YOU ARE INSTRUCTED TO READ THE FOLLOWING THOROUGHLY BEFORE PROCEEDING TO
|
|
|
- Beatrice Griffin
- 5 years ago
- Views:
Transcription
1 Title: OMB-MSOP 019 Processing Consent Forms and Patient Data Serial Number: OMB-MSOP 019 Version Approver: Enter name Version Number: 1.18 (Draft) Version Approval Date: [IN Version Effective: Two weeks from release Version Author: Gareth Bicknell Version Date: 08/08/2012 Area of Application: OMB Office Relevance: All OMB Staff Date Details of Review Version number No. of pages Name of Reviewer Next Review 22/02/12 08/08/12-12/09/14 New SOP formalising existing procedures Overhauled entire SOP in line with constantly-changing needs of the database; added adoption considerations Huma Zafar, Nick Kenny, Gareth Bicknell Gareth Bicknell, Karolina Kliskey February 2013 Sep 2015 YOU ARE INSTRUCTED TO READ THE FOLLOWING THOROUGHLY BEFORE PROCEEDING TO UNDERTAKE THE METHODS DESCRIBED. UNDER NO CIRCUMSTANCES ARE THESE INSTRUCTIONS TO BE AMENDED OR ALTERED IN ANY WAY OTHER THAN BY THE AUTHOR / APPROVER. OMB-MSOP 019 Version 1.18 (Draft) Page 1 of 15
2 Table Of Contents 1 PURPOSE SAFETY INFORMATION DEFINITIONS OMB NUMBER ADOPTION MRN REQUIREMENTS & RESPONSIBILITIES GENERALLY REFERENCES & RELATED DOCUMENTS PROCEDURE FAILURE TO CONSENT SUCCESSFUL CONSENT USE OF OMB PATIENT FOLDERS ALLOCATION OF AN OMB NUMBER ELECTRONIC REGISTRATION OF A NEW OMB PATIENT FOLDER SEARCHING THE OMB PATIENT MONITOR DATABASE UPDATING WITHDRAWAL, DECEASED, AND FOLLOW-UP/CONTACT PREFERENCES UPDATING PROCEDURE DETAILS UPDATING OMB FOLLOW-UP ATTEMPTS DUPLICATED CONSENTS IMPLEMENTATION TRAINING REQUIREMENTS STAFF RECORD OF ACKNOWLEDGEMENT OMB-MSOP 019 Version 1.18 (Draft) Page 2 of 15
3 1 Purpose 1.1 The purpose of this SOP is to standardise the processing of consent forms and patient data, particularly at the point of OMB Patient Folder creation, so that every consent form can be linked to an OMB tissue sample or dataset (and vice versa). 2 Safety Information 2.1 Specific reference should be made to the University Occupational Health Service and their advice on Display Screen Equipment use with respect to eye strain and ergonomics. 3 Definitions 3.1 OMB Number A number linking separate tissue samples/aliquots to a particular consent package. Equivalent to a trial number in a clinical trial. 3.2 Adoption The process of accepting consents and material from protocols other than OMB s. Within OMB, these protocols are often termed external ethics. 3.3 MRN Medical Record Number 4 Requirements & Responsibilities 4.1 Generally All OMB office and laboratory team members are responsible for ensuring that the link between consent, data and tissue is retained. 5 References & Related Documents OMB-MSOP 019 Version 1.18 (Draft) Page 3 of 15
4 5.1 OMB-F 002 OMB Consent Form 5.2 OUH Patient Agreement to Investigation or Treatment 5.3 OMB-F 003 Sample Tracker and Laboratory Checklist 5.4 OMB-F 009 Sample Tracker and Laboratory Checklist Externally Approved Projects 5.5 PROT-NDORMS 01 Oxford Musculoskeletal Biobank Management 5.6 OMB-MSOP 014 Identification and Control of Non-Conformities 5.7 OMB-MSOP 024 Withdrawal of Donor Consent 5.8 OMB-MSOP 029 Initiation and Receipt of Follow-ups 6 Procedure 6.1 Failure to Consent NB OMB prefers to record a bare minimum of tracability data when a patient fails to consent, so that consenters know whether or not to approach the patient again. However, it is essential that only a bare minimum of data is recorded, in order to avoid accidental use of unconsented data into a research context Open the OMB Patient Monitor Database. Trust it, if asked to Click the Open Non-Consents Form button Provide the following details: The date of failure to consent A reason for the failure (e.g. patient declined, no approach possible, appointment cancelled, patient ineligible) Your initials The MRN and, if available, the NHS number of the patient The patient s date of birth OMB-MSOP 019 Version 1.18 (Draft) Page 4 of 15
5 6.1.4 The record will be added when the form is closed. 6.2 Successful Consent Use of OMB Patient Folders NB A new OMB Patient Folder must be created each time a new OMB Number is allocated, i.e. every time a new patient care pathway is followed (as per OMB Management Protocol, PROT-NDORMS 01), or as determined by the adopted study s protocol. For OMB consents, when tissue originates from an episode of continuing treatment or a consent confirmation during continuing treatment, no new folder is required the existing one should be used OMB Patient Folders contain an archive of all paperwork relevant to a particular OMB Number, and folders are coloured according to the joint or pathway. The colour scheme is as follows: Red Operative Hip (non-oncological) Green Operative Knee (non-oncological) Blue Operative Shoulder (non-oncological) Yellow Operative Elbow (non-oncological) Black Oncological Orange Non-operative RA, Spine, Ankle, Wrist, Other On receipt of a consent form (whether OMB-F 002 or other), check for the following: In all cases, it must have been correctly signed and dated by the patient. In all cases, it must have been correctly signed and dated by the person obtaining consent. The version of the form should be one appropriate to the date of signing. All required boxes should have been initialled in the patient s handwriting as per relevant study protocol and patient wishes. NB some non-omb protocols may deem ticks acceptable. OMB-MSOP 019 Version 1.18 (Draft) Page 5 of 15
6 For non-omb consents being adopted, there must be a positive indication that samples and/or data can be stored for unspecified future research. For OMB-F 002 and NHS consent forms, the patient s name, NHS number and/or MRN and preferably a date of birth and gender must have been recorded. The name must match that of the person giving consent. For OMB-F 002, the person obtaining consent should be an approved OMB consenter (i.e. signed off by the OMB Manager) If there are any failures of the above conditions, report the matter immediately to the OMB Manager for consideration before continuing If the consent form is acceptable, secure it in the folder, along with any other relevant paperwork received. Consent forms should remain at the back of the folder throughout its lifespan in order to enable rapid consent audit at any time If the folder is a new one, secure a Sample Tracker and Laboratory Checklist into the front. There are two versions: OMB-F 003 (Sample Tracker and Laboratory Checklist), used for patients consented using OMB-F 002 (OMB Consent Form) or the OUH Patient Agreement to Investigation or Treatment ( NHS consent ) OMB-F 009 (Sample Tracker and Laboratory Checklist Externally Approved Projects), used for consents under non-omb protocols (i.e. consents for adoption) The Sample Tracker should remain at the front of the folder throughout its lifespan in order to allow rapid identification of the consent origin and care pathway Any other documentation should be sandwiched between these forms in reverse chronological order (i.e. most recent first), as per standard NHS practice. 6.3 Allocation of an OMB Number OMB-MSOP 019 Version 1.18 (Draft) Page 6 of 15
7 NB To avoid excessive bureaucracy, it is very important to allocate a new OMB Number only when the consent requires it. Always use the database to check for existing consents recorded against any particular patient, and always adhere to the OMB Management Protocol when classifying a consent. If you are in any doubt, consult the OMB Manager If you need a new OMB Number, locate the next available set of 20 stickers from the rolls in the OMB office. Avoid tearing off more sets than you are actually going to use, because all need to be accounted for. NB stickers are custom pre-printed in sets of 20. The format of each number is OMB YY/NNNN, where NNNN is a number in sequence from , and YY reflects the current financial year (e.g. 11 for 01/04/ /03/2012) Stick one sticker into the indicated box on the consent form (or somewhere prominent if there is no box) Stick one sticker to the top of the front-facing folder spine, orientated such that the bottom of the text points to the right of the folder: OMB XX / NNNN Lorem ipsum dolor sit amet, consectetur adipisicing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Ut enim ad minim veniam, quis nostrud exercitation ullamco laboris Using a stapler, attach the strip of remaining stickers to the Sample Tracker. 6.4 Electronic Registration of a New OMB Patient Folder NB It is very important to register a new OMB Patient Folder as soon as possible, and preferably on the day of consent. This allows other researchers to check the consent, thus avoiding over-burdening patients with consent attempts. It also avoids breaches of the OMB Management Protocol through accidental re-use of a consent beyond its limit Open the OMB Patient Monitor Database. Trust it, if asked to. OMB-MSOP 019 Version 1.18 (Draft) Page 7 of 15
8 6.4.2 Click the Add Record button At the top of the page, enter the following data: The OMB Number The NHS number, if available (the special number, , is valid and should be entered) The patient s date of birth, if available The patient s gender The category of procdure or treatment Any relevant MRN (i.e. old or new), if available On the Consent tab, enter the following data: The Date of consent The Version of consent form used (contact the OMB Manager if the required version is not available) The Location of consent, if at the OUH and if known The Consenter Initials if the consent is on OMB-F 002, or else select NHS for NHS consenters, or PROJ for consenters operating under external ethics The OMB Study reference for which consent has primarily been taken (if the consent is on OMB-F 002 or an NHS consent form), or the OMB reference under which OMB is adopting the consent (if being adopted), or nothing (otherwise) Any relevant comments, including any further study-specific consent comments Tick the Critical Initials Missing box only if: The consent form is OMB-F 002, and OMB-MSOP 019 Version 1.18 (Draft) Page 8 of 15
