FDP Meeting Summary. Compliance Unit Standard Procedure (CUSP) Project to Point of Contact Activities/Progress to Date
|
|
|
- Rosamond Fields
- 5 years ago
- Views:
Transcription
1 Compliance Unit Standard Procedure (CUSP) Project Point of Contact Activities/Progress to Date Agenda/Discussion Points Pending Decisions Participation Key Risks/Issues Meeting Summary Aubrey Schoenleben, University of Washington; Sally Thompson-Iritani, University of Washi The goal of this working group is to develop an online resource for sharing standard procedures used in animal care protocols. See below and session slides for progress to date. - Build out picklists for two procedure types (Euthanasia, Blood/Tissue Collection). - Define a process for how procedures will be renewed within the system. - Develop the procedure entry form. - Determine if single sign on (SSO) is a viable option to access the site. This session was attended by approximately 40 individuals, either in person or via web conference. See Key Decisions Pending above. Two new documents for the working group were introduced the Key Decision Log and the Site Scope and Spec Sheet. The Key Decision Log outlines project decisions and the rationale behind each decision. The Site Scope and Spec Sheet defines the scope for the first phase of the project and details the different requirements and characteristics of the CUSP site. This document will be used to support the development phase of the project. The working group is exploring whether use of single sign on (SSO) authentication is a viable option for the site. Initial evaluation revealed that 77% of FDP member institutions are members of InCommon Federation, an SSO solution that serves a broad range of education and research institutions. The group is currently evaluating what other SSO options might be available to leverage for the remaining 23% of FDP member institutions. It was recommended that we reach out to the era committee, as they have already done some work in this area. The group also reviewed and provided feedback on the initial wireframes for webpages that users with the roles of Designated Institutional Representative and Administrator will interact with. The pages reviewed include My Procedures, My Accounts, and Reports. The federal sponsor for this project provided an update on the Request for Information (RFI) that was issued in March 2018 in response to the 21st Century Cures Act. The purpose of this RFI is to seek input from the wider scientific community and public on potential measure that may be taken to reduce regulatory burden. A subset of working group members also met for an in-depth discussion on species and procedure types, and how to build the related pick lists. The picklists for the species was finalized, and the build out of picklists for two procedure types was started.
2 Volunteer Opportunities Please contact Aubrey or Sally if you are interested in joining this working group. The working group meets monthly.
3 Subawards Subcommittee Point of Contact Activities/Progress to Date Amanda Hamaker, Amanda Humphrey, Stephanie Scott Guidance Group - Two documents were produced since the last meeting. 1) Guidance document to share with internal institutional stakeholders (legal counsel, etc - those who may require added language to the templates). The document has background on FDP and the purpose of the templates, checklist of considerations prior to requesting language changes/additions, and process for making requests. 2) New form for requesting changes to the templates. Fixed Priced Subawards, Prior Approval, and Clinical Trials - Recap of the issue discussed in the January 2018 /September 2017 meetings. Draft language has been developed for requesting prior approval. Discussion over what is a Clinical trial - sub versus vendor, etc. Discussion of the Simplified acquisition threshold (SAT). Increased from $150K to $250K when the micro-purchase threshold increased. The group engaged with COGR Research Compliance & Administration (RCA) Committee. They met with NIH OPERA on 2/21/18 to present burdens and possible alternatives. The group with RCA provided draft language for GPS based on language found in DoD General Research Terms & Conditions. This is drafted and ready to be sent to NIH. If you are currently still having issues, Jennifer McAllister is still collecting information. She can also provide you with more information. Recommendation is to get OPERA involved in the meantime if you are dealing with one of these issues. Discussions from attendees show that there are lots of issues surrounding this topic. Agenda/Discussion Points Pending Decisions Research Integrity - Recap of the issue discussed at the January 2018 meeting. After last meeting, we gathered more information. A Research Integrity Officer meeting had a discussion wanting to know if institutions were ensuring subrecipients had applicable policies/assurances in place. Discussed the use of adding language specific to foreign/high risk template and/or adding something to the FDP Clearinghouse. Completed a survey to see how membership wanted to handle this issue. Suggested outcomes include: add language to foreign template; develop a welcome packet for foreign intuitions; explore FDP EC options; no language added to the FDP subaward templates; add guidance explaining choices. Also, engage RIO community for feedback. Poll of the audience is strong agreement with this plan. Certificates of Confidentiality - Recap of the issue discussed at the January 2018 and prior meetings. Sent out with the Research Integrity survey to gauge membership preference of whether or not to include language in the subaward templates and which of the two provided language options were preferred. Very mixed results and no clear concurrence from the survey. Very strong opinions on all options. Intuition types did not correlate to
4 Participation Open Questions at the end - the responses. Suggested outcomes: No clear suggestions. We have decided to post both versions of the language on the FDP Website and allow institutions to test over the next few months. Institutions should evaluate which work best for them, which get the least pushback, etc. We will ask for feedback at some point in late 2018 via another survey. These responses will help to determine what may get incorporated into the templates. The majority of attendees found this to be a reasonable approach. Subaward Templates Updates - Working on potential modifications to the code to stabilize the code. Asked the attendees if we should go ahead and post the updated templates with the code changes for bug fixes since this is an off-cycle update. Request to verify all links on attachment 2 to research terms and conditions are updated. 1. Discussion over DocuSign - one institution using Echosign (sp?), another using Docusign with no issues, still seeing pushback from state agencies/others who want hard signed blue ink, multiple copies, etc. 2. What if we want a new template - combining a couple of templates for a new scenario - should we use the change request form? Bigger requests like this should probably involve some pre-discussion with the co-chairs. 3. Single audit entities - Who has stopped sending out single audit certifications or not? Most comments indicated they are pushing back when they get a certification to complete. May be helpful to have a push back template language. Pamela will send out the UG language on the listserv. Key Risks/Issues Upcoming activities: Guidance Group - Finalize Change Request Form & Guidance Doc; New FAQs to include - Clarify Term Effective Date for Mods (how and when it is used), Certificates of Confidentiality - language options, Research Integrity, sirb - how it was incorporated into the templates, Revise FAQ7 - formatting change requests, New FAQs on template change requests, New FAQ on when it is appropriate for PTEs to request back-up of invoices, and the Carryover Doc - Total Amount of Federal Funds Obligated to Date (TAFFOD). Foreign Subaward Guidance/FAQs - new group. Foreign subawards may require different terms. As a result, guidance/faqs will be created to help membership with these subawards. Subcontract Working Group - finalizing the updated sample. Looking ahead: September meeting - Closeout, Back up for invoices, Foreign / high risk subrecipient informational packet, Collaboration agreement for federal entities Meeting Summary The session covered the following: Research Integrity - review of the survey results; action plan moving forward with audience approval Certificates of Confidentiality - review of the survey results; action plan moving forward
5 with audience approval Subaward Templates Update - Discussion of the need for off-cycle bug-fix release to stabilize code in Adobe templates Fixed Price/Clinical Trial Update - Update on progress since the January meeting. Joint COGR/FDP representative meeting with NIH OPERA. Draft language to be sent to NIH. Guidance Document Update/New Proposal Form - Shared information about the new change request form and related guidance doc for University stakeholders. Foreign Guidance Update - Discussion over need to develop guidance and FAQs. Volunteer Opportunities Guidance Document Group is looking for more volunteers to work on new and revised FAQs. Contact Stephanie Scott; new workgroup to be formed, led by Julie Renkas, to focus on guidance when issuing subawards to foreign entities.
