responsibilities
 The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
Roles & Responsibilities UR Office of Research and Project Administration
 Roles and Responsibilities - UR ORPA Roles & Responsibilities UR Office of Research and Project Administration The Office of Research and Projects Administration (ORPA) is the administrative office legally
Roles and Responsibilities - UR ORPA Roles & Responsibilities UR Office of Research and Project Administration The Office of Research and Projects Administration (ORPA) is the administrative office legally
East Carolina University Sponsored Projects Administration Roles and Responsibilities Matrix By Responsibility
 (See legend at end of matrix.) East Carolina University Sponsored Projects Administration Roles and Responsibilities Matrix By Responsibility This matrix is designed to provide general guidance on administrative
(See legend at end of matrix.) East Carolina University Sponsored Projects Administration Roles and Responsibilities Matrix By Responsibility This matrix is designed to provide general guidance on administrative
Grant Review and Submission Standard Operating Procedure
 Grant Review and Submission Standard Operating Procedure Stephen Hunt, MPA Assistant Director, Policy, Compliance & Reporting Standard Operating Procedures GRANT LIFECYCLE PRIMARY BUSINESS FUNCTIONS Application
Grant Review and Submission Standard Operating Procedure Stephen Hunt, MPA Assistant Director, Policy, Compliance & Reporting Standard Operating Procedures GRANT LIFECYCLE PRIMARY BUSINESS FUNCTIONS Application
SJSU Research Foundation
 SJSU Research Foundation The Nuts & Bolts of Proposal Preparation and Award Management Sponsored Programs Workshop February 16, 2017 Agenda Discussion of Common Issues and Solutions Terms and Conditions
SJSU Research Foundation The Nuts & Bolts of Proposal Preparation and Award Management Sponsored Programs Workshop February 16, 2017 Agenda Discussion of Common Issues and Solutions Terms and Conditions
EOP/SUPPORT ANNEX F/APPENDIX 14 EOC FINANCE SECTION APPENDIX 14 EOC FINANCE SECTION
 APPENDIX 14 APPENDIX 14-1 JUNE 2015 THIS PAGE INTENTIONALLY BLANK APPENDIX 14-2 JUNE 2015 OVERVIEW The Finance Section is responsible for providing accounting functions, including maintaining an audit
APPENDIX 14 APPENDIX 14-1 JUNE 2015 THIS PAGE INTENTIONALLY BLANK APPENDIX 14-2 JUNE 2015 OVERVIEW The Finance Section is responsible for providing accounting functions, including maintaining an audit
Grants Management Workshop. Proposal and Award Management Support
 Grants Management Workshop Proposal and Award Management Support Agenda Sponsored Project Administration UVM and RSENR Support Systems Provide an overview of the grant life cycle For each stage of the
Grants Management Workshop Proposal and Award Management Support Agenda Sponsored Project Administration UVM and RSENR Support Systems Provide an overview of the grant life cycle For each stage of the
General Administration GA STANDARD OPERATING PROCEDURE FOR Document Development and Change Control
 General Administration GA 104.00 STANDARD OPERATING PROCEDURE FOR Document Development and Change Control Approval: Nancy Paris, MS, FACHE President and CEO 08 March 2012 (Signature and Date) Approval:
General Administration GA 104.00 STANDARD OPERATING PROCEDURE FOR Document Development and Change Control Approval: Nancy Paris, MS, FACHE President and CEO 08 March 2012 (Signature and Date) Approval:
University of Colorado Denver
 University of Colorado Denver Campus Guidelines Title:, 4-13 Source: Prepared by: Approved by: Office of Grants and Contracts Director, Office of Grants and Contracts Vice Chancellor for Research Effective
University of Colorado Denver Campus Guidelines Title:, 4-13 Source: Prepared by: Approved by: Office of Grants and Contracts Director, Office of Grants and Contracts Vice Chancellor for Research Effective
Life Sciences Simons Collaboration on the Origins of Life Fellowship Policies and Procedures
 Life Sciences Simons Collaboration on the Origins of Life Fellowship Policies and Procedures Table of Contents 1. Our mission Page 2 2. Online fellowship management Page 2 3. Fellowship activation Page
Life Sciences Simons Collaboration on the Origins of Life Fellowship Policies and Procedures Table of Contents 1. Our mission Page 2 2. Online fellowship management Page 2 3. Fellowship activation Page
Outgoing Subaward Guide and F-A-Qs
 1. What is a subaward? Outgoing Subaward Guide and F-A-Qs 2. After an award is made to Rutgers, how is a subaward established? 3. What is the outgoing subaward process? 4. How can a subaward be changed/amended?
1. What is a subaward? Outgoing Subaward Guide and F-A-Qs 2. After an award is made to Rutgers, how is a subaward established? 3. What is the outgoing subaward process? 4. How can a subaward be changed/amended?
Cancer Prevention & Research Institute of Texas
 Cancer Prevention & Research Institute of Texas IA # 01-18 Internal Audit Report over Post-Award C O N T E N T S Page Internal Audit Report Transmittal Letter to the Oversight Committee... 1 Background...