9 Initials are missing from any statement except follow-up, contact-for-futurestudies, or implants when no implant is being removed Tick the Opt-out Preferences Unclear box only if The consent form is OMB-F 002, and Initials are missing from the follow-up and/or contact-for-future-studies statements, and There is also no clear indication elsewhere that the follow-up and/or contact is unwanted Tick the No Data Consent box only if the consent form does not include an explicit acknowledgement of consent to collect data from medical records (usually older NHS versions). NB this may be because the statement does not exist, or because the patient has failed to circle any option(s), or because the patient has circled one or more negative options Ensure the No Contact for Other Studies box has auto-ticked if the consent form does not specifically permit OMB to contact the patient for future studies Ensure the No Further Follow-up box has auto-ticked if the consent form does not specifically permit OMB to follow the patient up On the Procedure Details tab, enter the following data: The Clinician involved in the expected or actual procedure, if known The Expected Side, if known and available (or the Actual Side, if more appropriate) The Expected Procedure, if known and available (or the Actual Procedure, if more appropriate) The Expected Diagnosis, if known and available (or the Confirmed Diagnosis, if known) OMB-MSOP 019 Version 1.18 (Draft) Page 9 of 15
10 The remaining tabs are for recording other documentation and data available, and they should be completed as the information is added to the folder (usually at a later date) Click the Save button to commit the record to the database, or use your keyboard s Esc key to undo changes prior to closing the form. 6.5 Searching the OMB Patient Monitor Database Click the box representing the field that you wish to search Click the binoculars button Enter the search criterion into the Find What box, and click the Find Next button. NB a much broader search capability may be attained by selecting OMB Master Form from the Look In dropdown box and/or changing the value in the Match box Watch for changes being displayed in the form in the background. If a record is found, check that the details match those you are expecting, e.g. procedure. (You may switch data tabs with the Find window still open.) If there is not sufficient matching, click the Find Next button again Repeat the previous step until there is sufficient matching, or until a not found message is displayed Close the search form. 6.6 Updating Withdrawal, Deceased, and Follow-Up/Contact Preferences If a patient indicates that s/he wishes no further follow-up, tick the No Further Follow-up tick box, and record the pedigree of this information in the consent notes (e.g. the date and a reference to a phone-call) If a patient indicated that s/he wishes no further contact for other studies, tick the No Contact for Other Studies tick box, and record the pedigree of this information in the consent notes (e.g. the date and a reference to a phone-call). OMB-MSOP 019 Version 1.18 (Draft) Page 10 of 15
11 6.6.3 If a patient withdraws consent, enter the date of withdrawal and initials of the OMB Team member recording the withdrawal in the boxes provided by the main database form. You must then follow OMB-MSOP 024 (Withdrawal of Donor Consent) These features ensure that patients wishes are respected If a patient is deceased, tick the Deceased tick box and ensure that the No Further Follow-up and No Contact For Other Studies tick boxes are also ticked. Also record the pedigree of this information in the consent notes (e.g. the date and a reference to a phone-call) In all cases, file any provided documentation in the relevant OMB Patient Folder(s). If no documentation is available, best practice is to file your own dated note as a back-up to the electronic record. 6.7 Updating Procedure Details NB This is essential for correct follow-up of patients Click the Procedure Details tab Update the fields as follows: Actual Side the side of the procedure actually performed on the patient Actual Procedure the name of the procedure actually performed on the patient Confirmed Diagnosis the diagnosis that was actually given Actual Admission Date the date of actual operative admission Procedure Comments e.g. reason why operation was not performed If the Actual Procedure has changed from the Expected Procedure, you must ensure that the Category matches the Actual Procedure. Even if the project for which the patient was consented has not asked for follow-up, this is very important to avoid OMB sending out incorrect letters and questionnaires. OMB-MSOP 019 Version 1.18 (Draft) Page 11 of 15
12 6.8 Updating OMB Follow-Up Attempts This is covered by OMB-MSOP 029 (Initiation and Receipt of Follow-ups). 6.9 Duplicated Consents If you find a duplicate OMB consent for the same patient-side-joint, consult the OMB Manager for a decision on whether/how to merge OMB Patient Folders Folders from other studies monitored by the OMB Patient Monitor database must never be merged under any circumstances As a guide to merging decisions: If both folders refer to collected tissue or follow-ups, record the duplication of consent in the OMB Patient Monitor, stating that all future references should be made to [the newer OMB Number], and earlier references may exist as [the older OMB Number]. Place a sheet of paper stating the same principle into each folder. In the case of the older OMB Number folder, this sheet should be at the very front to prompt users not to add to the folder again. (This is equal folder precedence.) If one folder refers to collected tissue or follow-ups, but the other refers to neither, fully merge the documentation of the no-tissue/follow-ups folder into the documentation of the with-tissue/follow-ups folder. Retain the empty folder. Record the merger in both OMB Numbers in the OMB Patient Monitor database, and state its reason. Place a sheet stating the merger and its reason into both the newly merged folder and the newly empty folder. (This is folder content precedence.) If neither folder refers to collected tissue or follow-ups, and one consent is on a more recent consent version than the other, merge the earlier folder into the later folder. Record the merger in both OMB Numbers in the OMB Patient Monitor database, and state its reason. Place a sheet stating the merger and its reason into both the newly merged folder and the newly empty folder. (This is consent version precedence.) OMB-MSOP 019 Version 1.18 (Draft) Page 12 of 15
13 If neither folder refers to collected tissue or follow-ups, and both consents are on the same version, merge the later folder into the earlier one. Record the merger in both OMB Numbers in the OMB Patient Monitor database, and state its reason. Place a sheet stating the merger and its reason into both the newly merged folder and the newly empty folder. (This is consent date precedence.) Any variation from the above policies should be recorded as a Non-Conformity, as per OMB-MSOP 014, and a copy of the resulting note placed in both folders. 7 Implementation 7.1 This SOP describes the procedure for setting up an OMB Patient Folder and registering it on the database, and for updating the database in certain circumstances. 7.2 Although not affecting the actual OMB-critical data and tissue, this SOP describes how OMB intitiates the mechanism tissue/data-consent linking more rapidly than by paper archive alone. This SOP may therefore be referenced by SOPs covering a wide variety of scenarios, ranging from standard OMB samples to imported diagnostic samples, hosted samples to non-critical data. 7.3 Care should be taken when modifying this SOP to ensure that it still works in all situations. 8 Training Requirements 8.1 Basic office and computer training. Computer skills may be enhanced through course available from the University of Oxford or the OUH NHS Trust. OMB-MSOP 019 Version 1.18 (Draft) Page 13 of 15
14 9 Staff Record Of Acknowledgement 9.1 I understand the contents of this document. 9.2 I have received the training appropriate to the procedures and feel competent to undertake them. 9.3 My supervisor agrees that I am able to perform the work covered by this SOP. 9.4 I understand that further on the job or other training supervision may be required before working independently. 9.5 I understand that I may discuss my needs with my manager. Trainee Trainer (if applicable) Name Position Signature Date Signature Date OMB-MSOP 019 Version 1.18 (Draft) Page 14 of 15
15 OMB-MSOP 019 Version 1.18 (Draft) Page 15 of 15
Prepare for Spectator Events
 Unit Y501 Prepare for Spectator Events This unit is about preparing yourself as a steward and checking the facility, route or venue for hazards before an event. Preparing for spectator events is an essential
Unit Y501 Prepare for Spectator Events This unit is about preparing yourself as a steward and checking the facility, route or venue for hazards before an event. Preparing for spectator events is an essential
Version Approver: James Edwards Version Approval Date: 25/04/2012. Version Author: Huma Zafar (BMS) Version Date: 20/02/2012. No.