6 Emerging Research Institutions (ERI) Point of Contact Activities/Progress to Date Agenda/Discussion Points Pending Decisions Participation Susan Anderson In a continuing effort to support engagement by ERI members in FDP activities, a session at the May meeting was dedicated to an opportunity to have focused discussion with a Federal partner (NIH). Additional activities include recent administration of a survey to ERI Administrative Representatives; the survey is intended to identify areas in which ERI members would like to place attention. Survey results will be analyzed and discussed at a future meeting. Survey results will guide future project choices and meeting session topics/guests. In addition to the 5 representatives from NIH (Megan Columbus, Division of Communication and Outreach and NIH Program Manager for Electronic Submission of Grant Applications in the Office of Extramural Research, who led the discussion on behalf of NIH; along with Sheri Cummins, NIH Office of Extramural Research Communications and Scarlett Gibb, era Customer Relationship Manager; and Sam Ashe, Director, Division of Grants Policy, Stefanie Harris, Grants Policy Analyst, Division of Grants Policy), Federal representatives from NSF and USDA attended. Among the non-federal institutions, representatives from 10 ERI and 7 non-eri members participated. Of note was interest in the session among non-eri members, many of whom desired information to help them collaborate with ERI members. Key Risks/Issues Meeting Summary There was robust discussion of several items of interest to attendees and to NIH staff. NIH representatives took away several ideas, concerns, or suggestions that resulted from the discussion. Participants also left with a number of suggestions and resources to help them navigate NIH programs and systems. Challenges and risks: what makes ERIs unique? Limited support staff (e.g., JIT IRB turnaround challenging); limited experience with NIH (e.g., vocabulary; when/where to send JIT responses; appropriate programs). Concerns: competitiveness for some mechanisms; unsure who to talk to at NIH; how to get involved in peer review process (suggestion: NIH's Early Career Reviewer program); equity re: clinical trials definition/policies (e.g., AREA--CTs not allowed; small behavior studies no longer permitted re: CT issue); clean up of costs in PMS. How can NIH help? Single matrix of compliance requirements; provide systematic way to extract application data to avoid need to rekey; provide a resource with timelines for critical actions; indication in Commons if application will not be funded; best practice guidance for institutional policies; guidance for PIs on getting started with NIH. Additional resources noted: RePORT system; AREA mechanism; K12; SCORE program; RCMI
7 program; Diversity Program Consortium including BUILD initiative and NRMNetwork. Volunteer Opportunities NIH maintains a Commons Working Group listserv and holds meetings (associated with FDP but generally after). This group helps NIH ensure they design NIH era systems with the end user in mind and helps them identify opportunities to improve efficiencies for organizations. Megan Columbus can add you to the list; her at Megan.columbus@nih.gov. NIH s Office of Extramural Research is responsible for the resources about NIH grants processes and policies at grants.nih.gov. Please megan.columbus@nih.gov or oer@od.nih.gov with suggestions or questions.
8 FDP Expanded Clearinghouse Point of Contact Activities/Progress to Date Agenda/Discussion Points Pending Decisions Participation Key Risks/Issues Lynette Arias, Pamela Webb The Pilot has moved into an initiative and includes a Steering Committee. Publications/Communications: Final Pilot Report, Press Release, NCURA Article, NCURA regional meetings with more requests for publicity coming Almost all FDP members have joined the Expanded Clearinghouse profiles total todate! Sample pushback language has been updated and is available on the website. Website has been updated opilot information is being archived Instructional videos are available for the Expanded Clearinghouse with a few more coming Significant codebase updates, though users will not see any changes, in order to improve maintainability and sustainability Data Export feature extensively reviewed but will not be offered oduplicates API functionality oreduces references to website, increases risk of old/bad data oenhanced search tools will be coming to the website in the future API will go live this month o3 tokens have been issued to Beta testers oaccess may be requested through the Data Access tab on a published profile Financial Questionnaire ocurrent list on FDP website is exhaustive and is being consolidated oprogress to-date?goal to create a document that is widely usable?creating a survey to be sent to Pass through Entities to get a better understanding of interest and challenges Mandatory for all FDP members? At this point it is moot, but is something to consider in future phase. Inviting non-fdp organizations to join. More Federal Involvement? We d like to start conversations with SAM.gov and the Federal Audit Clearinghouse. Financial Questionnaire added to Clearinghouse? FCOI Clearinghouse to be added as well? Who should own the system/data? What data fields are irrelevant or not being utilized by the institutions? Approximately 100 attendees, actively participating in user group type discussions. Should we allow non-fdp organizations to join? A significant amount of thought must go
9 into ensuring the integrity of the data, among other things. Meeting Summary Volunteer Opportunities The session covered the following: Recent press releases and other communications and marketing of the Clearinghouse, Reviewed parameters for updating profiles and identify key areas currently causing confusion or delays Provided status of all FDP members who have become Participating Organizations System updates including: status of Automated Programming Interface (API); data dictionary; possible data export; SAM.gov mapping; and other enhancements Future planning including: status of adding additional members, financial questionnaire pilot, partnering with federal agencies on what we have learned Participate in Financial Questionnaire pilot. Please Sara Clough or Lesley Schmidt Sindberg
10 Faculty Administrator Collaboration Team (FACT) Point of Contact Activities/Progress to Date Agenda/Discussion Points Pending Decisions Participation Key Risks/Issues Meeting Summary Mark Haselkorn/Dave Reed Developed a charter Executive Committee endorsement Initiated website Established Communication and Marketing Subgroup Expanded membership Initiated two pilot projects to better understand the Faculty & Administrator collaboration oquantitative assessment of research administration with the FACT partner institutions oqualitative assessment of research administration with the FACT partner institutions Finalize quantitative metrics and gather qualitative interviews at nine participating institutions. Open to pairs of faculty/administrator. Contact Mark Haselkorn/Dave Reed Still gathering preliminary data. No obstacles to report to date. See FACT webpage, For detailed sessions notes Volunteer Opportunities Understand diversity of existing structures and strategies for Faculty-Administration research collaborations Identify best practices and opportunities for improvement of institutional structures and strategies for Faculty-Administration research collaborations Gain understanding of institutional definitions of research enterprise success Establish conditions for, and examples of, Faculty-Administration collaboration successes Establish examples of frequent challenges to Faculty-Administration collaboration
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM. December 2017
 Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE
 THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE Alexandra A. McKeown, Associate Dean Johns Hopkins University May 2, 2011 1 What is the FDP? The FDP is a cooperative effort among federal research
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE Alexandra A. McKeown, Associate Dean Johns Hopkins University May 2, 2011 1 What is the FDP? The FDP is a cooperative effort among federal research
FDP Meeting Summary. Pipelines Group. September 6-8, Point of Contact Activities/Progress to Date
 Pipelines Group Michele Masucci The pipelines group is reviewing a final manuscript of a report on work-life-balancesatisfaction approaches at member institutions, which can serve as an inventory of programs
Pipelines Group Michele Masucci The pipelines group is reviewing a final manuscript of a report on work-life-balancesatisfaction approaches at member institutions, which can serve as an inventory of programs
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE
 THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research organizations aimed at streamlining
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research organizations aimed at streamlining
FDP Expanded Clearinghouse
 FDP Expanded Clearinghouse Lynette Arias, University of Washington Pamela Webb, University of Minnesota Jennifer Barron, Johns Hopkins University Chris Renner, Vanderbilt University Medical Center Julie
FDP Expanded Clearinghouse Lynette Arias, University of Washington Pamela Webb, University of Minnesota Jennifer Barron, Johns Hopkins University Chris Renner, Vanderbilt University Medical Center Julie
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE
 THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE NECA Conference, August 21, 2012 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE NECA Conference, August 21, 2012 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research
FDP Meeting Summary. Controlled Unclassified Information (CUI) and FISM 5/10/2017-5/12/2017
 Controlled Unclassified Information (CUI) and FISM Makr Sweet This session was an update of some of the current issues surrounding Controlled Unclassified Information (CUI) and FISMA. The presenters provided
Controlled Unclassified Information (CUI) and FISM Makr Sweet This session was an update of some of the current issues surrounding Controlled Unclassified Information (CUI) and FISMA. The presenters provided
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates!
 FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
2017 SRA International Annual Meeting October 14-18, Federal Demonstration Partnership (FDP)/Council on Governmental Relations (COGR) Update
 2017 SRA International Annual Meeting October 14-18, 2017 Federal Demonstration Partnership (FDP)/Council on Governmental Relations (COGR) Update Who are we? Anthony DeCrappeo, President, Council on Governmental
2017 SRA International Annual Meeting October 14-18, 2017 Federal Demonstration Partnership (FDP)/Council on Governmental Relations (COGR) Update Who are we? Anthony DeCrappeo, President, Council on Governmental
Subrecipient Monitoring Procedures
 Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
OMB Uniform Guidance ( UG ) Briefing. ASRSP & OSR Brown Bag Tuesday, January 27 th
 OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
Emory Research A to Z ERAZ
 Emory Research A to Z ERAZ July 17, 2014 Whitehead Auditorium G01 Whitehead Building Agenda Controlled Substances Sub Award Processing RAS Updates SAM Kiosk Federal Updates OSP/OGCA Training Updates Christine
Emory Research A to Z ERAZ July 17, 2014 Whitehead Auditorium G01 Whitehead Building Agenda Controlled Substances Sub Award Processing RAS Updates SAM Kiosk Federal Updates OSP/OGCA Training Updates Christine
UNIFORM GUIDANCE IMPLEMENTATION
 UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
Procedure for Addressing PHS Animal Protocol-Proposal Congruency Requirements at the UMass Medical School
 Section I. Introduction A. In recent years, the NIH Office of Laboratory Animal Welfare has increasingly emphasized the importance of NIH-funded institutions proactively ensuring that their Investigators
Section I. Introduction A. In recent years, the NIH Office of Laboratory Animal Welfare has increasingly emphasized the importance of NIH-funded institutions proactively ensuring that their Investigators
Research Administrator Meeting Tuesday, January 23, :00-12:00 Illinois Room (348), IMU
 Research Administrator Meeting Tuesday, January 23, 2018 10:00-12:00 Illinois Room (348), IMU Agenda: 1. GAO Announcements 2. Costing Considerations 3. Quick Reference Guide Overview 4. DSP General Announcements
Research Administrator Meeting Tuesday, January 23, 2018 10:00-12:00 Illinois Room (348), IMU Agenda: 1. GAO Announcements 2. Costing Considerations 3. Quick Reference Guide Overview 4. DSP General Announcements
CURRENT COGR PRIORITIES - BY COMMITTEE (7/10/17)
 CURRENT COGR PRIORITIES - BY COMMITTEE (7/10/17) No. 1 Student Financial Aid and "Securing Student Information" and Compliance Supplement New single audit requirement. Delayed until 2018. MEDIUM Pending
CURRENT COGR PRIORITIES - BY COMMITTEE (7/10/17) No. 1 Student Financial Aid and "Securing Student Information" and Compliance Supplement New single audit requirement. Delayed until 2018. MEDIUM Pending
Research Administrators Round Table
 Research Administrators Round Table Recap Set framework for going forward Review new Uniform Guidance driven WVU policy changes Sponsored Agreement Closeouts (WVU-FS-23) http://adminfin.wvu.edu/policies/finance/financial-services/wvu-fs-23-policy
Research Administrators Round Table Recap Set framework for going forward Review new Uniform Guidance driven WVU policy changes Sponsored Agreement Closeouts (WVU-FS-23) http://adminfin.wvu.edu/policies/finance/financial-services/wvu-fs-23-policy
Expanded Clearinghouse Pilot Phase II. Will be part of 80 institutions nationwide to participate in the Clearinghouse.
 Expanded Clearinghouse Pilot Phase II Will be part of 80 institutions nationwide to participate in the Clearinghouse. 1 Goal of Pilot Reduce administrative burden associated with verifying standard information
Expanded Clearinghouse Pilot Phase II Will be part of 80 institutions nationwide to participate in the Clearinghouse. 1 Goal of Pilot Reduce administrative burden associated with verifying standard information
Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6
 Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6 KC-PRE28: Prepare Summary Proposal KC-AWD22: Research Gifts November 13, 2013 Goals and Outcomes Goals Obtain feedback on the proposed
Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6 KC-PRE28: Prepare Summary Proposal KC-AWD22: Research Gifts November 13, 2013 Goals and Outcomes Goals Obtain feedback on the proposed
Spartan RAN. Research Administrators Network Biannual Meeting April 23 rd, Before we get started, please follow the instructions on your table!
 Spartan RAN Research Administrators Network Biannual Meeting April 23 rd, 2015 Before we get started, please follow the instructions on your table! We re Going Green in 2015, so paper handouts will not
Spartan RAN Research Administrators Network Biannual Meeting April 23 rd, 2015 Before we get started, please follow the instructions on your table! We re Going Green in 2015, so paper handouts will not
The Lifecycle of a Sponsored Project
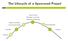 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
OMB Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards
 OMB Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Research Advisory Board What PIs Need to Know January 2015 1 Background o When and Why? o Overview Important
OMB Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Research Advisory Board What PIs Need to Know January 2015 1 Background o When and Why? o Overview Important
The Office for Sponsored Research (OSR): An Overview. Office for Sponsored Research
 The Office for Sponsored Research (OSR): An Overview Office for Sponsored Research OSR Mission The Office for Sponsored Research assists investigators in proposing and managing sponsored programs in support
The Office for Sponsored Research (OSR): An Overview Office for Sponsored Research OSR Mission The Office for Sponsored Research assists investigators in proposing and managing sponsored programs in support
Research Administrators Forum May 8th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum May 8th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Office of Research Administration OHRPP Retention of Research
Research Administrators Forum May 8th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Office of Research Administration OHRPP Retention of Research
Accelerated Translational Incubator Pilot (ATIP) Program. Frequently Asked Questions. ICTR Research Navigators January 19, 2017 Version 7.