Cancer Prevention & Research Institute of Texas IA # 01-18 Internal Audit Report over Post-Award C O N T E N T S Page Internal Audit Report Transmittal Letter to the Oversight Committee... 1 Background...
Research Administrators Forum August 14th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum August 14th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Associate Vice Chancellor, Office of Research Administration
Research Administrators Forum August 14th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Associate Vice Chancellor, Office of Research Administration
Pharmacy Practice Advancement Demonstration Grants
 Pharmacy Practice Advancement Demonstration Grants Application Policies and Guidelines The ASHP/ASHP Foundation Pharmacy Practice Advancement Demonstration grant was made possible through the generous
Pharmacy Practice Advancement Demonstration Grants Application Policies and Guidelines The ASHP/ASHP Foundation Pharmacy Practice Advancement Demonstration grant was made possible through the generous
Pharmacy Practice Advancement Demonstration Grants
 Pharmacy Practice Advancement Demonstration Grants Application Policies and Guidelines Administered by the ASHP Research and Education Foundation The ASHP/ASHP Foundation Pharmacy Practice Model Initiative
Pharmacy Practice Advancement Demonstration Grants Application Policies and Guidelines Administered by the ASHP Research and Education Foundation The ASHP/ASHP Foundation Pharmacy Practice Model Initiative
Policy on Principal Investigators Duties and Responsibilities on Sponsored Projects
 Office of Research and Sponsored Programs Foundation Administration Policy on Principal Investigators Duties and Responsibilities on Sponsored Projects Policy Index I. Introduction II. Policy Statement
Office of Research and Sponsored Programs Foundation Administration Policy on Principal Investigators Duties and Responsibilities on Sponsored Projects Policy Index I. Introduction II. Policy Statement
AURA Meeting. July 23, 2014
 AURA Meeting July 23, 2014 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored
AURA Meeting July 23, 2014 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored
Ramp up and Wrap up projects. Riding the Wave to Close Out
 Ramp up and Wrap up projects Riding the Wave to Close Out To maximize our time together today: Please turn off or silence your cell phones Limit side chatter so everyone can hear Best of all. Stay for
Ramp up and Wrap up projects Riding the Wave to Close Out To maximize our time together today: Please turn off or silence your cell phones Limit side chatter so everyone can hear Best of all. Stay for
Research Administrators Forum May 8th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum May 8th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Office of Research Administration OHRPP Retention of Research
Research Administrators Forum May 8th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Office of Research Administration OHRPP Retention of Research
INCENTIVE SCHEMES & SERVICE LEVEL AGREEMENTS
 MAY 2007 INCENTIVE SCHEMES & SERVICE LEVEL AGREEMENTS Practice Based Commissioning North and South Essex Local Medical Committees CLARIFYING THE RELATIONSHIP BETWEEN PBC GROUPS AND PCTS AIMS The aim of
MAY 2007 INCENTIVE SCHEMES & SERVICE LEVEL AGREEMENTS Practice Based Commissioning North and South Essex Local Medical Committees CLARIFYING THE RELATIONSHIP BETWEEN PBC GROUPS AND PCTS AIMS The aim of
FDA Perspective on the Pre- Submission Program: Updates from MDUFA IV
 FDA Perspective on the Pre- Submission Program: Updates from MDUFA IV Maureen Dreher, PhD Policy Analyst Clinical Trials Program/ Office of Device Evaluation/ CDRH Agenda Pre-Submission Program background
FDA Perspective on the Pre- Submission Program: Updates from MDUFA IV Maureen Dreher, PhD Policy Analyst Clinical Trials Program/ Office of Device Evaluation/ CDRH Agenda Pre-Submission Program background
Approved by WQGIT July 14, 2014
 Page 1 Approved by WQGIT July 14, 2014 Protocol for the Development, Review, and Approval of Loading and Effectiveness Estimates for Nutrient and Sediment Controls in the Chesapeake Bay Watershed Model
Page 1 Approved by WQGIT July 14, 2014 Protocol for the Development, Review, and Approval of Loading and Effectiveness Estimates for Nutrient and Sediment Controls in the Chesapeake Bay Watershed Model
The Office for Sponsored Research (OSR): An Overview. Office for Sponsored Research
 The Office for Sponsored Research (OSR): An Overview Office for Sponsored Research OSR Mission The Office for Sponsored Research assists investigators in proposing and managing sponsored programs in support
The Office for Sponsored Research (OSR): An Overview Office for Sponsored Research OSR Mission The Office for Sponsored Research assists investigators in proposing and managing sponsored programs in support
Sponsored Projects: Curriculum Plan Johns Hopkins University Panel Members:
 Sponsored Projects: Curriculum Plan Johns Hopkins University Panel Members: Mike Alexander, University Administration Jim Aumiller, University Administration Holly Benze, Engineering Don Cornely, University
Sponsored Projects: Curriculum Plan Johns Hopkins University Panel Members: Mike Alexander, University Administration Jim Aumiller, University Administration Holly Benze, Engineering Don Cornely, University
4.2. Clinical Trial Monitor (or Monitor): The person responsible for monitoring the data on behalf of the sponsor or contract research organization.