 Title: Processing of Urine Samples Serial Number: OMB-LSOP 036 Version Number: 1.0 Version Approver: James Edwards Version Approval Date: 25/04/2012 Version Effective: Two weeks after release Version Author:
Title: Processing of Urine Samples Serial Number: OMB-LSOP 036 Version Number: 1.0 Version Approver: James Edwards Version Approval Date: 25/04/2012 Version Effective: Two weeks after release Version Author:
PARTNERSHIP GRANT REQUEST FOR PROPOSAL REQUIREMENTS
 PARTNERSHIP GRANT REQUEST FOR PROPOSAL REQUIREMENTS I. BACKGROUND INFORMATION The State Budget Act allocates funds to the Equal Access Fund to improve equal access and the fair administration of justice.
PARTNERSHIP GRANT REQUEST FOR PROPOSAL REQUIREMENTS I. BACKGROUND INFORMATION The State Budget Act allocates funds to the Equal Access Fund to improve equal access and the fair administration of justice.
Using Your PSAT/NMSQT Scores to Increase College Readiness
 Using Your PSAT/NMSQT Scores to Increase College Readiness How Do I Access My PSAT/NMSQT Scores and Reports? 1. Review your Online reports 2. Review Your Paper Score Report 2 How Do I Access My Online
Using Your PSAT/NMSQT Scores to Increase College Readiness How Do I Access My PSAT/NMSQT Scores and Reports? 1. Review your Online reports 2. Review Your Paper Score Report 2 How Do I Access My Online
Alberta Health Services. PCS 5.67 Care Planning
 Alberta Health Services PCS 5.67 Care Planning 3/11/2015 Contents Care Planning in Central Zone... 5 Developing the Plan of Care... 7 Accessing the RAP Analysis Assessments... 8 Completing the RAP Analysis
Alberta Health Services PCS 5.67 Care Planning 3/11/2015 Contents Care Planning in Central Zone... 5 Developing the Plan of Care... 7 Accessing the RAP Analysis Assessments... 8 Completing the RAP Analysis
Surgical Appliance Walk-in patients
 APS02 Version 3.0 Appliance Services Operational Areas Included HCA Roles Responsible for Carrying out this Process All Other Areas Operational Areas Excluded GEN01 Logging into Lorenzo Associated Procedures
APS02 Version 3.0 Appliance Services Operational Areas Included HCA Roles Responsible for Carrying out this Process All Other Areas Operational Areas Excluded GEN01 Logging into Lorenzo Associated Procedures
RD SOP12 Research Passport Honorary Contracts / Letters of Access
 RD SOP12 Research Passport Honorary Contracts / Letters of Access Version Number: V2.1 Name of originator/author: Dr Andy Mee, R&I Manager Name of responsible committee: R&I Committee Name of executive
RD SOP12 Research Passport Honorary Contracts / Letters of Access Version Number: V2.1 Name of originator/author: Dr Andy Mee, R&I Manager Name of responsible committee: R&I Committee Name of executive
CONTENTS MESSAGE FROM THE CHAIRMAN WFDSA DIRECT SELLING WHY DIRECT SELLING GOVERNANCE STRATEGIC INITIATIVES ADVOCACY INITIATIVES
 ANNUAL REPORT 2017 CONTENTS MESSAGE FROM THE CHAIRMAN WFDSA DIRECT SELLING WHY DIRECT SELLING GOVERNANCE STRATEGIC INITIATIVES ADVOCACY INITIATIVES ASSOCIATION SERVICES INITIATIVES GLOBAL RESEARCH 2 WFDSA
ANNUAL REPORT 2017 CONTENTS MESSAGE FROM THE CHAIRMAN WFDSA DIRECT SELLING WHY DIRECT SELLING GOVERNANCE STRATEGIC INITIATIVES ADVOCACY INITIATIVES ASSOCIATION SERVICES INITIATIVES GLOBAL RESEARCH 2 WFDSA
Hello. Dan Bacchetta danbacchetta.com
 Hello Dan Bacchetta danbacchetta.com dmbacchetta@gmail.com PROJECT 2015 KCS Sports Awards - Voting Lead Designer Having a limited timeline before voting was open, the team and I streamlined the designs
Hello Dan Bacchetta danbacchetta.com dmbacchetta@gmail.com PROJECT 2015 KCS Sports Awards - Voting Lead Designer Having a limited timeline before voting was open, the team and I streamlined the designs
CLINICAL IMAGING REFERRAL PROTOCOL FOR REGISTERED NURSE PRACTITIONERS IN THE EMERGENCY DEPARTMENT, URGENT CARE CENTRE AND AMBULATORY CARE
 CLINICAL IMAGING REFERRAL PROTOCOL FOR REGISTERED NURSE PRACTITIONERS IN THE EMERGENCY DEPARTMENT, URGENT CARE CENTRE AND AMBULATORY CARE CLINICAL GUIDELINE V4. Summary. Start The non-medical practitioner
CLINICAL IMAGING REFERRAL PROTOCOL FOR REGISTERED NURSE PRACTITIONERS IN THE EMERGENCY DEPARTMENT, URGENT CARE CENTRE AND AMBULATORY CARE CLINICAL GUIDELINE V4. Summary. Start The non-medical practitioner
ACTIONS/PSOP/001 Version 1.0 Page 2 of 6
 1. The purpose of the Pharmacy Site File To enable the designated trust pharmacy to fulfil its role and exercise appropriate control over all aspects of study medication handling, an accurately maintained
1. The purpose of the Pharmacy Site File To enable the designated trust pharmacy to fulfil its role and exercise appropriate control over all aspects of study medication handling, an accurately maintained
Use the following to enter new patients into Horizon and to establish a patient for a pending admission. All referrals will be entered into Horizon.
 REFFERAL AND INTAKE SUMMARY Use the following to enter new patients into Horizon and to establish a patient for a pending admission. All referrals will be entered into Horizon. ROLES Supervisor/Nurse The
REFFERAL AND INTAKE SUMMARY Use the following to enter new patients into Horizon and to establish a patient for a pending admission. All referrals will be entered into Horizon. ROLES Supervisor/Nurse The
MEMBER HANDBOOK & BENEFITS GUIDE
 . n nt.i. ur e O et m R. r r lve u o Yoinv on MEMBER HANDBOOK & BENEFITS GUIDE WWW.TAMPABAYBEACHES.COM M ISSION The mission of your Tampa Bay Beaches Chamber of Commerce is to educate businesses and individuals,
. n nt.i. ur e O et m R. r r lve u o Yoinv on MEMBER HANDBOOK & BENEFITS GUIDE WWW.TAMPABAYBEACHES.COM M ISSION The mission of your Tampa Bay Beaches Chamber of Commerce is to educate businesses and individuals,
"The 40+ project: The GameChangers" 23/02/2017
 "The 40+ project: The GameChangers" 23/02/2017 Who we are The Advisory Council Adelina von Fürstenberg President of the Advisory Council https://en.wikipedia.org/wiki/adelina_von_f%c3%bcrstenberg Awarded
"The 40+ project: The GameChangers" 23/02/2017 Who we are The Advisory Council Adelina von Fürstenberg President of the Advisory Council https://en.wikipedia.org/wiki/adelina_von_f%c3%bcrstenberg Awarded
Informed Consent SOP Number: 25 Version Number: 6.0 Effective Date: 1 st September 2017 Review Date: 1 st September 2019
 Standard Operating Procedures (SOP) for: Informed Consent SOP Number: 25 Version Number: 6.0 Effective Date: 1 st September 2017 Review Date: 1 st September 2019 Author: Reviewer: Reviewer: Authorisation:
Standard Operating Procedures (SOP) for: Informed Consent SOP Number: 25 Version Number: 6.0 Effective Date: 1 st September 2017 Review Date: 1 st September 2019 Author: Reviewer: Reviewer: Authorisation:
DESTRUCTION AND RETENTION OF CLINICAL HEALTH RECORDS POLICY
 Directorate of Operations Central Operations Group Corporate Library Services DESTRUCTION AND RETENTION OF CLINICAL HEALTH RECORDS POLICY Reference: OPP023 Version: 1.7 This version issued: 02/05/12 Result
Directorate of Operations Central Operations Group Corporate Library Services DESTRUCTION AND RETENTION OF CLINICAL HEALTH RECORDS POLICY Reference: OPP023 Version: 1.7 This version issued: 02/05/12 Result
How to review applications in NHS Jobs
 How to review applications in NHS Jobs Click on Vacancies Click on the applications link under the vacancy you are shortlisting To Request reviews from Colleagues If you would like your colleagues to review
How to review applications in NHS Jobs Click on Vacancies Click on the applications link under the vacancy you are shortlisting To Request reviews from Colleagues If you would like your colleagues to review
Booking Elective Trauma Surgery for Inpatients
