Office of Sponsored Projects 5/31/ Volume 2, Issue 5
|
|
|
- Martin Dean
- 5 years ago
- Views:
Transcription
1 The OSP News & Updates, published by the Office of Sponsored Projects, is a bi-weekly subscription-based newsletter that provides OSP and sponsor updates and reminders, quick facts, guidance and training in all aspects of sponsored projects administration. To subscribe, visit i Table of Contents 1. New Workday Reports 1.1. Sponsored Award Recent Setups Yale 1.2. Grant Validator Yale 2. Presentation Hierarchy: Role Assignment to be Released in Workday 3. Registration is Open for June Brown Bag Session 4. Effort Reporting Principles Training 5. Upcoming OSP Communications and Training Opportunities 6. Sponsor-related Updates & Reminders 6.1. National Institutes of Health (NIH) Notice of Fiscal Policies in Effect for FY era Information: Make Sure to Update RPPR and Inclusion Data by June 8, 2018, Before IMS Retirement on June Notice of Legislative Mandates in Effect for FY National Science Foundation (NSF) Research.gov Now Accepts Proposal Submission 1 NEW WORKDAY REPORTS 1.1 SPONSORED AWARD RECENT SETUPS YALE For users who need to monitor Award setups, this report allows monitoring of Award modifications without getting notifications or to do items in Workday. It is not necessary to have the Grant Manager or 1 P age
2 Grant Budget Specialist role in order to run the report; however, you will need the Cost Center Financial Analyst role. Type Sponsored Award Recent Setups Yale into the search box to launch the report. Results will show original setup completion date, amendment date and reason, and correction date and comment for any Awards with setup, amendment, or correction dates in the 60 days prior to the date specified when running the report. 1.2 GRANT VALIDATOR YALE The Cost Center Financial Analyst role has been updated. Analysts with this role can now validate accurate grant start and end dates outside their cost center. Previously, users were unable to see start and end dates of grants associated with an award outside of their cost center. Type Grant Date Validator Yale into the search box to launch the report. 2 PRESENTATION HIERARCHY: ROLE ASSIGNMENT TO BE RELEASED IN WORKDAY Coming in June 2018, distributed users will be able to assign Grant Manager and Grant Budget Specialist roles using Workday s Assign Roles Business Process. To appropriately assign the roles, it is necessary to understand what Grant hierarchy does and how it s built. Please join us as we present what the Grant Hierarchy does and its basic structure. Date: June 07, 2018 from 2:00-3:00 p.m. via Zoom. Register for Grant Hierarchy presentation. 3 REGISTRATION IS OPEN FOR JUNE BROWN BAG SESSION Please join the June Brown Bag session hosted by the Office of Sponsored Projects on June 13 th. This session will be held at 12:30pm in 25 Science Park, Room 125 and can be attended remotely via Zoom. Topics for this session include: OSP Staff Updates OSP Reminders o Acknowledgement of Busy Season 2 P age
3 o Before you call, where to look for status Export Controls International Operations and Compliance Committee Update on PD for Clinical Trials Yale Continuous Improvements o Supplier Contracts o Subaward Invoicing o Subaward Budget Date o Award Closeout Visit the University s Training Management System (TMS) to view topics and register for the session. 4 EFFORT REPORTING PRINCIPLES TRAINING The Office of Sponsored Projects encourages you to register for an upcoming Effort Reporting Principles instructor-led training class. With the implementation of Workday (July 2017), the training presentation for Effort Reporting Principles has been updated to include an overview of the Effort Certification process in Workday, including administrative review and certification. Participants will be provided with a more comprehensive understanding of effort as it relates to sponsored projects. Topics covered include: Key terms, policies, procedures and regulations Effort management activities from proposal through closeout Key roles and responsibilities The effort reporting lifecycle, with case studies An overview of the effort certification process in Workday There will be a short break between Effort Reporting Principles and the overview of the Effort Certification process in Workday. Participants are welcome to depart during the break if the Workday Effort Certification process overview is not relevant. Visit the University s Training Management System (TMS) to view event times, locations and register for a session. 3 P age
4 5 UPCOMING OSP COMMUNICATIONS AND TRAINING OPPORTUNITIES JUNE 2018 MONDAY TUESDAY WEDNESDAY THURSDAY FRIDAY :30am 5:00pm Intro to SPA 25 Science Park, :30-4:30 Effort Reporting Principles 47 College, 206A 13 12:30pm OSP Brown Bag 25 Science Park, OSP News & Updates, Vol. 2, Iss :00pm Proposal Tracking (PT) Overview Training 25 Science Park, 371A OSP News & Updates, Vol. 2, Iss JULY 2018 MONDAY TUESDAY WEDNESDAY THURSDAY FRIDAY :30am 5:00pm Intro to SPA 25 Science Park, OSP News & Updates, Vol. 3, Iss :00pm 5:00pm Proposal Development (PD) 25 Science Park, 321 (Day 1 of 2) 26 1:00pm 5:00pm Proposal Development (PD) 25 Science Park, 321 (Day 2 of 2) OSP News & Updates, Vol. 3, Iss P age
5 6 SPONSOR-RELATED UPDATES & REMINDERS 6.1 NATIONAL INSTITUTES OF HEALTH (NIH) Notice of Fiscal Policies in Effect for FY 2018 NIH Notice Number: NOT-OD provided guidance about the NIH Fiscal Operations for FY 2018 and implements the Consolidated Appropriations Act, 2018 (Public Law ), signed into law on March 23, With the passage of the Act, NIH received a 9 percent increase over FY 2017, for a total of $ billion in program level funding, including $496,000,000 authorized under the 21st Century Cures Act. A general increase of over 5 percent was received, as well as specific increases for Opioid/Pain research, Alzheimer s disease, and other high priority research initiatives. The following NIH fiscal policies are instituted in FY 2018: FY 2018 Funding Levels: Non-competing continuation awards that have already been made in FY 2018 were generally funded at levels below that indicated on the most recent Notice of Award (generally up to 90% of the previously committed level) as described in NOT-OD In general, such reductions will be fully restored, and non-competing continuation grants (research and non-research) including those that remain to be issued in FY 2018 will be made at the commitment level indicated on the Notice of Award. Any exceptions will be posted at the site listed under "Additional Information" below. Out-year commitments for continuation awards in FY 2019 and beyond will remain unchanged. The NIH awarding Institutes/Centers (IC) will develop and post their fiscal policies consistent with overall NIH goals and available FY 2018 funds. Ruth L. Kirschstein National Research Service Awards (NRSA): Consistent with the recommendations of the Advisory Committee to the Director regarding the Biomedical Research Workforce, the NIH will increase NRSA stipends by approximately 2 percent on average. The full range of stipend adjustments for FY 2018 is described at NOT-OD Next Generation Researchers Initiative Policy: NIH will prioritize meritorious R01- equivalent applications from ESI PD/PIs. By providing funding priority for ESIs, NIH intends to encourage funding applications that involve researchers earlier in their career in accordance with the policy established in FY 2017 and described at 5 P age