 SOP #: MON-101 Page: 1 of 6 1. POLICY STATEMENT: The DF/HCC understands that external sponsors are required to monitor the progress of clinical investigations and ensure appropriate research data collection
SOP #: MON-101 Page: 1 of 6 1. POLICY STATEMENT: The DF/HCC understands that external sponsors are required to monitor the progress of clinical investigations and ensure appropriate research data collection
Post Award Functions. Department/ Principal Investigator. Sponsored Research
 Post Award Functions Department/ Principal Investigator HOBS Office of Sponsored Research Award Negotiation & Acceptance 1 Receives award document from sponsor. (May sometimes be received by PI.) 2 If
Post Award Functions Department/ Principal Investigator HOBS Office of Sponsored Research Award Negotiation & Acceptance 1 Receives award document from sponsor. (May sometimes be received by PI.) 2 If
FOR REINVESTMENT ACT. NILAH DEVANEY NWRO, AWARDING OFFICIAL
 1 PL 93-638 AWARD PROCESS FOR AMERICAN RECOVERY AND REINVESTMENT ACT (ARRA) NILAH DEVANEY NWRO, AWARDING OFFICIAL 503-872-2867 E-Mail: nilah.devaney@bia.gov 2 ARRA OVERVIEW ON DEADLINES FEBRUARY 17, 2010,
1 PL 93-638 AWARD PROCESS FOR AMERICAN RECOVERY AND REINVESTMENT ACT (ARRA) NILAH DEVANEY NWRO, AWARDING OFFICIAL 503-872-2867 E-Mail: nilah.devaney@bia.gov 2 ARRA OVERVIEW ON DEADLINES FEBRUARY 17, 2010,
University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures
 Summary 1. Subaward Definitions A. Subaward B. Subrecipient University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures C. Office of Contracts and Grants (OCG) 2. Distinguishing
Summary 1. Subaward Definitions A. Subaward B. Subrecipient University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures C. Office of Contracts and Grants (OCG) 2. Distinguishing
Life Sciences Simons Collaboration on the Global Brain (SCGB) Fellowships
 Life Sciences Simons Collaboration on the Global Brain (SCGB) Fellowships Fellowship Policies and Procedures Table of Contents 1. Our mission Page 2 2. Online fellowship management Page 2 3. Fellowship
Life Sciences Simons Collaboration on the Global Brain (SCGB) Fellowships Fellowship Policies and Procedures Table of Contents 1. Our mission Page 2 2. Online fellowship management Page 2 3. Fellowship
The purpose of this survey
 The purpose of this survey Dear Colleague: As part of our ongoing effort to strengthen grants administration across Boston University, we have created this survey to gain a better understanding of your
The purpose of this survey Dear Colleague: As part of our ongoing effort to strengthen grants administration across Boston University, we have created this survey to gain a better understanding of your
SAN DIEGO POLICE DEPARTMENT PROCEDURE
 SAN DIEGO POLICE DEPARTMENT PROCEDURE DATE: March 25, 2016 NUMBER: SUBJECT: RELATED POLICY: ORIGINATING DIVISION: 1.40 ADMINISTRATION GRANT PROCEDURES N/A ADMINISTRATIVE SERVICES PROCEDURE: PROCEDURAL
SAN DIEGO POLICE DEPARTMENT PROCEDURE DATE: March 25, 2016 NUMBER: SUBJECT: RELATED POLICY: ORIGINATING DIVISION: 1.40 ADMINISTRATION GRANT PROCEDURES N/A ADMINISTRATIVE SERVICES PROCEDURE: PROCEDURAL
Research Administration at Emory Foundation Program DAY TWO: The Financial Administration of Sponsored Programs
 Revised 05/2018 Research Administration at Emory Foundation Program DAY TWO: The Financial Administration of Sponsored Programs Presenter: Finance: Grants and Contracts (FGC) Jumionne Tiako AD Post Award
Revised 05/2018 Research Administration at Emory Foundation Program DAY TWO: The Financial Administration of Sponsored Programs Presenter: Finance: Grants and Contracts (FGC) Jumionne Tiako AD Post Award
NN SS 401 NEURONEXT NETWORK STANDARD OPERATING PROCEDURE FOR SITE SELECTION AND QUALIFICATION
 NN SS 401 NEURONEXT NETWORK STANDARD OPERATING PROCEDURE FOR SITE SELECTION AND QUALIFICATION SOP: NN SS 401 Version No.: 2.0 Effective Date: 21Oct2016 SITE SELECTION AND QUALIFICATION Supercedes Document:
NN SS 401 NEURONEXT NETWORK STANDARD OPERATING PROCEDURE FOR SITE SELECTION AND QUALIFICATION SOP: NN SS 401 Version No.: 2.0 Effective Date: 21Oct2016 SITE SELECTION AND QUALIFICATION Supercedes Document:
University of Pittsburgh
