Adverse Drug Event Monitoring with Clinical and Laboratory Data Using Arden Syntax
|
|
|
- Noah Simmons
- 6 years ago
- Views:
Transcription
1 Adverse Drug Event Monitoring with Clinical and Laboratory Data Using Arden Syntax Andrea Rappelsberger a, Klaus-Peter Adlassnig a,b, Jeroen S. de Bruin a, Manuela Plössnig c, Jochen Schuler d, Christina Hofer-Dückelmann e a Section for Artificial Intelligence and Decision Support, Center for Medical Statistics, Informatics, and Intelligent Systems, Medical University of Vienna, Spitalgasse 23, A-1090 Vienna, Austria b Medexter Healthcare GmbH, Borschkegasse 7/5, A-1090 Vienna, Austria c Salzburg Research Forschungsgesellschaft mbh, Jakob Haringer Strasse 5/3, A-5020 Salzburg, Austria d Paracelsus Medical Private University Salzburg, Institute of General Practice, Family Medicine and Preventive Medicine, Strubergasse 21, A-5020 Salzburg, Austria e Landesapotheke am St. Johanns-Spital Salzburg, Müllner Hauptstrasse 50, A-5020 Salzburg, Austria Abstract In times of steadily growing numbers of administered drugs, the detection of adverse drug events (ADEs) is an important aspect of improving patient safety. At present, only about 1 13% of detected ADEs are reported. Raising the number of reported ADEs will result in greater and more efficient support of pharmacovigilance. Therefore, potential ADE s must be identified early. In the imedication system, which is a rulebased application, triggers are used for computerized detection of possible ADEs. Creating a pilot system, we defined the relevant use cases hyperkalemia, hyponatremia, renal failure, and over-anticoagulation; knowledge bases were implemented in Arden Syntax for each use case. The objective of these knowledge bases is to interpret patient-specific clinical data and generate notifications based on a calculated ADE risk score, which may indicate possible ADEs. This will permit appropriate monitoring of potential ADE situations over time in the interest of patient care, quality assurance, and pharmacovigilance. Keywords: Drug Related Side Effects and Adverse Reactions; Drug Monitoring; Decision Support Systems, Clinical. Introduction Medical errors, i.e., unintended acts of omission or commission, or those that do not achieve their intended outcome [1] such as inaccurate medication do occur in hospitals. A recent study stated that medical errors are the thirdleading cause of death in the U.S. [2], making it more important than ever to prevent and mitigate medical errors, especially those causing damage to patients [3]. The fact that drugs are being administered in increasing numbers signifies a greater potential for drug-related harm, including adverse drug events (ADEs). Traditionally, ADEs are tracked and reported on a voluntary basis. Hence the success of ambulatory error reporting systems has been limited; approximately 10 20% of medication errors and only 1 13% of detected ADEs are reported [4]. Additionally, the process of ADE detection consumes a considerable amount of valuable time and money. Nevertheless, studies have shown that as many as 6% of all hospital admissions are due to ADEs, and this number is three-fold higher among elderly patients [5, 6]. Moreover, about 50% of these prescribing errors and ADEs are deemed avoidable [7]. Hospitals need a more efficient way to quantify the degree and severity of ADEs, such as automated or computerized detection. Identification of severe ADEs as well as measuring their frequency will enable pharmacists and physicians to take corrective measures. imedication supports the process of pharmacovigilance the pharmacological science relating to the detection, assessment, understanding and prevention of adverse effects, particularly the long-term and short-term side effects of medication [8] by identifying possible ADEs. With imedication, we aim to reduce the number and severity of ADEs over time, identifying potential ADEs as early as possible, supporting plausibility checks on suspected ADEs, and reporting verified ADEs in an appropriate and standardized manner. A secondary goal is to increase the motivation of physicians and pharmacists to report ADEs and last but not least save time and money during the reporting process. Existing approaches for computerized ADE detection employ methods such as data mining [9] and decision trees [10] to automatically generate ADE detection rules. Another strategy is to use ontologies [11] and derive ADE rules from existing data sources like SNOMED-CT [12]. Others approach the task by the automatic creation of rules with the aid of product label parsing [13]. In the imedication project, we integrate the operative knowledge of local and remote experts by linking distributed knowledge repositories, and manually derive specific rules from expert knowledge. This enables us to specify complex rules for the identification of ADEs. The system reports detected ADEs according to their severity; reports additionally include an explanation on how the knowledge base came to its conclusion to report an ADE. Furthermore, the report provides information that supports the physician or clinical pharmacist to take corrective therapeutic measures. If necessary, a report is forwarded to the Austrian Agency for Health and Food Safety (AGES), the agency responsible for pharmacovigilance in Austria. The workflow of ADE identification, verification, and reporting is depicted in Figure 1. In the present paper we report on the results of a pilot study performed in Using data on patients admitted to the
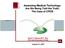
2 Figure 1 - Workflow of adverse drug event (ADE) detection and reporting. University Hospital of Salzburg (UHS) in 2007 and 2011, we analyze the effectiveness of the system in detail. Methods Theoretical foundations The imedication system is founded on the principles of the Institute for Healthcare Improvement (IHI) Global Trigger Tool method [3] and Morimoto s classification [14] for the detection of possible ADEs. The IHI Global Trigger Tool for Measuring Adverse Events is a method for identifying adverse events especially those leading to harm and measuring the rate of adverse events over time. The method employs triggers clues on possible adverse events to track adverse events, including ADEs. However, the tool is not meant to identify all adverse events, but rather employs a retrospective review of a random sample of inpatient data [3]. According to Morimoto et al. [14], irregular use of medication (referred to as incidents) can be classified in many ways: actual ADEs vs. potential; preventable vs. non-preventable; ameliorable vs. non-ameliorable; and errors vs. non-error. According to this method, an ADE is regarded as an injury due to medication. In general, incidents are identified by collecting practice data, soliciting incidents from patient caregivers, and surveying patients directly. These data are then independently reviewed by patient caregivers using various triggers, such as: Symptoms or actions that suggest a (potential) ADE or medication error, such as a new rash or new diarrhea. Diagnoses associated with (potential) ADEs or medication errors, such as poisoning by drugs. The use of specific drugs that suggests an ADE may have occurred. Drug combinations known to cause ADEs or the use of duplicate drugs. Combinations of drugs and symptoms that might indicate a (harmful) reaction to the drug, such as diarrhea or eruption due to antibiotics. Combinations of drugs and patient diagnoses, such as bleeding and antiplatelet agents or warfarin. Combinations of drugs and other factors such as patient age or sex, or pregnancy. Laboratory triggers, such as microbiology results that show improper use of antibiotics. Based on these triggers, incidents are classified as potential ADEs, medication errors without potential harm, ADE with medication error, and ADE without medication error. Study design, setting, and participants We conducted a retrospective single-center cohort study on sample data that were collected prospectively and validated. The study was performed at UHS, a tertiary-care and teaching hospital. Data from the UHS were collected from patients admitted in 2007 or 2011 to any ward of the Department of Internal Medicine (I+II). All female adult patients (age 18 years) admitted for at least 24 hours were eligible for the study.