6 Salary Limits: Section 202 of the Consolidated Appropriations Act, 2018 prohibits payments for salaries under grants and other extramural mechanisms in excess of Executive Level II previously set at $187,000, and effective January 7, 2018, increased to $189,600. See NOT-OD for additional information. Other Legislative Mandates: Other statutory requirements are described in NOT-OD Additional Information: Additional details on Fiscal Operations, including specific funding strategies for ICs will be posted at era Information: Make Sure to Update RPPR and Inclusion Data by June 8, 2018, Before IMS Retirement on June 9 If an RPPR is in progress on or before June 8, inclusion data cannot be submitted separately in IMS on or before June 8. In this situation, the entire RPPR must be submitted on or before June 8, or If the RPPR is submitted on or after June 9, any study data entered on or before June 8 in IMS must be reentered in HSS If inclusion data has not been completed in IMS on or before June 8: PI will need to reenter the inclusion data in HSS on or after June Notice of Legislative Mandates in Effect for FY 2018 NIH Notice Number: NOT-OD provides the current requirements outlined in the following statutory provisions that limits or conditions the use of funds on NIH grant, cooperative agreement, and contract awards for FY FY 2018 Legislative Mandates that remain in effect are as follows: Division H, Title II - Department of Health and Human Services Appropriations Act, P age
7 NEW: This title included a new requirement of two NIH Institutes that for-profit recipients of funds provided for research related to opioid addiction, development of opioid alternatives, pain management, and addiction treatment contribute a match of funds or documented in-kind contributions of not less than 50 percent of the total funds awarded to such entity. (1) Salary Limitation (Section 202) (2) Gun Control (Section 210) Division H, Title V - Departments of Labor, Health and Human Services, and Education, and Related Agencies Appropriations Act, 2018 (3) Anti-Lobbying (Section 503) (4) Acknowledgment of Federal Funding (Section 505) (5) Restriction on Abortions (Section 506) (6) Exceptions to Restriction on Abortions (Section 507) (7) Ban on Funding Human Embryo Research (Section 508) (8) Limitation on Use of Funds for Promotion of Legalization of Controlled Substances (Section 509) (9) Restriction on disclosure of political affiliation for Federal scientific advisory committee candidates (Section 515(a)) (10)Dissemination of False or Misleading Information (Section (515(b)) (11) Restriction on Distribution of Sterile Needles (Section 520) (12) Restriction of Pornography on Computer Networks (Section 521) Details: New: National Institute of Neurological Disorders and Stroke For carrying out section 301 and title IV of the PHS Act with respect to neurological disorders and stroke, $2,145,149,000: Provided, That $250,000,000 shall be available until September 30, 2019 for research related to opioid addiction, development of opioid alternatives, pain management, and addiction treatment: Provided further, That each for-profit recipient of funds provided in the previous proviso shall be subject to a matching requirement of funds or documented in-kind contributions of not less than 50 percent of the total funds awarded to such entity. New: National Institute on Drug Abuse For carrying out section 301 and title IV of the PHS Act with respect to drug abuse, $1,383,603,000: Provided, That $250,000,000 shall be available until September 30, 2019 for research related to opioid addiction, development of opioid alternatives, pain management, and addiction treatment: Provided further, That each for-profit recipient of funds provided in the previous proviso shall 7 P age
8 be subject to a matching requirement of funds or documented in-kind contributions of not less than 50 percent of the total funds awarded to such entity. 6.2 NATIONAL SCIENCE FOUNDATION (NSF) Research.gov Now Accepts Proposal Submission As of April 30, nonfederal entities submitting research proposals for awards from the National Science Foundation (NSF) can prepare and submit noncollaborative proposals in Research.gov rather than the former FastLane system. At a recent Federal Demonstration Partnership (FDP) meeting in Washington, D.C., Jean Feldman, head of the policy office in the NSF Division of Institution and Award Support told attendees that NSF has been working since federal fiscal year 2016 to enable the submission of research proposals via Research.gov. From February to April, NSF allowed users to preview the functionality at Research.gov and test the preparation of proposals (see Research.gov Soon To Offer Proposal Capabilities, February 2018). It now has made the site fully functional. Some of the new proposal preparation capabilities include compliance checks for page formatting, project summary and project description heading, and a completely redesigned display of the budget. The Research.gov site advises users to note that as of April 30, the Proposal Status page in FastLane is no longer accessible. Instead, proposers and NSF staff are redirected to Research.gov to view a proposal s status. Feldman added that separately submitted collaborative proposals, as well as collaborative proposals with subawards, cannot be submitted via Research.gov. NSF aims to continue to improve the Research.gov system and add functionality over time for other types of proposals. Meanwhile, FastLane will continue to operate for all types of proposals for now. International Collaboration Feldman reminded FDP attendees about updated provisions in the NSF Proposal and Award Policies and Procedures Guide (PAPPG) that became effective in January concerning the eligibility of foreign organizations in funding proposals. The PAPPG now states that if the proposal includes funding to be provided to an international branch campus of a U.S. institution of higher education (IHE) (including through use of subawards and consultant arrangements), the proposer must explain the benefit(s) to the 8 P age
9 project of performance at the international branch campus, and justify why the project activities cannot be performed at the U.S. campus. Further, NSF policy now states that in cases where the proposing institution considers the foreign organization s involvement to be essential to the project, the institution must explain why local support is not feasible and why the foreign organization can carry out the activity more effectively. The proposed activity also must demonstrate how one or more of the following conditions have been met: the foreign organization contributes a unique organization, facilities, geographic location and/or access to unique data resources not generally available to U.S. investigators (or which would require significant effort or time to duplicate) or other resources that are essential to the success of the proposed project; and/or the foreign organization to be supported offers significant science and engineering education, training or research opportunities to the U.S. This is not a deviation from where NSF has been going for many years, Feldman said. We absolutely support collaboration. This is just a reminder that for any proposal with an international branch campus, expect there to be an explanation as to the benefit to the project and a justification as to why this can t be conducted at a U.S. campus. This will be checked by reviewers and program officers so take this seriously when you are doing it. She added that future versions of the PAPPG will provide more instructions related to this topic. Research Terms and Conditions Feldman also said that grants now must incorporate the NSF Research Terms and Conditions dated March 14, 2017, and the related NSF Agency Specific Requirements, which have been revised and became effective May 14. The NSF Agency Specific Requirements contain the following significant changes from the previous version: Article 25, Equipment, now aligns with the equipment provision in the uniform grant guidance at for all equipment purchased under NSF awards, and provides clarity on reporting requirements for NSF-owned equipment. Article 26, Intangible Property, now implements governmentwide changes to the Department of Commerce Bayh-Dole regulations at 37 C.F.R Article 27, Copyright Ownership, Government License, was removed from the intangible property article and has been added as a standalone article. The content of the article has not changed. 9 P age