 I. Guideline Background University sponsors are increasingly relying on institutions for the development of adequate compliance programs for the financial administration of grants and contracts. An adequate
I. Guideline Background University sponsors are increasingly relying on institutions for the development of adequate compliance programs for the financial administration of grants and contracts. An adequate
Health Center Staff Documents Checklist
 Health Center Program Site Visit Protocol Health Center Staff Documents Checklist NOTE: This consolidated checklist contains documents used to assess multiple program requirements during Operational Site
Health Center Program Site Visit Protocol Health Center Staff Documents Checklist NOTE: This consolidated checklist contains documents used to assess multiple program requirements during Operational Site
SPONSORED PROJECTS: CLOSE-OUT
 SPONSORED PROJECTS: CLOSE-OUT Preterm: > 90 Days Prior to End Date OGCA - PreTerm Notifies department to review requests for PANs, NCE s, appointment forms & termination forms (if Training Grant) Reminds
SPONSORED PROJECTS: CLOSE-OUT Preterm: > 90 Days Prior to End Date OGCA - PreTerm Notifies department to review requests for PANs, NCE s, appointment forms & termination forms (if Training Grant) Reminds
2018 Corn Research and Education Request for Proposals
 2018 Corn Research and Education Request for Proposals Through the generous support of the NY Senate and Assembly, the New York Corn & Soybean Growers Association (NYCSGA) is pleased to announce their
2018 Corn Research and Education Request for Proposals Through the generous support of the NY Senate and Assembly, the New York Corn & Soybean Growers Association (NYCSGA) is pleased to announce their
Grants Financial Procedures (Post-Award) v. 2.0
 Grants Financial Procedures (Post-Award) v. 2.0 1 Grants Financial Procedures (Post Award) Version Number: 2.0 Procedures Identifier: Superseded Procedure(s): BU-PR0001 N/A Date Approved: 9/1/2013 Effective
Grants Financial Procedures (Post-Award) v. 2.0 1 Grants Financial Procedures (Post Award) Version Number: 2.0 Procedures Identifier: Superseded Procedure(s): BU-PR0001 N/A Date Approved: 9/1/2013 Effective
D. PROPOSAL DETAILS CREATE A NEW PROPOSAL GENERAL INFO ORGANIZATION ADD INVESTIGATORS AND KEY PERSONS CREDIT SPLIT SPECIAL REVIEW D.3.
 D. PROPOSAL DETAILS D. D. D.3. D.4. D.5. D.6. D.7. D.8. D.9. D.10. D.1 D.1 CREATE A NEW PROPOSAL GENERAL INFO ORGANIZATION ADD INVESTIGATORS AND KEY PERSONS CREDIT SPLIT SPECIAL REVIEW ABSTRACT OTHER YNQ
D. PROPOSAL DETAILS D. D. D.3. D.4. D.5. D.6. D.7. D.8. D.9. D.10. D.1 D.1 CREATE A NEW PROPOSAL GENERAL INFO ORGANIZATION ADD INVESTIGATORS AND KEY PERSONS CREDIT SPLIT SPECIAL REVIEW ABSTRACT OTHER YNQ
McKinsey Recommendations for Code Compliance and Economic Development. Status Report. Dallas City Council Briefing April 20, 2005 DRAFT 1
 McKinsey Recommendations for Code Compliance and Economic Development Status Report Dallas City Council Briefing April 20, 2005 DRAFT 1 PURPOSE To provide the City Council a status report on implementation
McKinsey Recommendations for Code Compliance and Economic Development Status Report Dallas City Council Briefing April 20, 2005 DRAFT 1 PURPOSE To provide the City Council a status report on implementation
STANDARD OPERATING PROCEDURE
 STANDARD OPERATING PROCEDURE Title Reference Number Corrective and Preventative Action SOP-QMS-008 Version Number 2 Issue Date 29 th Sep 2016 Effective Date 10 th Nov 2016 Review Date 10 th Nov 2018 Author(s)
STANDARD OPERATING PROCEDURE Title Reference Number Corrective and Preventative Action SOP-QMS-008 Version Number 2 Issue Date 29 th Sep 2016 Effective Date 10 th Nov 2016 Review Date 10 th Nov 2018 Author(s)
Post Award & Accounting Manual
 OFFICE OF RESEARCH & SPONSORED PROGRAMS Post Award & Accounting Manual Office of Research & Sponsored Programs University of New Orleans New Orleans, LA 70148-2105 CERM 460 http://www.uno.edu/orsp Phone:
OFFICE OF RESEARCH & SPONSORED PROGRAMS Post Award & Accounting Manual Office of Research & Sponsored Programs University of New Orleans New Orleans, LA 70148-2105 CERM 460 http://www.uno.edu/orsp Phone:
(Area Agency Name) B. Requirements of Section 287, Florida Statutes: These requirements are herein incorporated by reference.