3 An additional age constraint was imposed on patients admitted in 2007: all of them had to be older than 75 years. Data management and sample size Demographic patient information as well as clinical and laboratory values were obtained through systematic interrogation and sampling of the hospital information system (HIS). A total of 70 patient cases were selected for the study; 22 from 2007 and 48 from Data sources Patient data were collected from various sources, suhc as electronic health records (EHRs), HISs, and manually entered data. The following six main categories of data were used: Demographic data includes demographic information such as age, sex, weight, height, pregnancy, or epidemiological studies. Laboratory findings are provided by the HIS, for example serum creatinine, potassium, sodium, etc. Different time frames exist for absolute and relative findings. Absolute values are only taken into account within a time frame of three days prior to the data of calculation, whereas relative values permit a time frame of seven days. Symptoms occurred during the preceding seven days are integrated in the analysis. Diagnoses are specified by the 10 th revision of the International Statistical Classification of Diseases and Related Health Problems (ICD-10). A single diagnosis is identified by a 3 7 digit code (such as E87.5 for hyperkalemia). When more than one diagnosis from a diagnostic group is detected in the patient s chart, all of the diagnoses from this group are counted as a single trigger. Medications are specified by the Anatomical Therapeutic Chemical Classification System (ATC), according to which a single substance is defined by a 7-digit code. Hospital events denote any consultations of psychiatrists or trauma surgeons and internal accident reports during the hospital stay. Risk score calculation Risk score values are assigned to ADE triggers which are processed by an algorithm to calculate an overall ADE risk score on a given scale (1 5). Based on the ADE risk score, appropriate reminders are sent to the physicians and pharmacists. Furthermore, reporting forms are prepopulated with the relevant patient data and the ADE suspicion. The ADE risk score calculation consists of five main steps: 1. Patient data filtering. Only those data elements within a specified timeframe related to the calculation date and specified conditions are relevant for the calculation process. The time frames are based on clinical experience. Patient data shall be only integrated when the medication that may cause an ADE has been administered during the preceding three days. 2. The recognition of at least one medication which may cause the possible ADE is the prerequisite for the calculation of an ADE risk score and the specific rules. 3. Depending on the number of positive triggers from each category, a contribution to the ADE risk score is calculated. The maximum value over all categories is added to the ADE risk score (see Table 1). 4. The ADE risk score is adapted by a value that depends on the quantity of the patient s medication (see Table 2). 5. Standardizatino of the last ADE risk score is the last calculation step. The maximum value for the ADE risk score is 5. Table 1. The adverse drug event risk score increases, depending on the number of positive triggers For each category 1 2 triggers with an ADE risk score of 1: increase value by 1 3 triggers with an ADE risk score of 1: increase value by 2 1 trigger with an ADE risk score of 2: increase value by 2 1 trigger with an ADE risk score of 3: increase value by 3 new ADE risk score = old ADE risk score + (maximum of categories increase values) Table 2. The adverse drug event score increases, depending on the number of administered medications The occurrence of 2 4 different medications from the medication lists causing the ADE for the use case results in an increase of the ADE risk score by 1. The occurrence of >4 different medications from the medication lists causing the ADE for the use case results in an increase of the ADE risk score by 2. Knowledge base and data processing Four highly critical clinical situations, namely hyperkalemia, hyponatremia, renal failure, and over-anticoagulation, were defined as use cases for the imedication project. They represent significant ADEs for internal and geriatric medicine. The four knowledge bases in imedication are built upon these use cases, which are implemented in Arden Syntax, a knowledge representation and processing language supported by HL7 International [15]. Each knowledge base consists of several medical logic modules (MLMs) [16, 17], which are the basic knowledge representation and processing units in Arden Syntax and are executed by an Arden Syntax server [18]. In all there are 33 MLMs, considering 51 ADE triggers. Data are processed as follows: First, all relevant data are collected and aggregated to an information block a patient object and forwarded to the Arden Syntax server. The Arden Syntax server processes the obtained information and returns one result object for each knowledge base and each day. The return objects contain complete patient data, thus permitting the explanation and tracing of decisions made by the imedication system. Also, for each category, the fired triggers are stored and attached. The result object further includes information on the degree or severity of the detected ADE. This information determines the ADE risk score.
4 Result presentation We use patient demographic information (age, length of stay, number of verified ADEs) and treatment information (number of medications administered) to describe the patient population. We also discuss the number and risk score of ADE triggers during the study period. We define each ADE trigger with a risk score 4 as a positive test, and ADEs with lower risk scores as a negative test. Using this classification, we determine the effectiveness of the system with the sensitivity (SEN), specificity (SPEC), positive predictive value (PPV) and negative predictive value (NPV) metrics. Results The mean age of the patients 76.5 years (standard deviation 13.3 years, minimum age 43 years, maximum age 99 years). The mean duration of the stay was 11.2 days (standard deviation 10 days, minimum length of stay 2 days, maximum 53 days). On average, a patient received 8.5 medications during her stay (standard deviation 4.7; minimum number of administered medications 1, maximum numer 29). Of the 70 patients participating in the study, 16 (22,8%) experienced one or several ADEs confirmed by patient caregivers. Twelve patients with ADEs were confirmed in the study population of 2007, and 4 patients with ADEs among those examined in In all 26 ADEs were confirmed for the four medical situations implemented in the knowledge base: 2 for hyperkalemia, 13 for hyponatremia, 8 for renal failure, and 3 for over-anticoagulation. A total of 428 triggers were generated during the study period. An overview of these triggers and their scores are shown in Table 3. Table 3. Number of triggers generated during the study period and their associated scores. Trigger score Frequency Score Score 1 9 Score 2 9 Score 3 34 Score 4 34 Score 5 36 Total 428 Using the previously mentioned classification for a postitive test and a negative test, we constructed a 2x2 contingency table (Table 4). Based on the numbers in the contingency table, the system showed a SEN of 85%, a SPEC of 88%, a PPV of 31% and an NPV of 99%. Table 4. 2x2 Contingency table for the study results ADE confirmed ADE absent Total Positive test Negative test Total Discussion We present the imedication system, a computerized system that supports pharmacovigilance through the detection and reporting of potential ADEs, We outlined the underlying principles and mechanics of the system, and determined that the current pilot system showed good sensitivity and specificity. However, the system needs to be improved further before it can qualify as a trustworthy alarming system (PPV 31%). Computerized trigger tools for inpatient ADEs perform moderately well, are inexpensive to use, and already deployed in many hospitals [19]. The imedication system was able to correctly identify 85% of all ADEs, which is a multitude of the number of ADEs commonly reported (1 13%) [4]. This helps clinicians in many ways. First, a retrospective evaluation of clinical data permits quality assurance through statistical analysis of detected potential ADEs. Second, physicians are given active feedback (notifications) during the treatment of their patients, thus enabling them to take corrective measures in a timely manner. Finally, the imedication system supports (semi-)automated ADE reporting by notifications to the pharmacist with prepopulated forms. As a result, ADEs can be avoided or corrected and, when they do occur, their reporting is less resource intensive. The limitations of the study are worthy of mention. First, in the present evaluation phase, data input is accomplished semiautomatically because all relevant patient data are not available in electronic form. Furthermore, the four use cases currently implemented in the knowledge base have to be evaluated in a wider setting and improved in order to avoid alert fatigue. Finally, additional studies will be needed to evaluate the imedication phenomenon of much more frequent ADE reports to the AGES than done by conventional reporting. Conclusion We showed that a comprehensive solution for the (semi-) automated detection and reporting of ADEs is not only feasible but also effective. Given that tracking and reporting of ADEs is done only on a voluntary basis, the integration of such an automated computerized method in clinical routine would provide more information about the scope of the ADE problem at a minimal expense of resources. Acknowledgments The imedication project is funded by Research, Innovation, Technology Information Technology (FIT-IT [20]), an initiative of the Austrian Federal Ministry of Transport, Innovation, and Technology. The participants of this consortial project are Salzburg Research Forschungsgesellschaft mbh, Gemeinnützige Salzburger Landeskliniken Betriebsgesellschaft mbh, Paracelsus Medical Private University Salzburg, Landesapotheke am St. Johanns-Spital Salzburg, and Medexter Healthcare GmbH. References [1] L.L. Leape, Error in Medicine, JAMA 272 (1994), [2] M.A. Makary and M. Daniel, Medical Error The Third Leading Cause of Death in the US, BMJ 353 (2016), i2139.