 Accelerated Translational Incubator Pilot (ATIP) Program Frequently Asked Questions ICTR Research Navigators January 19, 2017 Version 7.0 TABLE OF CONTENTS Section # Title Page 1. ABOUT THE ATIP PROGRAM...
Accelerated Translational Incubator Pilot (ATIP) Program Frequently Asked Questions ICTR Research Navigators January 19, 2017 Version 7.0 TABLE OF CONTENTS Section # Title Page 1. ABOUT THE ATIP PROGRAM...
Reducing Regulatory and Institutional Burden Associated with Animal Research. June 8, 2017
 Reducing Regulatory and Institutional Burden Associated with Animal Research June 8, 2017 Matt Bailey, President, National Association for Biomedical Research Molly Greene, IACUC Advisor, Michigan State
Reducing Regulatory and Institutional Burden Associated with Animal Research June 8, 2017 Matt Bailey, President, National Association for Biomedical Research Molly Greene, IACUC Advisor, Michigan State
Electronic Research Administration (era) March 8, 2018
 Electronic Research Administration (era) March 8, 2018 Introductions We are. Terry Duperron, era Analyst Marlene Mooshian, Proposal Analyst Kassie Obelleiro, Training Officer Jinger Snyder, Award Analyst
Electronic Research Administration (era) March 8, 2018 Introductions We are. Terry Duperron, era Analyst Marlene Mooshian, Proposal Analyst Kassie Obelleiro, Training Officer Jinger Snyder, Award Analyst
FDP Expanded Clearinghouse
 FDP Expanded Clearinghouse Lynette Arias, University of Washington Pamela Webb, University of Minnesota Jennifer Barron, Johns Hopkins University Julie Thatcher, Institute for Systems Biology Chris Renner,
FDP Expanded Clearinghouse Lynette Arias, University of Washington Pamela Webb, University of Minnesota Jennifer Barron, Johns Hopkins University Julie Thatcher, Institute for Systems Biology Chris Renner,
Office of Sponsored Programs Workshop NIH Conflict of Interest Workshop 2
 Office of Sponsored Programs Workshop NIH Conflict of Interest Workshop 2 August 9, 2012 August 13, 2012-1 OSP Workshop Agenda Where we left off in Workshop 1 Who is an Investigator Conflicts of Interest
Office of Sponsored Programs Workshop NIH Conflict of Interest Workshop 2 August 9, 2012 August 13, 2012-1 OSP Workshop Agenda Where we left off in Workshop 1 Who is an Investigator Conflicts of Interest
OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015
 OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015 INTRODUCTION The University of Wisconsin-Milwaukee (UWM) is the prime recipient on a wide range of sponsored awards
OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015 INTRODUCTION The University of Wisconsin-Milwaukee (UWM) is the prime recipient on a wide range of sponsored awards
MEGAN COLUMBUS NIH OFFICE OF EXTRAMURAL RESEARCH (OER)
 MEGAN COLUMBUS NIH OFFICE OF EXTRAMURAL RESEARCH (OER) OCTOBER 2016 2 #1: Where s the funding? #2: What do I need to do? #3: Should I contact NIH before applying? #4: How long does it take to get funded?
MEGAN COLUMBUS NIH OFFICE OF EXTRAMURAL RESEARCH (OER) OCTOBER 2016 2 #1: Where s the funding? #2: What do I need to do? #3: Should I contact NIH before applying? #4: How long does it take to get funded?
Subaward Policies and Procedures Manual
 JOHNS HOPKINS UNIVERISTY Krieger School of Arts and Sciences Business and Research Administration Subaward Policies and Procedures Manual 2016 Contents Introduction...3 Differentiating types of relationships...3
JOHNS HOPKINS UNIVERISTY Krieger School of Arts and Sciences Business and Research Administration Subaward Policies and Procedures Manual 2016 Contents Introduction...3 Differentiating types of relationships...3
Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed:
 COEUS NIH-GRANTS.GOV PROPOSAL REVIEW CHECKLIST REVIEWER: Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed: Has the Program Announcement been reviewed? What is
COEUS NIH-GRANTS.GOV PROPOSAL REVIEW CHECKLIST REVIEWER: Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed: Has the Program Announcement been reviewed? What is
ORA Quarterly Meeting April
 ORA Quarterly Meeting April 19 2017 Agenda 1. Workday Grants Implementation Items General Information Blackout Period Sub K s - Set-up and Invoicing Advance Accounts 2. Jackson/ UM Partnership Master Agreement
ORA Quarterly Meeting April 19 2017 Agenda 1. Workday Grants Implementation Items General Information Blackout Period Sub K s - Set-up and Invoicing Advance Accounts 2. Jackson/ UM Partnership Master Agreement
The Uniform Guidance (2 CFR, Part 200)
 WCMC Implementation of The Uniform Guidance (2 CFR, Part 200) Tuesday, September 22, 2015 & Wednesday, September 23, 2015 UG Workshop Michelle A. Lewis, M.S. Director of Research Administration Interim
WCMC Implementation of The Uniform Guidance (2 CFR, Part 200) Tuesday, September 22, 2015 & Wednesday, September 23, 2015 UG Workshop Michelle A. Lewis, M.S. Director of Research Administration Interim
Sponsored Program Services. Ken Sandel Senior Director, Sponsored Program Services Date: August 18, 2016
 Sponsored Program Services Ken Sandel Senior Director, Sponsored Program Services Date: August 18, 2016 1 SPONSORED RESEARCH AWARDS Research awards $403M: RECORD $450 Sponsored Research Awards system-wide
Sponsored Program Services Ken Sandel Senior Director, Sponsored Program Services Date: August 18, 2016 1 SPONSORED RESEARCH AWARDS Research awards $403M: RECORD $450 Sponsored Research Awards system-wide
Postdoctoral Fellowships ( )
 Postdoctoral Fellowships (2018-2020) Guidelines and General Instructions for Application KEY DATES Application Release Date: May 24, 2017 Application Deadline: September 6, 2017 at 5:00 PM EST Peer Review
Postdoctoral Fellowships (2018-2020) Guidelines and General Instructions for Application KEY DATES Application Release Date: May 24, 2017 Application Deadline: September 6, 2017 at 5:00 PM EST Peer Review
Rebecca Trahan. Office of Sponsored Programs December 9, ORED Limited Submission Update
 Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
Research Administrators Forum October 10th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum October 10th, 2013 Marcia Smith Associate Vice Chancellor for Research Agenda Meeting Agenda Welcome and Announcements Marcia Smith, Office of Research Administration Contract
Research Administrators Forum October 10th, 2013 Marcia Smith Associate Vice Chancellor for Research Agenda Meeting Agenda Welcome and Announcements Marcia Smith, Office of Research Administration Contract
Sponsored Programs Roles & Responsibilities
 The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
Proposal Submission, Review and Acceptance
 Proposal Submission, Review and Acceptance October 2015 Peggy Bowe OGRD is now OROS Office of Research Operations and Support Recording date of this workshop is October 23, 2015 Some of the rules and procedures
Proposal Submission, Review and Acceptance October 2015 Peggy Bowe OGRD is now OROS Office of Research Operations and Support Recording date of this workshop is October 23, 2015 Some of the rules and procedures