 STANDARD CONTRACT AREA AGENCY ON AGING (Area Agency Name) THIS CONTRACT is entered into between the State of Florida, Department of Elder Affairs, hereinafter referred to as the "Department", and the,
STANDARD CONTRACT AREA AGENCY ON AGING (Area Agency Name) THIS CONTRACT is entered into between the State of Florida, Department of Elder Affairs, hereinafter referred to as the "Department", and the,
Appendix B-1. Feasibility Study Task Order Template
 Appendix B-1 Feasibility Study Task Order Template Task Order between and the Massachusetts Clean Energy Technology Center This Task Order dated (the Effective
Appendix B-1 Feasibility Study Task Order Template Task Order between and the Massachusetts Clean Energy Technology Center This Task Order dated (the Effective
Enter a Subcontract in erpm
 This procedure details the process of adding a subcontract (SUBK) to a project. I. PAF: indicating that a project will include a subcontract II. Subcontract Worksheet: Adding a subcontract III. PAF Workspace:
This procedure details the process of adding a subcontract (SUBK) to a project. I. PAF: indicating that a project will include a subcontract II. Subcontract Worksheet: Adding a subcontract III. PAF Workspace:
PLAN OF ACTION FOR IMPLEMENTATION OF 510(K) AND SCIENCE RECOMMENDATIONS
 PLAN OF ACTION FOR IMPLEMENTATION OF 510(K) AND SCIENCE RECOMMENDATIONS In August 2010, the Food and Drug Administration s Center for Devices and Radiological Health (CDRH or the Center) released for public
PLAN OF ACTION FOR IMPLEMENTATION OF 510(K) AND SCIENCE RECOMMENDATIONS In August 2010, the Food and Drug Administration s Center for Devices and Radiological Health (CDRH or the Center) released for public
(a) Article 1. Awards Covered by Research General T&Cs. REFERENCE: ARTICLE 2(a)
 U. S. ARMY RESEARCH OFFICE RESEARCH GENERAL TERMS AND CONDITIONS AGENCY SPECIFIC REQUIREMENTS 1 June 2009 Army Research Office Home Page http://www.aro.army.mil DoDGARS Part 32 http://www.dtic.mil/whs/directives/corres/html/321006r.htm
U. S. ARMY RESEARCH OFFICE RESEARCH GENERAL TERMS AND CONDITIONS AGENCY SPECIFIC REQUIREMENTS 1 June 2009 Army Research Office Home Page http://www.aro.army.mil DoDGARS Part 32 http://www.dtic.mil/whs/directives/corres/html/321006r.htm
Emory Research A to Z ERAZ
 Emory Research A to Z ERAZ July 17, 2014 Whitehead Auditorium G01 Whitehead Building Agenda Controlled Substances Sub Award Processing RAS Updates SAM Kiosk Federal Updates OSP/OGCA Training Updates Christine
Emory Research A to Z ERAZ July 17, 2014 Whitehead Auditorium G01 Whitehead Building Agenda Controlled Substances Sub Award Processing RAS Updates SAM Kiosk Federal Updates OSP/OGCA Training Updates Christine
Contract Administration Policy
 Contract Administration Policy Guidelines for contract approval March 15, 2010 5420 Old Orchard Road Skokie, Illinois 60077-1083 847.966.6200 Fax 847.966.9781 www.cement.org Guidelines for Contract Approval
Contract Administration Policy Guidelines for contract approval March 15, 2010 5420 Old Orchard Road Skokie, Illinois 60077-1083 847.966.6200 Fax 847.966.9781 www.cement.org Guidelines for Contract Approval
Division of Research
 Division of Research Quarterly Research Administration Meeting October 30, 2012 Page 1 1 Division of Research Quarterly Research Administration Meeting AGENDA Welcome and Introductions Pre Award Update
Division of Research Quarterly Research Administration Meeting October 30, 2012 Page 1 1 Division of Research Quarterly Research Administration Meeting AGENDA Welcome and Introductions Pre Award Update
Subaward Policies and Procedures Manual
 JOHNS HOPKINS UNIVERISTY Krieger School of Arts and Sciences Business and Research Administration Subaward Policies and Procedures Manual 2016 Contents Introduction...3 Differentiating types of relationships...3
JOHNS HOPKINS UNIVERISTY Krieger School of Arts and Sciences Business and Research Administration Subaward Policies and Procedures Manual 2016 Contents Introduction...3 Differentiating types of relationships...3
Standard Operating Procedure (SOP) for Reporting Serious Breaches in Clinical Research
 Standard Operating Procedure (SOP) for Reporting Serious Breaches in Clinical Research For Completion by SOP Author Reference Number PHT/RDSOP/002 Version V2.0 07 Apr 2016 Document Author(s) Document Reviewer(s)
Standard Operating Procedure (SOP) for Reporting Serious Breaches in Clinical Research For Completion by SOP Author Reference Number PHT/RDSOP/002 Version V2.0 07 Apr 2016 Document Author(s) Document Reviewer(s)
Electronic Research Administration (era) March 8, 2018
 Electronic Research Administration (era) March 8, 2018 Introductions We are. Terry Duperron, era Analyst Marlene Mooshian, Proposal Analyst Kassie Obelleiro, Training Officer Jinger Snyder, Award Analyst
Electronic Research Administration (era) March 8, 2018 Introductions We are. Terry Duperron, era Analyst Marlene Mooshian, Proposal Analyst Kassie Obelleiro, Training Officer Jinger Snyder, Award Analyst
SUMMARY. Extramural Funds Accounting effectively conducts post award financial management:
 SUMMARY Extramural Funds Accounting assists campus faculty and staff in meeting their teaching, research, and operational needs by providing quality financial services for externally funded projects. Protecting
SUMMARY Extramural Funds Accounting assists campus faculty and staff in meeting their teaching, research, and operational needs by providing quality financial services for externally funded projects. Protecting
Office of Business and Financial Services Procurement and Contracts Division Section SUBJECT: PROCUREMENT OF CONSTRUCTION SERVICES
 2510.8 SUBJECT: PROCUREMENT OF CONSTRUCTION SERVICES :1 OBJECTIVE: To establish a uniform policy and procedure for the acquisition of construction services for the City of Orlando (City), including, but
2510.8 SUBJECT: PROCUREMENT OF CONSTRUCTION SERVICES :1 OBJECTIVE: To establish a uniform policy and procedure for the acquisition of construction services for the City of Orlando (City), including, but
Accelerated Translational Incubator Pilot (ATIP) Program. Frequently Asked Questions. ICTR Research Navigators January 19, 2017 Version 7.
 Accelerated Translational Incubator Pilot (ATIP) Program Frequently Asked Questions ICTR Research Navigators January 19, 2017 Version 7.0 TABLE OF CONTENTS Section # Title Page 1. ABOUT THE ATIP PROGRAM...
Accelerated Translational Incubator Pilot (ATIP) Program Frequently Asked Questions ICTR Research Navigators January 19, 2017 Version 7.0 TABLE OF CONTENTS Section # Title Page 1. ABOUT THE ATIP PROGRAM...
Single Audit Entrance Conference Uniform Guidance Refresher
 Single Audit Entrance Conference Uniform Guidance Refresher MGO Audit Partner Annie Louie 31 Uniform Guidance Effective Date Federal Agencies Implement policies and procedures by promulgating regulations
Single Audit Entrance Conference Uniform Guidance Refresher MGO Audit Partner Annie Louie 31 Uniform Guidance Effective Date Federal Agencies Implement policies and procedures by promulgating regulations
Sponsored Program Administration Policy Approved by Academic Senate on 4/4/06
 Sponsored Program Administration Policy Approved by Academic Senate on 4/4/06 Page 1 of 10 Article 1. Definitions 1.1. Auxiliary means an Auxiliary Organization as defined in Executive Order No. 698. 1.2.
Sponsored Program Administration Policy Approved by Academic Senate on 4/4/06 Page 1 of 10 Article 1. Definitions 1.1. Auxiliary means an Auxiliary Organization as defined in Executive Order No. 698. 1.2.
Complaints and Suggestions for Improvement Handling Procedure
 Complaints and Suggestions for Improvement Handling Procedure Date of most recent review: 20 June 2013 Date of next review: August 2016 Responsibility: Quality Officer Approved by: Learning, Teaching and
Complaints and Suggestions for Improvement Handling Procedure Date of most recent review: 20 June 2013 Date of next review: August 2016 Responsibility: Quality Officer Approved by: Learning, Teaching and
LISD Procedures For contract monitoring and compliance
 LISD Procedures For contract monitoring and compliance Source of Policy CH Legal and Chapter 44 of the Texas Education Code CH Local Board Policies Financial Guidelines Required Documents Copy of contract
LISD Procedures For contract monitoring and compliance Source of Policy CH Legal and Chapter 44 of the Texas Education Code CH Local Board Policies Financial Guidelines Required Documents Copy of contract
SJSU Research Foundation
 SJSU Research Foundation Grant Proposal Development Taking a Bite Out of Forms Sponsored Programs Workshop November 19, 2015 Overview of Workshop Why We Have Forms Forms from OSP Forms from Human Resources
SJSU Research Foundation Grant Proposal Development Taking a Bite Out of Forms Sponsored Programs Workshop November 19, 2015 Overview of Workshop Why We Have Forms Forms from OSP Forms from Human Resources
BRISTOL-MYERS SQUIBB DATA SHARING INDEPENDENT REVIEW COMMITTEE (IRC) CHARTER
 BRISTOL-MYERS SQUIBB DATA SHARING INDEPENDENT REVIEW COMMITTEE (IRC) CHARTER Charter Effective Date: October 13, 2017 Release v2.0 Page 1 of 6 Introduction This Charter describes the roles and responsibilities
BRISTOL-MYERS SQUIBB DATA SHARING INDEPENDENT REVIEW COMMITTEE (IRC) CHARTER Charter Effective Date: October 13, 2017 Release v2.0 Page 1 of 6 Introduction This Charter describes the roles and responsibilities
Project Request and Approval Process
 The University of the District of Columbia Information Technology Project Request and Approval Process Kia Xiong Information Technology Projects Manager 13 June 2017 Table of Contents Project Management
The University of the District of Columbia Information Technology Project Request and Approval Process Kia Xiong Information Technology Projects Manager 13 June 2017 Table of Contents Project Management
Policies and Procedures for CRA Funded Grants
 Policies and Procedures for CRA Funded Grants Table of Contents 1. Our mission... 2 2. Glossary of CRA terms... 2 3. Online grant management... 3 4. Grant activation... 3 5. International awards... 3 6.