5 [3] F.A. Griffin and R.K. Resar, IHI Global Trigger Tool for Measuring Adverse Events (Second Edition), IHI Innovation Series White Paper, Institute for Healthcare Improvement, Cambridge, MA., Available from last access: 17 December [4] A. Zafar, J. Hickner, W. Pace, and W. Tierney, An Adverse Drug Event and Medication Error Reporting System for Ambulatory Care (MEADERS), AMIA Annu Symp Proc 2008 (2008), [5] A. Bobb, K. Gleason, M. Husch, J. Feinglass, P.R. Yarnold, and G.A. Noskin, The Epidemiology of Prescribing Errors: The Potential Impact of Computerized Prescriber Order Entry, Arch Intern Med 164 (2004), [6] J.S. de Bruin, W. Seeling, and C. Schuh, Data Use and Effectiveness in Electronic Surveillance of Healthcare Associated Infections in the 21st Century: A Systematic Review, J Am Med Inform Assoc 21 (2014), [7] A.J. Forster, H.J. Murff, J.F. Peterson, T.K. Gandhi, and D.W. Bates, The Incidence and Severity of Adverse Events Affecting Patients after Discharge from the Hospital, Ann Intern Med 138 (2003), [8] Definition of pharmacovigilance (WHO, 2002) [17] G. Hripcsak, Writing Arden Syntax Medical Logic Modules, Comput Biol Med 24 (1994), [18] K.-P. Adlassnig and K. Fehre, Service-Oriented Fuzzy- Arden-Syntax-Based Clinical Decision Support, Indian Journal of Medical Informatics 8 (2014), [19] H.J. Mull, J.R. Nebeker, S.L. Shimada, H.M. Kaafarani, P.E. Rivard, and A.K. Rosen, Consensus Building for Development of Outpatient Adverse Drug Event Triggers, J Patient Saf 7 (2011), [20] FIT-IT, last access: 17 December Address for correspondence Univ.-Prof. DI Dr. Klaus-Peter Adlassnig Section for Artificial Intelligence and Decision Support, Center for Medical Statistics, Informatics, and Intelligent Systems, Medical University of Vienna, Spitalgasse 23, 1090 Vienna, Austria klaus-peter.adlassnig@meduniwien.ac.at and Medexter Healthcare GmbH, Borschkegasse 7/5, 1090 Vienna, Austria kpa@medexter.com [9] E. Chazard, C. Preda, B. Merlin, G. Ficheur, and R. Beuscart, Data-Mining-Based Detection of Adverse Drug Events, Stud Health Technol Inform 150 (2009), [10] E. Chazard, A. Baceanu, R. Marcilly, S. Bernonville, G. Ficheur, and R. Beuscart, A Web Tool for Automated Adverse Drug Events Detection: The ADE Scorecards, Stud Health Technol Inform 169 (2011), [11] F. Cao, X. Sun, X. Wang, B. Li, J. Li, and Y. Pan, Ontology-Based Knowledge Management for Personalized Adverse Drug Events Detection, Stud Health Technol Inform 169 (2011), [12] Snomed, last access: 17 December [13] J.D. Duke and J. Friedlin, ADESSA: A Real-Time Decision Support Service for Delivery of Semantically Coded Adverse Drug Event Data, AMIA Annu Symp Proc 2010 (2010), [14] T. Morimoto, T.K. Gandhi, A.C. Seger, T.C. Hsieh, and D.W. Bates, Adverse Drug Events and Medication Errors: Detection and Classification Methods, Qual Saf Health Care 13 (2004), [15] Health Level Seven, Arden Syntax v2.10 (Health Level Seven Arden Syntax for Medical Logic Systems, Version 2.10), Available at: last access: 8 November [16] K.-P. Adlassnig and A. Rappelsberger, Medical Knowledge Packages and Their Integration into Health- Care Information Systems and the World Wide Web, Stud Health Technol Inform 136 (2008),
COMPUTERIZED PHYSICIAN ORDER ENTRY (CPOE)
 COMPUTERIZED PHYSICIAN ORDER ENTRY (CPOE) Ahmed Albarrak 301 Medical Informatics albarrak@ksu.edu.sa 1 Outline Definition and context Why CPOE? Advantages of CPOE Disadvantages of CPOE Outcome measures
COMPUTERIZED PHYSICIAN ORDER ENTRY (CPOE) Ahmed Albarrak 301 Medical Informatics albarrak@ksu.edu.sa 1 Outline Definition and context Why CPOE? Advantages of CPOE Disadvantages of CPOE Outcome measures
Using Electronic Health Records for Antibiotic Stewardship
 Using Electronic Health Records for Antibiotic Stewardship STRENGTHEN YOUR LONG-TERM CARE STEWARDSHIP PROGRAM BY TRACKING AND REPORTING ELECTRONIC DATA Introduction Why Use Electronic Systems for Stewardship?
Using Electronic Health Records for Antibiotic Stewardship STRENGTHEN YOUR LONG-TERM CARE STEWARDSHIP PROGRAM BY TRACKING AND REPORTING ELECTRONIC DATA Introduction Why Use Electronic Systems for Stewardship?
Quality Data Model (QDM) Style Guide. QDM (version MAT) for Meaningful Use Stage 2
 Quality Data Model (QDM) Style Guide QDM (version MAT) for Meaningful Use Stage 2 Introduction to the QDM Style Guide The QDM Style Guide provides guidance as to which QDM categories, datatypes, and attributes
Quality Data Model (QDM) Style Guide QDM (version MAT) for Meaningful Use Stage 2 Introduction to the QDM Style Guide The QDM Style Guide provides guidance as to which QDM categories, datatypes, and attributes
Improving patient discharge process using electronic medication input tool and on-line guide to arranging follow-ups
 BMJ Quality Improvement Reports 2013; u756.w711 doi: 10.1136/bmjquality.u756.w711 Improving patient discharge process using electronic medication input tool and on-line guide to arranging follow-ups Rory
BMJ Quality Improvement Reports 2013; u756.w711 doi: 10.1136/bmjquality.u756.w711 Improving patient discharge process using electronic medication input tool and on-line guide to arranging follow-ups Rory
Measuring Medication Harm: Advantages of Using a Trigger Tool. Frank Federico Executive Director
 Measuring Medication Harm: Advantages of Using a Trigger Tool Frank Federico Executive Director ffederico@ihi.org Objectives Review the use of the trigger tool Discuss how to use the trigger tool for high-alert
Measuring Medication Harm: Advantages of Using a Trigger Tool Frank Federico Executive Director ffederico@ihi.org Objectives Review the use of the trigger tool Discuss how to use the trigger tool for high-alert
Adverse Drug Events and Readmissions: The Global Picture
 Adverse Drug Events and Readmissions: The Global Picture Kyle E. Hultgren, PharmD Managing Director Center for Medication Safety Advancement Purdue University College of Pharmacy Indianapolis, IN 4 Learning
Adverse Drug Events and Readmissions: The Global Picture Kyle E. Hultgren, PharmD Managing Director Center for Medication Safety Advancement Purdue University College of Pharmacy Indianapolis, IN 4 Learning
W e were aware that optimising medication management
 207 QUALITY IMPROVEMENT REPORT Improving medication management for patients: the effect of a pharmacist on post-admission ward rounds M Fertleman, N Barnett, T Patel... See end of article for authors affiliations...
207 QUALITY IMPROVEMENT REPORT Improving medication management for patients: the effect of a pharmacist on post-admission ward rounds M Fertleman, N Barnett, T Patel... See end of article for authors affiliations...
Measuring Harm. Objectives and Overview
 Patient Safety Research Introductory Course Session 3 Measuring Harm Albert W Wu, MD, MPH Former Senior Adviser, WHO Professor of Health Policy & Management, Johns Hopkins Bloomberg School of Public Health
Patient Safety Research Introductory Course Session 3 Measuring Harm Albert W Wu, MD, MPH Former Senior Adviser, WHO Professor of Health Policy & Management, Johns Hopkins Bloomberg School of Public Health
Patient Safety Research Introductory Course Session 3. Measuring Harm
 Patient Safety Research Introductory Course Session 3 Measuring Harm Albert W Wu, MD, MPH Former Senior Adviser, WHO Professor of Health Policy & Management, Johns Hopkins Bloomberg School of Public Health
Patient Safety Research Introductory Course Session 3 Measuring Harm Albert W Wu, MD, MPH Former Senior Adviser, WHO Professor of Health Policy & Management, Johns Hopkins Bloomberg School of Public Health
Admissions and Readmissions Related to Adverse Events, NMCPHC-EDC-TR
 Admissions and Readmissions Related to Adverse Events, 2007-2014 By Michael J. Hughes and Uzo Chukwuma December 2015 Approved for public release. Distribution is unlimited. The views expressed in this
Admissions and Readmissions Related to Adverse Events, 2007-2014 By Michael J. Hughes and Uzo Chukwuma December 2015 Approved for public release. Distribution is unlimited. The views expressed in this
A23/B23: Patient Harm in US Hospitals: How Much? Objectives
 A23/B23: Patient Harm in US Hospitals: How Much? 23rd Annual National Forum on Quality Improvement in Health Care December 6, 2011 Objectives Summarize the findings of three recent studies measuring adverse
A23/B23: Patient Harm in US Hospitals: How Much? 23rd Annual National Forum on Quality Improvement in Health Care December 6, 2011 Objectives Summarize the findings of three recent studies measuring adverse
Computer Provider Order Entry (CPOE)
 Computer Provider Order Entry (CPOE) Use computerized provider order entry (CPOE) for medication orders directly entered by any licensed healthcare professional who can enter orders into the medical record
Computer Provider Order Entry (CPOE) Use computerized provider order entry (CPOE) for medication orders directly entered by any licensed healthcare professional who can enter orders into the medical record
Adverse Events in Hospitals: How Many and Why Not Reported. Fran Griffin Senior Manager Clinical Programs, BD
 Adverse Events in Hospitals: How Many and Why Not Reported Fran Griffin Senior Manager Clinical Programs, BD Disclosure Currently full time employed at BD and faculty at The Institute for Healthcare Improvement
Adverse Events in Hospitals: How Many and Why Not Reported Fran Griffin Senior Manager Clinical Programs, BD Disclosure Currently full time employed at BD and faculty at The Institute for Healthcare Improvement
Pharmacists Role in Care Transitions
 Pharmacists Role in Care Transitions SHE A FA NNING, PHA RMD, PGY 1 PHA RMA C Y RE SIDENT ST. PETER S HOSPITAL HE LE NA, MT Disclosures Co-investigators: Thomas Richardson, PharmD, BCPS AQ-ID; Brad Hornung,
Pharmacists Role in Care Transitions SHE A FA NNING, PHA RMD, PGY 1 PHA RMA C Y RE SIDENT ST. PETER S HOSPITAL HE LE NA, MT Disclosures Co-investigators: Thomas Richardson, PharmD, BCPS AQ-ID; Brad Hornung,
Fundamentals of Self-Limiting Conditions Prescribing for Manitoba Pharmacists. Ronald F. Guse Registrar College of Pharmacists of Manitoba (CPhM)
 Fundamentals of Self-Limiting Conditions Prescribing for Manitoba Pharmacists Ronald F. Guse Registrar College of Pharmacists of Manitoba (CPhM) 1 Learning Objectives Upon successful completion of this
Fundamentals of Self-Limiting Conditions Prescribing for Manitoba Pharmacists Ronald F. Guse Registrar College of Pharmacists of Manitoba (CPhM) 1 Learning Objectives Upon successful completion of this
SHRI GURU RAM RAI INSTITUTE OF TECHNOLOGY AND SCIENCE MEDICATION ERRORS
 MEDICATION ERRORS Patients depend on health systems and health professionals to help them stay healthy. As a result, frequently patients receive drug therapy with the belief that these medications will
MEDICATION ERRORS Patients depend on health systems and health professionals to help them stay healthy. As a result, frequently patients receive drug therapy with the belief that these medications will
Medication Reconciliation Review
 The Medication Reconciliation Review tool provides step-by-step instructions for conducting a review of closed patient records to identify errors related to unreconciled medications. Organizations that
The Medication Reconciliation Review tool provides step-by-step instructions for conducting a review of closed patient records to identify errors related to unreconciled medications. Organizations that
The Role of the Agency for Healthcare Research and Quality (AHRQ) in the US Drug Safety System
 The Role of the Agency for Healthcare Research and Quality (AHRQ) in the US Drug Safety System Scott R. Smith, MSPH, PhD Center for Outcomes & Evidence Agency for Healthcare Research & Quality July 20,
The Role of the Agency for Healthcare Research and Quality (AHRQ) in the US Drug Safety System Scott R. Smith, MSPH, PhD Center for Outcomes & Evidence Agency for Healthcare Research & Quality July 20,
IMPACT OF TECHNOLOGY ON MEDICATION SAFETY
 Continuous Quality Improvement IMPACT OF Steven R. Abel, PharmD, FASHP TECHNOLOGY ON Nital Patel, PharmD. MBA MEDICATION SAFETY Sheri Helms, PharmD Candidate Brian Heckman, PharmD Candidate Ismaila D Badjie
Continuous Quality Improvement IMPACT OF Steven R. Abel, PharmD, FASHP TECHNOLOGY ON Nital Patel, PharmD. MBA MEDICATION SAFETY Sheri Helms, PharmD Candidate Brian Heckman, PharmD Candidate Ismaila D Badjie
One or More Errors in 67% of the IV Infusions: Insights from a Study of IV Medication Administration
 One or More Errors in 67% of the IV Infusions: Insights from a Study of IV Medication Administration Presented by: Marla Husch Northwestern Memorial Hospital Northwestern Memorial Hospital Chicago, Illinois
One or More Errors in 67% of the IV Infusions: Insights from a Study of IV Medication Administration Presented by: Marla Husch Northwestern Memorial Hospital Northwestern Memorial Hospital Chicago, Illinois
Health Management Information Systems: Computerized Provider Order Entry
 Health Management Information Systems: Computerized Provider Order Entry Lecture 2 Audio Transcript Slide 1 Welcome to Health Management Information Systems: Computerized Provider Order Entry. The component,
Health Management Information Systems: Computerized Provider Order Entry Lecture 2 Audio Transcript Slide 1 Welcome to Health Management Information Systems: Computerized Provider Order Entry. The component,
Optimizing pharmaceutical care via Health Information Technology:
 Optimizing pharmaceutical care via Health Information Technology: The Epic Challenge Rilwan Badamas, PharmD, CAHIMS Pharmacy Grand Rounds 01/03/2017 2011 MFMER slide-1 The medication management team requests
Optimizing pharmaceutical care via Health Information Technology: The Epic Challenge Rilwan Badamas, PharmD, CAHIMS Pharmacy Grand Rounds 01/03/2017 2011 MFMER slide-1 The medication management team requests
Impact of a Pharmacy-Led Medication Reconciliation Program
 Impact of a Pharmacy-Led Medication Reconciliation Program Naomi Digiantonio, PharmD, BCPS; Jeremy Lund, PharmD, MS, BCCCP, BCPS; and Samantha Bastow, PharmD, BCPS ABSTRACT Objective: To determine the
Impact of a Pharmacy-Led Medication Reconciliation Program Naomi Digiantonio, PharmD, BCPS; Jeremy Lund, PharmD, MS, BCCCP, BCPS; and Samantha Bastow, PharmD, BCPS ABSTRACT Objective: To determine the
Evidence-Based Quality Improvement: A recipe for improving medication safety and handover of care Smeulers, Marian
 UvA-DARE (Digital Academic Repository) Evidence-Based Quality Improvement: A recipe for improving medication safety and handover of care Smeulers, Marian Link to publication Citation for published version
UvA-DARE (Digital Academic Repository) Evidence-Based Quality Improvement: A recipe for improving medication safety and handover of care Smeulers, Marian Link to publication Citation for published version
Medication Control and Distribution. Minor/technical revision of existing policy. ± Major revision of existing policy Reaffirmation of existing policy
 Name of Policy: Policy Number: 3364-133-17 Department: Pharmacy Approvingofficer: Chief Executive Officer THE unrversity OF TOLEDO MEDICAL CERITER Responsible Agent: Scope: Director of Pharmacy University
Name of Policy: Policy Number: 3364-133-17 Department: Pharmacy Approvingofficer: Chief Executive Officer THE unrversity OF TOLEDO MEDICAL CERITER Responsible Agent: Scope: Director of Pharmacy University
Medication Reconciliation Project Edmonton Zone Steps To MedRec Success Across Multiple Programs and Sites in a Large Urban Setting
 Medication Reconciliation Project Edmonton Zone Steps To MedRec Success Across Multiple Programs and Sites in a Large Urban Setting Natalie McMurtry, BSc Pharm, Sr. Medication Consultant; Vanessa Moorgen,
Medication Reconciliation Project Edmonton Zone Steps To MedRec Success Across Multiple Programs and Sites in a Large Urban Setting Natalie McMurtry, BSc Pharm, Sr. Medication Consultant; Vanessa Moorgen,
Impact of an Innovative ADC System on Medication Administration
 Impact of an Innovative ADC System on Medication Administration March 1, 2016 Nilesh Desai, BS, RPh, MBA Administrator Pharmacy and Clinical Operations Hackensack University Medical Center Conflict of
Impact of an Innovative ADC System on Medication Administration March 1, 2016 Nilesh Desai, BS, RPh, MBA Administrator Pharmacy and Clinical Operations Hackensack University Medical Center Conflict of
Introductions. Welcome to the APAC Global Trigger Tool Session. Dr Carol Haraden IHI Gillian Robb CMDHB. Carol Haraden.
 Welcome to the APAC Global Trigger Tool Session Dr Carol Haraden IHI Gillian Robb CMDHB Carol Haraden Introductions Gillian Robb Outline for this session Introduction to the Global Trigger Tool What is
Welcome to the APAC Global Trigger Tool Session Dr Carol Haraden IHI Gillian Robb CMDHB Carol Haraden Introductions Gillian Robb Outline for this session Introduction to the Global Trigger Tool What is
IMPROVING INPATIENT TO OUTPATIENT TRANSITION FOR GENERAL MEDICINE CLINIC PATIENTS
 IMPROVING INPATIENT TO OUTPATIENT TRANSITION FOR GENERAL MEDICINE CLINIC PATIENTS JULIE GILBREATH, MD, FACP; RAMON GALLEGOS, RN; PATRICIA REYES 8/2016-1/2017 1 THE TEAM CSE Participants: Julie Gilbreath,
IMPROVING INPATIENT TO OUTPATIENT TRANSITION FOR GENERAL MEDICINE CLINIC PATIENTS JULIE GILBREATH, MD, FACP; RAMON GALLEGOS, RN; PATRICIA REYES 8/2016-1/2017 1 THE TEAM CSE Participants: Julie Gilbreath,
Quality ID #46 (NQF 0097): Medication Reconciliation Post-Discharge National Quality Strategy Domain: Communication and Care Coordination
 Quality ID #46 (NQF 0097): Medication Reconciliation Post-Discharge National Quality Strategy Domain: Communication and Care Coordination 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE:
Quality ID #46 (NQF 0097): Medication Reconciliation Post-Discharge National Quality Strategy Domain: Communication and Care Coordination 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE:
Medication Management: Is It in Your Toolbox?
 Medication Management: Is It in Your Toolbox? Brian K. Esterly, MBA, SVP, Corporate Development, excellerx, Inc. O: 215.282.1676, besterly@excellerx.com What has been your Medication Management experience?
Medication Management: Is It in Your Toolbox? Brian K. Esterly, MBA, SVP, Corporate Development, excellerx, Inc. O: 215.282.1676, besterly@excellerx.com What has been your Medication Management experience?
enotification: Adapting ereferral for Public Health Notifiable Disease Reporting in New Zealand
 Case Report Healthc Inform Res. 2012 September;18(3):225-230. pissn 2093-3681 eissn 2093-369X enotification: Adapting ereferral for Public Health Notifiable Disease Reporting in New Zealand Nicholas F.
Case Report Healthc Inform Res. 2012 September;18(3):225-230. pissn 2093-3681 eissn 2093-369X enotification: Adapting ereferral for Public Health Notifiable Disease Reporting in New Zealand Nicholas F.
Strategies for Successful Medication Reconciliation and Management. Steve Kennedy, PharmD Paula Zelle, PharmD, FASHP
 Strategies for Successful Medication Reconciliation and Management Steve Kennedy, PharmD Paula Zelle, PharmD, FASHP Disclosures The speakers declare no conflicts of interest or financial interest in any
Strategies for Successful Medication Reconciliation and Management Steve Kennedy, PharmD Paula Zelle, PharmD, FASHP Disclosures The speakers declare no conflicts of interest or financial interest in any
Strategies for Successful Medication Reconciliation and Management
 Strategies for Successful Medication Reconciliation and Management Steve Kennedy, PharmD Paula Zelle, PharmD, FASHP Disclosures The speakers declare no conflicts of interest or financial interest in any
Strategies for Successful Medication Reconciliation and Management Steve Kennedy, PharmD Paula Zelle, PharmD, FASHP Disclosures The speakers declare no conflicts of interest or financial interest in any
Reviewing Methods Used in Patient Safety Research: Advantages and Disadvantages. This SPSRN work is funded by
 Reviewing Methods Used in Patient Safety Research: Advantages and Disadvantages Dr Jeanette Jackson (j.jackson@abdn.ac.uk) This SPSRN work is funded by Introduction Effective management of patient safety
Reviewing Methods Used in Patient Safety Research: Advantages and Disadvantages Dr Jeanette Jackson (j.jackson@abdn.ac.uk) This SPSRN work is funded by Introduction Effective management of patient safety
Kupu Taurangi Hauora o Aotearoa
 Kupu Taurangi Hauora o Aotearoa National GTT Workshop 2014 Using Data for Improvement Update Global Trigger Tool (GTT) Targeted chart reviews using triggers as flags for patient harm Provides a high level
Kupu Taurangi Hauora o Aotearoa National GTT Workshop 2014 Using Data for Improvement Update Global Trigger Tool (GTT) Targeted chart reviews using triggers as flags for patient harm Provides a high level
Maryland Patient Safety Center s Annual MEDSAFE Conference: Taking Charge of Your Medication Safety Challenges November 3, 2011 The Conference Center
 Maryland Patient Safety Center s Annual MEDSAFE Conference: Taking Charge of Your Medication Safety Challenges November 3, 2011 The Conference Center at the Maritime Institute Reducing Hospital Readmissions
Maryland Patient Safety Center s Annual MEDSAFE Conference: Taking Charge of Your Medication Safety Challenges November 3, 2011 The Conference Center at the Maritime Institute Reducing Hospital Readmissions
MEANINGFUL USE STAGE 2
 MEANINGFUL USE STAGE 2 PHASED-IN IMPLEMENTATION PROCESS DECEMBER 2014 - PREPARATION MONTH Start this process as early as possible WATCH VIDEO TRAINING SESSIONS: (Sessions available starting December 1,
MEANINGFUL USE STAGE 2 PHASED-IN IMPLEMENTATION PROCESS DECEMBER 2014 - PREPARATION MONTH Start this process as early as possible WATCH VIDEO TRAINING SESSIONS: (Sessions available starting December 1,
Pediatric Nurse Buddy Program Cohort 3 The impact of EMR on Healthcare Provider Wellness
 Pediatric Nurse Buddy Program Cohort 3 The impact of EMR on Healthcare Provider Wellness Disclosure Kayeleigh Higgerson, DO, UT Health SA, UHS has no relationships with commercial companies to disclose.
Pediatric Nurse Buddy Program Cohort 3 The impact of EMR on Healthcare Provider Wellness Disclosure Kayeleigh Higgerson, DO, UT Health SA, UHS has no relationships with commercial companies to disclose.
Introduction of EPMA in paediatric practice in UK:
 Introduction of EPMA in paediatric practice in UK: REALISING THE CLINICAL BENEFITS AND ENGAGING CLINICAL STAFF Stephen Marks Consultant Paediatric Nephrologist and EPMA lead Great Ormond Street Hospital
Introduction of EPMA in paediatric practice in UK: REALISING THE CLINICAL BENEFITS AND ENGAGING CLINICAL STAFF Stephen Marks Consultant Paediatric Nephrologist and EPMA lead Great Ormond Street Hospital
Quality Management Building Blocks
 Quality Management Building Blocks Quality Management A way of doing business that ensures continuous improvement of products and services to achieve better performance. (General Definition) Quality Management
Quality Management Building Blocks Quality Management A way of doing business that ensures continuous improvement of products and services to achieve better performance. (General Definition) Quality Management
Review Process. Introduction. InterQual Level of Care Criteria Long-Term Acute Care Criteria
 InterQual Level of Care Criteria Long-Term Acute Care Criteria Review Process Introduction InterQual Level of Care Criteria support determining the appropriateness of Long-Term Acute Care (LTAC) admission,
InterQual Level of Care Criteria Long-Term Acute Care Criteria Review Process Introduction InterQual Level of Care Criteria support determining the appropriateness of Long-Term Acute Care (LTAC) admission,
The Impact of CPOE and CDS on the Medication Use Process and Pharmacist Workflow
 The Impact of CPOE and CDS on the Medication Use Process and Pharmacist Workflow Conflict of Interest Disclosure The speaker has no real or apparent conflicts of interest to report. Anne M. Bobb, R.Ph.,
The Impact of CPOE and CDS on the Medication Use Process and Pharmacist Workflow Conflict of Interest Disclosure The speaker has no real or apparent conflicts of interest to report. Anne M. Bobb, R.Ph.,
Mental Health Care and OpenVista
 Medsphere Systems Corporation Mental and OpenVista Version 2.0 The OpenVista Platform: Integrated Support for Mental Designed by clinicians from all healthcare disciplines, OpenVista is guided by the principle
Medsphere Systems Corporation Mental and OpenVista Version 2.0 The OpenVista Platform: Integrated Support for Mental Designed by clinicians from all healthcare disciplines, OpenVista is guided by the principle
Mental Health Care and OpenVista
 Medsphere Systems Corporation Mental and OpenVista Version 2.0 The OpenVista Platform: Integrated Support for Mental Designed by clinicians from all healthcare disciplines, OpenVista is guided by the principle
Medsphere Systems Corporation Mental and OpenVista Version 2.0 The OpenVista Platform: Integrated Support for Mental Designed by clinicians from all healthcare disciplines, OpenVista is guided by the principle
Improving patient safety and infection. Patient Safety Forum Dr J Coleman 1 ELECTRONIC PRESCRIBING AND CLINICAL DECISION SUPPORT (CDS)
 Improving Patient Safety and Infection Control Through Electronic Prescribing Dr Jamie Coleman Senior Lecturer in Clinical Pharmacology / Honorary Consultant Physician The brief Clinical computing technologies
Improving Patient Safety and Infection Control Through Electronic Prescribing Dr Jamie Coleman Senior Lecturer in Clinical Pharmacology / Honorary Consultant Physician The brief Clinical computing technologies
Measure #46 (NQF 0097): Medication Reconciliation Post-Discharge National Quality Strategy Domain: Communication and Care Coordination
 Measure #46 (NQF 0097): Medication Reconciliation Post-Discharge National Quality Strategy Domain: Communication and Care Coordination 2017 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY MEASURE TYPE: Process
Measure #46 (NQF 0097): Medication Reconciliation Post-Discharge National Quality Strategy Domain: Communication and Care Coordination 2017 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY MEASURE TYPE: Process
Background and Methodology
 Study Sites and Investigators Emergency Department Pharmacists Improve Patient Safety: Results of a Multicenter Study Supported by the ASHP Foundation Jeffrey Rothschild, MD, MPH-Principal Investigator
Study Sites and Investigators Emergency Department Pharmacists Improve Patient Safety: Results of a Multicenter Study Supported by the ASHP Foundation Jeffrey Rothschild, MD, MPH-Principal Investigator
Acute Care Workflow Solutions
 Acute Care Workflow Solutions 2016 North American General Acute Care Workflow Solutions Product Leadership Award The Philips IntelliVue Guardian solution provides general floor, medical-surgical units,
Acute Care Workflow Solutions 2016 North American General Acute Care Workflow Solutions Product Leadership Award The Philips IntelliVue Guardian solution provides general floor, medical-surgical units,
a. General E Code Coding Guidelines
 19. Supplemental Classification of External Causes of Injury and Poisoning (E-codes, E800-E999) Introduction: These guidelines are provided for those who are currently collecting E codes in order that
19. Supplemental Classification of External Causes of Injury and Poisoning (E-codes, E800-E999) Introduction: These guidelines are provided for those who are currently collecting E codes in order that
Assessing Medical Technology- Are We Being Told the Truth. The Case of CPOE. David C Classen M.D., M.S. FCG and University of Utah
 Assessing Medical Technology- Are We Being Told the Truth. The Case of CPOE David C Classen M.D., M.S. FCG and University of Utah August 21, 2007 FCG 2006 Slide 1 November 2006 CPOE Adoption Growing Despite
Assessing Medical Technology- Are We Being Told the Truth. The Case of CPOE David C Classen M.D., M.S. FCG and University of Utah August 21, 2007 FCG 2006 Slide 1 November 2006 CPOE Adoption Growing Despite
Measure #46 (NQF 0097): Medication Reconciliation Post-Discharge National Quality Strategy Domain: Communication and Care Coordination
 Measure #46 (NQF 0097): Medication Reconciliation Post-Discharge National Quality Strategy Domain: Communication and Care Coordination 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE:
Measure #46 (NQF 0097): Medication Reconciliation Post-Discharge National Quality Strategy Domain: Communication and Care Coordination 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE:
Qualifying for Medicare Incentive Payments with Crystal Practice Management. Version 1.0
 Qualifying for Medicare Incentive Payments with Crystal Practice Management Version 1.0 July 18, Table of Contents Qualifying for Medicare Incentive Payments with... 1 General Information... 3 Links to
Qualifying for Medicare Incentive Payments with Crystal Practice Management Version 1.0 July 18, Table of Contents Qualifying for Medicare Incentive Payments with... 1 General Information... 3 Links to
Using Predictive Analytics to Improve Sepsis Outcomes 4/23/2014
 Using Predictive Analytics to Improve Sepsis Outcomes 4/23/2014 Ryan Arnold, MD Department of Emergency Medicine and Value Institute Christiana Care Health System, Newark, DE Susan Niemeier, RN Chief Nursing
Using Predictive Analytics to Improve Sepsis Outcomes 4/23/2014 Ryan Arnold, MD Department of Emergency Medicine and Value Institute Christiana Care Health System, Newark, DE Susan Niemeier, RN Chief Nursing
Real Time Alert System: A Disease Management System Leveraging Health Information Exchange
 Real Time Alert System: A Disease Management System Leveraging Health Information Exchange Vibha Anand 1, 2, Meena E. Sheley 1, Shawn Xu 1,Stephen M. Downs 1, 2 1 Children s Health Services Research, Indiana
Real Time Alert System: A Disease Management System Leveraging Health Information Exchange Vibha Anand 1, 2, Meena E. Sheley 1, Shawn Xu 1,Stephen M. Downs 1, 2 1 Children s Health Services Research, Indiana
Improving Sign-Outs in Hospital Medicine
 Improving Sign-Outs in Hospital Medicine Arpana R. Vidyarthi, MD Assistant Professor of Medicine Division of Hospital Medicine Director of Quality, Division of Hospital Medicine Director, Patient Safety
Improving Sign-Outs in Hospital Medicine Arpana R. Vidyarthi, MD Assistant Professor of Medicine Division of Hospital Medicine Director of Quality, Division of Hospital Medicine Director, Patient Safety
Order Source Misattribution: The Impact on CPOE Metrics
 Order Source Misattribution: The Impact on CPOE Metrics Linda Catzoela, RN, BSN, Clinical Informaticist George Gellert, MD, MPH, MPA, Associate System CMIO CHRISTUS Health March 3, 2016 Co-authors and
Order Source Misattribution: The Impact on CPOE Metrics Linda Catzoela, RN, BSN, Clinical Informaticist George Gellert, MD, MPH, MPA, Associate System CMIO CHRISTUS Health March 3, 2016 Co-authors and
Advancing Care Information Performance Category Fact Sheet
 Fact Sheet The Medicare Access and CHIP Reauthorization Act of 2015 (MACRA) replaced three quality programs (the Medicare Electronic Health Record (EHR) Incentive program, the Physician Quality Reporting
Fact Sheet The Medicare Access and CHIP Reauthorization Act of 2015 (MACRA) replaced three quality programs (the Medicare Electronic Health Record (EHR) Incentive program, the Physician Quality Reporting
3. Does the institution have a dedicated hospital-wide committee geared towards the improvement of laboratory test stewardship? a. Yes b.
 Laboratory Stewardship Checklist: Governance Leadership Commitment It is extremely important that the Laboratory Stewardship Committee is sanctioned by the hospital leadership. This may be recognized by
Laboratory Stewardship Checklist: Governance Leadership Commitment It is extremely important that the Laboratory Stewardship Committee is sanctioned by the hospital leadership. This may be recognized by
Update on ACG Guidelines Stephen B. Hanauer, MD President American College of Gastroenterology
 Update on ACG Guidelines Stephen B. Hanauer, MD President American College of Gastroenterology Clifford Joseph Barborka Professor of Medicine Northwestern University Feinberg School of Medicine Guideline
Update on ACG Guidelines Stephen B. Hanauer, MD President American College of Gastroenterology Clifford Joseph Barborka Professor of Medicine Northwestern University Feinberg School of Medicine Guideline
Leapfrog Group Report on CPOE Evaluation Tool Results June 2008 to January 2010
 Leapfrog Group Report on CPOE Evaluation Tool Results June 2008 to January 2010 Executive Summary Using The Leapfrog Group s web based simulation tool, 214 hospitals tested their computerized physician
Leapfrog Group Report on CPOE Evaluation Tool Results June 2008 to January 2010 Executive Summary Using The Leapfrog Group s web based simulation tool, 214 hospitals tested their computerized physician
SNOMED CT AND ICD-10-BE: TWO OF A KIND?
 Federal Public Service of Health, Food Chain Safety and Environment Directorate-General Health Care Department Datamanagement Arabella D Havé, chief of Terminology, Classification, Grouping & Audit arabella.dhave@health.belgium.be
Federal Public Service of Health, Food Chain Safety and Environment Directorate-General Health Care Department Datamanagement Arabella D Havé, chief of Terminology, Classification, Grouping & Audit arabella.dhave@health.belgium.be
PROPOSED CHANGES TO THE 2018 LEAPFROG HOSPITAL SURVEY
 PROPOSED CHANGES TO THE 2018 LEAPFROG HOSPITAL SURVEY OPEN FOR PUBLIC COMMENT Each year, The Leapfrog Group s team of researchers reviews the literature and convenes expert panels to ensure the Leapfrog
PROPOSED CHANGES TO THE 2018 LEAPFROG HOSPITAL SURVEY OPEN FOR PUBLIC COMMENT Each year, The Leapfrog Group s team of researchers reviews the literature and convenes expert panels to ensure the Leapfrog
Nova Scotia Drug Information System
 Nova Scotia Drug Information System INTRODUCTION Presentation Details: Slides: 21 Duration: 00:22:44 Filename: Module1.Introduction.ppt Presenter Details: Slide 1 Nova Scotia Drug Information System Duration:
Nova Scotia Drug Information System INTRODUCTION Presentation Details: Slides: 21 Duration: 00:22:44 Filename: Module1.Introduction.ppt Presenter Details: Slide 1 Nova Scotia Drug Information System Duration:
Hospital Guidance Webinar
 Hospital Guidance Webinar Using the CPOE Tool Results for Quality Improvement PRESENTED BY: DAVID BATES, MD, MS C AND DAVID CLASSEN, MD, MS C Overview Introduction What the current test looks like and
Hospital Guidance Webinar Using the CPOE Tool Results for Quality Improvement PRESENTED BY: DAVID BATES, MD, MS C AND DAVID CLASSEN, MD, MS C Overview Introduction What the current test looks like and
PHARMACY SERVICES/MEDICATION USE
 25.01. 10 Drug Reactions & Administration Errors & Incompatibilities. Drug administration errors, adverse drug reactions and incompatibilities must be immediately reported to the attending physician and
25.01. 10 Drug Reactions & Administration Errors & Incompatibilities. Drug administration errors, adverse drug reactions and incompatibilities must be immediately reported to the attending physician and
Medication Reconciliation with Pharmacy Technicians
 Technician Education Day March 29, 2014 Jacksonville, FL Outline with Pharmacy Technicians Roma Merrick RPhT., CPhT. Pharmacy Technician Coordinator St. Vincent s Medical Center Southside Jacksonville,
Technician Education Day March 29, 2014 Jacksonville, FL Outline with Pharmacy Technicians Roma Merrick RPhT., CPhT. Pharmacy Technician Coordinator St. Vincent s Medical Center Southside Jacksonville,
Automation and Information Technology
 4 Automation and Information Technology Positions Automation and Information Technology Ensuring Patient Safety and Data Integrity During Cyber-attacks (1701) To advocate that healthcare organizations
4 Automation and Information Technology Positions Automation and Information Technology Ensuring Patient Safety and Data Integrity During Cyber-attacks (1701) To advocate that healthcare organizations
MorCare Infection Prevention prevent hospital-acquired infections proactively
 Infection Prevention prevent hospital-acquired infections proactively Enterprise Software and Consulting Solutions for Improved Population Health s Enterprise Software and Consulting Solutions Healthcare
Infection Prevention prevent hospital-acquired infections proactively Enterprise Software and Consulting Solutions for Improved Population Health s Enterprise Software and Consulting Solutions Healthcare
Protocol Applies To: UW Health Clinics: all adult outpatients with an active order for warfarin
 Protocol Number: 7 Protocol Title: Ambulatory Initiation and Management of Warfarin for Adults Protocol Applies To: UW Health Clinics: all adult outpatients with an active order for warfarin Target Patient
Protocol Number: 7 Protocol Title: Ambulatory Initiation and Management of Warfarin for Adults Protocol Applies To: UW Health Clinics: all adult outpatients with an active order for warfarin Target Patient
DEPARTMENT OF HEALTH AND HUMAN SERVICES CENTERS FOR MEDICARE & MEDICAID SERVICES
 Use for a resident who has potentially unnecessary medications, is prescribed psychotropic medications or has the potential for an adverse outcome to determine whether facility practices are in place to
Use for a resident who has potentially unnecessary medications, is prescribed psychotropic medications or has the potential for an adverse outcome to determine whether facility practices are in place to
Medication Safety Action Bundle Adverse Drug Events (ADE) All High-Risk Medication Safety
 Medication Safety Action Bundle Adverse Drug Events (ADE) All High-Risk Medication Safety Background The Institute of medicine (IOM) estimates that 1.5 million preventable Adverse Drug Events (ADE) occur
Medication Safety Action Bundle Adverse Drug Events (ADE) All High-Risk Medication Safety Background The Institute of medicine (IOM) estimates that 1.5 million preventable Adverse Drug Events (ADE) occur
BMHI Internship Presentation. Saba Akbar UNC Chapel Hill Apr 11, 2018
 BMHI Internship Presentation Saba Akbar UNC Chapel Hill Apr 11, 2018 2 Centre for Healthcare Resilience and Implementation Science Centre for Health Informatics Centre for Health Systems and Safety Research
BMHI Internship Presentation Saba Akbar UNC Chapel Hill Apr 11, 2018 2 Centre for Healthcare Resilience and Implementation Science Centre for Health Informatics Centre for Health Systems and Safety Research
Essential Skills for Evidence-based Practice: Evidence Access Tools
 Essential Skills for Evidence-based Practice: Evidence Access Tools Jeanne Grace Corresponding author: J. Grace E-mail: Jeanne_Grace@urmc.rochester.edu Jeanne Grace RN PhD Emeritus Clinical Professor of
Essential Skills for Evidence-based Practice: Evidence Access Tools Jeanne Grace Corresponding author: J. Grace E-mail: Jeanne_Grace@urmc.rochester.edu Jeanne Grace RN PhD Emeritus Clinical Professor of
A Pharmacist Network for Integrated Medication Management in the Medical Home
 A Pharmacist Network for Integrated Medication Management in the Medical Home Marie Smith, PharmD UConn School of Pharmacy Professor/Dept. Head Pharmacy Practice Asst. Dean, Practice and Public Policy
A Pharmacist Network for Integrated Medication Management in the Medical Home Marie Smith, PharmD UConn School of Pharmacy Professor/Dept. Head Pharmacy Practice Asst. Dean, Practice and Public Policy
Using Data to Inform Quality Improvement
 20 15 10 5 0 Using Data to Inform Quality Improvement Ethan Kuperman, MD FHM Aparna Kamath, MD MS Justin Glasgow, MD PhD Disclosures None of the presenters today have relevant personal or financial conflicts
20 15 10 5 0 Using Data to Inform Quality Improvement Ethan Kuperman, MD FHM Aparna Kamath, MD MS Justin Glasgow, MD PhD Disclosures None of the presenters today have relevant personal or financial conflicts
Predicting 30-day Readmissions is THRILing
 2016 CLINICAL INFORMATICS SYMPOSIUM - CONNECTING CARE THROUGH TECHNOLOGY - Predicting 30-day Readmissions is THRILing OUT OF AN OLD MODEL COMES A NEW Texas Health Resources 25 hospitals in North Texas
2016 CLINICAL INFORMATICS SYMPOSIUM - CONNECTING CARE THROUGH TECHNOLOGY - Predicting 30-day Readmissions is THRILing OUT OF AN OLD MODEL COMES A NEW Texas Health Resources 25 hospitals in North Texas
Health Management Information Systems
 Health Management Information Systems Computerized Provider Order Entry (CPOE) Computerized Provider Order Entry (CPOE) Learning Objectives 1. Describe the purpose, attributes and functions of CPOE 2.
Health Management Information Systems Computerized Provider Order Entry (CPOE) Computerized Provider Order Entry (CPOE) Learning Objectives 1. Describe the purpose, attributes and functions of CPOE 2.
Long-Stay Alternate Level of Care in Ontario Mental Health Beds
 Health System Reconfiguration Long-Stay Alternate Level of Care in Ontario Mental Health Beds PREPARED BY: Jerrica Little, BA John P. Hirdes, PhD FCAHS School of Public Health and Health Systems University
Health System Reconfiguration Long-Stay Alternate Level of Care in Ontario Mental Health Beds PREPARED BY: Jerrica Little, BA John P. Hirdes, PhD FCAHS School of Public Health and Health Systems University
Update in Medical Informatics
 Update in Medical Informatics (Some random papers that we found) PCC Ojai, California June 2014 Colin Banas, CMIO, Virginia Commonwealth University Bill Galanter, Associate CHIO, University of Illinois
Update in Medical Informatics (Some random papers that we found) PCC Ojai, California June 2014 Colin Banas, CMIO, Virginia Commonwealth University Bill Galanter, Associate CHIO, University of Illinois
Pharmaceutical Services Report to Joint Conference Committee September 2010
 Pharmaceutical Services Report to Joint Conference Committee September 21 Background: Pharmaceutical Services staffing has increased by 31 FTE from 26 due to program changes and to comply with regulatory
Pharmaceutical Services Report to Joint Conference Committee September 21 Background: Pharmaceutical Services staffing has increased by 31 FTE from 26 due to program changes and to comply with regulatory
European Association of Hospital Pharmacists (EAHP)
 European Association of Hospital Pharmacists (EAHP) Consultation Response Delegated Act on the detailed rules for a unique identifier for medicinal products for human use, and its verification. April 2012
European Association of Hospital Pharmacists (EAHP) Consultation Response Delegated Act on the detailed rules for a unique identifier for medicinal products for human use, and its verification. April 2012
Survey about Venous Thrombo-Embolism (VTE) Prophylaxis. Nurses
 Survey about Venous Thrombo-Embolism (VTE) Prophylaxis Nurses Dear staff member, This is a short survey about venous thromboembolism (VTE) at your hospital organization. Venous Thromboembolism (VTE) is
Survey about Venous Thrombo-Embolism (VTE) Prophylaxis Nurses Dear staff member, This is a short survey about venous thromboembolism (VTE) at your hospital organization. Venous Thromboembolism (VTE) is
Preventing Adverse Drug Events and Harm
 Preventing Adverse Drug Events and Harm Frank Federico, RPh, IHI Executive Director Steve Meisel, PharmD, IHI Faculty March 27th,2012 12:00-1:00pm ET Beth O Donnell, MPH Beth O Donnell, MPH, Institute
Preventing Adverse Drug Events and Harm Frank Federico, RPh, IHI Executive Director Steve Meisel, PharmD, IHI Faculty March 27th,2012 12:00-1:00pm ET Beth O Donnell, MPH Beth O Donnell, MPH, Institute
Measuring Comprehensiveness of Primary Care: Past, Present, and Future
 Measuring Comprehensiveness of Primary Care: Past, Present, and Future Mathematica Policy Research Washington, DC June 27, 2014 Welcome Moderator Eugene Rich, M.D. Mathematica Policy Research 2 About CHCE
Measuring Comprehensiveness of Primary Care: Past, Present, and Future Mathematica Policy Research Washington, DC June 27, 2014 Welcome Moderator Eugene Rich, M.D. Mathematica Policy Research 2 About CHCE
Adverse Drug Events: A Focus on Anticoagulation Steve Meisel, Pharm.D., CPPS Director of Patient Safety Fairview Health Services, Minneapolis, MN
 Adverse Drug Events: A Focus on Anticoagulation Steve Meisel, Pharm.D., CPPS Director of Patient Safety Fairview Health Services, Minneapolis, MN Fairview Health Services 6 hospitals, ranging from rural
Adverse Drug Events: A Focus on Anticoagulation Steve Meisel, Pharm.D., CPPS Director of Patient Safety Fairview Health Services, Minneapolis, MN Fairview Health Services 6 hospitals, ranging from rural
Efficacy of Tympanostomy Tubes for Children with Recurrent Acute Otitis Media Randomization Phase
 CONSENT FOR A CHILD TO BE A SUBJECT IN MEDICAL RESEARCH AND AUTHORIZATION TO PERMIT THE USE AND SHARING OF IDENTIFIABLE MEDICAL INFORMATION FOR RESEARCH PURPOSES TITLE Efficacy of Tympanostomy Tubes for
CONSENT FOR A CHILD TO BE A SUBJECT IN MEDICAL RESEARCH AND AUTHORIZATION TO PERMIT THE USE AND SHARING OF IDENTIFIABLE MEDICAL INFORMATION FOR RESEARCH PURPOSES TITLE Efficacy of Tympanostomy Tubes for
Improvement Activities for ACI Bonus Measures
 Improvement Activity Performance Category Subcategory Expanded Practice Activity Name Activity Improvement Activity Performance Category Weight Provide 24/7 access to eligible clinicians or groups, who
Improvement Activity Performance Category Subcategory Expanded Practice Activity Name Activity Improvement Activity Performance Category Weight Provide 24/7 access to eligible clinicians or groups, who
3M Health Information Systems. 3M Clinical Risk Groups: Measuring risk, managing care
 3M Health Information Systems 3M Clinical Risk Groups: Measuring risk, managing care 3M Clinical Risk Groups: Measuring risk, managing care Overview The 3M Clinical Risk Groups (CRGs) are a population
3M Health Information Systems 3M Clinical Risk Groups: Measuring risk, managing care 3M Clinical Risk Groups: Measuring risk, managing care Overview The 3M Clinical Risk Groups (CRGs) are a population
Understanding and improving the quality of medication use: Research in Clinical Pharmacy starting from Academia. Anne Spinewine
 Understanding and improving the quality of medication use: Research in Clinical Pharmacy starting from Academia Clinical Pharmacy Research Group (CLIP) Anne Spinewine 1 04.10.2011 WBI- UCL Research activities
Understanding and improving the quality of medication use: Research in Clinical Pharmacy starting from Academia Clinical Pharmacy Research Group (CLIP) Anne Spinewine 1 04.10.2011 WBI- UCL Research activities
Novel combined patient instruction and discharge summary tool improves timeliness of documentation and outpatient provider satisfaction
 701053SMO0010.1177/2050312117701053SAGE Open MedicineGilliam et al. research-article2017 Original Article SAGE Open Medicine Novel combined patient instruction and discharge summary tool improves timeliness
701053SMO0010.1177/2050312117701053SAGE Open MedicineGilliam et al. research-article2017 Original Article SAGE Open Medicine Novel combined patient instruction and discharge summary tool improves timeliness
Medication Reconciliation
 Medication Reconciliation Where are we now? Angie Powell, PharmD Director of Pharmacy Baxter Regional Medical Center Disclosures I, Angie Powell, have no relevant financial relationships to disclose. Learning
Medication Reconciliation Where are we now? Angie Powell, PharmD Director of Pharmacy Baxter Regional Medical Center Disclosures I, Angie Powell, have no relevant financial relationships to disclose. Learning
Safe Medication Practices
 Safe Medication Practices Patient Safety: Preventing Adverse Events OHA Conference Renaissance Toronto Hotel at SkyDome Toronto June 14, 2004 David U President & CEO, ISMP Canada Agenda ISMP Canada Patient
Safe Medication Practices Patient Safety: Preventing Adverse Events OHA Conference Renaissance Toronto Hotel at SkyDome Toronto June 14, 2004 David U President & CEO, ISMP Canada Agenda ISMP Canada Patient
Electronic Prescribing of Chemotherapy-It s Not a Video Game!
 Faculty Disclosures Electronic Prescribing of Chemotherapy-It s Not a Video Game! Mary Mably has no disclosures Mary S. Mably, RPh, BCOP Pharmacy Oncology Coordinator, University of Wisconsin Hospital
Faculty Disclosures Electronic Prescribing of Chemotherapy-It s Not a Video Game! Mary Mably has no disclosures Mary S. Mably, RPh, BCOP Pharmacy Oncology Coordinator, University of Wisconsin Hospital
A Layered Learning Medication Reconciliation Program
 A Layered Learning Medication Reconciliation Program Brittany Bates, PharmD, BCPS Clinical Pharmacist, Lima Memorial Health System Clinical Assistant Professor, Ohio Northern University Jana Randolph,
A Layered Learning Medication Reconciliation Program Brittany Bates, PharmD, BCPS Clinical Pharmacist, Lima Memorial Health System Clinical Assistant Professor, Ohio Northern University Jana Randolph,
Belgian Meaningful Use Criteria for Mental Healthcare Hospitals and other non-general Hospitals
 Belgian Meaningful Use Criteria for Mental Healthcare Hospitals and other non-general Hospitals Introduction This document is the result the conclusion of the WG Belgian Meaningful Use Criteria for Mental
Belgian Meaningful Use Criteria for Mental Healthcare Hospitals and other non-general Hospitals Introduction This document is the result the conclusion of the WG Belgian Meaningful Use Criteria for Mental
POLICY BRIEF. Identifying Adverse Drug Events in Rural Hospitals: An Eight-State Study. May rhrc.umn.edu. Background.
 POLICY BRIEF Identifying Adverse Drug Events in Rural Hospitals: An Eight-State Study Michelle Casey, MS Peiyin Hung, MSPH Emma Distel, MPH Shailendra Prasad, MBBS, MPH Key Findings In 2013, Critical Access
POLICY BRIEF Identifying Adverse Drug Events in Rural Hospitals: An Eight-State Study Michelle Casey, MS Peiyin Hung, MSPH Emma Distel, MPH Shailendra Prasad, MBBS, MPH Key Findings In 2013, Critical Access
Bulletin Independent prescribing information for NHS Wales
 Bulletin Independent prescribing information for NHS Wales Medicines-related admissions February 2015 Although medicines play an important role in the management of chronic and acute illnesses, they can
Bulletin Independent prescribing information for NHS Wales Medicines-related admissions February 2015 Although medicines play an important role in the management of chronic and acute illnesses, they can
Engaging Staff in EHR Implementation and Reducing Risk: Making Your Laboratory Data SAFER
 Engaging Staff in EHR Implementation and Reducing Risk: Making Your Laboratory Data SAFER Megan E. Sawchuk, MT(ASCP) Health Scientist CLMA KnowledgeLab 2015 Orlando, FL March 31, 2015 Center for Surveillance,
Engaging Staff in EHR Implementation and Reducing Risk: Making Your Laboratory Data SAFER Megan E. Sawchuk, MT(ASCP) Health Scientist CLMA KnowledgeLab 2015 Orlando, FL March 31, 2015 Center for Surveillance,
Chapter 14 Regina Qu Appelle Regional Health Authority Safe and Timely Discharge of Hospital Patients 1.0 MAIN POINTS
 Chapter 14 Regina Qu Appelle Regional Health Authority Safe and Timely Discharge of Hospital Patients 1.0 MAIN POINTS Safe and timely discharge of patients from hospitals helps ensure patients well-being
Chapter 14 Regina Qu Appelle Regional Health Authority Safe and Timely Discharge of Hospital Patients 1.0 MAIN POINTS Safe and timely discharge of patients from hospitals helps ensure patients well-being