Sponsored Programs Roles & Responsibilities
 The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
UNIFORM GUIDANCE UPDATE
 UNIFORM GUIDANCE UPDATE CINDY KIEL Executive Associate Vice Chancellor Office of Research Michael Allred Associate Vice Chancellor for Finance/Controller What is the Uniform Guidance? Uniform Administrative
UNIFORM GUIDANCE UPDATE CINDY KIEL Executive Associate Vice Chancellor Office of Research Michael Allred Associate Vice Chancellor for Finance/Controller What is the Uniform Guidance? Uniform Administrative
AURA Meeting. July 23, 2014
 AURA Meeting July 23, 2014 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored
AURA Meeting July 23, 2014 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored
Title: OFFICE OF SPONSORED PROGRAMS/IRB/ORIP COORDINATION
 SYRACUSE UNIVERSITY HUMAN RESEARCH PROTECTION PROGRAM STANDARD OPERATING PROCEDURES TITLE: OFFICE OF SPONSORED PROGRAMS/IRB/ORIP COORDINATION DOCUMENT NUMBER: 033 REVISION NUMBER REVISION DATE (SUPERSEDES
SYRACUSE UNIVERSITY HUMAN RESEARCH PROTECTION PROGRAM STANDARD OPERATING PROCEDURES TITLE: OFFICE OF SPONSORED PROGRAMS/IRB/ORIP COORDINATION DOCUMENT NUMBER: 033 REVISION NUMBER REVISION DATE (SUPERSEDES
Uniform Guidance Working Group
 Uniform Guidance Working Group Section: FDP Subaward Templates January 13, 2015 Agenda Working Group Charge Working Group Membership Process UG-related Changes Applicability of FDP Subaward Templates Working
Uniform Guidance Working Group Section: FDP Subaward Templates January 13, 2015 Agenda Working Group Charge Working Group Membership Process UG-related Changes Applicability of FDP Subaward Templates Working
USF HRPP Updates October 2017
 USF HRPP Updates October 2017 Julie Moore, J.D., M.S. PA, CIP Associate Director, Research Integrity & Compliance Research Integrity and Compliance Revised Common Rule 45 CFR 46 Effective date and compliance
USF HRPP Updates October 2017 Julie Moore, J.D., M.S. PA, CIP Associate Director, Research Integrity & Compliance Research Integrity and Compliance Revised Common Rule 45 CFR 46 Effective date and compliance
Administrative Burden of Research Compliance
 Administrative Burden of Research Compliance Measuring and Minimizing David L. Wynes, Ph.D. Vice President for Research Administration Emory University 1 FDP Faculty Burden Survey (X2) PIs estimated that
Administrative Burden of Research Compliance Measuring and Minimizing David L. Wynes, Ph.D. Vice President for Research Administration Emory University 1 FDP Faculty Burden Survey (X2) PIs estimated that
Research Administrators Forum August 14th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum August 14th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Associate Vice Chancellor, Office of Research Administration
Research Administrators Forum August 14th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Associate Vice Chancellor, Office of Research Administration
PEER (PAPERLESS ENVIRONMENT FOR ELECTRONIC REVIEW)
 PEER (PAPERLESS ENVIRONMENT FOR ELECTRONIC REVIEW) 1 What is PEER? PEER is a web portal maintained by Vanderbilt University which handles internal processing of grant related requests and contract submission
PEER (PAPERLESS ENVIRONMENT FOR ELECTRONIC REVIEW) 1 What is PEER? PEER is a web portal maintained by Vanderbilt University which handles internal processing of grant related requests and contract submission
October 30 th, :00PM EDT. Special Cases in Proposal Development: Large-Scale, Multidisciplinary and/or Multi-Organizational Proposals
 October 30 th, 2013 1:00PM EDT Large-Scale, Multidisciplinary and/or Introduction Questions: Please use the Q&A pod located at the bottom of your screen Audio: Participants can choose to use the microphone
October 30 th, 2013 1:00PM EDT Large-Scale, Multidisciplinary and/or Introduction Questions: Please use the Q&A pod located at the bottom of your screen Audio: Participants can choose to use the microphone
October electronic Research Administration (era) OER, OD, National Institutes of Health
 October 2016 electronic Research Administration (era) OER, OD, National Institutes of Health Scarlett Gibb era Customer Relationship Manager, era Commons Joe Schumaker era Communications and Outreach 2
October 2016 electronic Research Administration (era) OER, OD, National Institutes of Health Scarlett Gibb era Customer Relationship Manager, era Commons Joe Schumaker era Communications and Outreach 2
Proposal Preparation and Submission February 2018
 Proposal Preparation and Submission February 2018 Introductions We are. Chris Dye-Hixenbaugh, C&G Officer, Proposals Brooke Herevia, C&G Analyst, Proposals Shanna Nation Jose, C&G Analyst, Proposals Kassie
Proposal Preparation and Submission February 2018 Introductions We are. Chris Dye-Hixenbaugh, C&G Officer, Proposals Brooke Herevia, C&G Analyst, Proposals Shanna Nation Jose, C&G Analyst, Proposals Kassie
Outgoing Subaward Basics - Subawards Issued by UCLA under Extramurally Funded Grants/Cooperative Agreements
 Outgoing Subaward Basics - Subawards Issued by UCLA under Extramurally Funded Grants/Cooperative Agreements PREVIEW: Master Training (Mary Haskins, OCGA Subaward Officer) When: Wednesday, April 18, 2018
Outgoing Subaward Basics - Subawards Issued by UCLA under Extramurally Funded Grants/Cooperative Agreements PREVIEW: Master Training (Mary Haskins, OCGA Subaward Officer) When: Wednesday, April 18, 2018
FAQ S FOR UNIFORM GUIDANCE
 FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP)
 Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Complex Award Management Program Projects/Multi-Fund Awards- Post Award NUMBER: RAS SOP 2001 VERSION: 4.0
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Complex Award Management Program Projects/Multi-Fund Awards- Post Award NUMBER: RAS SOP 2001 VERSION: 4.0
Outgoing Subagreements: Subawards and Subcontracts
 Effective Date: March 1, 2014 Version: 1 Page: 1 of 6 Overview Sponsored programs may include partnerships with other organizations or institutions whereby a portion of the project is conducted by investigators
Effective Date: March 1, 2014 Version: 1 Page: 1 of 6 Overview Sponsored programs may include partnerships with other organizations or institutions whereby a portion of the project is conducted by investigators
Subrecipient Monitoring Under 2 CFR 200. Kris Rhodes, MS Director, Higher Education Practice
 Subrecipient Monitoring Under 2 CFR 200 Kris Rhodes, MS Director, Higher Education Practice krisrhodes@maximus.com 1 MAXIMUS Higher Education Practice Headquartered in Northbrook, IL Satellite Offices
Subrecipient Monitoring Under 2 CFR 200 Kris Rhodes, MS Director, Higher Education Practice krisrhodes@maximus.com 1 MAXIMUS Higher Education Practice Headquartered in Northbrook, IL Satellite Offices
Reducing Investigators Administrative Workload for Federally-Funded Research
 Reducing Investigators Administrative Workload for Federally-Funded Research National Science Board National Research Council Committee on Federal Research Regulations and Reporting Requirements February