Policies and Procedures for CRA Funded Grants Table of Contents 1. Our mission... 2 2. Glossary of CRA terms... 2 3. Online grant management... 3 4. Grant activation... 3 5. International awards... 3 6.
Guidance for Industry ANDA Submissions Prior Approval Supplements Under GDUFA
 Guidance for Industry ANDA Submissions Prior Approval Supplements Under GDUFA DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this
Guidance for Industry ANDA Submissions Prior Approval Supplements Under GDUFA DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this
Office of Internal Audit
 Office of Internal Audit July 5, 2017 Dr. Kirk A. Calhoun, President UT Health Northeast 11937 U. S. Hwy 271 Tyler, TX 75708 Dr. Calhoun, We have completed the that was part of our Audit Plan. The objective
Office of Internal Audit July 5, 2017 Dr. Kirk A. Calhoun, President UT Health Northeast 11937 U. S. Hwy 271 Tyler, TX 75708 Dr. Calhoun, We have completed the that was part of our Audit Plan. The objective
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018
 CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
Tier A Cycle 1 Pipeline to Proposal Awards Application Guidelines. Published March 1, 2017
 Tier A Cycle 1 Pipeline to Proposal Awards Application Guidelines Published March 1, 2017 About PCORI The Patient-Centered Outcomes Research Institute (PCORI) is committed to transparency and a rigorous
Tier A Cycle 1 Pipeline to Proposal Awards Application Guidelines Published March 1, 2017 About PCORI The Patient-Centered Outcomes Research Institute (PCORI) is committed to transparency and a rigorous
OSP Subaward Request Process. To utilize the system to initiate and complete a request, follow the steps below.
 Effective Date: March 1, 2014 Version: 1 Page: 1 of 13 Overview All Sponsored Awards, whether grants, contracts or cooperative agreements, which are received by Emory and which involve participation by
Effective Date: March 1, 2014 Version: 1 Page: 1 of 13 Overview All Sponsored Awards, whether grants, contracts or cooperative agreements, which are received by Emory and which involve participation by
NOVA SOUTHEASTERN UNIVERSITY
 NOVA SOUTHEASTERN UNIVERSITY DIVISION OF RESPONSIBILITIES FOR RESEARCH AND SPONSORED PROGRAMS Vice President of Research & Technology Transfer: The responsibilities of the Vice President of Research &
NOVA SOUTHEASTERN UNIVERSITY DIVISION OF RESPONSIBILITIES FOR RESEARCH AND SPONSORED PROGRAMS Vice President of Research & Technology Transfer: The responsibilities of the Vice President of Research &
PPEA Guidelines and Supporting Documents
 PPEA Guidelines and Supporting Documents APPENDIX 1: DEFINITIONS "Affected jurisdiction" means any county, city or town in which all or a portion of a qualifying project is located. "Appropriating body"
PPEA Guidelines and Supporting Documents APPENDIX 1: DEFINITIONS "Affected jurisdiction" means any county, city or town in which all or a portion of a qualifying project is located. "Appropriating body"
PERFORMANCE IN INITIATING CLINICAL RESEARCH (PI) CLINICAL TRIAL ADJUSTMENT PROCESS HRA APPROVED TRIALS
 PERFORMANCE IN INITIATING CLINICAL RESEARCH (PI) CLINICAL TRIAL ADJUSTMENT PROCESS HRA APPROVED TRIALS Version Control Pages 7 9 Notes/action column updated 1 Acronyms CTP DSC DSS FPR HRA MHRA REC SOP
PERFORMANCE IN INITIATING CLINICAL RESEARCH (PI) CLINICAL TRIAL ADJUSTMENT PROCESS HRA APPROVED TRIALS Version Control Pages 7 9 Notes/action column updated 1 Acronyms CTP DSC DSS FPR HRA MHRA REC SOP
EFFICIENCY MAINE TRUST REQUEST FOR PROPOSALS FOR Forward Capacity Market Support Services RFP NUMBER EM
 EFFICIENCY MAINE TRUST REQUEST FOR PROPOSALS FOR Forward Capacity Market Support Services RFP NUMBER EM-006-2018 Date Issued: 11/20/2017 TABLE OF CONTENTS SECTION 1 RFP INFORMATION AND INSTRUCTIONS SECTION
EFFICIENCY MAINE TRUST REQUEST FOR PROPOSALS FOR Forward Capacity Market Support Services RFP NUMBER EM-006-2018 Date Issued: 11/20/2017 TABLE OF CONTENTS SECTION 1 RFP INFORMATION AND INSTRUCTIONS SECTION
Sponsorship Agreement/Sub-Grant Posted Date June 6, 2016 Due Date for Applications Cycle 1: Cycle 2: July 15, 2016 January 13, 2017
 REQUEST FOR PROPOSALS Xcel Energy Renewable Development Fund MnSCU Block Grant Award Type Sponsorship Agreement/Sub-Grant Posted Date June 6, 2016 Due Date for Applications Cycle 1: Cycle 2: July 15, 2016
REQUEST FOR PROPOSALS Xcel Energy Renewable Development Fund MnSCU Block Grant Award Type Sponsorship Agreement/Sub-Grant Posted Date June 6, 2016 Due Date for Applications Cycle 1: Cycle 2: July 15, 2016
REQUEST FOR PROPOSAL FOR: Grant Writing & Post-Award/Project Implementation Technical Assistance River Valley Community College BID #RVC15-09
 REQUEST FOR PROPOSAL FOR: Grant Writing & Post-Award/Project Implementation Technical Assistance River Valley Community College BID #RVC15-09 PURPOSE: River Valley Community College seeks professional
REQUEST FOR PROPOSAL FOR: Grant Writing & Post-Award/Project Implementation Technical Assistance River Valley Community College BID #RVC15-09 PURPOSE: River Valley Community College seeks professional
Clinical Trials Where World Class Care Begins
 Office of Clinical Trials Clinical Trials Where World Class Care Begins We seek to attract novel, innovative and transformational research and devices to our medical center and its stakeholders, and serve
Office of Clinical Trials Clinical Trials Where World Class Care Begins We seek to attract novel, innovative and transformational research and devices to our medical center and its stakeholders, and serve
Agreement. Between: The Research Foundation, Cerebral Palsy Alliance. And: (Name of Institution) Project: (Name of project)
 Agreement Between: The Research Foundation, Cerebral Palsy Alliance And: (Name of Institution) Project: (Name of project) Contents 1. Defined meanings... 1 2. Administration of Award... 1 3. Funding...
Agreement Between: The Research Foundation, Cerebral Palsy Alliance And: (Name of Institution) Project: (Name of project) Contents 1. Defined meanings... 1 2. Administration of Award... 1 3. Funding...
Florida MIECHV Initiative Provider Quality Assurance Monitoring Procedure Manual
 2016 Florida MIECHV Initiative Provider Quality Assurance Monitoring Procedure Manual Florida MIECHV Initiative This project is/was supported by the Health Resources and Services Administration (HRSA)
2016 Florida MIECHV Initiative Provider Quality Assurance Monitoring Procedure Manual Florida MIECHV Initiative This project is/was supported by the Health Resources and Services Administration (HRSA)
COMMISSIONING SUPPORT PROGRAMME. Standard operating procedure
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE COMMISSIONING SUPPORT PROGRAMME Standard operating procedure April 2018 1. Introduction The Commissioning Support Programme (CSP) at NICE supports the
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE COMMISSIONING SUPPORT PROGRAMME Standard operating procedure April 2018 1. Introduction The Commissioning Support Programme (CSP) at NICE supports the
Policy Type. Rationale. Terms. New Proposed Title Grants and Sponsored Programs. Proposed Title. Revision. Existing Title. Existing Index No.
 Policy Type Please provide any applicable information below. Leave blank what is not applicable. New Proposed Title Grants and Sponsored Programs Proposed Title Revision Existing Title Existing Index No.
Policy Type Please provide any applicable information below. Leave blank what is not applicable. New Proposed Title Grants and Sponsored Programs Proposed Title Revision Existing Title Existing Index No.
Northern Ireland Social Care Council Quality Assurance Framework for Education and Training Regulated by the Northern Ireland Social Care Council
 Northern Ireland Social Care Council Quality Assurance Framework for Education and Training Regulated by the Northern Ireland Social Care Council Approval, Monitoring, Review and Inspection Arrangements
Northern Ireland Social Care Council Quality Assurance Framework for Education and Training Regulated by the Northern Ireland Social Care Council Approval, Monitoring, Review and Inspection Arrangements
Principal Investigator
 Appendix A: Roles and Responsibilities Details Principal Investigator 1. Proposal Preparation 1.1 Has an overall responsibility to assemble a submission ready proposal meaning a complete, accurate proposal
Appendix A: Roles and Responsibilities Details Principal Investigator 1. Proposal Preparation 1.1 Has an overall responsibility to assemble a submission ready proposal meaning a complete, accurate proposal
