Faculty Overview of the September 10-11, 2007 FDP meeting
|
|
|
- Lambert Thompson
- 5 years ago
- Views:
Transcription
1 Faculty Overview of the September 10-11, 2007 FDP meeting Overall, the September FDP meeting was a very forward-looking meeting, which included many discussions of the plans for Phase V of the FDP. The framework developing for Phase V of the FDP presents continued opportunities for increasing faculty participation in FDP activities and priorities. The draft administrative policies for Phase V state that The FDP s Purpose is to promote increased research productivity in the conduct of federally supported research and education, provide for enhanced stewardship under federally supported programs, and achieve reductions in administrative burdens and costs associated with sponsored research and education. During Phase IV, the findings from of the Faculty Burden Survey have shown the negative impact on productivity resulting from the increased the administrative burdens on the faculty. In addition, the activities of the faculty representatives have become more focused and more closely interwoven into the overall fabric of the FDP. There seems to be a general expectation that the voice and viewpoints of the faculty members will be critical in planning and carrying out the activities of the FDP during the remainder of Phase IV and during the implementation and execution of Phase V. The Sessions began with Welcoming Remarks from the FDP Chair, Nancy Wray and from the Vice Chair of the FDP and Chair of the Faculty Committee, Sara Rockwell. Nancy Wray welcomed the participants to the FDP meeting. She then reminded the participants that this was the last year of Phase IV of the FDP and mentioned that Phase V would bring some changes in the structure of the FDP (e.g. new members) and would lead to considerations of new areas (e.g. problems in oversight of international research projects). Sara Rockwell then welcomed the participants and reminded the faculty that the faculty voice was very important to the FDP and was one of the unique features of the FDP. There are many forums in which university administrators meet with one another and with representatives of federal agencies, but few of these venues include significant numbers of faculty members. A unique feature of the FDP is therefore that faculty members are present and engaged in the discussions and in the development of the projects and demonstrations of the organization. Dr Rockwell then reminded faculty of the two faculty sessions, pointed out some other sessions of special interest to faculty members, and stressed the importance of broad faculty participation in all sessions of the meeting. Dick Seligman and Joanna Rom then discussed the Phase V Transition. Phase V will last 5 years, and the FDP will be open to new members as the phase begins. There will be some changes in the administration of the FDP at the beginning of Phase V. These include the possible formation of an oversight board including senior federal officials to help guide and review the activities of the FDP. Elections of the next chair and vice chair will be timed so that they begin their service with the initiation of Phase V, in September The incoming chair and vice chair will serve 3 year terms. The Federal Updates Session was disappointing from a faculty perspective, as several federal agencies did not present, even though some had representatives present in the audience. Joe Ellis presented an update from NIH which began with a reminder that NIH would be on continuing resolution until November or December, and that this would impact both competitive and continuing awards. He also noted that award notifications are now 90% electronic and that this would soon reach 100%, underscoring the importance of to investigators of using the NIH commons. He also noted that although the NIH 398 was to expire at the end of September, it was still active and that NIH would continue to use it. As of October1, he reported, NIH would no longer accept hard copies of financial reports. Finally, he reported on the NIH RFI for comments 1
2 and suggestions concerning peer review, and noted that NIH had received >1000 comments over the web or by , plus additional comments in the Regional Meetings. Jean Feldman the presented an update from the NSF, which is well captured in the PowerPoint mounted on the FDP website, describing some important changes and potential changes in NSF programs, review procedures, cost sharing policies and grant applications, as well as some new web resources for faculty. Frank O Day reported briefly for ONR on the PKI requirement and on some internal infrastructure restructuring, which should not affect faculty. Merrilea Mayo reported for GUIRR on a workshop considering the special problems of faculty submitting applications through Emerging Research Institutions, which result from the lack of time and lack of administrative infrastructure at their institutions, and presented some potential approaches to mitigating these problems. Her report is well captured by the PowerPoint on the FDP website. Morning Breakout Sessions The session on Administration of Awards and Subawards Conducted in Foreign Countries, led by Richard Ortisi of ONR and Deloris Pettis of Harvard, discussed agency expectations and institutional processes. Concerns and problems related to the oversight of complex projects in regions with limited administrative infrastructure and very limited financial resources were discussed, as were problems arising from difficulties in communication and diverse cultural expectations. PowerPoints of both presentations are on the FDP website. The Session on the A-133 Audit Demonstration, led by James Becker and Lynn Johnson, described a pilot involving 9 FDP Institutions that has recently been completed. The group is soliciting interest from additional FDP Institutions to move into a full demonstration regarding audit findings on sub-recipient awards. The goal is to show OMB that there are minimal audit findings and therefore inter-organizational monitoring is an unnecessary exercise. The demonstration would be to test electronic forms to respond to A-133 audit findings broken down into three major headings, namely 1) Questioned Costs; 2) Systemic Findings; and 3) Compliance Findings. The potential impact to faculty is that if such monitoring is deemed superfluous, faculty would no longer have to review and attest to sub-recipient charges as usual and customary. The reduction in administrative burden for faculty would probably be small relative to the reduction in institutional burden. The session on NSF s Grants Management Line of Business (GMLoB) consortium on Research.gov outlined the progress and plans for this new resource. The PowerPoint from the research.gov team describing this new resource, which is scheduled for a fall 2007 release, is available on the on the FDP website. The session on the Grants Management Line of Business (GMLoB) provided updated information on the attempt to streamline access to grant information on grant.gov and to provide services for multiple agencies. PowerPoints on the FDP website from the presenters, Tony Cavataio of the Department of Education and Michael Curtis of HHS cover these presentations nicely. 2
3 Lunch Discussion Sessions The Lunch Sessions included the Faculty Lunch. A number of issues were discussed at this meeting. Norka Ruiz-Bravo, Deputy Director for Extramural Research at NIH, followed up on her plenary presentation at the May FDP meeting to discuss further the problems related to the use of animals in research. She showed draft versions of potential NIH web resources and asked for feedback from the faculty. She also described resources and communications infrastructure needed by universities and described information and assistance available to NIH-funded institutions from the NIH. Sandra Schneider announced that she is leaving NSF to return to Academia, and introduced Mark Weiss, who will take her place as the NSF representative on the Human Subjects Research Subcommittee. The faculty expressed their willingness to continue working with the HSRS on the problems of mutual interest to the two groups. Tom Weber discussed his efforts to have NSF consider revisiting their policies of requiring detailed budgets at the time of grant submission, and described the negative reaction of the associate directors at NSF to a proposed demonstration project on modular budgets. The faculty offered to write a letter in support of this concept and in support of a pilot project demonstration testing the merit/feasibility of this approach with NSF grants. Joe Konstan was assigned the task of drafting this letter. Parallel lunch session aimed primarily at administrative representatives included a discussion session led by Debi Rafi (ONR) to follow up on the morning session on Administration of Awards and Subawards Conducted in Foreign Countries and a session led by John Harris (Northeastern), Deb Carmel (Harvard) and Karen Seward (ONR) discussing the possible streamlining of A-110/A-21 inventory requirements. A session devoted to the special problems at Emerging Research Institutions drew both faculty and administrative representatives. Plenary Session In the Plenary Session Gil Tran (OMB), Terry Hurst (HHS), Barbara Dorf (HUD) and Earl Warrington (GSA) discussed various aspects of the Federal Funding and Accountability Transparency Act (FFATA), which mandates the development of a searchable website that allows the public to ascertain the amount of federal money awarded to specific entities and to download the results of such searches. The funding types to be posted include grants, subgrants, loans, awards, cooperative agreements, and other forms of financial assistance as well as contracts, subcontracts, purchase orders, task orders, and delivery orders; they do not include individual transactions below $25,000. The systems to be used in this reporting are still under development. The concern for faculty is how much these new reporting requirements will increase the administrative burden on faculty and laboratory staff associated with their federally sponsored research and therefore how much they will increase the administrative costs they pay from the direct dollars on their research grants. The information provided (described in the slides on the FDP website) describes the systems being developed, but did not address the concerns of the faculty. Early Afternoon Breakout Sessions The four early afternoon breakout sessions included a session led by Jean Feldman (NSF) continuing the discussion of the implementation of the FFATA, and describing the new SF424 forms/procedures being developed to meet the requirements of this legislation. The slides for this presentation are on the website. In a simultaneous session entitled Best Practices Agency Matrix Steve Dowdy (MIT), Tammy 3
4 Custer (Cornell), Michelle Powell (Georgia Tech) and Ron Splittgerber (Colorado State) described their progress in establishing a template that summarized each Federal Agency s position on grants.gov issues. The slides (posted on the website) emphasize that very significant differences will still exist in agency requirements and practices despite the harmonization of forms and practices envisioned for the overarching grants.gov system. The Contracts Task Force session, co-chaired by Alexandra McKeown (Johns Hopkins) and Carol Zuiches (University of Washington) and Rosemary Hamill (NIH) considered current federal contracting issues, provided updates, and discussed the troublesome clauses demonstration. Unfortunately, no presentation or report from this session is yet available on the website. The session on Streamlining at NSF and NIH, led by Tom Weber (NSF) and Anne McNicol (NIH), discussed a proposed budget streamlining project at NSF that would allow just in time submission of detailed budgets and also discussed continued NIH streamlining efforts and activities. The faculty at the session expressed their continued support for streamlining efforts in all areas and their continued willingness to participate in information gathering, pilot projects and demonstrations that would facilitate streamlining and harmonization of processes at the funding agencies, citing the negative impact of unnecessary faculty administrative burdens and the value of reducing those burdens by streamlining and standardizing forms and procedures. Late Afternoon Breakout Sessions The Late Afternoon Breakout Sessions included a meeting on Project Certification led by Joe Ellis (NIH) and Andy Rudczynski (Yale). Discussions focused largely on the aspects of current standards, policies, and practices that the session participants felt could be improved by clarifications, streamlining, and refining. The Faculty Burden Survey was cited as showing the negative effects on productivity of the current level of regulatory burden. Problems associated with effort reporting were discussed, as was the need to clarify and streamline aspects of A21 and the principles behind costing, and the need for clarification on voluntary uncommitted cost sharing. The possibility of a faculty survey on time and effort reporting was discussed. Elements might include: is accountability currently in place; do the expectations reflect how science is optimally and actually done; is the process working. This was envisioned as raising the question of whether there was an unnecessarily large burden associated with current processes and whether there was a need for change. This discussion will be continued by session participants and agency representatives over the fall and a plan for proceeding will be brought to the January meeting if appropriate. Participation of faculty representatives in these discussions was encouraged. The session on the NSF Merit Review and Award Mechanism provided a report by Joanne Tornow (NSF) on trends in NSF funding, including a breakdown of the relative impact of those trends on different types of applications and investigators. The report, available on the website, shows that the success rate of NSF applications has fallen from 30% to 21% over the last decade. The decrease in the success rate was similar across the metrics presented (new and established investigators, solicited and unsolicited proposals, type of institution, etc). Applications increased by 50% over this period, reflecting both an increase in the number of applicants and an increase in the number of proposals submitted per applicant. Importantly, the proportion of highly rated proposals did not decline as a result of this increase in submissions, but the funding rate for the highly rated proposals decreased. The report also discussed the stress on the NSF peer review system as a result of these changes, the perception of the review and funding situation by the research community, and the increased use of submission 4
5 limitations (by PI and by institution) to manage the number of submissions received by the agency. The session on Terms and Conditions, led by Mike Ludwig (Purdue) and Jean Feldman (NSF) continued the ongoing discussions of problems in this area. Tuesday September 11, 2007 Tuesday Morning began with an early meeting of the Membership Standing Committee, led by Joanna Rom (NSF) and Susan Sedwick (University of Texas) to discuss current membership issues (e.g. requests for memberships; members that have not yet paid their dues) and to discuss membership issues related to Phase V (e.g. solicitation of members; requirements for membership). Early Morning Concurrent Sessions The Faculty Standing Committee meeting continued several discussions from the lunch meeting on Monday. There was continued interest in and discussion of the possibility of a session on the reasons why some institutions require detailed budgets even when agencies do not. David Robinson will continue to work on such a session for the January meeting, and (as he could not attend the September FDP meeting) ask that faculty who had ideas for the session and for possible participants contact him. He would like to have a team including a faculty member and an administrator present for each of the participating institutions. The faculty were asked to suggest other sessions for future meetings. Bill Osborne reported on the workshop on the Common Progress Reports, where he represented the FPD faculty as a speaker and member of the panel. Marilea Mayo reported on the Emerging Research Institution lunch, which occurred in parallel with the Monday Faculty lunch. She noted that faculty at ERI report that one of their major problems is obtaining protected time for research, which reflects not only the emphasis of these institutions on teaching and the heavy teaching load of these faculty but also the lack of robust administrative and academic infrastructures to support faculty research research efforts. Joe Konstan reported on progress in the development of a proposal for a pilot project aimed at providing PIs with support for certain administrative tasks related to their research. In the Faculty Burden Survey, the faculty noted that some of the burden associated with their research could be delegated to highly trained and skilled assistants. Joe is developing a proposal to test whether allowing PIs to budget for such support (or to rebudget small amounts of salary on existing grants to this purpose) would decrease the burden on faculty, without increasing the costs of the research, and thereby increase overall productivity. A white paper describing such a proposal is being drafted, to be distributed when appropriate with copies of the Faculty Burden Survey. The white paper will also include a discussion of the need for clarification from OMB of the level of complexity needed to allow budgeting of administrative support as a direct cost under A21. Plans for further distribution of the faculty Burden Survey were also discussed. Faculty were reminded to give Sara Rockwell, Nancy Wray or David Wright the names of organizations (e.g. federal agencies, professional societies, or scholarly publications) who should receive copies of the Faculty Burden Survey, with the name of a contact person if possible. 5
6 Joe Konstan also volunteered to write the draft of a letter to go to the NSF from the FDP describing the success of the NIH modular budget concept in reducing the administrative burden for PIs and proposing an FDP/NSF pilot project testing a similar requirement for a detailed budget only for projects likely to be funded, at the beginning of the funding negotiation process. Two sessions ran in parallel to the faculty session. In the Subawards session, Susan Boone (University of Chicago) and George Gardner (NIH) continued discussions of subawards to foreign recipients and reviewed the template for these awards that had been vetted recently by NSF. In the session on Streamlining Federal Agency Invoicing Systems, Debi Rafi led a continuation of the discussions of this issue. Late Morning Concurrent Sessions The ERA Grants.gov Update session presented by Terry Nicolosi and the grants.gov team updated the FDP on the current status and progress with this system and provided an opportunity for discussion and questions from institutions and investigators using grants.gov. The FFATA pilot Discussion Session continued discussions and planning of the pilot discussion begun in the session on Monday and described above. The session on the FASEB COI Toolkit, led by Carol Liedtke and Susan Sedwick, described and discussed a valuable new toolkit for investigators, institutions, editors, professional societies, and industry to use to help in the management of conflicts of interest. The toolkit is an unusual resource, because it considers the different needs and viewpoints of the different stakeholders, recognizes both the potential benefits of interactions between academic researchers and industry and also the need to manage conflicts of interest that could arise because of these interactions, and considers the principles involved in managing potential conflicts. The PowerPoints from the session are available on the FDP website. The Toolkit is available at Plenary Session At the final Plenary Session, Diane DiEuliis (OSTP) described the Research Business Models Subcommittee which reports to the NCST, a cabinet level council of advisors to the President on science and technology. She also described the priority areas of the RBM Subcommittee (facilitating collaborative multidisciplinary research, improving consistency of agency practices, and harmonizing accountability), and discussed its past results, recent progress, and developing activities in these areas. The PowerPoint of this presentation is available on the website. Respectfully Submitted, Sara Rockwell, PhD Chair, Faculty Standing Committee Vice Chair, FDP 6
Cooperative Framework of Institutions and Funding Agencies to Improve Administrative Burden: The story of the Federal Demonstration Partnership (FDP)
 Cooperative Framework of Institutions and Funding Agencies to Improve Administrative Burden: The story of the Federal Demonstration Partnership (FDP) Tokyo, Japan February 22, 2008 Joanna Rom Executive
Cooperative Framework of Institutions and Funding Agencies to Improve Administrative Burden: The story of the Federal Demonstration Partnership (FDP) Tokyo, Japan February 22, 2008 Joanna Rom Executive
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE
 THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE Alexandra A. McKeown, Associate Dean Johns Hopkins University May 2, 2011 1 What is the FDP? The FDP is a cooperative effort among federal research
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE Alexandra A. McKeown, Associate Dean Johns Hopkins University May 2, 2011 1 What is the FDP? The FDP is a cooperative effort among federal research
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE
 THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research organizations aimed at streamlining
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research organizations aimed at streamlining
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE
 THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE NECA Conference, August 21, 2012 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE NECA Conference, August 21, 2012 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research
Jean Feldman. Policy Office. Head, Policy Office, Policy Office, Division of Institution & Award Support
 NATIONAL SCIENCE FOUNDATION UPDATE Federal Demonstration Partnership Meeting January 26, 2011 Washington, DC Ask Early, Ask Often Jean Feldman Head, Policy Office, Policy Office, Division of Institution
NATIONAL SCIENCE FOUNDATION UPDATE Federal Demonstration Partnership Meeting January 26, 2011 Washington, DC Ask Early, Ask Often Jean Feldman Head, Policy Office, Policy Office, Division of Institution
2017 SRA International Annual Meeting October 14-18, Federal Demonstration Partnership (FDP)/Council on Governmental Relations (COGR) Update
 2017 SRA International Annual Meeting October 14-18, 2017 Federal Demonstration Partnership (FDP)/Council on Governmental Relations (COGR) Update Who are we? Anthony DeCrappeo, President, Council on Governmental
2017 SRA International Annual Meeting October 14-18, 2017 Federal Demonstration Partnership (FDP)/Council on Governmental Relations (COGR) Update Who are we? Anthony DeCrappeo, President, Council on Governmental
Reducing Investigators Administrative Workload for Federally-Funded Research
 Reducing Investigators Administrative Workload for Federally-Funded Research National Science Board National Research Council Committee on Federal Research Regulations and Reporting Requirements February
Reducing Investigators Administrative Workload for Federally-Funded Research National Science Board National Research Council Committee on Federal Research Regulations and Reporting Requirements February
2012 Faculty Workload Survey
 FEDERAL DEMONSTRATION PARTNERSHIP (FDP) 2012 Faculty Workload Survey EXECUTIVE SUMMARY Sandra L. Schneider, Principal Investigator. University of South Florida Kirsten K. Ness, St. Jude Children s Research
FEDERAL DEMONSTRATION PARTNERSHIP (FDP) 2012 Faculty Workload Survey EXECUTIVE SUMMARY Sandra L. Schneider, Principal Investigator. University of South Florida Kirsten K. Ness, St. Jude Children s Research
OSR Monthly Meeting. August 16, 2018 (Chicago) August 21, 2018 (Evanston)
 OSR Monthly Meeting August 16, 2018 (Chicago) August 21, 2018 (Evanston) Agenda OSR Announcements/Staffing Updates ESPR Replacement Project Update Year-end Procedures NSF Updates NIH Biosketch Reminder
OSR Monthly Meeting August 16, 2018 (Chicago) August 21, 2018 (Evanston) Agenda OSR Announcements/Staffing Updates ESPR Replacement Project Update Year-end Procedures NSF Updates NIH Biosketch Reminder
Recent Legislative Actions Taken to Reduce Research Regulatory Burden. 21st Century Cures (Passed House and Senate. Signed into law Dec.
 . Signed into law Dec. 13) Link to PDF Link to PDF Link to PDF Research Policy Board - A public-private entity recommended by the National Academies "to foster more effective conception, development and
. Signed into law Dec. 13) Link to PDF Link to PDF Link to PDF Research Policy Board - A public-private entity recommended by the National Academies "to foster more effective conception, development and
Sponsored Program Administration Meeting. September 2016
 Sponsored Program Administration Meeting September 2016 Agenda NCURA Internal Controls Mike Unplugged: Training/Education Specialist position Budgeting for Post doc increases FDP Update ETS Update Effort
Sponsored Program Administration Meeting September 2016 Agenda NCURA Internal Controls Mike Unplugged: Training/Education Specialist position Budgeting for Post doc increases FDP Update ETS Update Effort
NIH Update. Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH. FDP January 11, 2016
 NIH Update Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH FDP January 11, 2016 1 2 NIH FY 2016 Budget News NIH is currently funded under the Consolidated
NIH Update Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH FDP January 11, 2016 1 2 NIH FY 2016 Budget News NIH is currently funded under the Consolidated
National Science Foundation Update. SRA Annual Meeting October 20, 2015
 National Science Foundation Update SRA Annual Meeting October 20, 2015 Speakers Jean Feldman Head, Policy Office Division of Institution & Award Support Office of Budget, Finance & Award Management 703.292.8243;
National Science Foundation Update SRA Annual Meeting October 20, 2015 Speakers Jean Feldman Head, Policy Office Division of Institution & Award Support Office of Budget, Finance & Award Management 703.292.8243;
Help Is on the Way: The Federal Demonstration Partnership and Emerging Research Institutions
 Help Is on the Way: The Federal Demonstration Partnership and Emerging Research Institutions David Wright Executive Director Federal Demonstration Partnership (FDP) Federal Demonstration Partnership (FDP)
Help Is on the Way: The Federal Demonstration Partnership and Emerging Research Institutions David Wright Executive Director Federal Demonstration Partnership (FDP) Federal Demonstration Partnership (FDP)
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM. December 2017
 Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Rebecca Trahan. Office of Sponsored Programs December 9, ORED Limited Submission Update
 Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
Document Downloaded: Tuesday November 10, EPA Interim Conflict of Interest Policy. Author: Research Compliance and Administration Committee
 Document Downloaded: Tuesday November 10, 2015 EPA Interim Conflict of Interest Policy Author: Research Compliance and Administration Committee Published Date: 02/20/2015 COGR COUNCIL ON GOVERNMENTAL RELATIONS
Document Downloaded: Tuesday November 10, 2015 EPA Interim Conflict of Interest Policy Author: Research Compliance and Administration Committee Published Date: 02/20/2015 COGR COUNCIL ON GOVERNMENTAL RELATIONS
COGR COUNCIL ON GOVERNMENTAL RELATIONS. July 8, Mr. Gilbert Tran Ms. Rhea Hubbard Ms. Bridget Miller
 COGR COUNCIL ON GOVERNMENTAL RELATIONS an organization of research universities 1200 New York Avenue, N.W., Suite 460, Washington, D.C. 20005 (202) 289-6655/(202) 289-6698 (FAX) BOARD OF DIRECTORS JAMES
COGR COUNCIL ON GOVERNMENTAL RELATIONS an organization of research universities 1200 New York Avenue, N.W., Suite 460, Washington, D.C. 20005 (202) 289-6655/(202) 289-6698 (FAX) BOARD OF DIRECTORS JAMES
OMB Uniform Guidance ( UG ) Briefing. ASRSP & OSR Brown Bag Tuesday, January 27 th
 OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
Request for Information (RFI): Reducing Investigator s Administrative Workload for Federally Funded Research
 Request for Information (RFI): Reducing Investigator s Administrative Workload for Federally Funded Research Key Dates Release Date: March 25, 2013 Response Date: May 24, 2013 Issued by National Science
Request for Information (RFI): Reducing Investigator s Administrative Workload for Federally Funded Research Key Dates Release Date: March 25, 2013 Response Date: May 24, 2013 Issued by National Science
Outgoing Subagreements: Subawards and Subcontracts
 Effective Date: March 1, 2014 Version: 1 Page: 1 of 6 Overview Sponsored programs may include partnerships with other organizations or institutions whereby a portion of the project is conducted by investigators
Effective Date: March 1, 2014 Version: 1 Page: 1 of 6 Overview Sponsored programs may include partnerships with other organizations or institutions whereby a portion of the project is conducted by investigators
UNIFORM GUIDANCE IMPLEMENTATION
 UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
Implementation of the NSTC Presidential Review Directive-4: Renewing the Federal Government-University Research Partnership for the 21 st Century
 Implementation of the NSTC Presidential Review Directive-4: Renewing the Federal Government-University Research Partnership for the 21 st Century Committee on Science National Science and Technology Council
Implementation of the NSTC Presidential Review Directive-4: Renewing the Federal Government-University Research Partnership for the 21 st Century Committee on Science National Science and Technology Council
Reducing Regulatory and Institutional Burden Associated with Animal Research. June 8, 2017
 Reducing Regulatory and Institutional Burden Associated with Animal Research June 8, 2017 Matt Bailey, President, National Association for Biomedical Research Molly Greene, IACUC Advisor, Michigan State
Reducing Regulatory and Institutional Burden Associated with Animal Research June 8, 2017 Matt Bailey, President, National Association for Biomedical Research Molly Greene, IACUC Advisor, Michigan State
Document Downloaded: Tuesday November 10, June 30, Revisions to Procurement Standards and Grace Period Extension.
 Document Downloaded: Tuesday November 10, 2015 June 30, 2015 - Revisions to Procurement Standards and Grace Period Extension Author: COGR Published Date: 06/30/2015 COGR COUNCIL ON GOVERNMENTAL RELATIONS
Document Downloaded: Tuesday November 10, 2015 June 30, 2015 - Revisions to Procurement Standards and Grace Period Extension Author: COGR Published Date: 06/30/2015 COGR COUNCIL ON GOVERNMENTAL RELATIONS
University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures
 Summary 1. Subaward Definitions A. Subaward B. Subrecipient University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures C. Office of Contracts and Grants (OCG) 2. Distinguishing
Summary 1. Subaward Definitions A. Subaward B. Subrecipient University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures C. Office of Contracts and Grants (OCG) 2. Distinguishing
The Lifecycle of a Sponsored Project
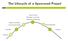 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
Policy and Compliance: Working Together Like Hand in Glove
 Policy and Compliance: Working Together Like Hand in Glove Samuel Ashe, Director, Division of Grants Policy, OPERA, OER Diane W. Dean, Director, Division of Grants Compliance and Oversight, OPERA, OER
Policy and Compliance: Working Together Like Hand in Glove Samuel Ashe, Director, Division of Grants Policy, OPERA, OER Diane W. Dean, Director, Division of Grants Compliance and Oversight, OPERA, OER
FDP Meeting Summary. Controlled Unclassified Information (CUI) and FISM 5/10/2017-5/12/2017
 Controlled Unclassified Information (CUI) and FISM Makr Sweet This session was an update of some of the current issues surrounding Controlled Unclassified Information (CUI) and FISMA. The presenters provided
Controlled Unclassified Information (CUI) and FISM Makr Sweet This session was an update of some of the current issues surrounding Controlled Unclassified Information (CUI) and FISMA. The presenters provided
UPDATES. Meet the Proposal Deadline. NIH: Public Access to Research Results OFFICE OF SPONSORED INSIDE THIS ISSUE:
 UPDATES INSIDE THIS ISSUE: Meet the Proposal Deadline Responsible Conduct of Research New Federal Regulations on Conflict of Interest NSF s New Proposal Guidelines and Project Reporting Changes Rutgers
UPDATES INSIDE THIS ISSUE: Meet the Proposal Deadline Responsible Conduct of Research New Federal Regulations on Conflict of Interest NSF s New Proposal Guidelines and Project Reporting Changes Rutgers
Research Administrators Forum August 14th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum August 14th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Associate Vice Chancellor, Office of Research Administration
Research Administrators Forum August 14th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Associate Vice Chancellor, Office of Research Administration
UNIFORM ADMINISTRATIVE REQUIREMENTS, COST PRINCIPLES, AND AUDIT REQUIREMENTS FOR FEDERAL AWARDS - UPDATE FEBRUARY 2015
 UNIFORM ADMINISTRATIVE REQUIREMENTS, COST PRINCIPLES, AND AUDIT REQUIREMENTS FOR FEDERAL AWARDS - UPDATE FEBRUARY 2015 AOA Conference Pasadena, CA February 9, 2015 Agenda 1. Introduction / Disclaimer 2.
UNIFORM ADMINISTRATIVE REQUIREMENTS, COST PRINCIPLES, AND AUDIT REQUIREMENTS FOR FEDERAL AWARDS - UPDATE FEBRUARY 2015 AOA Conference Pasadena, CA February 9, 2015 Agenda 1. Introduction / Disclaimer 2.
Performance audit report. Department of Internal Affairs: Administration of two grant schemes
 Performance audit report Department of Internal Affairs: Administration of two grant schemes Office of of the the Auditor-General PO PO Box Box 3928, Wellington 6140 Telephone: (04) (04) 917 9171500 Facsimile:
Performance audit report Department of Internal Affairs: Administration of two grant schemes Office of of the the Auditor-General PO PO Box Box 3928, Wellington 6140 Telephone: (04) (04) 917 9171500 Facsimile:
Administrative Burden of Research Compliance
 Administrative Burden of Research Compliance Measuring and Minimizing David L. Wynes, Ph.D. Vice President for Research Administration Emory University 1 FDP Faculty Burden Survey (X2) PIs estimated that
Administrative Burden of Research Compliance Measuring and Minimizing David L. Wynes, Ph.D. Vice President for Research Administration Emory University 1 FDP Faculty Burden Survey (X2) PIs estimated that
RSP 101: Introduction to Research and Sponsored Programs Policies and Procedures. Marcus W. Shute, P.E., Ph.D.
 RSP 101: Introduction to Research and Sponsored Programs Policies and Procedures Marcus W. Shute, P.E., Ph.D. mwshute@cau.edu, x6990 2008-2009 Shute & Clark Atlanta University. All Rights Reserved Introduction
RSP 101: Introduction to Research and Sponsored Programs Policies and Procedures Marcus W. Shute, P.E., Ph.D. mwshute@cau.edu, x6990 2008-2009 Shute & Clark Atlanta University. All Rights Reserved Introduction
CURRENT COGR PRIORITIES - BY COMMITTEE (7/10/17)
 CURRENT COGR PRIORITIES - BY COMMITTEE (7/10/17) No. 1 Student Financial Aid and "Securing Student Information" and Compliance Supplement New single audit requirement. Delayed until 2018. MEDIUM Pending
CURRENT COGR PRIORITIES - BY COMMITTEE (7/10/17) No. 1 Student Financial Aid and "Securing Student Information" and Compliance Supplement New single audit requirement. Delayed until 2018. MEDIUM Pending
Basic Grants Overview WELCOME
 Basic Grants Overview WELCOME Introduction This grants Webinar session is an overview that will focus on the Federal Grant Process. A lot of the information presented during this session can be used when
Basic Grants Overview WELCOME Introduction This grants Webinar session is an overview that will focus on the Federal Grant Process. A lot of the information presented during this session can be used when
East Carolina University Sponsored Projects Administration Roles and Responsibilities Matrix By Responsibility
 (See legend at end of matrix.) East Carolina University Sponsored Projects Administration Roles and Responsibilities Matrix By Responsibility This matrix is designed to provide general guidance on administrative
(See legend at end of matrix.) East Carolina University Sponsored Projects Administration Roles and Responsibilities Matrix By Responsibility This matrix is designed to provide general guidance on administrative
Subrecipient Risk Assessment and Monitoring of Northeastern University Issued Subawards
 Subrecipient Risk Assessment and Monitoring of Northeastern University Issued Subawards What is a Subaward? A Subaward is a contractual agreement between Northeastern University and a third party organization
Subrecipient Risk Assessment and Monitoring of Northeastern University Issued Subawards What is a Subaward? A Subaward is a contractual agreement between Northeastern University and a third party organization
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards. AU SPAN Martha Taylor Larry Hankins
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards AU SPAN 6 23 2014 Martha Taylor Larry Hankins Grants Reform February 2011 President directed OMB to reduce
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards AU SPAN 6 23 2014 Martha Taylor Larry Hankins Grants Reform February 2011 President directed OMB to reduce
DFA Conversations Research Administration Forum
 DFA Conversations Research Administration Forum Fall 2017 10/23/2017 Agenda What is Financial Research Administration? Our Issues Recent Audits Your Issues Traveling for Cornell on Sponsored Awards Recent
DFA Conversations Research Administration Forum Fall 2017 10/23/2017 Agenda What is Financial Research Administration? Our Issues Recent Audits Your Issues Traveling for Cornell on Sponsored Awards Recent
Research Administrators Forum October 10th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum October 10th, 2013 Marcia Smith Associate Vice Chancellor for Research Agenda Meeting Agenda Welcome and Announcements Marcia Smith, Office of Research Administration Contract
Research Administrators Forum October 10th, 2013 Marcia Smith Associate Vice Chancellor for Research Agenda Meeting Agenda Welcome and Announcements Marcia Smith, Office of Research Administration Contract
Grant/Sponsor Related Systems. Department and OSP Perspectives on ERA
 Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
Subrecipient Monitoring Procedures
 Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
Uniform Guidance. Overview and Implementation Plan. November 21, 2014
 Uniform Guidance Overview and Implementation Plan November 21, 2014 Uniform Guidance. Change is coming! College or Department name here 1 Overarching goal of the reform is to: streamline the rules and
Uniform Guidance Overview and Implementation Plan November 21, 2014 Uniform Guidance. Change is coming! College or Department name here 1 Overarching goal of the reform is to: streamline the rules and
Office of Sponsored Projects 5/31/ Volume 2, Issue 5
 The OSP News & Updates, published by the Office of Sponsored Projects, is a bi-weekly subscription-based newsletter that provides OSP and sponsor updates and reminders, quick facts, guidance and training
The OSP News & Updates, published by the Office of Sponsored Projects, is a bi-weekly subscription-based newsletter that provides OSP and sponsor updates and reminders, quick facts, guidance and training
The Uniform Guidance (2 CFR, Part 200)
 WCMC Implementation of The Uniform Guidance (2 CFR, Part 200) Tuesday, September 22, 2015 & Wednesday, September 23, 2015 UG Workshop Michelle A. Lewis, M.S. Director of Research Administration Interim
WCMC Implementation of The Uniform Guidance (2 CFR, Part 200) Tuesday, September 22, 2015 & Wednesday, September 23, 2015 UG Workshop Michelle A. Lewis, M.S. Director of Research Administration Interim
FDP Expanded Clearinghouse
 FDP Expanded Clearinghouse Lynette Arias, University of Washington Pamela Webb, University of Minnesota Jennifer Barron, Johns Hopkins University Chris Renner, Vanderbilt University Medical Center Julie
FDP Expanded Clearinghouse Lynette Arias, University of Washington Pamela Webb, University of Minnesota Jennifer Barron, Johns Hopkins University Chris Renner, Vanderbilt University Medical Center Julie
Effort Reporting Again
 Effort Reporting Again KIM MORELAND UNIVERSITY OF WISCONSIN MADISON JULIE JARVIS UNIVERSITY OF ILLINOIS, URBANA-CHAMPAIGN NCURA REGION IV 2010 Agenda 2 Audit Findings Common Policy Issues FDP Project Certification
Effort Reporting Again KIM MORELAND UNIVERSITY OF WISCONSIN MADISON JULIE JARVIS UNIVERSITY OF ILLINOIS, URBANA-CHAMPAIGN NCURA REGION IV 2010 Agenda 2 Audit Findings Common Policy Issues FDP Project Certification
Subaward Policies and Procedures Manual
 JOHNS HOPKINS UNIVERISTY Krieger School of Arts and Sciences Business and Research Administration Subaward Policies and Procedures Manual 2016 Contents Introduction...3 Differentiating types of relationships...3
JOHNS HOPKINS UNIVERISTY Krieger School of Arts and Sciences Business and Research Administration Subaward Policies and Procedures Manual 2016 Contents Introduction...3 Differentiating types of relationships...3
Minimizing Your Institution's Administrative Burdens Imprint. Susan Wyatt Sedwick Courtney Frazier Swaney Christopher Jenkins
 Minimizing Your Institution's Administrative Burdens Imprint Susan Wyatt Sedwick Courtney Frazier Swaney Christopher Jenkins Panelists SUSAN WYATT SEDWICK, PhD, CRA Consulting Associate COURTNEY FRAZIER
Minimizing Your Institution's Administrative Burdens Imprint Susan Wyatt Sedwick Courtney Frazier Swaney Christopher Jenkins Panelists SUSAN WYATT SEDWICK, PhD, CRA Consulting Associate COURTNEY FRAZIER
Cindy Gilbert Assistant Director January 14, 2016
 Cindy Gilbert Assistant Director cgilbert@research.ucla.edu January 14, 2016 OCGA era Help Email S2S Grants Help (s2sgrants@research.ucla.edu) retired Replaced by OCGA era Help (erahelp@research.ucla.edu)
Cindy Gilbert Assistant Director cgilbert@research.ucla.edu January 14, 2016 OCGA era Help Email S2S Grants Help (s2sgrants@research.ucla.edu) retired Replaced by OCGA era Help (erahelp@research.ucla.edu)
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates!
 FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
THE NATIONAL ACADEMIES FEDERAL DEMONSTRATION PARTNERSHIP. ELECTRONIC GRANTS BUSINESS FORUM Friday, July 29, 2005
 THE NATIONAL ACADEMIES FEDERAL DEMONSTRATION PARTNERSHIP Please see following documents appended: 1.) DUNS&CCR 2.) NGP White Paper ELECTRONIC GRANTS BUSINESS FORUM Friday, July 29, 2005 The National Academies
THE NATIONAL ACADEMIES FEDERAL DEMONSTRATION PARTNERSHIP Please see following documents appended: 1.) DUNS&CCR 2.) NGP White Paper ELECTRONIC GRANTS BUSINESS FORUM Friday, July 29, 2005 The National Academies
Special Cases in Proposal Development: Large-Scale, Multidisciplinary and/or Multi-Organizational Proposals
 WEBINAR BRIEFING Special Cases in Proposal Development: Large-Scale, Multidisciplinary and/or Featuring Hanover Research Grants Consultant Bryan DeBusk, PhD, GPC And Hanover Research Grants Consultant
WEBINAR BRIEFING Special Cases in Proposal Development: Large-Scale, Multidisciplinary and/or Featuring Hanover Research Grants Consultant Bryan DeBusk, PhD, GPC And Hanover Research Grants Consultant
Sonia Esperança Program Director; Directorate for Geosciences; Division of Earth Sciences
 Proposal Preparation Panelists David Campbell Acting Division Director and Program Director; Directorate for Education & Human Resources; Division of Research on Learning in Formal & Informal Settings
Proposal Preparation Panelists David Campbell Acting Division Director and Program Director; Directorate for Education & Human Resources; Division of Research on Learning in Formal & Informal Settings
Expanded Clearinghouse Pilot Phase II. Will be part of 80 institutions nationwide to participate in the Clearinghouse.
 Expanded Clearinghouse Pilot Phase II Will be part of 80 institutions nationwide to participate in the Clearinghouse. 1 Goal of Pilot Reduce administrative burden associated with verifying standard information
Expanded Clearinghouse Pilot Phase II Will be part of 80 institutions nationwide to participate in the Clearinghouse. 1 Goal of Pilot Reduce administrative burden associated with verifying standard information
COST SHARING POLICY COST SHARING POLICY PAGE 1 OF 8
 COST SHARING POLICY Effective Date: December 4, 2013 Replaces: Cost Sharing dated January 1, 2010 Approved by: Steve McNally, Senior Associate Vice Chancellor for Budget, Finance and Enrollment Services
COST SHARING POLICY Effective Date: December 4, 2013 Replaces: Cost Sharing dated January 1, 2010 Approved by: Steve McNally, Senior Associate Vice Chancellor for Budget, Finance and Enrollment Services
Kathy Hancock, Assistant Grants Compliance Officer, DGCO, OPERA, Office of Extramural Research, NIH, HHS
 Division of Grants Compliance and Oversight (DGCO) Office of Policy for Extramural Research Administration (OPERA), OER National Institutes of Health (NIH), DHHS 19 th Annual SBIR/STTR Conference Milwaukee,
Division of Grants Compliance and Oversight (DGCO) Office of Policy for Extramural Research Administration (OPERA), OER National Institutes of Health (NIH), DHHS 19 th Annual SBIR/STTR Conference Milwaukee,
RAIS AND REPORTING USER GROUP RESEARCH ADMINISTRATION INFORMATION SYSTEMS (RAIS)
 RAIS AND REPORTING USER GROUP RESEARCH ADMINISTRATION INFORMATION SYSTEMS (RAIS) Agenda Coeus Coeus Access Form Update Coeus User Access Roles New Grants.gov Forms NSF Submissions Proposal Person Addresses
RAIS AND REPORTING USER GROUP RESEARCH ADMINISTRATION INFORMATION SYSTEMS (RAIS) Agenda Coeus Coeus Access Form Update Coeus User Access Roles New Grants.gov Forms NSF Submissions Proposal Person Addresses
ARRA Stimulus Weekly Update, May 7, 2009
 ARRA Stimulus Weekly Update, May 7, 2009 I. Reporting Weekly Submissions to UNC-GA. Campuses have been submitting information on ARRArelated proposals, preliminary notices of award (i.e. JIT submissions)
ARRA Stimulus Weekly Update, May 7, 2009 I. Reporting Weekly Submissions to UNC-GA. Campuses have been submitting information on ARRArelated proposals, preliminary notices of award (i.e. JIT submissions)
NSF Grants Conference NSF Policies and Procedures Update
 Slide 1 NSF Grants Conference NSF Policies and Procedures Update March 11-12, 2013 Hosted by Howard University, Arlington, Virginia Slide 2 Panelist Jean Feldman Head, Policy Office, Division of Institution
Slide 1 NSF Grants Conference NSF Policies and Procedures Update March 11-12, 2013 Hosted by Howard University, Arlington, Virginia Slide 2 Panelist Jean Feldman Head, Policy Office, Division of Institution
Higher Education Coordinating Committee September 11, 2015 Conference Call 10:00 a.m. 12:00 p.m.
 Higher Education Coordinating Committee September 11, 2015 Conference Call 10:00 a.m. 12:00 p.m. Members present Tom Kuntz Marshall Criser Curtis Austin Ken Burke Ed Moore Susan Pareigis Madeline Pumariega
Higher Education Coordinating Committee September 11, 2015 Conference Call 10:00 a.m. 12:00 p.m. Members present Tom Kuntz Marshall Criser Curtis Austin Ken Burke Ed Moore Susan Pareigis Madeline Pumariega
Any observations not included in this report were discussed with your staff at the informal exit conference and may be subject to follow-up.
 Larry Mandel Vice Chancellor and Chief Audit Officer Office of Audit and Advisory Services 401 Golden Shore, 4th Floor Long Beach, CA 90802-4210 562-951-4430 562-951-4955 (Fax) lmandel@calstate.edu September
Larry Mandel Vice Chancellor and Chief Audit Officer Office of Audit and Advisory Services 401 Golden Shore, 4th Floor Long Beach, CA 90802-4210 562-951-4430 562-951-4955 (Fax) lmandel@calstate.edu September
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards. AUSPAN Martha Taylor
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards AUSPAN 2 16 2015 Martha Taylor Grants Reform February 2011 President directed OMB to reduce unnecessary regulatory
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards AUSPAN 2 16 2015 Martha Taylor Grants Reform February 2011 President directed OMB to reduce unnecessary regulatory
CLASP TOPICS OF INTEREST: Q&A DOCUMENT March 2015
 1 CLASP TOPICS OF INTEREST: Q&A DOCUMENT March 2015 SUBJECT: NATIONAL SCIENCE FOUNDATION POLICY & GUIDELINES NSF Cost-Sharing Policy Compensation vs. Time Commitment 1. PIs may be required to reduce or
1 CLASP TOPICS OF INTEREST: Q&A DOCUMENT March 2015 SUBJECT: NATIONAL SCIENCE FOUNDATION POLICY & GUIDELINES NSF Cost-Sharing Policy Compensation vs. Time Commitment 1. PIs may be required to reduce or
Sponsored Program Administration. October 10, 2017
 Sponsored Program Administration October 10, 2017 Agenda Procurement Services FDP Updates Jaggaer Pre-Award Federal Agency Updates Grants.gov Transition to Workspace NIH FORMS-E Post-Award GONE Act Certificate
Sponsored Program Administration October 10, 2017 Agenda Procurement Services FDP Updates Jaggaer Pre-Award Federal Agency Updates Grants.gov Transition to Workspace NIH FORMS-E Post-Award GONE Act Certificate
FAQ S FOR UNIFORM GUIDANCE
 FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
December 2015 Research Administration Working Group WELCOME OFFICE OF THE VICE PRESIDENT FOR RESEARCH
 December 2015 Research Administration Working Group WELCOME Open Mike Fringe Rates FY17 proposed Budget v. Charging Distribution of Indirects PI award letter out & accounts funded Departments- this month
December 2015 Research Administration Working Group WELCOME Open Mike Fringe Rates FY17 proposed Budget v. Charging Distribution of Indirects PI award letter out & accounts funded Departments- this month
Federal Demonstration Partnership (FDP) DATA Act Section 5 Grants Pilot Update and CDER Library Test Model Brief. May 5, 2016
 Federal Demonstration Partnership (FDP) DATA Act Section 5 Grants Pilot Update and CDER Library Test Model Brief May 5, 2016 Presenter Christopher Zeleznik Department of Health and Human Services DATA
Federal Demonstration Partnership (FDP) DATA Act Section 5 Grants Pilot Update and CDER Library Test Model Brief May 5, 2016 Presenter Christopher Zeleznik Department of Health and Human Services DATA
Finding Funding, Budget Preparation, and Proposal Submission for Sponsored Research
 Finding Funding, Budget Preparation, and Proposal Submission for Sponsored Research Laura Johnston, Office of Sponsored Programs, Asst. Director February 24, 2017 1 Agenda A Walkthrough of MSU s Proposal
Finding Funding, Budget Preparation, and Proposal Submission for Sponsored Research Laura Johnston, Office of Sponsored Programs, Asst. Director February 24, 2017 1 Agenda A Walkthrough of MSU s Proposal
MUSC Center for Global Health Request for Applications (RFA) for Faculty Pilot Project Grants
 MUSC Center for Global Health Request for Applications (RFA) for Faculty Pilot Project Grants RFA Release Date: Monday, April 10, 2017 Full Proposal (6 page limit) Deadline: May 16, 2017 12 p.m. (noon)
MUSC Center for Global Health Request for Applications (RFA) for Faculty Pilot Project Grants RFA Release Date: Monday, April 10, 2017 Full Proposal (6 page limit) Deadline: May 16, 2017 12 p.m. (noon)
Slide 1. NSF Grants Conference. Proposal Preparation. March 11-12, 2013 Hosted by Howard University, Arlington, Virginia
 Slide 1 NSF Grants Conference Proposal Preparation March 11-12, 2013 Hosted by Howard University, Arlington, Virginia Slide 2 Panelists Sonia Esperança Program Director, Directorate for Geosciences, Division
Slide 1 NSF Grants Conference Proposal Preparation March 11-12, 2013 Hosted by Howard University, Arlington, Virginia Slide 2 Panelists Sonia Esperança Program Director, Directorate for Geosciences, Division
Developing Proposal Budgets
 Grant Writing & Developing Proposal Budgets Sandra H. Harpole Associate VP for Research Sandy Williamson ORED Fiscal Officer January 8, 2008 Idea First or Opportunity First? Idea Have an idea for a project
Grant Writing & Developing Proposal Budgets Sandra H. Harpole Associate VP for Research Sandy Williamson ORED Fiscal Officer January 8, 2008 Idea First or Opportunity First? Idea Have an idea for a project
Effort Certification Training
 Effort Certification Training Please Sign In. We will be emailing the slides after today s meeting. http://finance.uark.edu/pdfs/efforttraining.pdf What is effort? The total work done to achieve a particular
Effort Certification Training Please Sign In. We will be emailing the slides after today s meeting. http://finance.uark.edu/pdfs/efforttraining.pdf What is effort? The total work done to achieve a particular
REQUEST FOR PROPOSALS JAMES H. ZUMBERGE FACULTY RESEARCH & INNOVATION FUND ZUMBERGE INDIVIDUAL RESEARCH AWARD
 REQUEST FOR PROPOSALS JAMES H. ZUMBERGE FACULTY RESEARCH & INNOVATION FUND ZUMBERGE INDIVIDUAL RESEARCH AWARD APPLICATION DEADLINE: 5 pm, Monday, January 8, 2018 PURPOSE The primary purpose of the Zumberge
REQUEST FOR PROPOSALS JAMES H. ZUMBERGE FACULTY RESEARCH & INNOVATION FUND ZUMBERGE INDIVIDUAL RESEARCH AWARD APPLICATION DEADLINE: 5 pm, Monday, January 8, 2018 PURPOSE The primary purpose of the Zumberge
Subrecipient Profile Questionnaire
 Subrecipient Profile Questionnaire How to use: The questionnaire is used to help determine a subrecipient organization s financial and management strength, which helps assess risk and dictates the monitoring
Subrecipient Profile Questionnaire How to use: The questionnaire is used to help determine a subrecipient organization s financial and management strength, which helps assess risk and dictates the monitoring
Proposal Submission, Review and Acceptance
 Proposal Submission, Review and Acceptance October 2015 Peggy Bowe OGRD is now OROS Office of Research Operations and Support Recording date of this workshop is October 23, 2015 Some of the rules and procedures
Proposal Submission, Review and Acceptance October 2015 Peggy Bowe OGRD is now OROS Office of Research Operations and Support Recording date of this workshop is October 23, 2015 Some of the rules and procedures
Jean Feldman Head, Policy Office, Office of Budget, Finance & Award Management; Division of Institution & Award Support
 NSF Grants Conference Proposal Preparation October 6-7, 2014 Hosted by The George Washington University, Arlington, Virginia Panelists Jean Feldman Head, Policy Office, Office of Budget, Finance & Award
NSF Grants Conference Proposal Preparation October 6-7, 2014 Hosted by The George Washington University, Arlington, Virginia Panelists Jean Feldman Head, Policy Office, Office of Budget, Finance & Award
Navigating the New Uniform Grant Guidance. Jack Reagan, Audit Partner Grant Thornton LLP. Grant Thornton. All rights reserved.
 Navigating the New Uniform Grant Guidance Jack Reagan, Audit Partner Grant Thornton LLP Objectives What s New with OMB: Uniform Administrative Requirements, Cost Principles, and Audit requirements for
Navigating the New Uniform Grant Guidance Jack Reagan, Audit Partner Grant Thornton LLP Objectives What s New with OMB: Uniform Administrative Requirements, Cost Principles, and Audit requirements for
AFOSR GRANTS OVERVIEW AFOSR. Air Force Office of Scientific Research. RINA MARTINEZ and MATT SLOWIK Air Force Office of Scientific Research
 AFOSR GRANTS RINA MARTINEZ and MATT SLOWIK Air Force Office of Scientific Research Distribution A. Approved for Public Release OVERVIEW AFOSR Process Types of Awards Award Terms & Conditions Pre-award
AFOSR GRANTS RINA MARTINEZ and MATT SLOWIK Air Force Office of Scientific Research Distribution A. Approved for Public Release OVERVIEW AFOSR Process Types of Awards Award Terms & Conditions Pre-award
3 rd Annual Symposium for Research Administrators
 3 rd Annual Symposium for Research Administrators Cost Transfer Policy Martha Martin, CRA, Grants Administration, School of Information and Library Science Sarah Van Heusen, MHA, Director of Research Administration,
3 rd Annual Symposium for Research Administrators Cost Transfer Policy Martha Martin, CRA, Grants Administration, School of Information and Library Science Sarah Van Heusen, MHA, Director of Research Administration,
2017 UC Multicampus Research Funding Opportunities
 2017 UC Multicampus Research Funding Opportunities The UC Multicampus Research Programs and Initiatives The President s Research Catalyst Award REQUEST FOR PROPOSALS v. March 9, 2016 (draft; proposals
2017 UC Multicampus Research Funding Opportunities The UC Multicampus Research Programs and Initiatives The President s Research Catalyst Award REQUEST FOR PROPOSALS v. March 9, 2016 (draft; proposals
International Regulatory Harmonization Amid Globalization of Biomedical Research & Medical Product Development: An Institute of Medicine Workshop
 Board on Health Sciences Policy Forum on Drug Discovery, Development, and Translation International Regulatory Harmonization Amid Globalization of Biomedical Research & Medical Product Development: An
Board on Health Sciences Policy Forum on Drug Discovery, Development, and Translation International Regulatory Harmonization Amid Globalization of Biomedical Research & Medical Product Development: An
University of Pittsburgh SPONSORED PROJECT FINANCIAL GUIDELINE Subject: SUBRECIPIENT MONITORING
 I. Scope Subrecipient monitoring is the process by which the University selects, monitors, controls, and accounts for University subcontractors or subrecipients utilized on sponsored projects in accordance
I. Scope Subrecipient monitoring is the process by which the University selects, monitors, controls, and accounts for University subcontractors or subrecipients utilized on sponsored projects in accordance
Grants and Agreements: Choosing the Right Tool to Ensure Partnership Success A Partnership Network Peer Learning Session
 Tanner Ridge, Columbia Gorge, OR Grants and Agreements: Choosing the Right Tool to Ensure Partnership Success A Partnership Network Peer Learning Session Alison Léiman, WO G&A Policy Thursday, April 13,
Tanner Ridge, Columbia Gorge, OR Grants and Agreements: Choosing the Right Tool to Ensure Partnership Success A Partnership Network Peer Learning Session Alison Léiman, WO G&A Policy Thursday, April 13,
Division of Research
 Division of Research Quarterly Research Administration Meeting October 30, 2012 Page 1 1 Division of Research Quarterly Research Administration Meeting AGENDA Welcome and Introductions Pre Award Update
Division of Research Quarterly Research Administration Meeting October 30, 2012 Page 1 1 Division of Research Quarterly Research Administration Meeting AGENDA Welcome and Introductions Pre Award Update
University of Colorado Denver
 University of Colorado Denver Campus Guidelines Title:, 4-13 Source: Prepared by: Approved by: Office of Grants and Contracts Director, Office of Grants and Contracts Vice Chancellor for Research Effective
University of Colorado Denver Campus Guidelines Title:, 4-13 Source: Prepared by: Approved by: Office of Grants and Contracts Director, Office of Grants and Contracts Vice Chancellor for Research Effective
Electronic Research Administration (era) March 8, 2018
 Electronic Research Administration (era) March 8, 2018 Introductions We are. Terry Duperron, era Analyst Marlene Mooshian, Proposal Analyst Kassie Obelleiro, Training Officer Jinger Snyder, Award Analyst
Electronic Research Administration (era) March 8, 2018 Introductions We are. Terry Duperron, era Analyst Marlene Mooshian, Proposal Analyst Kassie Obelleiro, Training Officer Jinger Snyder, Award Analyst
Research.gov. A Partnership to Provide Transparency and Meet the Needs of the Research Community. May 2010
 Research.gov A Partnership to Provide Transparency and Meet the Needs of the Research Community May 2010 Research.gov Supports the needs of institutions and researchers Modernization of FastLane providing
Research.gov A Partnership to Provide Transparency and Meet the Needs of the Research Community May 2010 Research.gov Supports the needs of institutions and researchers Modernization of FastLane providing
MONDAY, JULY 11, 2016
 AGENDA A Workshop on the Institute of Medicine * Report, Strategies to Improve Cardiac Arrest Survival: A Time to Act July 11-12, 2016 National Academies of Sciences Building 2101 Constitution Ave., NW,
AGENDA A Workshop on the Institute of Medicine * Report, Strategies to Improve Cardiac Arrest Survival: A Time to Act July 11-12, 2016 National Academies of Sciences Building 2101 Constitution Ave., NW,
BC Capacity Initiative
 BC Capacity Initiative 2018/2019 PROPOSAL GUIDELINES PROPOSAL CLOSING DATE: 4:30 PM on Monday, December 11, 2017 EMAIL your proposal and all supporting documents to BCMail@aandc.gc.ca. 1. Copy the following
BC Capacity Initiative 2018/2019 PROPOSAL GUIDELINES PROPOSAL CLOSING DATE: 4:30 PM on Monday, December 11, 2017 EMAIL your proposal and all supporting documents to BCMail@aandc.gc.ca. 1. Copy the following
The Lifecycle of a Sponsored Project
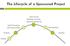 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where does the
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where does the
NSF FUNDAMENTALS WORKSHOP. Thomas Jefferson University December 2017
 NSF FUNDAMENTALS WORKSHOP Thomas Jefferson University December 2017 SPEAKERS Jean Feldman Head, Policy Office Division of Institution and Award Support Office of Budget, Finance and Award Management 703.292.8243;
NSF FUNDAMENTALS WORKSHOP Thomas Jefferson University December 2017 SPEAKERS Jean Feldman Head, Policy Office Division of Institution and Award Support Office of Budget, Finance and Award Management 703.292.8243;
UNIVERSITY RESEARCH ADMINISTRATION FINANCIAL ROLES AND RESPONSIBILITIES MATRIX - WORK IN PROGRESS 10/03/2013 Roles.
 UNIVERSITY RESEARCH ADMINISTRATION Roles Business Internal Controller's Clinical Responsibilities PI Office Chair Dean Audit Office OCR GCFA GCA PROVOST Trials Office I. GENERAL RESEARCH ADMINISTRATION
UNIVERSITY RESEARCH ADMINISTRATION Roles Business Internal Controller's Clinical Responsibilities PI Office Chair Dean Audit Office OCR GCFA GCA PROVOST Trials Office I. GENERAL RESEARCH ADMINISTRATION
Cost Sharing Administrative Guidelines
 Southern Illinois University Carbondale Cost Sharing Administrative Guidelines Summary Cost sharing refers to the resources contributed or allocated by the University to an externally sponsored project,
Southern Illinois University Carbondale Cost Sharing Administrative Guidelines Summary Cost sharing refers to the resources contributed or allocated by the University to an externally sponsored project,
FINANCE-315 7/1/2017 SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a proposal to UACES. It provides a checklist of documents and certifications required by sponsors, as well as an
SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a proposal to UACES. It provides a checklist of documents and certifications required by sponsors, as well as an
Course Planners: Anne Klibanski, MD Donna Lawton, MS Nancy Rigotti, MD Ann Skoczenski, PhD Theodore Stern, MD. Course Faculty: Theodore Stern, MD
 The following planners, speakers, and content reviewers, on behalf of themselves and their spouse or partner, have reported no relevant financial relationships with any entity producing, marketing, re-selling,
The following planners, speakers, and content reviewers, on behalf of themselves and their spouse or partner, have reported no relevant financial relationships with any entity producing, marketing, re-selling,
Please fill out the survey forms 2/9/2012. Upcoming ORA Training Opportunities. Agenda
 Agenda Welcome and Announcements Marcia Smith Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Data Management Tool Sharon Farb, Associate University Librarian,
Agenda Welcome and Announcements Marcia Smith Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Data Management Tool Sharon Farb, Associate University Librarian,
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018
 CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