Reducing Investigators Administrative Workload for Federally-Funded Research National Science Board National Research Council Committee on Federal Research Regulations and Reporting Requirements February
Sponsored Program Administration Meeting. September 2016
 Sponsored Program Administration Meeting September 2016 Agenda NCURA Internal Controls Mike Unplugged: Training/Education Specialist position Budgeting for Post doc increases FDP Update ETS Update Effort
Sponsored Program Administration Meeting September 2016 Agenda NCURA Internal Controls Mike Unplugged: Training/Education Specialist position Budgeting for Post doc increases FDP Update ETS Update Effort
NURAP: COI Update. New edisclosure System. January 21, 2016 Julia Campbell Director, Conflict of Interest
 NURAP: COI Update New edisclosure System January 21, 2016 Julia Campbell Director, Conflict of Interest Topics for Discussion Overview of new system Impacts on research-related processes Timeline Training
NURAP: COI Update New edisclosure System January 21, 2016 Julia Campbell Director, Conflict of Interest Topics for Discussion Overview of new system Impacts on research-related processes Timeline Training
Clinical Investigator Career Development Award ( )
 Clinical Investigator Career Development Award (2019-2022) Guidelines and General Instructions for Application KEY DATES Application Release Date: May 30, 2018 Application Deadline: September 6, 2018 at
Clinical Investigator Career Development Award (2019-2022) Guidelines and General Instructions for Application KEY DATES Application Release Date: May 30, 2018 Application Deadline: September 6, 2018 at
Recent Legislative Actions Taken to Reduce Research Regulatory Burden. 21st Century Cures (Passed House and Senate. Signed into law Dec.
 . Signed into law Dec. 13) Link to PDF Link to PDF Link to PDF Research Policy Board - A public-private entity recommended by the National Academies "to foster more effective conception, development and
. Signed into law Dec. 13) Link to PDF Link to PDF Link to PDF Research Policy Board - A public-private entity recommended by the National Academies "to foster more effective conception, development and
TRIAL INNOVATION NETWORK Key Terminology and Definitions
 Organization of the Trial Innovation Network Trial Innovation Network Key organizational components include the TICs, RIC, and CTSA Program Hubs All are key partners of the Trial Innovation Network and
Organization of the Trial Innovation Network Trial Innovation Network Key organizational components include the TICs, RIC, and CTSA Program Hubs All are key partners of the Trial Innovation Network and
Outgoing Subaward Guide and F-A-Qs
 1. What is a subaward? Outgoing Subaward Guide and F-A-Qs 2. After an award is made to Rutgers, how is a subaward established? 3. What is the outgoing subaward process? 4. How can a subaward be changed/amended?
1. What is a subaward? Outgoing Subaward Guide and F-A-Qs 2. After an award is made to Rutgers, how is a subaward established? 3. What is the outgoing subaward process? 4. How can a subaward be changed/amended?
Step Two: Download & complete the Grants.gov application
 Instructions: This guide will walk you through the creation and submission of a NIH proposal through Grants.gov. In the mygrant section we will guide you through all the required fields. There are many
Instructions: This guide will walk you through the creation and submission of a NIH proposal through Grants.gov. In the mygrant section we will guide you through all the required fields. There are many
Help Is on the Way: The Federal Demonstration Partnership and Emerging Research Institutions
 Help Is on the Way: The Federal Demonstration Partnership and Emerging Research Institutions David Wright Executive Director Federal Demonstration Partnership (FDP) Federal Demonstration Partnership (FDP)
Help Is on the Way: The Federal Demonstration Partnership and Emerging Research Institutions David Wright Executive Director Federal Demonstration Partnership (FDP) Federal Demonstration Partnership (FDP)
Funding Focus: The New NIH Biosketch. Presenter: Rachel Dresbeck Date: June 19, 2014
 Funding Focus: The New NIH Biosketch Presenter: Rachel Dresbeck Date: June 19, 2014 New Biosketch Pilot phase 5 pages Allows you to describe up to five of your most significant contributions to science
Funding Focus: The New NIH Biosketch Presenter: Rachel Dresbeck Date: June 19, 2014 New Biosketch Pilot phase 5 pages Allows you to describe up to five of your most significant contributions to science
Office of Sponsored Projects 5/31/ Volume 2, Issue 5
 The OSP News & Updates, published by the Office of Sponsored Projects, is a bi-weekly subscription-based newsletter that provides OSP and sponsor updates and reminders, quick facts, guidance and training
The OSP News & Updates, published by the Office of Sponsored Projects, is a bi-weekly subscription-based newsletter that provides OSP and sponsor updates and reminders, quick facts, guidance and training
Policy and Compliance: Working Together Like Hand in Glove
 Policy and Compliance: Working Together Like Hand in Glove Samuel Ashe, Director, Division of Grants Policy, OPERA, OER Diane W. Dean, Director, Division of Grants Compliance and Oversight, OPERA, OER
Policy and Compliance: Working Together Like Hand in Glove Samuel Ashe, Director, Division of Grants Policy, OPERA, OER Diane W. Dean, Director, Division of Grants Compliance and Oversight, OPERA, OER
Navigating Grant Resources. 10/3/2018 Dr. Susan Fell
 Navigating Grant Resources 10/3/2018 Dr. Susan Fell sfell@fsu.edu 850-645-2172 Sponsored Research Administration https://www.research.fsu.edu/ Applying for a Grant Opportunity Does this require sign-off
Navigating Grant Resources 10/3/2018 Dr. Susan Fell sfell@fsu.edu 850-645-2172 Sponsored Research Administration https://www.research.fsu.edu/ Applying for a Grant Opportunity Does this require sign-off
Clinical Investigator Career Development Award ( )
 Clinical Investigator Career Development Award (2018-2021) Guidelines and General Instructions for Application KEY DATES Application Release Date: May 24, 2017 Application Deadline: September 6, 2017 at
Clinical Investigator Career Development Award (2018-2021) Guidelines and General Instructions for Application KEY DATES Application Release Date: May 24, 2017 Application Deadline: September 6, 2017 at
CAYUSE Research Suite
 CAYUSE Research Suite Internal Clearance & Submission Process Principal Investigator/Program Director Cayuse SP is used by ORSP to process Internal Clearance and when applicable to submit a proposal for
CAYUSE Research Suite Internal Clearance & Submission Process Principal Investigator/Program Director Cayuse SP is used by ORSP to process Internal Clearance and when applicable to submit a proposal for
OSP Subaward Request Process. To utilize the system to initiate and complete a request, follow the steps below.