 ADT31 Version 3.1 Trauma Team Operational Areas Included Trauma Co-ordinator Roles Responsible for Carrying out this Process All other areas Operational Areas Excluded GEN01 Logging into Lorenzo GEN02
ADT31 Version 3.1 Trauma Team Operational Areas Included Trauma Co-ordinator Roles Responsible for Carrying out this Process All other areas Operational Areas Excluded GEN01 Logging into Lorenzo GEN02
MARITIME AND PORT SECTORS IN THE AMERICAS: CHALLENGES AND OPPORTUNITIES IN LIGHT OF THE SDG S
 Dirección General de Capitanías y Guardacostas del Perú (DICAPI) and the World Maritime University WMU Regional Conference for the Americas Lima, Peru 23-25 October, 2017 MARITIME AND PORT SECTORS IN THE
Dirección General de Capitanías y Guardacostas del Perú (DICAPI) and the World Maritime University WMU Regional Conference for the Americas Lima, Peru 23-25 October, 2017 MARITIME AND PORT SECTORS IN THE
Access to Health Records Procedure
 Access to Health Records Procedure Version: 1.0 Ratified by: Date ratified: 11/03/2015 Name of originator/author: Name of responsible individual: Information Governance Group Medical Records Manager, Jackie
Access to Health Records Procedure Version: 1.0 Ratified by: Date ratified: 11/03/2015 Name of originator/author: Name of responsible individual: Information Governance Group Medical Records Manager, Jackie
Standard Operating Procedure (SOP) Research and Development Office
 Standard Operating Procedure (SOP) Research and Development Office Title of SOP: Routine Project Audit SOP Number: 6 Version Number: 2.0 Supercedes: 1.0 Effective date: August 2013 Review date: August
Standard Operating Procedure (SOP) Research and Development Office Title of SOP: Routine Project Audit SOP Number: 6 Version Number: 2.0 Supercedes: 1.0 Effective date: August 2013 Review date: August
Handover of Care (Maternity) Guidelines Author s job title Lead Clinical Midwife Department Ladywell Unit. Comment / Changes / Approval
 Document Control Title Author Directorate Surgery Date Version Issued 0.1 Oct 2009 0.2 Nov 2009 1.0 Nov 2009 1.1 Feb 2010 2.0 Feb 2010 2.1 Aug 2011 2.2 Oct 2011 Handover of Care (Maternity) Guidelines
Document Control Title Author Directorate Surgery Date Version Issued 0.1 Oct 2009 0.2 Nov 2009 1.0 Nov 2009 1.1 Feb 2010 2.0 Feb 2010 2.1 Aug 2011 2.2 Oct 2011 Handover of Care (Maternity) Guidelines
CHRISP STREET HEALTH CENTRE EMIS WEB IN-HOUSE MODULAR TRAINING
 CHRISP STREET HEALTH CENTRE EMIS WEB IN-HOUSE MODULAR TRAINING Module 2 Finding Patients As you all are aware we are planning to go live with EMIS Web on Wednesday 12 th October 2011. To prepare for this
CHRISP STREET HEALTH CENTRE EMIS WEB IN-HOUSE MODULAR TRAINING Module 2 Finding Patients As you all are aware we are planning to go live with EMIS Web on Wednesday 12 th October 2011. To prepare for this
CLINICAL GUIDELINE FOR CLINICAL IMAGING REFERRAL PROTOCOL FOR NURSE SPECIALISTS IN HEART FUNCTION WITHIN RCHT Summary. Start
 CLINICAL GUIDELINE FOR CLINICAL IMAGING REFERRAL PROTOCOL FOR NURSE SPECIALISTS IN HEART FUNCTION WITHIN RCHT Summary. Start The non-medical practitioner has received sufficient training to make clinical
CLINICAL GUIDELINE FOR CLINICAL IMAGING REFERRAL PROTOCOL FOR NURSE SPECIALISTS IN HEART FUNCTION WITHIN RCHT Summary. Start The non-medical practitioner has received sufficient training to make clinical
Data Entry onto the National Immunoglobulin Database
 number SCOPE RESPONSIBILITY NHS enter board name here Pharmaceutical Service Populate the National immunoglobulin Database Lead Procurement Officer/Senior Technician Enter local details Data Entry onto
number SCOPE RESPONSIBILITY NHS enter board name here Pharmaceutical Service Populate the National immunoglobulin Database Lead Procurement Officer/Senior Technician Enter local details Data Entry onto
Dear Colleague. Performers List National Application Arrangements. Summary
 NHS Circular: PCA(M)(2016)(4) Directorate for Population Health Primary Care Division Dear Colleague Performers List National Application Arrangements Summary 1. This Circular directs 1 NHS Boards in relation
NHS Circular: PCA(M)(2016)(4) Directorate for Population Health Primary Care Division Dear Colleague Performers List National Application Arrangements Summary 1. This Circular directs 1 NHS Boards in relation
MHRA Findings Dissemination Joint Office Launch Jan Presented by: Carolyn Maloney UHL R&D Manager
 MHRA Findings Dissemination Joint Office Launch Jan. 2012 Presented by: Carolyn Maloney UHL R&D Manager Purpose of presentation To feed back abridged findings from March 2011 MHRA Statutory Systems Inspection
MHRA Findings Dissemination Joint Office Launch Jan. 2012 Presented by: Carolyn Maloney UHL R&D Manager Purpose of presentation To feed back abridged findings from March 2011 MHRA Statutory Systems Inspection
Hi Tech Software Solutions Are You Still Handwriting Care Plans?
 Are You Still Handwriting Care Plans? Care Plans/Service Plans... 2 Overview... 2 Edit Care Plan Edit Service Plan... 4 Auto RAP/CAA Driven (for Nursing Care)... 5 Auto RAP/CAA Driven: Edit Resident Care
Are You Still Handwriting Care Plans? Care Plans/Service Plans... 2 Overview... 2 Edit Care Plan Edit Service Plan... 4 Auto RAP/CAA Driven (for Nursing Care)... 5 Auto RAP/CAA Driven: Edit Resident Care
Document Title: Study Data SOP (CRFs and Source Data)
 Document Title: Study Data SOP (CRFs and Source Data) Document Number: SOP047 Staff involved in development: Job titles only Document author/owner: Directorate: Department: For use by: RM&G Manager, R&D
Document Title: Study Data SOP (CRFs and Source Data) Document Number: SOP047 Staff involved in development: Job titles only Document author/owner: Directorate: Department: For use by: RM&G Manager, R&D
Guideline Complex Care Management Documentation in Valence Care Manager
 Title: RN, Complex Care Management Purpose: Provide consistent and systematic documentation of all contact with Complex Care Management cases. Scope: Complex Care Management referrals, encounters, enrollments,
Title: RN, Complex Care Management Purpose: Provide consistent and systematic documentation of all contact with Complex Care Management cases. Scope: Complex Care Management referrals, encounters, enrollments,
Please upload a copy of your 501(c)(3) status OR a letter of agreement and 501(c)(3) documentation from your fiscal agent.
 SECTION 1: Eligibility Requirements About your organization My organization is a current non-profit 501(c)(3) Please upload a copy of your 501(c)(3) status OR a letter of agreement and 501(c)(3) documentation
SECTION 1: Eligibility Requirements About your organization My organization is a current non-profit 501(c)(3) Please upload a copy of your 501(c)(3) status OR a letter of agreement and 501(c)(3) documentation
ANTI-COAGULATION MONITORING
 ANTI-COAGULATION MONITORING 2016-17 a) Purpose of Agreement This Agreement outlines the service to be provided by the Provider, called an Anti-coagulation monitoring service. b) Duration of Agreement This
ANTI-COAGULATION MONITORING 2016-17 a) Purpose of Agreement This Agreement outlines the service to be provided by the Provider, called an Anti-coagulation monitoring service. b) Duration of Agreement This
SUPPORTING DATA QUALITY NJR STRATEGY 2014/16
 SUPPORTING DATA QUALITY NJR STRATEGY 2014/16 CONTENTS Supporting data quality 2 Introduction 2 Aim 3 Governance 3 Overview: NJR-healthcare provider responsibilities 3 Understanding current 4 data quality
SUPPORTING DATA QUALITY NJR STRATEGY 2014/16 CONTENTS Supporting data quality 2 Introduction 2 Aim 3 Governance 3 Overview: NJR-healthcare provider responsibilities 3 Understanding current 4 data quality
A step by step guide to using IRAS to apply to conduct research in or through the NHS/HSC.
 A step by step guide to using IRAS to apply to conduct research in or through the NHS/HSC. 28 June 2018 Summary All project-based research conducted in or through the NHS/HSC in England, Northern Ireland,
A step by step guide to using IRAS to apply to conduct research in or through the NHS/HSC. 28 June 2018 Summary All project-based research conducted in or through the NHS/HSC in England, Northern Ireland,
Document Title: Informed Consent for Research Studies
 Document Title: Informed Consent for Research Studies Document Number: SOP003 Staff involved in development: Job titles only Document author/owner: Directorate: Department: For use by: RM&G Manager, R&D
Document Title: Informed Consent for Research Studies Document Number: SOP003 Staff involved in development: Job titles only Document author/owner: Directorate: Department: For use by: RM&G Manager, R&D
HCAI Data Capture System User Manual. Case Capture: Main Data Collections
 User Manual Case Capture: Main Data Collections About Public Health England Public Health England exists to protect and improve the nation's health and wellbeing, and reduce health inequalities. It does
User Manual Case Capture: Main Data Collections About Public Health England Public Health England exists to protect and improve the nation's health and wellbeing, and reduce health inequalities. It does
Trial set-up, conduct and Trial Master File for HEY-sponsored CTIMPs
 R&D Department Trial set-up, conduct and Trial Master File for HEY-sponsored CTIMPs Hull And East Yorkshire Hospitals NHS Trust 2010 All Rights Reserved No part of this document may be reproduced, stored
R&D Department Trial set-up, conduct and Trial Master File for HEY-sponsored CTIMPs Hull And East Yorkshire Hospitals NHS Trust 2010 All Rights Reserved No part of this document may be reproduced, stored
Applicant Reviewer Quick Guide
 Applicant Reviewer Quick Guide Creating a Hiring Proposal January 22, 2016 Contents Applicant Reviewer... 3 Logging In... 3 Changing User Groups... 3 Viewing Postings... 4 Viewing Applicants... 4 Hiring
Applicant Reviewer Quick Guide Creating a Hiring Proposal January 22, 2016 Contents Applicant Reviewer... 3 Logging In... 3 Changing User Groups... 3 Viewing Postings... 4 Viewing Applicants... 4 Hiring
HELLO HEALTH TRAINING MANUAL
 HELLO HEALTH TRAINING MANUAL Please note: As with all training materials, the names and data used in this training manual are purely fictitious and for information and training purposes only Login/What
HELLO HEALTH TRAINING MANUAL Please note: As with all training materials, the names and data used in this training manual are purely fictitious and for information and training purposes only Login/What
Certification of Employee Time and Effort
 Procedure: Policy: Number: Completing a Personnel Activity Report (PAR) Certification of Employee Time and Effort GP1200.3 ( ) Complete Revision Supersedes: Page: ( ) Partial Revision Page 1 of 21 ( X
Procedure: Policy: Number: Completing a Personnel Activity Report (PAR) Certification of Employee Time and Effort GP1200.3 ( ) Complete Revision Supersedes: Page: ( ) Partial Revision Page 1 of 21 ( X
MGH Research Lab Orders Date: 08/10/2016
 MGH Research Lab Orders Date: 08/10/2016 Try It Out MGH Ordering Research Labs A. Research Coordinator Draws Blood (for Research) Scenario #1- No SQ printer and No Clinical Visit MGH Lab processing and
MGH Research Lab Orders Date: 08/10/2016 Try It Out MGH Ordering Research Labs A. Research Coordinator Draws Blood (for Research) Scenario #1- No SQ printer and No Clinical Visit MGH Lab processing and
Office of Clinical Research. CTMS Reference Guide Patient Entry & Visit Tracking
 Se Office of Clinical Research CTMS Reference Guide Patient Entry & Visit Tracking Table of Contents Logging into CTMS... 3 Search and Recruitment / Quick Search... 4 How to Configure Quick Search Fields...
Se Office of Clinical Research CTMS Reference Guide Patient Entry & Visit Tracking Table of Contents Logging into CTMS... 3 Search and Recruitment / Quick Search... 4 How to Configure Quick Search Fields...
PROGRAMMES IMPLEMENTATION PLATFORM (PIP) Community Childcare Subvention 2015/2016 HOW TO GUIDE
 PROGRAMMES IMPLEMENTATION PLATFORM (PIP) Community Childcare Subvention 2015/2016 HOW TO GUIDE Release Date 11/01/2016 1 Contents 1. Logging-in to the PIP Portal... 4 1.1 How to log-in to the PIP Portal...
PROGRAMMES IMPLEMENTATION PLATFORM (PIP) Community Childcare Subvention 2015/2016 HOW TO GUIDE Release Date 11/01/2016 1 Contents 1. Logging-in to the PIP Portal... 4 1.1 How to log-in to the PIP Portal...
Gaining NHS Trust R&D Approvals
 Version 1.1 Effective date: 1 October 2012 Author: Approved by: Claire Daffern, QA Manager Dr Sarah Duggan, CTU Manager Revision Chronology: Effective Date Reason for change Version 1.1 1 October 2012
Version 1.1 Effective date: 1 October 2012 Author: Approved by: Claire Daffern, QA Manager Dr Sarah Duggan, CTU Manager Revision Chronology: Effective Date Reason for change Version 1.1 1 October 2012
The Clinical Research Center Research Practice Manual. Guideline for Study Document and Data Handling RPG-08. Guideline. Purpose.
 The Clinical Research Center Research Practice Manual Guideline for Study Document and Data Handling RPG-08 Purpose Guideline This Guideline provides functional definitions for common data management terms;
The Clinical Research Center Research Practice Manual Guideline for Study Document and Data Handling RPG-08 Purpose Guideline This Guideline provides functional definitions for common data management terms;
Registrations 2017/18
 Registrations 2017/18 A guide for centre administrators In this guide you will find information on how to create groups and upload files for registrations, add students to existing groups, and view your
Registrations 2017/18 A guide for centre administrators In this guide you will find information on how to create groups and upload files for registrations, add students to existing groups, and view your
A list of authorised referrers will be retained by the Colposcopy team and the Clinical Imaging Department.
 Clinical Guideline for Clinical Imaging Referral Protocol for Nurse Colposcopist within Colposcopy Dept. Royal Cornwall Hospital 1. Aim/Purpose of this Guideline 1.1 This protocol applies to Nurse Colposcopist
Clinical Guideline for Clinical Imaging Referral Protocol for Nurse Colposcopist within Colposcopy Dept. Royal Cornwall Hospital 1. Aim/Purpose of this Guideline 1.1 This protocol applies to Nurse Colposcopist
NHS Summary Care Record. Guide for GP Practice Staff
 NHS Summary Care Record Guide for GP Practice Staff NHS Summary Care Record Guide for GP Practice Staff v1.2 October 2012 Table of Contents 1 Introduction to this guide...3 2 Overview of the Summary Care
NHS Summary Care Record Guide for GP Practice Staff NHS Summary Care Record Guide for GP Practice Staff v1.2 October 2012 Table of Contents 1 Introduction to this guide...3 2 Overview of the Summary Care
Diagnostic Testing Procedures for Ophthalmic Science
 V4.0 01/08/17 Table of Contents 1. Introduction... 3 2. Purpose of this Policy... 3 3. Scope... 3 4. Definitions / Glossary... 3 5. Ownership and Responsibilities... 3 5.2. Role of the Managers... 3 5.3.
V4.0 01/08/17 Table of Contents 1. Introduction... 3 2. Purpose of this Policy... 3 3. Scope... 3 4. Definitions / Glossary... 3 5. Ownership and Responsibilities... 3 5.2. Role of the Managers... 3 5.3.
EMDR AUSTRALIA ACCREDITED PRACTITIONER COMPETENCY BASED FRAMEWORK
 EMDR AUSTRALIA ACCREDITED PRACTITIONER COMPETENCY BASED FRAMEWORK APPLICATION FORM TO BE RETURNED TO: EMDR Association of Australia Email Address accred@emdraa.org Section I: Section II: Section III: Section
EMDR AUSTRALIA ACCREDITED PRACTITIONER COMPETENCY BASED FRAMEWORK APPLICATION FORM TO BE RETURNED TO: EMDR Association of Australia Email Address accred@emdraa.org Section I: Section II: Section III: Section
Go! Guide: Adding Medication Administration History
 Go! Guide: Adding Medication Administration History Introduction Past medication administrations are often an integral part of a patient scenario. It may be important for students to review the patient
Go! Guide: Adding Medication Administration History Introduction Past medication administrations are often an integral part of a patient scenario. It may be important for students to review the patient
MEDICAL SPECIALISTS OF THE PALM BEACHES, INC. Chronic Care Management (CCM) Program Training Manual
 MEDICAL SPECIALISTS OF THE PALM BEACHES, INC. Chronic Care Management (CCM) Program Training Manual September 2017 Table of Contents CCM PROGRAM OVERVIEW... 4 3 STEPS TO BEGIN CCM:... 5 Identify the Patient...
MEDICAL SPECIALISTS OF THE PALM BEACHES, INC. Chronic Care Management (CCM) Program Training Manual September 2017 Table of Contents CCM PROGRAM OVERVIEW... 4 3 STEPS TO BEGIN CCM:... 5 Identify the Patient...
COMMUNITY PHARMACY MINOR AILMENTS SERVICE
 COMMUNITY PHARMACY MINOR AILMENTS SERVICE SUPPORTING SELF-CARE OCTOBER 2010 CONTENTS Index Page No 1 Introduction 3 2 Service Specification 4 3 Consultation Procedure 7 4 Re-ordering Documentation 10 Appendices
COMMUNITY PHARMACY MINOR AILMENTS SERVICE SUPPORTING SELF-CARE OCTOBER 2010 CONTENTS Index Page No 1 Introduction 3 2 Service Specification 4 3 Consultation Procedure 7 4 Re-ordering Documentation 10 Appendices
Care360 EHR Frequently Asked Questions
 Care360 EHR Frequently Asked Questions Table of Contents Care360 EHR... 4 What is Care360 EHR?... 4 What are the current capabilities of Care 360 EHR?... 4 Is Care 360 EHR an EMR?... 5 Can I have Care360
Care360 EHR Frequently Asked Questions Table of Contents Care360 EHR... 4 What is Care360 EHR?... 4 What are the current capabilities of Care 360 EHR?... 4 Is Care 360 EHR an EMR?... 5 Can I have Care360
Care Manager Guide SPIRIT CMTS. Care Management Tracking System. University of Washington aims.uw.edu
 Care Manager Guide SPIRIT CMTS Care Management Tracking System University of Washington aims.uw.edu rev. 12/4/2017 Table of Contents TOP TIPS & TRICKS... 1 INTRODUCTION... 2 CARE MANAGER ACCOUNT ROLE...
Care Manager Guide SPIRIT CMTS Care Management Tracking System University of Washington aims.uw.edu rev. 12/4/2017 Table of Contents TOP TIPS & TRICKS... 1 INTRODUCTION... 2 CARE MANAGER ACCOUNT ROLE...
Consultation on proposals to introduce independent prescribing by radiographers across the United Kingdom
 Consultation on proposals to introduce independent prescribing by radiographers across the United Kingdom Response by the Royal College of Radiologists (RCR) The RCR is the UK professional body for the
Consultation on proposals to introduce independent prescribing by radiographers across the United Kingdom Response by the Royal College of Radiologists (RCR) The RCR is the UK professional body for the
MANAGEMENT OF PROTOCOL AND GCP DEVIATIONS AND VIOLATIONS
 MANAGEMENT OF PROTOCOL AND GCP DEVIATIONS AND VIOLATIONS DOCUMENT NO.: CR010 v4.0 AUTHOR: Heather Charles ISSUE DATE: 01 September 2016 EFFECTIVE DATE: 15 September 2016 1 INTRODUCTION 1.1 The Academic
MANAGEMENT OF PROTOCOL AND GCP DEVIATIONS AND VIOLATIONS DOCUMENT NO.: CR010 v4.0 AUTHOR: Heather Charles ISSUE DATE: 01 September 2016 EFFECTIVE DATE: 15 September 2016 1 INTRODUCTION 1.1 The Academic
QOF queries in SystmOne
 QOF queries in SystmOne For further help with QOF: 1. See the Primary Care Contracting (www.primarycarecontracting.nhs.uk) website for more information 2. Contact your PCT Information or Data Quality team
QOF queries in SystmOne For further help with QOF: 1. See the Primary Care Contracting (www.primarycarecontracting.nhs.uk) website for more information 2. Contact your PCT Information or Data Quality team
STANDARD OPERATING PROCEDURE SOP 715. Principles of Clinical Research Laboratory Practice
 STANDARD OPERATING PROCEDURE SOP 715 Principles of Clinical Research Laboratory Practice Version 1.2 Version date 13.11.2015 Effective date 24.04.2017 Number of pages 9 Review date June 2018 Author Role
STANDARD OPERATING PROCEDURE SOP 715 Principles of Clinical Research Laboratory Practice Version 1.2 Version date 13.11.2015 Effective date 24.04.2017 Number of pages 9 Review date June 2018 Author Role
Using PowerChart: Organizer View
 Slide Agenda Caption 3 1. Finding and logging into PowerChart 2. The Millennium Message Box 3. Toolbar Basics 4. The Organizer Toolbar 5. The Actions Toolbar 4 6. The Links toolbar 7. Patient Search Options
Slide Agenda Caption 3 1. Finding and logging into PowerChart 2. The Millennium Message Box 3. Toolbar Basics 4. The Organizer Toolbar 5. The Actions Toolbar 4 6. The Links toolbar 7. Patient Search Options
Level Two Provisional Accreditation As a Cognitive Behavioural Psychotherapist Criteria and Guidelines
 Level Two Provisional Accreditation As a Cognitive Behavioural Psychotherapist Criteria and Guidelines Provisional Accreditation as a Cognitive Behavioural Psychotherapist with BABCP means that the entry
Level Two Provisional Accreditation As a Cognitive Behavioural Psychotherapist Criteria and Guidelines Provisional Accreditation as a Cognitive Behavioural Psychotherapist with BABCP means that the entry
Irradiated blood products - Pathway for requesting To provide healthcare professionals with clear guidance on the use of irradiated blood products.
 Document Title: Document Purpose: Document Statement: Document Application: Responsible for Implementation: Irradiated blood products - Pathway for requesting To provide healthcare professionals with clear
Document Title: Document Purpose: Document Statement: Document Application: Responsible for Implementation: Irradiated blood products - Pathway for requesting To provide healthcare professionals with clear
Guidance for MRC units on HTA licence applications for storage of human samples for research purposes
 Guidance for MRC units on HTA licence applications for storage of human samples for research purposes Summary In England, Wales and Northern Ireland the Human Tissue Authority (HTA) is licensing premises
Guidance for MRC units on HTA licence applications for storage of human samples for research purposes Summary In England, Wales and Northern Ireland the Human Tissue Authority (HTA) is licensing premises
abcdefghijklmnopqrstu
 Directorate of Chief Medical Officer, Public Health and Sport abcdefghijklmnopqrstu T: 0131-244 2655 F: 0131-244 2285 E: craig.gilbert@scotland.gsi.gov.uk Dear Colleague ACCREDITATION SCHEME FOR THE COLLECTION
Directorate of Chief Medical Officer, Public Health and Sport abcdefghijklmnopqrstu T: 0131-244 2655 F: 0131-244 2285 E: craig.gilbert@scotland.gsi.gov.uk Dear Colleague ACCREDITATION SCHEME FOR THE COLLECTION
Guidance for organisations applying for both registration and licensing as a new service provider
 Guidance for organisations applying for both registration and licensing as a new service provider CQC and Monitor have combined the separate application forms to apply for a CQC registration and an NHS
Guidance for organisations applying for both registration and licensing as a new service provider CQC and Monitor have combined the separate application forms to apply for a CQC registration and an NHS
User Manual. MDAnalyze A Reference Guide
 User Manual MDAnalyze A Reference Guide Document Status The controlled master of this document is available on-line. Hard copies of this document are for information only and are not subject to document
User Manual MDAnalyze A Reference Guide Document Status The controlled master of this document is available on-line. Hard copies of this document are for information only and are not subject to document
MA/Office Staff: Proposing Surgical Procedure Orders and PowerPlans (Order Sets)
 Acute Surgical Procedure Orders and PowerPlans Affiliated MA/Office Staff: Proposing Surgical Procedure Orders and PowerPlans (Order Sets) This document walks you through: 1. Requesting a FIN (Financial
Acute Surgical Procedure Orders and PowerPlans Affiliated MA/Office Staff: Proposing Surgical Procedure Orders and PowerPlans (Order Sets) This document walks you through: 1. Requesting a FIN (Financial
POLICY ON THE HANDLING OF CHEMOTHERAPY BY STAFF WHO ARE PREGNANT OR BREASTFEEDING
 Policy on the handling of chemotherapy by staff who are pregnant/breastfeeding, v2.1 POLICY ON THE HANDLING OF CHEMOTHERAPY BY STAFF WHO ARE PREGNANT OR BREASTFEEDING Version: 2.1 Ratified by: Date ratified:
Policy on the handling of chemotherapy by staff who are pregnant/breastfeeding, v2.1 POLICY ON THE HANDLING OF CHEMOTHERAPY BY STAFF WHO ARE PREGNANT OR BREASTFEEDING Version: 2.1 Ratified by: Date ratified:
RETRIEVAL AND CRITICAL HEALTH INFORMATION SYSTEM
 RETRIEVAL AND CRITICAL HEALTH INFORMATION SYSTEM USER GUIDE November 2014 Contents Introduction... 4 Access to REACH... 4 Homepage... 4 Roles within REACH... 5 Hospital Administrator... 5 Hospital User...
RETRIEVAL AND CRITICAL HEALTH INFORMATION SYSTEM USER GUIDE November 2014 Contents Introduction... 4 Access to REACH... 4 Homepage... 4 Roles within REACH... 5 Hospital Administrator... 5 Hospital User...
Vetting and Barring Scheme and Independent Safeguarding Authority
 Vetting and Barring Scheme and Independent Safeguarding Authority Royal College of Nursing briefing Publication number: 003576 (March 2010) 1 Vetting and Barring Scheme and Independent Safeguarding Authority
Vetting and Barring Scheme and Independent Safeguarding Authority Royal College of Nursing briefing Publication number: 003576 (March 2010) 1 Vetting and Barring Scheme and Independent Safeguarding Authority
Application Grant Instructions
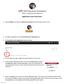 Application Grant Instructions 1. Log into MyKaty, and select the Education Foundation tab at the bottom of the screen. 2. To begin an application, click the Foundation Grant Application tile. 3. The Foundation
Application Grant Instructions 1. Log into MyKaty, and select the Education Foundation tab at the bottom of the screen. 2. To begin an application, click the Foundation Grant Application tile. 3. The Foundation
Policy for Patient Access
 Policy for Patient Access DOCUMENT CONTROL Revision Date Old Version 10/12/2014 1.0 01/07/2016 1.1 30/04/17 1.2 Amendment General Management Review General Management Review General Management Review Authored
Policy for Patient Access DOCUMENT CONTROL Revision Date Old Version 10/12/2014 1.0 01/07/2016 1.1 30/04/17 1.2 Amendment General Management Review General Management Review General Management Review Authored
Paragon Clinician Hub for Physicians (PCH) Reference
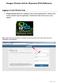 Paragon Clinician Hub for Physicians (PCH) Reference Logging in to the Clinician Hub Paragon Clinician Hub (PCH) is available on any Carroll Hospital Network. VMWare View must be utilized to open the application.
Paragon Clinician Hub for Physicians (PCH) Reference Logging in to the Clinician Hub Paragon Clinician Hub (PCH) is available on any Carroll Hospital Network. VMWare View must be utilized to open the application.
Clinical Practice Guideline Development Manual
 Clinical Practice Guideline Development Manual Publication Date: September 2016 Review Date: September 2021 Table of Contents 1. Background... 3 2. NICE accreditation... 3 3. Patient Involvement... 3 4.
Clinical Practice Guideline Development Manual Publication Date: September 2016 Review Date: September 2021 Table of Contents 1. Background... 3 2. NICE accreditation... 3 3. Patient Involvement... 3 4.
Announced Inspection Report care for older people in acute hospitals
 Announced Inspection Report care for older people in acute hospitals Hairmyres Hospital NHS Lanarkshire Healthcare Improvement Scotland is committed to equality. We have assessed the inspection function
Announced Inspection Report care for older people in acute hospitals Hairmyres Hospital NHS Lanarkshire Healthcare Improvement Scotland is committed to equality. We have assessed the inspection function
RAPID. Health SOFTWARE PHYSICIAN REFERRAL. Call:
 Health RAPID SOFTWARE PHYSICIAN REFERRAL www.acomhealth.com Call: 866.286.5315 Email: acomhealth@acom.com 2455 Meadowbrook Pkwy NW, Duluth, GA 30096 Tel: (866) 286-5315 Fax: (770) 814-7011 email: rapidsupport@acom.com
Health RAPID SOFTWARE PHYSICIAN REFERRAL www.acomhealth.com Call: 866.286.5315 Email: acomhealth@acom.com 2455 Meadowbrook Pkwy NW, Duluth, GA 30096 Tel: (866) 286-5315 Fax: (770) 814-7011 email: rapidsupport@acom.com
Dr Jonathan Davies GMC Colleague Survey Results
 Dr Jonathan Davies GMC Colleague Survey Results Published: 09/07/2013 19/06/2013 2 SURVEY OVERVIEW 3 DESCRIPTION 3 INSTRUCTIONS PROVIDED TO RESPONDENTS 3 RESPONDENT METRICS 3 SURVEY RESULTS 4 SECTION -
Dr Jonathan Davies GMC Colleague Survey Results Published: 09/07/2013 19/06/2013 2 SURVEY OVERVIEW 3 DESCRIPTION 3 INSTRUCTIONS PROVIDED TO RESPONDENTS 3 RESPONDENT METRICS 3 SURVEY RESULTS 4 SECTION -
DANA-FARBER / HARVARD CANCER CENTER STANDARD OPERATING PROCEDURES FOR HUMAN SUBJECT RESEARCH
 SOP #: CON-100 Page: 1 of 9 Effective Date: 2/28/17 1. POLICY STATEMENT: The research team is responsible for obtaining and documenting the informed consent of each subject who participates in research.
SOP #: CON-100 Page: 1 of 9 Effective Date: 2/28/17 1. POLICY STATEMENT: The research team is responsible for obtaining and documenting the informed consent of each subject who participates in research.
RETRIEVAL AND CRITICAL HEALTH INFORMATION SYSTEM
 RETRIEVAL AND CRITICAL HEALTH INFORMATION SYSTEM USER GUIDE May 2017 Contents Introduction... 3 Access to REACH... 3 Homepage... 3 Roles within REACH... 4 Hospital Administrator... 4 Hospital User... 4
RETRIEVAL AND CRITICAL HEALTH INFORMATION SYSTEM USER GUIDE May 2017 Contents Introduction... 3 Access to REACH... 3 Homepage... 3 Roles within REACH... 4 Hospital Administrator... 4 Hospital User... 4
Review Student Applicants and Create Hiring Proposal
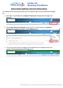 Review Student Applicants and Create Hiring Proposal **All applicant review and hiring proposals must be completed using the Student Department Manager role. 1. Upon login, user will default to the Applicant
Review Student Applicants and Create Hiring Proposal **All applicant review and hiring proposals must be completed using the Student Department Manager role. 1. Upon login, user will default to the Applicant
Chapter 13. Documenting Clinical Activities
 Chapter 13. Documenting Clinical Activities INTRODUCTION Documenting clinical activities is required for one or more of the following: clinical care of individual patients -sharing information with other
Chapter 13. Documenting Clinical Activities INTRODUCTION Documenting clinical activities is required for one or more of the following: clinical care of individual patients -sharing information with other
Quanum Electronic Health Record Frequently Asked Questions
 Quanum Electronic Health Record Frequently Asked Questions Table of Contents... 4 What is Quanum EHR?... 4 What are the current capabilities of Quanum EHR?... 4 Is Quanum EHR an EMR?... 5 Can I have Quanum
Quanum Electronic Health Record Frequently Asked Questions Table of Contents... 4 What is Quanum EHR?... 4 What are the current capabilities of Quanum EHR?... 4 Is Quanum EHR an EMR?... 5 Can I have Quanum
PERFORMANCE IN INITIATING CLINICAL RESEARCH (PI) CLINICAL TRIAL ADJUSTMENT PROCESS HRA APPROVED TRIALS
 PERFORMANCE IN INITIATING CLINICAL RESEARCH (PI) CLINICAL TRIAL ADJUSTMENT PROCESS HRA APPROVED TRIALS Version Control Pages 7 9 Notes/action column updated 1 Acronyms CTP DSC DSS FPR HRA MHRA REC SOP
PERFORMANCE IN INITIATING CLINICAL RESEARCH (PI) CLINICAL TRIAL ADJUSTMENT PROCESS HRA APPROVED TRIALS Version Control Pages 7 9 Notes/action column updated 1 Acronyms CTP DSC DSS FPR HRA MHRA REC SOP
Linda Cutter / Dr Charles Heatley. GP Practices and Community Pharmacies
 Schedule 2 Part A Service Specification Service Specification No. 04 Service Anti-coagulation Monitoring Levels 3, 4 & 5 Commissioner Lead Provider Lead Linda Cutter / Dr Charles Heatley GP Practices and
Schedule 2 Part A Service Specification Service Specification No. 04 Service Anti-coagulation Monitoring Levels 3, 4 & 5 Commissioner Lead Provider Lead Linda Cutter / Dr Charles Heatley GP Practices and
Document Title: Research Database Application (ReDA) Document Number: 043
 Document Title: Research Database Application (ReDA) Document Number: 043 Version: 1.1 Ratified by: Committee Date ratified: 23 February 2017 Name of originator/author: Rachel Fay Directorate: Medical
Document Title: Research Database Application (ReDA) Document Number: 043 Version: 1.1 Ratified by: Committee Date ratified: 23 February 2017 Name of originator/author: Rachel Fay Directorate: Medical
18 Weeks Referral to Treatment Guidance (Access Policy)
 18 Weeks Referral to Treatment Guidance (Access Policy) CATEGORY: Guidelines CLASSIFICATION: Clinical PURPOSE: To provide guidance on the management of the 18 week referral to treatment pathway Controlled
18 Weeks Referral to Treatment Guidance (Access Policy) CATEGORY: Guidelines CLASSIFICATION: Clinical PURPOSE: To provide guidance on the management of the 18 week referral to treatment pathway Controlled
Preceptorship Framework for Newly Qualified Nurses, Midwives and Allied Health Professionals
 Preceptorship Framework for Newly Qualified Nurses, Midwives and Allied Health Professionals : May 2012 Review date: May 2014 Author: Deborah Dent with acknowledgments to the members of the task & finish
Preceptorship Framework for Newly Qualified Nurses, Midwives and Allied Health Professionals : May 2012 Review date: May 2014 Author: Deborah Dent with acknowledgments to the members of the task & finish
Marie-Claire Rickard, Governance and GCP Manager Jimena Lovos, Quality Assurance Manager Elizabeth Clough, R&D Governance Operations Manager
 Standard Operating Procedures (SOP) for: Reporting of Serious Breaches of or the Trial Protocol SOP Number: 037 Version Number: 5.0 Effective Date: 17/6/16 Review Date: 17/6/18 Author: Reviewer: Reviewer
Standard Operating Procedures (SOP) for: Reporting of Serious Breaches of or the Trial Protocol SOP Number: 037 Version Number: 5.0 Effective Date: 17/6/16 Review Date: 17/6/18 Author: Reviewer: Reviewer
APPROVALS PANEL ENGLAND SOUTH APPLICATION FOR APPROVAL AS AN APPROVED CLINICIAN UNDER THE MENTAL HEALTH ACT 1983 (AS AMENDED 2007)
 APPROVALS PANEL ENGLAND SOUTH APPLICATION FOR APPROVAL AS AN APPROVED CLINICIAN UNDER THE MENTAL HEALTH ACT 1983 (AS AMENDED 2007) PLEASE ENSURE THE APPLICATION FORM IS COMPLETED IN FULL AND WITHOUT ERROR
APPROVALS PANEL ENGLAND SOUTH APPLICATION FOR APPROVAL AS AN APPROVED CLINICIAN UNDER THE MENTAL HEALTH ACT 1983 (AS AMENDED 2007) PLEASE ENSURE THE APPLICATION FORM IS COMPLETED IN FULL AND WITHOUT ERROR
CLINICIAN S GUIDE TO HIPAA PRIVACY
 CLINICIAN S GUIDE TO HIPAA PRIVACY Introduction... 2 What is HIPAA?... 2 Health Information Privacy... 2 Protected Health Information... 3 Identifiers... 3 HIPAA s Impact on Clinical Practice, Treatment,
CLINICIAN S GUIDE TO HIPAA PRIVACY Introduction... 2 What is HIPAA?... 2 Health Information Privacy... 2 Protected Health Information... 3 Identifiers... 3 HIPAA s Impact on Clinical Practice, Treatment,
City of Richmond. Health, Social and Safety (HSS) Grant Program. Parks, Recreation and Community Events (PRCE) Grant Program
 Health, Social and Safety (HSS) Grant Program Parks, Recreation and Community Events (PRCE) Grant Program City Grants Web-based System 3907928 REVISION CONTROL Reference Document(s) Version # Date Prepared
Health, Social and Safety (HSS) Grant Program Parks, Recreation and Community Events (PRCE) Grant Program City Grants Web-based System 3907928 REVISION CONTROL Reference Document(s) Version # Date Prepared
Implementing a wound assessment and management system (WAMS)
 Southern Cross University From the SelectedWorks of Dr Joanne Rowley Fall May, 2006 Implementing a wound assessment and management system (WAMS) Karen Saunders Joanne Rowley, Southern Cross University
Southern Cross University From the SelectedWorks of Dr Joanne Rowley Fall May, 2006 Implementing a wound assessment and management system (WAMS) Karen Saunders Joanne Rowley, Southern Cross University
NOTICE OF PRIVACY PRACTICES
 VII-07B Notice of Privacy Practices (p) The MetroHealth System 2500 MetroHealth Drive Cleveland, OH 44109-1998 NOTICE OF PRIVACY PRACTICES THIS NOTICE DESCRIBES HOW WE MAY USE AND DISCLOSE YOUR PROTECTED
VII-07B Notice of Privacy Practices (p) The MetroHealth System 2500 MetroHealth Drive Cleveland, OH 44109-1998 NOTICE OF PRIVACY PRACTICES THIS NOTICE DESCRIBES HOW WE MAY USE AND DISCLOSE YOUR PROTECTED
NHS e-referral Service (e-rs) Frequently Asked Questions for Referrers
 NHS e-referral Service (e-rs) Frequently Asked Questions for Referrers Purpose Primary Care colleagues are sometimes faced with situations regarding referrals and may not necessarily know the correct action
NHS e-referral Service (e-rs) Frequently Asked Questions for Referrers Purpose Primary Care colleagues are sometimes faced with situations regarding referrals and may not necessarily know the correct action
Wednesday, 17 May, 2017
 Submission Guidelines for NHMRC Postgraduate Research Scholarships for 2018 Closing Date for Initial Submission: Wednesday, 17 May, 2017 Draft Submission: Final Submission: By email: (1) The Assessor Snapshot
Submission Guidelines for NHMRC Postgraduate Research Scholarships for 2018 Closing Date for Initial Submission: Wednesday, 17 May, 2017 Draft Submission: Final Submission: By email: (1) The Assessor Snapshot
EMAR Medication Pass
 EMAR Medication Pass This manual includes recording of resident medication passes on a computer. To begin your Medication Pass, click on the EMAR icon, then select a Med Provider. The listing of Med Providers
EMAR Medication Pass This manual includes recording of resident medication passes on a computer. To begin your Medication Pass, click on the EMAR icon, then select a Med Provider. The listing of Med Providers
Once the feasibility assessment has been conducted the study team will be notified via (Appendix 3) of the outcome and whether the study is;
 1. INTRODUCTION 2. SCOPE Feasibility assessments will ascertain any operational concerns about a research study which may delay NHS Permission or cause issues with study delivery. By conducting an assessment
1. INTRODUCTION 2. SCOPE Feasibility assessments will ascertain any operational concerns about a research study which may delay NHS Permission or cause issues with study delivery. By conducting an assessment
School Based Health Services Consent Form
 MRN: PCP: Teacher: Grade: School Based Health Services Consent Form Before your child sees a provider, we are asking you to authorize medical and/ or dental treatment. We will work with you to improve
MRN: PCP: Teacher: Grade: School Based Health Services Consent Form Before your child sees a provider, we are asking you to authorize medical and/ or dental treatment. We will work with you to improve
Site Manager Guide CMTS. Care Management Tracking System. University of Washington aims.uw.edu
 Site Manager Guide CMTS Care Management Tracking System University of Washington aims.uw.edu rev. 8/13/2018 Table of Contents INTRODUCTION... 1 SITE MANAGER ACCOUNT ROLE... 1 ACCESSING CMTS... 2 SITE NAVIGATION
Site Manager Guide CMTS Care Management Tracking System University of Washington aims.uw.edu rev. 8/13/2018 Table of Contents INTRODUCTION... 1 SITE MANAGER ACCOUNT ROLE... 1 ACCESSING CMTS... 2 SITE NAVIGATION
CRITERIA AND GUIDELINES FOR FULL ACCREDITATION AS A BEHAVIOURAL AND/OR COGNITIVE PSYCHOTHERAPIST
 CRITERIA AND GUIDELINES FOR FULL ACCREDITATION AS A BEHAVIOURAL AND/OR COGNITIVE PSYCHOTHERAPIST Full Accreditation is dependent on submission, 12 months after the date Provisional Accreditation, of an
CRITERIA AND GUIDELINES FOR FULL ACCREDITATION AS A BEHAVIOURAL AND/OR COGNITIVE PSYCHOTHERAPIST Full Accreditation is dependent on submission, 12 months after the date Provisional Accreditation, of an