10 Lastly, Feldman explained that NSF has released for 60 days of public comments a draft of the revised PAPPG that would potentially become effective in January Changes that would be made to the current PAPPG are highlighted in yellow. She said, Please take advantage of the fact that we are one of the few agencies that gives an opportunity for you to tell us about unexpected consequences or if something is not clear. For More Information The NSF Research Terms and Conditions and NSF Agency Specific Requirements are available at The draft 2019 PAPPG is available at i Thank you to all who have contributed to this newsletter. Questions about this newsletter should be directed to osp.communications@yale.edu. To unsubscribe, visit For archived issues, visit OSP News & Updates archives. 10 P age
Sponsored Programs New Developments and Important Reminders
 Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs January 23, 2018 1 NSF Updates NIH Updates Federal Updates Defense Base Act (DBA) Insurance OSP Updates
Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs January 23, 2018 1 NSF Updates NIH Updates Federal Updates Defense Base Act (DBA) Insurance OSP Updates
Sponsored Program Administration. October 10, 2017
 Sponsored Program Administration October 10, 2017 Agenda Procurement Services FDP Updates Jaggaer Pre-Award Federal Agency Updates Grants.gov Transition to Workspace NIH FORMS-E Post-Award GONE Act Certificate
Sponsored Program Administration October 10, 2017 Agenda Procurement Services FDP Updates Jaggaer Pre-Award Federal Agency Updates Grants.gov Transition to Workspace NIH FORMS-E Post-Award GONE Act Certificate
Federal Demonstration Partnership, NIH Updates, & Grants/Cooperative Agreement Reminders November 13, 2015
 Federal Demonstration Partnership, NIH Updates, & Grants/Cooperative Agreement Reminders November 13, 2015 Federal Demonstration Partnership BACKGROUND 154 participating institutions Goal: Improve Government
Federal Demonstration Partnership, NIH Updates, & Grants/Cooperative Agreement Reminders November 13, 2015 Federal Demonstration Partnership BACKGROUND 154 participating institutions Goal: Improve Government
Presentation. Title. Presented Name. Office of Policy for Extramural Research Administration, OER, NIH. Title. Office
 Presentation Title Michelle Presented G. Bulls, By: Director Name Office of Policy for Extramural Research Administration, OER, NIH Title Office Federal Demonstration Partnership, January 2015 NIH is currently
Presentation Title Michelle Presented G. Bulls, By: Director Name Office of Policy for Extramural Research Administration, OER, NIH Title Office Federal Demonstration Partnership, January 2015 NIH is currently
2017 NIH Update. Presented by Stephanie Smith and Stacey Wade
 2017 NIH Update Presented by Stephanie Smith and Stacey Wade PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs NIH INSTITUTES National
2017 NIH Update Presented by Stephanie Smith and Stacey Wade PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs NIH INSTITUTES National
Presentation. Title. Presented Name. Office of Policy for Extramural Research Administration, OER, NIH. Title. Office
 Presentation Title Michelle Presented G. Bulls, By: Director Name Office of Policy for Extramural Research Administration, OER, NIH Title Office Federal Demonstration Partnership, May 2015 NIH is currently
Presentation Title Michelle Presented G. Bulls, By: Director Name Office of Policy for Extramural Research Administration, OER, NIH Title Office Federal Demonstration Partnership, May 2015 NIH is currently
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018
 CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
Today s Topics. OCGA Quick Updates Kim Duiker and Heather Winters Updates from NIH Regional Seminar Kim Duiker S2S Grants Updates Cindy Gilbert
 August 8, 2013 Today s Topics OCGA Quick Updates Kim Duiker and Heather Winters Updates from NIH Regional Seminar Kim Duiker S2S Grants Updates Cindy Gilbert Quick Updates OCGA Staffing Update Three retirements
August 8, 2013 Today s Topics OCGA Quick Updates Kim Duiker and Heather Winters Updates from NIH Regional Seminar Kim Duiker S2S Grants Updates Cindy Gilbert Quick Updates OCGA Staffing Update Three retirements
office of research administration newsletter
 july/august 2010 vol. 5 no. 1 office of research administration newsletter Yale has a clear obligation to comply with all regulations pertaining to the administration of federal grants, and we will spare
july/august 2010 vol. 5 no. 1 office of research administration newsletter Yale has a clear obligation to comply with all regulations pertaining to the administration of federal grants, and we will spare
The Uniform Guidance (2 CFR, Part 200)
 WCMC Implementation of The Uniform Guidance (2 CFR, Part 200) Tuesday, September 22, 2015 & Wednesday, September 23, 2015 UG Workshop Michelle A. Lewis, M.S. Director of Research Administration Interim
WCMC Implementation of The Uniform Guidance (2 CFR, Part 200) Tuesday, September 22, 2015 & Wednesday, September 23, 2015 UG Workshop Michelle A. Lewis, M.S. Director of Research Administration Interim
NIH Update. Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH. FDP January 11, 2016
 NIH Update Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH FDP January 11, 2016 1 2 NIH FY 2016 Budget News NIH is currently funded under the Consolidated
NIH Update Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH FDP January 11, 2016 1 2 NIH FY 2016 Budget News NIH is currently funded under the Consolidated
Patti Manheim - Director February 13, 2014
 Patti Manheim - Director February 13, 2014 Today s Topics NIH Fiscal Operations for FY 2014 Updates from the January 2014 FDP Meeting Other OCGA Updates NIH Fiscal Operations for FY2014 NIH Budget $30.15
Patti Manheim - Director February 13, 2014 Today s Topics NIH Fiscal Operations for FY 2014 Updates from the January 2014 FDP Meeting Other OCGA Updates NIH Fiscal Operations for FY2014 NIH Budget $30.15
Rebecca Trahan. Office of Sponsored Programs December 9, ORED Limited Submission Update
 Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
Research Administration Updates. May 16, :00 am IOP Auditorium
 Research Administration Updates May 16, 2018 11:00 am IOP Auditorium Topics Covered Using ClinCard (Greenphire) for Federally Sponsored Clinical Research Studies Ryan Mulligan, SCTR Institute OCR & ORSP
Research Administration Updates May 16, 2018 11:00 am IOP Auditorium Topics Covered Using ClinCard (Greenphire) for Federally Sponsored Clinical Research Studies Ryan Mulligan, SCTR Institute OCR & ORSP
UPDATES. Meet the Proposal Deadline. NIH: Public Access to Research Results OFFICE OF SPONSORED INSIDE THIS ISSUE:
 UPDATES INSIDE THIS ISSUE: Meet the Proposal Deadline Responsible Conduct of Research New Federal Regulations on Conflict of Interest NSF s New Proposal Guidelines and Project Reporting Changes Rutgers
UPDATES INSIDE THIS ISSUE: Meet the Proposal Deadline Responsible Conduct of Research New Federal Regulations on Conflict of Interest NSF s New Proposal Guidelines and Project Reporting Changes Rutgers
Notice of Grant Award (NGA): STANDARD Terms and Conditions
 After the Award is Made Then What? Cheryl Chick Chief Grants Management Officer National Human Genome Research Institute Roger G. Sorensen, Ph.D., MPA Division of Neuroscience and Behavior National Institute
After the Award is Made Then What? Cheryl Chick Chief Grants Management Officer National Human Genome Research Institute Roger G. Sorensen, Ph.D., MPA Division of Neuroscience and Behavior National Institute
Sponsored Programs New Developments and Important Reminders
 Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs March 7, 2012 1 Training Session Overview National Institutes of Health (NIH) Update NSF Proposal
Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs March 7, 2012 1 Training Session Overview National Institutes of Health (NIH) Update NSF Proposal
Research Administration Forum March 9, Marcia Smith Associate Vice Chancellor for Research
 Research Administration Forum March 9, 2017 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements - Marcia Smith Immigration Update and Q & A Sam Nahidi, Assistant Director,
Research Administration Forum March 9, 2017 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements - Marcia Smith Immigration Update and Q & A Sam Nahidi, Assistant Director,
Agenda. NIH Update. Other Updates. Proposal & Progress Report Statistics Research Administration Training Topic:
 Agenda NIH Update Salary Cap Guidance NOT OD 17 037: NIH Implementation of the Interim RPPR While a Renewal Application is Under Consideration Prior Approval for No Cost Extensions via the era Commons
Agenda NIH Update Salary Cap Guidance NOT OD 17 037: NIH Implementation of the Interim RPPR While a Renewal Application is Under Consideration Prior Approval for No Cost Extensions via the era Commons
Overview of the NIH Career Development Programs
 NIH Office of Extramural Research EB2010/FASEB April 2010 Overview of the NIH Career Development Programs Henry Khachaturian, Ph.D. NIH Extramural Program Policy Officer Office of Extramural Research,
NIH Office of Extramural Research EB2010/FASEB April 2010 Overview of the NIH Career Development Programs Henry Khachaturian, Ph.D. NIH Extramural Program Policy Officer Office of Extramural Research,
Sponsored Program Administration
 Sponsored Program Administration March, 2018 research.uconn.edu AGENDA OVPR/SPS News and Information IPR Form FCOI SPS Procedures for Fund Set Up Banner NIH News and Information Federal Executive Pay Scale
Sponsored Program Administration March, 2018 research.uconn.edu AGENDA OVPR/SPS News and Information IPR Form FCOI SPS Procedures for Fund Set Up Banner NIH News and Information Federal Executive Pay Scale
Navigating Grant Resources. 10/3/2018 Dr. Susan Fell
 Navigating Grant Resources 10/3/2018 Dr. Susan Fell sfell@fsu.edu 850-645-2172 Sponsored Research Administration https://www.research.fsu.edu/ Applying for a Grant Opportunity Does this require sign-off
Navigating Grant Resources 10/3/2018 Dr. Susan Fell sfell@fsu.edu 850-645-2172 Sponsored Research Administration https://www.research.fsu.edu/ Applying for a Grant Opportunity Does this require sign-off
Grant/Sponsor Related Systems. Department and OSP Perspectives on ERA
 Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
Policy and Compliance: Working Together Like Hand in Glove
 Policy and Compliance: Working Together Like Hand in Glove Samuel Ashe, Director, Division of Grants Policy, OPERA, OER Diane W. Dean, Director, Division of Grants Compliance and Oversight, OPERA, OER
Policy and Compliance: Working Together Like Hand in Glove Samuel Ashe, Director, Division of Grants Policy, OPERA, OER Diane W. Dean, Director, Division of Grants Compliance and Oversight, OPERA, OER
Gina Billiot, CRA Grant/Contract Specialist 2 (ERA) Office of Sponsored Programs (OSP)
 Gina Billiot, CRA Grant/Contract Specialist 2 (ERA) Office of Sponsored Programs (OSP) SPS - System Features SPS - System Overview Workspace Proposal Creation Demonstration Resources LOUISIANA STATE UNIVERSITY
Gina Billiot, CRA Grant/Contract Specialist 2 (ERA) Office of Sponsored Programs (OSP) SPS - System Features SPS - System Overview Workspace Proposal Creation Demonstration Resources LOUISIANA STATE UNIVERSITY
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Preparing campus for what is changing and what is not What is Grants Reform and when is it effective? What
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Preparing campus for what is changing and what is not What is Grants Reform and when is it effective? What
Patti Manheim, Director April 11, 2013
 Patti Manheim, Director April 11, 2013 Today s Topics NIH Research Performance Project Report (RPPR) Update Patti Manheim NIH Public Access Policy Reporting Requirements Patti Manheim and Lisa Federer,
Patti Manheim, Director April 11, 2013 Today s Topics NIH Research Performance Project Report (RPPR) Update Patti Manheim NIH Public Access Policy Reporting Requirements Patti Manheim and Lisa Federer,
Sponsored Program Administration Meeting. September 2016
 Sponsored Program Administration Meeting September 2016 Agenda NCURA Internal Controls Mike Unplugged: Training/Education Specialist position Budgeting for Post doc increases FDP Update ETS Update Effort
Sponsored Program Administration Meeting September 2016 Agenda NCURA Internal Controls Mike Unplugged: Training/Education Specialist position Budgeting for Post doc increases FDP Update ETS Update Effort
Presentation 2/26/2013. Director, TitleOPERA
 Presentation Title Michelle Presented G. Bulls By: Name Director, TitleOPERA Office OER AASCU Washington Conference February 21, 2013 2 NIH is operating under a continuing resolution. See NOT-OD- 13-018
Presentation Title Michelle Presented G. Bulls By: Name Director, TitleOPERA Office OER AASCU Washington Conference February 21, 2013 2 NIH is operating under a continuing resolution. See NOT-OD- 13-018
Uniform Guidance. Overview and Implementation Plan. November 21, 2014
 Uniform Guidance Overview and Implementation Plan November 21, 2014 Uniform Guidance. Change is coming! College or Department name here 1 Overarching goal of the reform is to: streamline the rules and
Uniform Guidance Overview and Implementation Plan November 21, 2014 Uniform Guidance. Change is coming! College or Department name here 1 Overarching goal of the reform is to: streamline the rules and
INTRODUCTION TO RESEARCH ADMINISTRATION. Office of Grants and Contracts Administration August 2015
 INTRODUCTION TO RESEARCH ADMINISTRATION Office of Grants and Contracts Administration August 2015 THE RESEARCH VESSEL SIKULIAQ FUNDED BY THE NATIONAL SCIENCE FOUNDATION Objectives To understand the UAF
INTRODUCTION TO RESEARCH ADMINISTRATION Office of Grants and Contracts Administration August 2015 THE RESEARCH VESSEL SIKULIAQ FUNDED BY THE NATIONAL SCIENCE FOUNDATION Objectives To understand the UAF
National Institute of Health (NIH)
 National Institute of Health (NIH) Funding Presented by: Samantha J. Taylor Senior Research Officer Wyss Institute for Biologically Inspired Engineering, Harvard University What is NIH s primary mission?
National Institute of Health (NIH) Funding Presented by: Samantha J. Taylor Senior Research Officer Wyss Institute for Biologically Inspired Engineering, Harvard University What is NIH s primary mission?
National Science Foundation Update. SRA Annual Meeting October 20, 2015
 National Science Foundation Update SRA Annual Meeting October 20, 2015 Speakers Jean Feldman Head, Policy Office Division of Institution & Award Support Office of Budget, Finance & Award Management 703.292.8243;
National Science Foundation Update SRA Annual Meeting October 20, 2015 Speakers Jean Feldman Head, Policy Office Division of Institution & Award Support Office of Budget, Finance & Award Management 703.292.8243;
OMB Uniform Guidance ( UG ) Briefing. ASRSP & OSR Brown Bag Tuesday, January 27 th
 OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
APPLIES TO: ALL DDPSC Scientific Employees, Students, Visiting Scientists RETENTION OF AND ACCESS TO RESEARCH DATA
 APPLIES TO: ALL DDPSC Scientific Employees, Students, Visiting Scientists TITLE: RETENTION OF AND ACCESS TO RESEARCH DATA PURPOSE: Maintaining accurate and appropriate research records is an essential
APPLIES TO: ALL DDPSC Scientific Employees, Students, Visiting Scientists TITLE: RETENTION OF AND ACCESS TO RESEARCH DATA PURPOSE: Maintaining accurate and appropriate research records is an essential
Federal Demonstration Partnership May 13, 2013
 Current News From NIH Michelle Bulls Director Office of Policy for Extramural Research Administration Federal Demonstration Partnership May 13, 2013 1 NIH Fiscal Policy for Grant FY 2013 Consolidated and
Current News From NIH Michelle Bulls Director Office of Policy for Extramural Research Administration Federal Demonstration Partnership May 13, 2013 1 NIH Fiscal Policy for Grant FY 2013 Consolidated and
Clarification on Salary Supplementation and Compensation During Entire Period of K Award
 Clarification on Salary Supplementation and Compensation During Entire Period of K Award 100 % Full-Time Professional Percent Effort 75% minimum devoted to K award (50% minimum for certain clinical specialties).
Clarification on Salary Supplementation and Compensation During Entire Period of K Award 100 % Full-Time Professional Percent Effort 75% minimum devoted to K award (50% minimum for certain clinical specialties).
Rebecca Trahan Office of Sponsored Programs January 10, 2017
 Rebecca Trahan Office of Sponsored Programs January 10, 2017 1 NSF Update NIH Update OSP Updates and Reminders Q & A 2 1 3 4 2 NSF 17-1 effective 1/30/2017 Effective for proposals submitted or due on or
Rebecca Trahan Office of Sponsored Programs January 10, 2017 1 NSF Update NIH Update OSP Updates and Reminders Q & A 2 1 3 4 2 NSF 17-1 effective 1/30/2017 Effective for proposals submitted or due on or
Outgoing Subagreements: Subawards and Subcontracts
 Effective Date: March 1, 2014 Version: 1 Page: 1 of 6 Overview Sponsored programs may include partnerships with other organizations or institutions whereby a portion of the project is conducted by investigators
Effective Date: March 1, 2014 Version: 1 Page: 1 of 6 Overview Sponsored programs may include partnerships with other organizations or institutions whereby a portion of the project is conducted by investigators
ORA Quarterly Meeting April
 ORA Quarterly Meeting April 19 2017 Agenda 1. Workday Grants Implementation Items General Information Blackout Period Sub K s - Set-up and Invoicing Advance Accounts 2. Jackson/ UM Partnership Master Agreement
ORA Quarterly Meeting April 19 2017 Agenda 1. Workday Grants Implementation Items General Information Blackout Period Sub K s - Set-up and Invoicing Advance Accounts 2. Jackson/ UM Partnership Master Agreement
Research Administrators Forum October 10th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum October 10th, 2013 Marcia Smith Associate Vice Chancellor for Research Agenda Meeting Agenda Welcome and Announcements Marcia Smith, Office of Research Administration Contract
Research Administrators Forum October 10th, 2013 Marcia Smith Associate Vice Chancellor for Research Agenda Meeting Agenda Welcome and Announcements Marcia Smith, Office of Research Administration Contract
Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32)
 Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32) Overview: This document is intended to help you complete a KC System to System (S2S) proposal submission
Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32) Overview: This document is intended to help you complete a KC System to System (S2S) proposal submission
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM. December 2017
 Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
I am an INVESTIGATOR. Should I consider adding a diversity supplement (via PA ) to my ongoing studies?
 Last Updated 9/10/2017 THE NIH DIVERSITY SUPPLEMENT PROGRAM & THE RUTH L. KIRSCHSTEIN NATIONAL RESEARCH SERVICE AWARD INDIVIDUAL PREDOCTORAL FELLOWSHIP TO PROMOTE DIVERSITY IN HEALTH-RELATED RESEARCH SCHOOL
Last Updated 9/10/2017 THE NIH DIVERSITY SUPPLEMENT PROGRAM & THE RUTH L. KIRSCHSTEIN NATIONAL RESEARCH SERVICE AWARD INDIVIDUAL PREDOCTORAL FELLOWSHIP TO PROMOTE DIVERSITY IN HEALTH-RELATED RESEARCH SCHOOL
SJSU Research Foundation
 SJSU Research Foundation Grant Proposal Development Taking a Bite Out of Forms Sponsored Programs Workshop November 19, 2015 Overview of Workshop Why We Have Forms Forms from OSP Forms from Human Resources
SJSU Research Foundation Grant Proposal Development Taking a Bite Out of Forms Sponsored Programs Workshop November 19, 2015 Overview of Workshop Why We Have Forms Forms from OSP Forms from Human Resources
NIH Update FDP September 2009 Office of Policy for Extramural Research Administration
 NIH Update FDP September 2009 Office of Policy for Extramural Research Administration OPERA New NIH Director Francis S. Collins, M.D., Ph.D., was officially sworn in on Monday, August 17, 2009 as the 16th
NIH Update FDP September 2009 Office of Policy for Extramural Research Administration OPERA New NIH Director Francis S. Collins, M.D., Ph.D., was officially sworn in on Monday, August 17, 2009 as the 16th
HS SPPO Contacts Meeting December 8, 2016
 HS SPPO Contacts Meeting December 8, 2016 Agenda HS SPPO Updates NIH Updates & Reminders OCGA Updates HS SPPO Updates 2016 Holiday Closure HS SPPO, along with the Vice Chancellor Health Sciences/Dean s
HS SPPO Contacts Meeting December 8, 2016 Agenda HS SPPO Updates NIH Updates & Reminders OCGA Updates HS SPPO Updates 2016 Holiday Closure HS SPPO, along with the Vice Chancellor Health Sciences/Dean s
Research Administrators Round Table
 Research Administrators Round Table Recap Set framework for going forward Review new Uniform Guidance driven WVU policy changes Sponsored Agreement Closeouts (WVU-FS-23) http://adminfin.wvu.edu/policies/finance/financial-services/wvu-fs-23-policy
Research Administrators Round Table Recap Set framework for going forward Review new Uniform Guidance driven WVU policy changes Sponsored Agreement Closeouts (WVU-FS-23) http://adminfin.wvu.edu/policies/finance/financial-services/wvu-fs-23-policy
The Office for Sponsored Research (OSR): An Overview. Office for Sponsored Research
 The Office for Sponsored Research (OSR): An Overview Office for Sponsored Research OSR Mission The Office for Sponsored Research assists investigators in proposing and managing sponsored programs in support
The Office for Sponsored Research (OSR): An Overview Office for Sponsored Research OSR Mission The Office for Sponsored Research assists investigators in proposing and managing sponsored programs in support
Research Administrators Forum August 14th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum August 14th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Associate Vice Chancellor, Office of Research Administration
Research Administrators Forum August 14th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Associate Vice Chancellor, Office of Research Administration
Agenda. Workday Training Session for Grants. Tuesday, December 15, :30 am 12:00pm. Office Depot Conference Room OD 101. Time Topic Presenter
 Agenda Workday Training Session for Grants Tuesday, December 15, 2015 9:30 am 12:00pm Office Depot Conference Room OD 101 Time Topic Presenter 9:30 9:40 am Welcome and Introductions Heather Saunders 9:40
Agenda Workday Training Session for Grants Tuesday, December 15, 2015 9:30 am 12:00pm Office Depot Conference Room OD 101 Time Topic Presenter 9:30 9:40 am Welcome and Introductions Heather Saunders 9:40
IRB Approval of Research Using Survey Procedures Kerry Agnitsch, Office for Responsible Research October 1, 2009
 IRB Approval of Research Using Survey Procedures Kerry Agnitsch, Office for Responsible Research October 1, 2009 Key Definitions (per 45 CFR 46.102(d) & (f): Research a systematic investigation, including
IRB Approval of Research Using Survey Procedures Kerry Agnitsch, Office for Responsible Research October 1, 2009 Key Definitions (per 45 CFR 46.102(d) & (f): Research a systematic investigation, including
NSF Update: 17-1 Proposal & Award Policies & Procedures Guide (PAPPG)
 NSF Update: 17-1 Proposal & Award Policies & Procedures Guide (PAPPG) Daniel Lewis Sponsored Programs Administrator dlewis15@utk.edu 865-974-8873 Raymond Norris Sponsored Programs Administrator rnorris6@utk.edu
NSF Update: 17-1 Proposal & Award Policies & Procedures Guide (PAPPG) Daniel Lewis Sponsored Programs Administrator dlewis15@utk.edu 865-974-8873 Raymond Norris Sponsored Programs Administrator rnorris6@utk.edu
Jean Feldman. Policy Office. Head, Policy Office, Policy Office, Division of Institution & Award Support
 NATIONAL SCIENCE FOUNDATION UPDATE Federal Demonstration Partnership Meeting January 26, 2011 Washington, DC Ask Early, Ask Often Jean Feldman Head, Policy Office, Policy Office, Division of Institution
NATIONAL SCIENCE FOUNDATION UPDATE Federal Demonstration Partnership Meeting January 26, 2011 Washington, DC Ask Early, Ask Often Jean Feldman Head, Policy Office, Policy Office, Division of Institution
Electronic Research Administration (era) March 8, 2018
 Electronic Research Administration (era) March 8, 2018 Introductions We are. Terry Duperron, era Analyst Marlene Mooshian, Proposal Analyst Kassie Obelleiro, Training Officer Jinger Snyder, Award Analyst
Electronic Research Administration (era) March 8, 2018 Introductions We are. Terry Duperron, era Analyst Marlene Mooshian, Proposal Analyst Kassie Obelleiro, Training Officer Jinger Snyder, Award Analyst
Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6
 Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6 KC-PRE28: Prepare Summary Proposal KC-AWD22: Research Gifts November 13, 2013 Goals and Outcomes Goals Obtain feedback on the proposed
Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6 KC-PRE28: Prepare Summary Proposal KC-AWD22: Research Gifts November 13, 2013 Goals and Outcomes Goals Obtain feedback on the proposed
ORA Closeout Process for NIH Awards
 Office of Research Administration ORA Closeout Process for NIH Awards ORA CLOSEOUT GUIDELINES ORA is responsible for making sure that necessary closeout documents are submitted to NIH within 90 days of
Office of Research Administration ORA Closeout Process for NIH Awards ORA CLOSEOUT GUIDELINES ORA is responsible for making sure that necessary closeout documents are submitted to NIH within 90 days of
OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015
 OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015 INTRODUCTION The University of Wisconsin-Milwaukee (UWM) is the prime recipient on a wide range of sponsored awards
OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015 INTRODUCTION The University of Wisconsin-Milwaukee (UWM) is the prime recipient on a wide range of sponsored awards
Financial Conflict of Interest: Investigator Procedures. Office of Research, Innovation, and Economic Development Research Integrity and Compliance
 Financial Conflict of Interest: Investigator Procedures Office of Research, Innovation, and Economic Development Research Integrity and Compliance June 2018 2 Table of Contents Introduction... 3 Private,
Financial Conflict of Interest: Investigator Procedures Office of Research, Innovation, and Economic Development Research Integrity and Compliance June 2018 2 Table of Contents Introduction... 3 Private,
The Lifecycle of a Sponsored Project
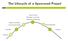 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
Research Administration Network Agenda. SUB Lobo A&B April 11th, :00am to 10:00am
 Research Administration Network Agenda SUB Lobo A&B April 11th, 2017 9:00am to 10:00am 1. Introductions 2. Presentations Audrey Pineda, Office of Research and Compliance Topic: Industrial Security and
Research Administration Network Agenda SUB Lobo A&B April 11th, 2017 9:00am to 10:00am 1. Introductions 2. Presentations Audrey Pineda, Office of Research and Compliance Topic: Industrial Security and
RESEARCH ADMINISTRATION NEWSLETTER
 RESEARCH ADMINISTRATION NEWSLETTER http://www.brown.edu/research/research-administration-newsletters SEPTEMBER 2016 The monthly newsletter provides timely information on agency updates; sponsor and University
RESEARCH ADMINISTRATION NEWSLETTER http://www.brown.edu/research/research-administration-newsletters SEPTEMBER 2016 The monthly newsletter provides timely information on agency updates; sponsor and University
Weekly NIH Funding Opportunities and Notices NIH Guide for Grants and Contracts April 27, 2018 Table of Contents (TOC) Web Version
 Weekly NIH Funding Opportunities and Notices NIH Guide for Grants and Contracts April 27, 2018 Table of Contents (TOC) Web Version Notices Request for Information (RFI): Inviting Scientific Interest in
Weekly NIH Funding Opportunities and Notices NIH Guide for Grants and Contracts April 27, 2018 Table of Contents (TOC) Web Version Notices Request for Information (RFI): Inviting Scientific Interest in
CURRENT COGR PRIORITIES - BY COMMITTEE (7/10/17)
 CURRENT COGR PRIORITIES - BY COMMITTEE (7/10/17) No. 1 Student Financial Aid and "Securing Student Information" and Compliance Supplement New single audit requirement. Delayed until 2018. MEDIUM Pending
CURRENT COGR PRIORITIES - BY COMMITTEE (7/10/17) No. 1 Student Financial Aid and "Securing Student Information" and Compliance Supplement New single audit requirement. Delayed until 2018. MEDIUM Pending
NIH Agency Specifics August 11, 2015
 NIH Agency Specifics August 11, 2015 Maria Skinner Office of Sponsored Programs Stacy Salisbury Contract & Grant Administration NIH Pre-Award Presented by: Maria Skinner Office of Sponsored Programs (OSP)
NIH Agency Specifics August 11, 2015 Maria Skinner Office of Sponsored Programs Stacy Salisbury Contract & Grant Administration NIH Pre-Award Presented by: Maria Skinner Office of Sponsored Programs (OSP)
UPDATES WELCOME BACK! OFFICE OF SPONSORED
 UPDATES INSIDE THIS ISSUE: Meet the Proposal Deadline RU s New Endorsement Form Rutgers Budget Template NIH Changes to Form Packages NIH Changes for Reports NSF MRI Program Limited Submission Pre-proposal
UPDATES INSIDE THIS ISSUE: Meet the Proposal Deadline RU s New Endorsement Form Rutgers Budget Template NIH Changes to Form Packages NIH Changes for Reports NSF MRI Program Limited Submission Pre-proposal
The NIH Public Access Policy
 The NIH Public Access Policy NIH Regionals October 2016 Neil Thakur, Bart Trawick Posted at communications.htm Today s Discussion: The NIH Public Access Policy 1. The Basics 2. Awardee Tasks 3. NIHMS:
The NIH Public Access Policy NIH Regionals October 2016 Neil Thakur, Bart Trawick Posted at communications.htm Today s Discussion: The NIH Public Access Policy 1. The Basics 2. Awardee Tasks 3. NIHMS:
UNIFORM GUIDANCE IMPLEMENTATION
 UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
October electronic Research Administration (era) OER, OD, National Institutes of Health
 October 2016 electronic Research Administration (era) OER, OD, National Institutes of Health Scarlett Gibb era Customer Relationship Manager, era Commons Joe Schumaker era Communications and Outreach 2
October 2016 electronic Research Administration (era) OER, OD, National Institutes of Health Scarlett Gibb era Customer Relationship Manager, era Commons Joe Schumaker era Communications and Outreach 2
The Association of Universities for Research in Astronomy. Award Management Policies Manual
 The Association of Universities for Research in Astronomy Award Management Policies Manual May 1, 2014 The Association of Universities for Research in Astronomy Award Management Policies Manual Table of
The Association of Universities for Research in Astronomy Award Management Policies Manual May 1, 2014 The Association of Universities for Research in Astronomy Award Management Policies Manual Table of
After the Award is Made THEN WHAT?
 After the Award is Made THEN WHAT? George Tucker Chief Grants Management Officer National Institute of Environmental Health Sciences October 2016 TOPICS OF DISCUSSION The ins & outs of the NoA Accessing
After the Award is Made THEN WHAT? George Tucker Chief Grants Management Officer National Institute of Environmental Health Sciences October 2016 TOPICS OF DISCUSSION The ins & outs of the NoA Accessing
Research Administrator Meeting Tuesday, January 23, :00-12:00 Illinois Room (348), IMU
 Research Administrator Meeting Tuesday, January 23, 2018 10:00-12:00 Illinois Room (348), IMU Agenda: 1. GAO Announcements 2. Costing Considerations 3. Quick Reference Guide Overview 4. DSP General Announcements
Research Administrator Meeting Tuesday, January 23, 2018 10:00-12:00 Illinois Room (348), IMU Agenda: 1. GAO Announcements 2. Costing Considerations 3. Quick Reference Guide Overview 4. DSP General Announcements
TEXAS GENERAL LAND OFFICE COMMUNITY DEVELOPMENT & REVITALIZATION PROCUREMENT GUIDANCE FOR SUBRECIPIENTS UNDER 2 CFR PART 200 (UNIFORM RULES)
 TEXAS GENERAL LAND OFFICE COMMUNITY DEVELOPMENT & REVITALIZATION PROCUREMENT GUIDANCE FOR SUBRECIPIENTS UNDER 2 CFR PART 200 (UNIFORM RULES) The Texas General Land Office Community Development & Revitalization
TEXAS GENERAL LAND OFFICE COMMUNITY DEVELOPMENT & REVITALIZATION PROCUREMENT GUIDANCE FOR SUBRECIPIENTS UNDER 2 CFR PART 200 (UNIFORM RULES) The Texas General Land Office Community Development & Revitalization
NIH Mission Improve human health through biomedical and behavioral research, research training and communications.
 Overview of the NIH SBIR/STTR Program NCET2 March 21, 2013 Matthew Portnoy, Ph.D. SBIR/STTR Program Coordinator, NIH 1 NIH Mission Improve human health through biomedical and behavioral research, research
Overview of the NIH SBIR/STTR Program NCET2 March 21, 2013 Matthew Portnoy, Ph.D. SBIR/STTR Program Coordinator, NIH 1 NIH Mission Improve human health through biomedical and behavioral research, research
NSF FUNDAMENTALS WORKSHOP. Thomas Jefferson University December 2017
 NSF FUNDAMENTALS WORKSHOP Thomas Jefferson University December 2017 SPEAKERS Jean Feldman Head, Policy Office Division of Institution and Award Support Office of Budget, Finance and Award Management 703.292.8243;
NSF FUNDAMENTALS WORKSHOP Thomas Jefferson University December 2017 SPEAKERS Jean Feldman Head, Policy Office Division of Institution and Award Support Office of Budget, Finance and Award Management 703.292.8243;
FAQ S FOR UNIFORM GUIDANCE
 FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
National Science Foundation (NSF) Update --- Spring Federal Demonstration Partnership Meeting May 13, 2013
 National Science Foundation (NSF) Update --- Spring 2013 Federal Demonstration Partnership Meeting May 13, 2013 Ask Early, Ask Often Jean Feldman Head, Policy Office Division of Institution & Award Support
National Science Foundation (NSF) Update --- Spring 2013 Federal Demonstration Partnership Meeting May 13, 2013 Ask Early, Ask Often Jean Feldman Head, Policy Office Division of Institution & Award Support
RESEARCH POLICY MANUAL
 POLICY MANUAL RESEARCH Number 588 Subject: Research Data Covered Employees: USU Employees and Students Date of Origin: May 5, 2017 588.1 INTRODUCTION Research data are an essential component of any research
POLICY MANUAL RESEARCH Number 588 Subject: Research Data Covered Employees: USU Employees and Students Date of Origin: May 5, 2017 588.1 INTRODUCTION Research data are an essential component of any research
National Institutes of Health
 National Institutes of Health NIH NIH s mission is to seek fundamental knowledge about the nature and behavior of living systems and the application of that knowledge to enhance health, lengthen life,
National Institutes of Health NIH NIH s mission is to seek fundamental knowledge about the nature and behavior of living systems and the application of that knowledge to enhance health, lengthen life,
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates!
 FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
Current Issues in Sponsored Projects Administration. March 11 10:30-11:45 3:15 4:30
 Current Issues in Sponsored Projects Administration March 11 10:30-11:45 3:15 4:30 Workshop Presenters Name: Linda Learned Title: Associate Director/Interim Director, Office of Sponsored Programs and Research
Current Issues in Sponsored Projects Administration March 11 10:30-11:45 3:15 4:30 Workshop Presenters Name: Linda Learned Title: Associate Director/Interim Director, Office of Sponsored Programs and Research
Kathy Hancock, Assistant Grants Compliance Officer, DGCO, OPERA, Office of Extramural Research, NIH, HHS
 Division of Grants Compliance and Oversight (DGCO) Office of Policy for Extramural Research Administration (OPERA), OER National Institutes of Health (NIH), DHHS 19 th Annual SBIR/STTR Conference Milwaukee,
Division of Grants Compliance and Oversight (DGCO) Office of Policy for Extramural Research Administration (OPERA), OER National Institutes of Health (NIH), DHHS 19 th Annual SBIR/STTR Conference Milwaukee,
Research Administrators Forum September 13, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum September 13, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith OCGA Initiatives Patti Manheim, Kim Duiker Proposal Intake
Research Administrators Forum September 13, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith OCGA Initiatives Patti Manheim, Kim Duiker Proposal Intake
National Academy of Sciences Committee on University IP Management
 National Academy of Sciences Committee on University IP Management June 30, 2008 Robert Hardy Director, Contracts and IP Management Council on Governmental Relations A Word About COGR Council on Governmental
National Academy of Sciences Committee on University IP Management June 30, 2008 Robert Hardy Director, Contracts and IP Management Council on Governmental Relations A Word About COGR Council on Governmental
Division of Research
 Division of Research Quarterly Research Administration Meeting October 30, 2012 Page 1 1 Division of Research Quarterly Research Administration Meeting AGENDA Welcome and Introductions Pre Award Update
Division of Research Quarterly Research Administration Meeting October 30, 2012 Page 1 1 Division of Research Quarterly Research Administration Meeting AGENDA Welcome and Introductions Pre Award Update
+ Pre and Post Award Grants Management for Mailman School Researchers
 + Pre and Post Award Grants Management for Mailman School Researchers Rosa Rivera, Associate Dean, Grants & Contracts and Director of MSPH Operations, Office of Sponsored Projects Administration Katherine
+ Pre and Post Award Grants Management for Mailman School Researchers Rosa Rivera, Associate Dean, Grants & Contracts and Director of MSPH Operations, Office of Sponsored Projects Administration Katherine
MENTORED CAREER DEVELOPMENT (KL2) AWARD REQUEST FOR PROPOSALS
 MENTORED CAREER DEVELOPMENT (KL2) AWARD REQUEST FOR PROPOSALS BACKGROUND A major goal of the University of Massachusetts Center for Clinical and Translational Science (UMCCTS) is to develop and support
MENTORED CAREER DEVELOPMENT (KL2) AWARD REQUEST FOR PROPOSALS BACKGROUND A major goal of the University of Massachusetts Center for Clinical and Translational Science (UMCCTS) is to develop and support
Office of Sponsored Programs Budgetary and Cost Accounting Procedures
 Office of Sponsored Programs Budgetary and Cost Accounting Procedures Table of Contents 1. Purpose and Services 2. Definitions of Terms 3. Budget Items 4. Travel 5. Effort Certification Reporting 6. Costing
Office of Sponsored Programs Budgetary and Cost Accounting Procedures Table of Contents 1. Purpose and Services 2. Definitions of Terms 3. Budget Items 4. Travel 5. Effort Certification Reporting 6. Costing
IDeA Post Award Management
 IDeA Post Award Management Lisa Hildred Grants Management Specialist Grants Administration Branch National Institute of General Medical Sciences, NIH Meet the Grants Management Team Christy Leake Sam Farrell
IDeA Post Award Management Lisa Hildred Grants Management Specialist Grants Administration Branch National Institute of General Medical Sciences, NIH Meet the Grants Management Team Christy Leake Sam Farrell
Template D Plain-crimson-dark 1
 WSU Research Administration Series: Research Administration Update Dan Nordquist, Kim Small, Sponsored Programs Heather Lopez, Internal Audit September 12, 2014 Recording date of this workshop is September
WSU Research Administration Series: Research Administration Update Dan Nordquist, Kim Small, Sponsored Programs Heather Lopez, Internal Audit September 12, 2014 Recording date of this workshop is September
Finding Funding, Budget Preparation, and Proposal Submission for Sponsored Research
 Finding Funding, Budget Preparation, and Proposal Submission for Sponsored Research Laura Johnston, Office of Sponsored Programs, Asst. Director Craig O Neill, Office of Sponsored Programs, Manager February
Finding Funding, Budget Preparation, and Proposal Submission for Sponsored Research Laura Johnston, Office of Sponsored Programs, Asst. Director Craig O Neill, Office of Sponsored Programs, Manager February
Expense Review Process and Internal Controls for Award Closeout. Semi-annual Networking Event February 2017
 ACCOUNTING SERVICES FOR RESEARCH AND SPONSORED PROGRAMS (ASRSP) Expense Review Process and Internal Controls for Award Closeout Semi-annual Networking Event February 2017 Kathy Mustea, Assistant Director
ACCOUNTING SERVICES FOR RESEARCH AND SPONSORED PROGRAMS (ASRSP) Expense Review Process and Internal Controls for Award Closeout Semi-annual Networking Event February 2017 Kathy Mustea, Assistant Director
Research Administrators Group (RAG) Meeting July 2015
 Research Administrators Group (RAG) Meeting July 2015 Office of Sponsored Projects 1 Agenda Office of Sponsored Projects 2 Introduction of New Staff Proposal/PD Update Proposal Metrics RPPR Transition
Research Administrators Group (RAG) Meeting July 2015 Office of Sponsored Projects 1 Agenda Office of Sponsored Projects 2 Introduction of New Staff Proposal/PD Update Proposal Metrics RPPR Transition
Rosemarie Filart, MD MPH MBA NIH Program Officer National Center of Research Resources, NIH NCRR
 Na ationa al Ins stitut tes of Hea alth The Basics of SBIR/STTR Grants and Other Public & Private Funding Sources for Innovation and Technology Weill Cornell CTSC February 27, 2009 New York, NY Rosemarie
Na ationa al Ins stitut tes of Hea alth The Basics of SBIR/STTR Grants and Other Public & Private Funding Sources for Innovation and Technology Weill Cornell CTSC February 27, 2009 New York, NY Rosemarie
UC Davis Policy and Procedure Manual
 UC Davis Policy and Procedure Manual Chapter 230, Sponsored Programs Section 07, Public Health Service Regulations on Objectivity in Research Date: Supersedes: 8/24/12 Responsible Department: Office of
UC Davis Policy and Procedure Manual Chapter 230, Sponsored Programs Section 07, Public Health Service Regulations on Objectivity in Research Date: Supersedes: 8/24/12 Responsible Department: Office of
AURA Meeting. July 23, 2014
 AURA Meeting July 23, 2014 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored
AURA Meeting July 23, 2014 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored
Step 2 From the Modifications Summary in the Award Workspace click the link to the newly created Modification.
 No Cost Extensions When a project requires additional time to complete the scope of work a Modification for a No Cost Extension must be requested. Depending upon the terms and conditions of the Award there
No Cost Extensions When a project requires additional time to complete the scope of work a Modification for a No Cost Extension must be requested. Depending upon the terms and conditions of the Award there
COST PRINCIPLES AND PROCEDURAL STATEMENTS FAQs
 COST PRINCIPLES AND PROCEDURAL STATEMENTS FAQs Cost Principles Policy FAQs Question: What is required for a cost to be adequately documented? Answer: Receipts or copies of receipts for all purchases (electronic
COST PRINCIPLES AND PROCEDURAL STATEMENTS FAQs Cost Principles Policy FAQs Question: What is required for a cost to be adequately documented? Answer: Receipts or copies of receipts for all purchases (electronic
Presentation. Title. Presented By: Name. Title
 Presentation Title Presented By: Name Title Lisa Moeller, CRA, Grants Administration OfficeBranch, National Institute of General Medical Sciences, National Institutes of Health Why? Protected time for
Presentation Title Presented By: Name Title Lisa Moeller, CRA, Grants Administration OfficeBranch, National Institute of General Medical Sciences, National Institutes of Health Why? Protected time for
INDIRECT COST POLICY
 UNIVERSITY OF LOUISIANA AT LAFAYETTE OFFICE OF THE VICE PRESIDENT FOR RESEARCH INDIRECT COST POLICY Revision Date: 8/11/2014 Original Effective Date: 11/08/2006 Responsible Office: Reference: Vice President
UNIVERSITY OF LOUISIANA AT LAFAYETTE OFFICE OF THE VICE PRESIDENT FOR RESEARCH INDIRECT COST POLICY Revision Date: 8/11/2014 Original Effective Date: 11/08/2006 Responsible Office: Reference: Vice President