 Effective Date: March 1, 2014 Version: 1 Page: 1 of 13 Overview All Sponsored Awards, whether grants, contracts or cooperative agreements, which are received by Emory and which involve participation by
Effective Date: March 1, 2014 Version: 1 Page: 1 of 13 Overview All Sponsored Awards, whether grants, contracts or cooperative agreements, which are received by Emory and which involve participation by
Quickguide to Creating a Proposal in Kuali Coeus
 Proposal Tab v.1 Revised 7/18/2014 Panel Field Action Notes Proposal Development Guide Reference Page(s) Enter a description of the proposal in this format: Document Overview Description Due Date xx/xx/xx_pi
Proposal Tab v.1 Revised 7/18/2014 Panel Field Action Notes Proposal Development Guide Reference Page(s) Enter a description of the proposal in this format: Document Overview Description Due Date xx/xx/xx_pi
Pre Award Table of Contents
 Pre Award Table of Contents ORA Internal Procedures for Account Establishment 2 Conflict of Interest: Thomas Jefferson University Conflict of Interest (COI) Policy 4 Cost Sharing and Matching Funds 6 Developing
Pre Award Table of Contents ORA Internal Procedures for Account Establishment 2 Conflict of Interest: Thomas Jefferson University Conflict of Interest (COI) Policy 4 Cost Sharing and Matching Funds 6 Developing
Narration: Welcome to the Anatomy of an Administrative Shell mini course.
 Welcome to the Anatomy of an Administrative Shell mini course. 1 If you have previously joined us for other sponsored project mini courses, you will be familiar with the Sponsored Project Life Cycle. In
Welcome to the Anatomy of an Administrative Shell mini course. 1 If you have previously joined us for other sponsored project mini courses, you will be familiar with the Sponsored Project Life Cycle. In
Trigger / Timing / Frequency: When a new award is received by the University and OSP determines that the award can be accepted.
 Kuali Research User Guide: Create a New Parent Award Version October 06 Purpose: To create a new parent award record in the system. Trigger / Timing / Frequency: When a new award is received by the University
Kuali Research User Guide: Create a New Parent Award Version October 06 Purpose: To create a new parent award record in the system. Trigger / Timing / Frequency: When a new award is received by the University
MAXIMUS Higher Education Practice
 Federal Compliance Webinar Series 2 CFR 200 Uniform Guidance Kris Rhodes, Director, MAXIMUS Jason Guilbeault, Senior Consultant, MAXIMUS 1 MAXIMUS Higher Education Practice Headquartered in Northbrook,
Federal Compliance Webinar Series 2 CFR 200 Uniform Guidance Kris Rhodes, Director, MAXIMUS Jason Guilbeault, Senior Consultant, MAXIMUS 1 MAXIMUS Higher Education Practice Headquartered in Northbrook,
Milestones. RFAs announced November 29, Letter of intent due January 31, Application due March 30, Award announcement June 1, 2018
 Milestones RFAs announced November 29, 2017 Letter of intent due January 31, 2018 Application due March 30, 2018 Award announcement June 1, 2018 Funds made available (pending IRB approval) July 2, 2018
Milestones RFAs announced November 29, 2017 Letter of intent due January 31, 2018 Application due March 30, 2018 Award announcement June 1, 2018 Funds made available (pending IRB approval) July 2, 2018
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards. AU SPAN Martha Taylor Larry Hankins
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards AU SPAN 6 23 2014 Martha Taylor Larry Hankins Grants Reform February 2011 President directed OMB to reduce
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards AU SPAN 6 23 2014 Martha Taylor Larry Hankins Grants Reform February 2011 President directed OMB to reduce
D. PROPOSAL DETAILS CREATE A NEW PROPOSAL GENERAL INFO ORGANIZATION ADD INVESTIGATORS AND KEY PERSONS CREDIT SPLIT SPECIAL REVIEW D.3.
 D. PROPOSAL DETAILS D. D. D.3. D.4. D.5. D.6. D.7. D.8. D.9. D.10. D.1 D.1 CREATE A NEW PROPOSAL GENERAL INFO ORGANIZATION ADD INVESTIGATORS AND KEY PERSONS CREDIT SPLIT SPECIAL REVIEW ABSTRACT OTHER YNQ
D. PROPOSAL DETAILS D. D. D.3. D.4. D.5. D.6. D.7. D.8. D.9. D.10. D.1 D.1 CREATE A NEW PROPOSAL GENERAL INFO ORGANIZATION ADD INVESTIGATORS AND KEY PERSONS CREDIT SPLIT SPECIAL REVIEW ABSTRACT OTHER YNQ
Page 1 of 10 1/31/2008
 Page 1 of 10 1/31/2008 Step by Step Tips to Avoid Errors and Warnings o Full instructions can be found at: http://grants1.nih.gov/grants/funding/424/index.htm. o This document is intended to assist you
Page 1 of 10 1/31/2008 Step by Step Tips to Avoid Errors and Warnings o Full instructions can be found at: http://grants1.nih.gov/grants/funding/424/index.htm. o This document is intended to assist you
Research Administration Updates. May 16, :00 am IOP Auditorium
 Research Administration Updates May 16, 2018 11:00 am IOP Auditorium Topics Covered Using ClinCard (Greenphire) for Federally Sponsored Clinical Research Studies Ryan Mulligan, SCTR Institute OCR & ORSP
Research Administration Updates May 16, 2018 11:00 am IOP Auditorium Topics Covered Using ClinCard (Greenphire) for Federally Sponsored Clinical Research Studies Ryan Mulligan, SCTR Institute OCR & ORSP
Finding Funding, Budget Preparation, and Proposal Submission for Sponsored Research
 Finding Funding, Budget Preparation, and Proposal Submission for Sponsored Research Laura Johnston, Office of Sponsored Programs, Asst. Director Craig O Neill, Office of Sponsored Programs, Manager February
Finding Funding, Budget Preparation, and Proposal Submission for Sponsored Research Laura Johnston, Office of Sponsored Programs, Asst. Director Craig O Neill, Office of Sponsored Programs, Manager February
Costing and Procurement Updates
 Costing and Procurement Updates Jim Luther, Duke University Edwin Bemmel, University of Miami Doug Backman, University of South Florida Sara Bible, Stanford University January 8, 2018 Agenda Update (Costing
Costing and Procurement Updates Jim Luther, Duke University Edwin Bemmel, University of Miami Doug Backman, University of South Florida Sara Bible, Stanford University January 8, 2018 Agenda Update (Costing
Responsible Conduct of Research. Information Session March 2, 2011 Summary
 Responsible Conduct of Research Information Session March 2, 2011 Summary Outline What is RCR? Why is Cornell Requiring It? NSF Requirements NIH Requirements Responsibilities ORIA, OSP, Units Resources
Responsible Conduct of Research Information Session March 2, 2011 Summary Outline What is RCR? Why is Cornell Requiring It? NSF Requirements NIH Requirements Responsibilities ORIA, OSP, Units Resources
Welcome to RAIN! Research Administration Information Network
 Welcome to RAIN! Research Administration Information Network DATE: March 17, 2016 HOSTED BY: Andrew Watson RAIN Agenda with Andrew Watson Introduction Updates and timely information Special guest: CFS
Welcome to RAIN! Research Administration Information Network DATE: March 17, 2016 HOSTED BY: Andrew Watson RAIN Agenda with Andrew Watson Introduction Updates and timely information Special guest: CFS
CCSG 101. Teresa Christiansen John Gricoski Dr. Robert Houlihan Robert Mitchell Lowell Smith
 CCSG 101 Teresa Christiansen John Gricoski Dr. Robert Houlihan Robert Mitchell Lowell Smith Characteristics Organizational Type Research Conducted Description Comprehensive, Free-Standing Temple University
CCSG 101 Teresa Christiansen John Gricoski Dr. Robert Houlihan Robert Mitchell Lowell Smith Characteristics Organizational Type Research Conducted Description Comprehensive, Free-Standing Temple University
UNIFORM GUIDANCE OVERVIEW. Budget Officers Meeting January 28, 2015
 UNIFORM GUIDANCE OVERVIEW Budget Officers Meeting January 28, 2015 OMB Circulars Before and After Eight circulars condensed to one new comprehensive set of requirements for recipients of federal awards
UNIFORM GUIDANCE OVERVIEW Budget Officers Meeting January 28, 2015 OMB Circulars Before and After Eight circulars condensed to one new comprehensive set of requirements for recipients of federal awards
CCSG 101 4/29/2017. Teresa Christiansen John Gricoski Dr. Robert Houlihan Robert Mitchell Lowell Smith. Guidelines: Timelines / Internal Review
 Teresa Christiansen John Gricoski Dr. Robert Houlihan Robert Mitchell Lowell Smith Characteristics Organizational Type Research Conducted Description Comprehensive, Free-Standing Temple University Health
Teresa Christiansen John Gricoski Dr. Robert Houlihan Robert Mitchell Lowell Smith Characteristics Organizational Type Research Conducted Description Comprehensive, Free-Standing Temple University Health
Medicare and Medicaid Programs: Electronic Health Record Incentive Program -- Stage 3 and Modifications to Meaningful Use in 2015 through 2017
 Medicare and Medicaid Programs: Electronic Health Record Incentive Program -- Stage 3 and Modifications to Meaningful Use in 2015 through 2017 and 2015 Edition Health Information Technology Certification
Medicare and Medicaid Programs: Electronic Health Record Incentive Program -- Stage 3 and Modifications to Meaningful Use in 2015 through 2017 and 2015 Edition Health Information Technology Certification
Results of the 2012 Faculty Workload Survey (FWS)
 Original PowerPoint slides available upon request at fdp@nas.edu Results of the 2012 Faculty Workload Survey (FWS) Sandra L. Schneider, Ph.D. FDP Vice Chair; FWS PI and Task Force Chair University of South
Original PowerPoint slides available upon request at fdp@nas.edu Results of the 2012 Faculty Workload Survey (FWS) Sandra L. Schneider, Ph.D. FDP Vice Chair; FWS PI and Task Force Chair University of South
NIH ASSIST Multi-Component NIH Submissions Updated: August 7, 2018
 NIH ASSIST Multi-Component NIH Submissions Updated: August 7, 2018 Training Outline Class Overview Multi-Component NIH Submission Definition Submitting a Multi-Component Application to NIH Resources Learning
NIH ASSIST Multi-Component NIH Submissions Updated: August 7, 2018 Training Outline Class Overview Multi-Component NIH Submission Definition Submitting a Multi-Component Application to NIH Resources Learning
RAS What s New for Grants?
 Finance Division Compass RAS What s New for Grants? Welcome & Introductions About me What have you heard about 9.2? 2 Agenda Ground Rules Course Objectives Value of Compass 9.2 Pre-Award changes Award
Finance Division Compass RAS What s New for Grants? Welcome & Introductions About me What have you heard about 9.2? 2 Agenda Ground Rules Course Objectives Value of Compass 9.2 Pre-Award changes Award
Proposal Development Guide
 Proposal Development Guide Contents The Proposal Tab... 3 Document Overview Panel... 3 Required Fields for Saving Document Panel... 3 Institutional Fields (Conditionally Required)... 8 Sponsor & Program
Proposal Development Guide Contents The Proposal Tab... 3 Document Overview Panel... 3 Required Fields for Saving Document Panel... 3 Institutional Fields (Conditionally Required)... 8 Sponsor & Program
SYSTEM REQUIREMENTS AND USEFUL INFORMATION LOGGING INTO THE PERIS PORTAL
 SYSTEM REQUIREMENTS AND USEFUL INFORMATION ------------------------------------------------- LOGGING INTO THE PERIS PORTAL -------------------------------------------------------------------------- CREATING
SYSTEM REQUIREMENTS AND USEFUL INFORMATION ------------------------------------------------- LOGGING INTO THE PERIS PORTAL -------------------------------------------------------------------------- CREATING
Subawards and Subrecipient Monitoring
 Subawards and Subrecipient Monitoring H O L LY S O M M E R S D I R E C T O R, O S P G R A N T S J O S H R O S E N B E R G A C T I N G D I R E C T O R, O G C A Objectives 2 Distinguish Between: Procurement
Subawards and Subrecipient Monitoring H O L LY S O M M E R S D I R E C T O R, O S P G R A N T S J O S H R O S E N B E R G A C T I N G D I R E C T O R, O G C A Objectives 2 Distinguish Between: Procurement
University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures
 Summary 1. Subaward Definitions A. Subaward B. Subrecipient University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures C. Office of Contracts and Grants (OCG) 2. Distinguishing
Summary 1. Subaward Definitions A. Subaward B. Subrecipient University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures C. Office of Contracts and Grants (OCG) 2. Distinguishing
SUBRECIPIENT COMMITMENT FORM
 SECTION A SUBRECIPIENT INFORMATION SUBRECIPIENT COMMITMENT FORM Phone: Email: SUBRECIPIENT: CU PI: PROPOSAL TITLE: TOTAL FUNDING REQUESTED: ORIGINATING SPONSOR, IF A PASS THRU APPLICATION: PRIME SPONSOR:
SECTION A SUBRECIPIENT INFORMATION SUBRECIPIENT COMMITMENT FORM Phone: Email: SUBRECIPIENT: CU PI: PROPOSAL TITLE: TOTAL FUNDING REQUESTED: ORIGINATING SPONSOR, IF A PASS THRU APPLICATION: PRIME SPONSOR:
