Sponsored Program Administration. October 10, 2017
|
|
|
- Adrian Harrison
- 6 years ago
- Views:
Transcription
1 Sponsored Program Administration October 10, 2017
2 Agenda Procurement Services FDP Updates Jaggaer Pre-Award Federal Agency Updates Grants.gov Transition to Workspace NIH FORMS-E Post-Award GONE Act Certificate of Confidentiality Greenphire Update Adjustments/Credits on Grants Effort Reporting Updates and Reminders New Screens UConn Salary Calculator Updated Calculator for Furlough Days
3 Procurement Services
4 September FDP Conference Key Updates Procurement Rules Go-Live Date 5/17: OMB announces the one year extension for the grace period for implementation of the Procurement section of Uniform Guidance. 7/1/18 is the official go-live date. Micro-purchase Threshold National Defense Authorization Act & American Innovation and Competitiveness Act seem to increase threshold to 10k. HOWEVER this is not reflected in the official UG text yet. When can IHEs start using the 10k threshold? How can IHEs request a MPT higher than 10k? (discussion at other IHEs, not UConn, 10k matches state statutes) What is considered a clean audit? (one-off or systemic issue?) What internal controls will be acceptable? How will this be determined? What kind of risk analysis would be acceptable? Who should the appeal for a higher threshold be sent to?
5 September FDP Conference Key Updates (cont.) Unanswered Questions Posed at FDP Procurement Session How should IHEs document negotiating for profit when the vendor refuses to share the necessary information? With the new COI, is there any way for managed conflicts to be allowed? How should the new COI rules be communicated to the campus (i.e. faculty buying from their own companies is NOT allowable under UG)? How strict should the Sole Source exemptions be interpreted? How should the exemptions be communicated to the campus community? Procurement agent is now responsible for their transactions.
6 Uniform Guidance Federal Register New UG updates should be coming soon in the Federal Register UPDATE: 9/17 STILL no update from OMB on when the Federal Register update will occur. FDP and COGR are unsure on when this will occur since it is so late. Procurement Services will keep you up to date on its website:
7 Jaggaer Project Kickoff: September 6 th, 2017 Procurement will be seeking research community involvement Creating a Jaggaer-specific website to keep community informed ETA October Creating a Readiness Council Each department or division will have a representative Targeted Hard Go-Live : July 2 nd, 2018
8 Lab Suppliers Contract Portfolio Update Fisher Scientific contract has been signed VWR (secondary supplier) negotiations just finishing Westnet negotiations have been initiated A communication launch will be circulated after VWR is signed Lab Supplies website to launch in November
9 One University Initiative Procurement Services is working with the OVPR to better align the researcher experience at UCH with that of Storrs/Regionals
10 Pre-Award
11 Federal Agency Updates All Federal Agencies Although some agencies (e.g., NIH and NSF) extend the deadline to the next business day if it falls on a weekend or federal holiday, this is not true for all agencies (e.g., NOAA). Unless stated in the solicitation or agency policy, assume that the deadline is hard. NIH Some Institutes/Centers are prohibiting salary increases in proposal budgets and will reduce the budget accordingly at time of award. This impacts all proposals awarded by NHLBI and may impact other areas as well. Pre-Award recommends no salary escalations for NHBLI budgets and will respect the decision not to escalate in NIH proposals in general. NSF Research.gov transition update (initial launch planned for winter/spring 2018)
12 Grants.gov Transition to Workspace Now Workspace is available for use January 1, 2018 PDF forms no longer available to download April 1, 2018 Only Workspace applications will be accepted, PDF forms will not be accepted Steps to use Workspace Create a grants.gov account (enter UConn s DUNS ): Search for the application package Select the option to apply using Workspace
13 Grants.gov Transition to Workspace Resources Creating a Workspace package: Completing a Workspace package: Current Issues/Concerns SPS will not have the ability to view the attachments, until this is resolved, attachments must also be ed to SPS NIH NIH applications may be submitted through either ASSIST or Workspace
14 NIH FORMS-E FORMS-E must be used for all applications with due dates on or after January 25, 2018 FORMS-D must be used for all applications with due dates on or before January 24, 2018 Major Changes Removed Human Subjects section (protection of human subjects, data safety and monitoring, inclusion of women and minorities and inclusion of children) Timeline 10/25/17 or earlier: Application guides available 11/10/17 or earlier: Application packages available
15 Post-Award
16 GONE Act The Grants Oversight and New Efficiency Act (GONE Act) was signed into law on January 28, 2016 in response to the increasing amount of expired grants in the Department of Health and Human Services (HHS) Payment Management System (PMS)
17 Certificate of Confidentiality NIH has updated their Certificate of Confidentiality (CoC) policy. New Policy, NOT- OD , effective October 1, 2017, which offers: Privacy protection for individuals participating in NIH funded research studies. Elimination of NIH funded investigators having to apply for a CoC. All ongoing or new research funded by NIH as of December 13, 2016 that is collecting or using identifiable, sensitive information is automatically issued a CoC. Compliance requirements are outlined in the NIH Grants Policy Statement, which is a term and condition of all NIH awards. NIH will no longer issue a physical certificate. You may point to your Notice of Award and the NIH Grants Policy Statement as documentation of the CoC protection. The awardee must ensure that anyone who is conducting research as a subawardee or receives a copy of identifiable sensitive information protected by the policy understand they are they are also subject to the disclosure restrictions, even if they are not funded directly by NIH.
18 Certificate of Confidentiality Recipient Responsibilities Applying the Policy To determine if this COC Policy applies to research conducted or supported by NIH, investigators will need to ask, and answer the following question: Is the activity biomedical, behavioral, clinical, or other research? If the answer to this question is no, then the activity is not issued a Certificate.
19 Certificate of Confidentiality Recipient Responsibilities - Applying the Policy (Continued) If the answer is yes, then investigators will need to answer the following questions: Does the research involve Human Subjects as defined by 45 CFR Part 46? Are you collecting or using biospecimens that are identifiable to an individual as part of the research? If collecting or using biospecimens as part of the research, is there a small risk that some combination of the biospecimen, a request for the biospecimen, and other available data sources could be used to deduce the identity of an individual? Does the research involve the generation of individual level, human genomic data? If the answer to any one of these questions is yes, then this Policy will apply to the research.
20 Certificate of Confidentiality Recipient Responsibilities - Will I ever need to extend or amend my CoC? If your NIH-funding will or has ended but the collection of new data from research participants will continue without NIH-funding you will need to apply for a CoC for continuity of protections using the CoC application system. If your NIH funding will or has ended but your study has completed all enrollment and data collection, there is no need to extend the Certificate. Sensitive, identifiable research information maintained by investigators during any time a Certificate is in effect, is protected permanently.
21 Greenphire System Human Subject Payment System (Debit Card based system) Contract was signed by the CT Attorney General Office on 10/5/17 A Pilot Group will be formally contacted to be trained and begin using the systems for payments. Procedures and Quick Guides are planned to be developed during pilot. Full rollout and training is anticipated to begin in approximately eight weeks.
22 Adjustments/Credits on Sponsored Accounts o Vendor Credits o Return of purchases o Corrections of billing errors o Payroll Credits o Payroll after the end date of Grant o Overpayments to employees (e.g., late processing of separation) o Escheatments o Unclaimed payments (e.g., non-cashing of checks for travel reimbursements) How to handle: o If Grant is still active, credits should be applied to the account the original charge was allocated to; and o If Grant has ended, please contact SPS for guidance.
23 Effort Reporting
24 Effort Reporting Updates Spring 2017 report Due back from departments by 11/1/2017 New Excel Export on Report Period Overview School level screen New Quick Guides to be launched Commitment Comparison Screen (formally Effort Reporting Details) Commitment Information by Project Commitment Information by Person Don t forget to check the commitments! New wording on screens to help certifiers remember as well.
25 Commitment Comparison Screen
26 New Commitment Screens
27 Changes to Effort Report Screen Same screen, new name Links to Commitment Comparison
28 Changes to Effort Report Screen Easy for PI to compare the Commitment and Actual Effort entered on report
29 Updates to UConn Salary Calculator
30 Updated UCONN Salary Calculator UConn Salary Calculator Version 3.1 Now available on the SPS website (coming soon on the ESS and Payroll websites) Now defaults to AY New calculations for furlough adjustments and related accrual rate changes Funding End Date can now continue until 8/22
31 Updated UCONN Salary Calculator Change to No for employees that are not impacted by furloughs The full-time salary adjusted for three furlough days (26 pay periods). This shows the bi-weekly pay rate in place for the majority of the funding period (actual pay rate may vary in AY ). The number of pay periods for which the Funding is in place. Pay periods between Funding Start and End Dates (can now run through 8/22).
32 Furlough Adjustments For those impacted by furloughs, calculations were done as follows: Salary for 3 Furlough Days 21 Pay Periods = Bi-Weekly Salary Reduction for Remainder of FY2018 NOTE: the first pay period with furloughs was the pay period ending 8/31/2017 this pay period crossed two academic years.
33 Furlough Adjustments For the 9 & 10 month faculty with accruals: To avoid accrual adjustments for AY , the entire biweekly furlough adjustment was recognized in the AY portion of PPE 8/31/2017. This results in three pay rates for accrual purposes for 9 & 10 month faculty in AY /23/2017-8/31/2017: where the full bi-weekly furlough adjustment was taken over 7 days 9/1/2017-6/7/2018: the regular furlough adjusted pay rate 6/8/2018-8/22/2018: the standard pay rate without furloughs
34 Updated UCONN Salary Calculator Use the UConn Salary Calculator to determine the financial impact of your payroll authorizations Other payroll changes (FTE reductions, salary increases, unpaid leaves) will result in a nonstandard accrual rate for that faculty member The UConn Salary Calculator will not be 100% accurate in these cases If you have any questions about the calculator or the accruals, contact Jen Pelletier or facultyaccrual@uconn.edu
35 Questions?
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Preparing campus for what is changing and what is not What is Grants Reform and when is it effective? What
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Preparing campus for what is changing and what is not What is Grants Reform and when is it effective? What
Sponsored Program Administration Meeting. September 2016
 Sponsored Program Administration Meeting September 2016 Agenda NCURA Internal Controls Mike Unplugged: Training/Education Specialist position Budgeting for Post doc increases FDP Update ETS Update Effort
Sponsored Program Administration Meeting September 2016 Agenda NCURA Internal Controls Mike Unplugged: Training/Education Specialist position Budgeting for Post doc increases FDP Update ETS Update Effort
Office of Sponsored Projects 5/31/ Volume 2, Issue 5
 The OSP News & Updates, published by the Office of Sponsored Projects, is a bi-weekly subscription-based newsletter that provides OSP and sponsor updates and reminders, quick facts, guidance and training
The OSP News & Updates, published by the Office of Sponsored Projects, is a bi-weekly subscription-based newsletter that provides OSP and sponsor updates and reminders, quick facts, guidance and training
CURRENT COGR PRIORITIES - BY COMMITTEE (7/10/17)
 CURRENT COGR PRIORITIES - BY COMMITTEE (7/10/17) No. 1 Student Financial Aid and "Securing Student Information" and Compliance Supplement New single audit requirement. Delayed until 2018. MEDIUM Pending
CURRENT COGR PRIORITIES - BY COMMITTEE (7/10/17) No. 1 Student Financial Aid and "Securing Student Information" and Compliance Supplement New single audit requirement. Delayed until 2018. MEDIUM Pending
2017 SRA International Annual Meeting October 14-18, Federal Demonstration Partnership (FDP)/Council on Governmental Relations (COGR) Update
 2017 SRA International Annual Meeting October 14-18, 2017 Federal Demonstration Partnership (FDP)/Council on Governmental Relations (COGR) Update Who are we? Anthony DeCrappeo, President, Council on Governmental
2017 SRA International Annual Meeting October 14-18, 2017 Federal Demonstration Partnership (FDP)/Council on Governmental Relations (COGR) Update Who are we? Anthony DeCrappeo, President, Council on Governmental
UNIFORM GUIDANCE IMPLEMENTATION
 UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM. December 2017
 Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
OMB Uniform Guidance ( UG ) Briefing. ASRSP & OSR Brown Bag Tuesday, January 27 th
 OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards (New Uniform Guidance)
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards (New Uniform Guidance) Spring Research Administrators Series March 19, 2015 1 Agenda Background UW-Madison
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards (New Uniform Guidance) Spring Research Administrators Series March 19, 2015 1 Agenda Background UW-Madison
Costing and Procurement Updates
 Costing and Procurement Updates Jim Luther, Duke University Edwin Bemmel, University of Miami Doug Backman, University of South Florida Sara Bible, Stanford University January 8, 2018 Agenda Update (Costing
Costing and Procurement Updates Jim Luther, Duke University Edwin Bemmel, University of Miami Doug Backman, University of South Florida Sara Bible, Stanford University January 8, 2018 Agenda Update (Costing
Recent Legislative Actions Taken to Reduce Research Regulatory Burden. 21st Century Cures (Passed House and Senate. Signed into law Dec.
 . Signed into law Dec. 13) Link to PDF Link to PDF Link to PDF Research Policy Board - A public-private entity recommended by the National Academies "to foster more effective conception, development and
. Signed into law Dec. 13) Link to PDF Link to PDF Link to PDF Research Policy Board - A public-private entity recommended by the National Academies "to foster more effective conception, development and
USF HRPP Updates October 2017
 USF HRPP Updates October 2017 Julie Moore, J.D., M.S. PA, CIP Associate Director, Research Integrity & Compliance Research Integrity and Compliance Revised Common Rule 45 CFR 46 Effective date and compliance
USF HRPP Updates October 2017 Julie Moore, J.D., M.S. PA, CIP Associate Director, Research Integrity & Compliance Research Integrity and Compliance Revised Common Rule 45 CFR 46 Effective date and compliance
UNIFORM ADMINISTRATIVE REQUIREMENTS, COST PRINCIPLES, AND AUDIT REQUIREMENTS FOR FEDERAL AWARDS - UPDATE FEBRUARY 2015
 UNIFORM ADMINISTRATIVE REQUIREMENTS, COST PRINCIPLES, AND AUDIT REQUIREMENTS FOR FEDERAL AWARDS - UPDATE FEBRUARY 2015 AOA Conference Pasadena, CA February 9, 2015 Agenda 1. Introduction / Disclaimer 2.
UNIFORM ADMINISTRATIVE REQUIREMENTS, COST PRINCIPLES, AND AUDIT REQUIREMENTS FOR FEDERAL AWARDS - UPDATE FEBRUARY 2015 AOA Conference Pasadena, CA February 9, 2015 Agenda 1. Introduction / Disclaimer 2.
SPONSORED PROGRAM ADMINISTRATION MEETING March 2017 WELCOME
 SPONSORED PROGRAM ADMINISTRATION MEETING March 2017 WELCOME OVPR/SPS News and Information Technology Commercialization Services Andrew Zehner 2017 Individual Financial Disclosure in Research Internal Routing
SPONSORED PROGRAM ADMINISTRATION MEETING March 2017 WELCOME OVPR/SPS News and Information Technology Commercialization Services Andrew Zehner 2017 Individual Financial Disclosure in Research Internal Routing
RAIS AND REPORTING USER GROUP RESEARCH ADMINISTRATION INFORMATION SYSTEMS (RAIS)
 RAIS AND REPORTING USER GROUP RESEARCH ADMINISTRATION INFORMATION SYSTEMS (RAIS) Agenda Coeus Coeus Access Form Update Coeus User Access Roles New Grants.gov Forms NSF Submissions Proposal Person Addresses
RAIS AND REPORTING USER GROUP RESEARCH ADMINISTRATION INFORMATION SYSTEMS (RAIS) Agenda Coeus Coeus Access Form Update Coeus User Access Roles New Grants.gov Forms NSF Submissions Proposal Person Addresses
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018
 CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
Uniform Guidance Subpart D Administrative Requirements. Why This Session Is Needed. Lesson Overview & Module Objectives
 Uniform Guidance Subpart D Administrative Requirements 1 Why This Session Is Needed Changes impact grant operations and policies Changes to recipient and subrecipient responsibilities 2 Lesson Overview
Uniform Guidance Subpart D Administrative Requirements 1 Why This Session Is Needed Changes impact grant operations and policies Changes to recipient and subrecipient responsibilities 2 Lesson Overview
Please fill out the survey forms 2/9/2012. Upcoming ORA Training Opportunities. Agenda
 Agenda Welcome and Announcements Marcia Smith Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Data Management Tool Sharon Farb, Associate University Librarian,
Agenda Welcome and Announcements Marcia Smith Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Data Management Tool Sharon Farb, Associate University Librarian,
INTRODUCTION TO RESEARCH ADMINISTRATION. Office of Grants and Contracts Administration August 2015
 INTRODUCTION TO RESEARCH ADMINISTRATION Office of Grants and Contracts Administration August 2015 THE RESEARCH VESSEL SIKULIAQ FUNDED BY THE NATIONAL SCIENCE FOUNDATION Objectives To understand the UAF
INTRODUCTION TO RESEARCH ADMINISTRATION Office of Grants and Contracts Administration August 2015 THE RESEARCH VESSEL SIKULIAQ FUNDED BY THE NATIONAL SCIENCE FOUNDATION Objectives To understand the UAF
Presenter. Changes to Federal Programs & Single Audits (A-87, A-21, A-122, A-102, A-110, A-89, A-133 & A-50) The New OMB Uniform Guidance
 Changes to Federal Programs & Single Audits (A-87, A-21, A-122, A-102, A-110, A-89, A-133 & A-50) The New OMB Uniform Guidance Presenter Richard Cunningham Quality Assurance & Technical Specialist Center
Changes to Federal Programs & Single Audits (A-87, A-21, A-122, A-102, A-110, A-89, A-133 & A-50) The New OMB Uniform Guidance Presenter Richard Cunningham Quality Assurance & Technical Specialist Center
GATA GRANT ACCOUNTABILITY AND TRANSPARENCY ACT OVERVIEW T.H.E. CONFERENCE
 GATA GRANT ACCOUNTABILITY AND TRANSPARENCY ACT OVERVIEW T.H.E. CONFERENCE 2.28.17 Topics of Discussion GATA Myths Applicability of GATA GATA Overview What s New Roles and Responsibilities Consequences
GATA GRANT ACCOUNTABILITY AND TRANSPARENCY ACT OVERVIEW T.H.E. CONFERENCE 2.28.17 Topics of Discussion GATA Myths Applicability of GATA GATA Overview What s New Roles and Responsibilities Consequences
December 2015 Research Administration Working Group WELCOME OFFICE OF THE VICE PRESIDENT FOR RESEARCH
 December 2015 Research Administration Working Group WELCOME Open Mike Fringe Rates FY17 proposed Budget v. Charging Distribution of Indirects PI award letter out & accounts funded Departments- this month
December 2015 Research Administration Working Group WELCOME Open Mike Fringe Rates FY17 proposed Budget v. Charging Distribution of Indirects PI award letter out & accounts funded Departments- this month
NECA Update The New Uniform Guidance 2 CFR 200
 NECA Update The New Uniform Guidance 2 CFR 200 September 23, 2014 Increase in Federal Grants Activity The Catalog of Federal Domestic Assistance lists over 2,000 Federal grant programs $600B $200B $7B
NECA Update The New Uniform Guidance 2 CFR 200 September 23, 2014 Increase in Federal Grants Activity The Catalog of Federal Domestic Assistance lists over 2,000 Federal grant programs $600B $200B $7B
Rebecca Trahan. Office of Sponsored Programs December 9, ORED Limited Submission Update
 Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
The Uniform Guidance (2 CFR, Part 200)
 WCMC Implementation of The Uniform Guidance (2 CFR, Part 200) Tuesday, September 22, 2015 & Wednesday, September 23, 2015 UG Workshop Michelle A. Lewis, M.S. Director of Research Administration Interim
WCMC Implementation of The Uniform Guidance (2 CFR, Part 200) Tuesday, September 22, 2015 & Wednesday, September 23, 2015 UG Workshop Michelle A. Lewis, M.S. Director of Research Administration Interim
Grants Financial Procedures (Post-Award) v. 2.0
 Grants Financial Procedures (Post-Award) v. 2.0 1 Grants Financial Procedures (Post Award) Version Number: 2.0 Procedures Identifier: Superseded Procedure(s): BU-PR0001 N/A Date Approved: 9/1/2013 Effective
Grants Financial Procedures (Post-Award) v. 2.0 1 Grants Financial Procedures (Post Award) Version Number: 2.0 Procedures Identifier: Superseded Procedure(s): BU-PR0001 N/A Date Approved: 9/1/2013 Effective
Federal Rules for Sponsored Programs. Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards 2 CFR 200
 Federal Rules for Sponsored Programs Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards 2 CFR 200 Uniform Guidance (UG) The Basics Presented by Dan Evon Director
Federal Rules for Sponsored Programs Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards 2 CFR 200 Uniform Guidance (UG) The Basics Presented by Dan Evon Director
UPDATES. Meet the Proposal Deadline. NIH: Public Access to Research Results OFFICE OF SPONSORED INSIDE THIS ISSUE:
 UPDATES INSIDE THIS ISSUE: Meet the Proposal Deadline Responsible Conduct of Research New Federal Regulations on Conflict of Interest NSF s New Proposal Guidelines and Project Reporting Changes Rutgers
UPDATES INSIDE THIS ISSUE: Meet the Proposal Deadline Responsible Conduct of Research New Federal Regulations on Conflict of Interest NSF s New Proposal Guidelines and Project Reporting Changes Rutgers
Grant/Sponsor Related Systems. Department and OSP Perspectives on ERA
 Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
OMB Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards
 OMB Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Research Advisory Board What PIs Need to Know January 2015 1 Background o When and Why? o Overview Important
OMB Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Research Advisory Board What PIs Need to Know January 2015 1 Background o When and Why? o Overview Important
Diane Dean, Director Kathy Hancock, Assistant Grants Compliance Officer Joel Snyderman, Assistant Grants Compliance Officer
 Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar San Diego, CA October 2015 Diane Dean,
Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar San Diego, CA October 2015 Diane Dean,
Research Administration Frequently Asked Questions
 Research Administration Frequently Asked Questions I am submitting a proposal to NIH, but I can t download the grants.gov package, how do I submit? A. Use an old grants.gov package from a previous submission
Research Administration Frequently Asked Questions I am submitting a proposal to NIH, but I can t download the grants.gov package, how do I submit? A. Use an old grants.gov package from a previous submission
Financial Oversight of Sponsored Projects
 Financial Oversight of Sponsored Projects Today s Agenda Introduction Reasons for Principal Investigator Oversight Sponsor and University Policies General Considerations Unallowable Costs Re-Budgeting
Financial Oversight of Sponsored Projects Today s Agenda Introduction Reasons for Principal Investigator Oversight Sponsor and University Policies General Considerations Unallowable Costs Re-Budgeting
Research Administrators Round Table
 Research Administrators Round Table Recap Set framework for going forward Review new Uniform Guidance driven WVU policy changes Sponsored Agreement Closeouts (WVU-FS-23) http://adminfin.wvu.edu/policies/finance/financial-services/wvu-fs-23-policy
Research Administrators Round Table Recap Set framework for going forward Review new Uniform Guidance driven WVU policy changes Sponsored Agreement Closeouts (WVU-FS-23) http://adminfin.wvu.edu/policies/finance/financial-services/wvu-fs-23-policy
FINANCE-315 7/1/2017 SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a proposal to UACES. It provides a checklist of documents and certifications required by sponsors, as well as an
SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a proposal to UACES. It provides a checklist of documents and certifications required by sponsors, as well as an
Paul H. Calabrese Rubino & Company, CPAs & Consultants Senior Manager. Tel:
 Federal Agencies Implementation of the Super Circular & the JIR Presenter: Paul H. Calabrese Rubino & Company, CPAs & Consultants Senior Manager Tel: 301-214-4137 pcalabrese@rubino.com www.linkedin.com/pub/paul-calabrese/20/293/645/
Federal Agencies Implementation of the Super Circular & the JIR Presenter: Paul H. Calabrese Rubino & Company, CPAs & Consultants Senior Manager Tel: 301-214-4137 pcalabrese@rubino.com www.linkedin.com/pub/paul-calabrese/20/293/645/
2017 NIH Update. Presented by Stephanie Smith and Stacey Wade
 2017 NIH Update Presented by Stephanie Smith and Stacey Wade PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs NIH INSTITUTES National
2017 NIH Update Presented by Stephanie Smith and Stacey Wade PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs NIH INSTITUTES National
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE
 THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research organizations aimed at streamlining
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research organizations aimed at streamlining
ORA Quarterly Meeting April
 ORA Quarterly Meeting April 19 2017 Agenda 1. Workday Grants Implementation Items General Information Blackout Period Sub K s - Set-up and Invoicing Advance Accounts 2. Jackson/ UM Partnership Master Agreement
ORA Quarterly Meeting April 19 2017 Agenda 1. Workday Grants Implementation Items General Information Blackout Period Sub K s - Set-up and Invoicing Advance Accounts 2. Jackson/ UM Partnership Master Agreement
Welcome to A Beginner s Guide to Sponsored Project Solicitations. This is one of the introductory mini courses in Northwestern s Sponsored Project
 Welcome to A Beginner s Guide to Sponsored Project Solicitations. This is one of the introductory mini courses in Northwestern s Sponsored Project Online Training series. 1 If you have previously joined
Welcome to A Beginner s Guide to Sponsored Project Solicitations. This is one of the introductory mini courses in Northwestern s Sponsored Project Online Training series. 1 If you have previously joined
Research Administration Updates. May 16, :00 am IOP Auditorium
 Research Administration Updates May 16, 2018 11:00 am IOP Auditorium Topics Covered Using ClinCard (Greenphire) for Federally Sponsored Clinical Research Studies Ryan Mulligan, SCTR Institute OCR & ORSP
Research Administration Updates May 16, 2018 11:00 am IOP Auditorium Topics Covered Using ClinCard (Greenphire) for Federally Sponsored Clinical Research Studies Ryan Mulligan, SCTR Institute OCR & ORSP
Cost Sharing Administrative Guidelines
 Southern Illinois University Carbondale Cost Sharing Administrative Guidelines Summary Cost sharing refers to the resources contributed or allocated by the University to an externally sponsored project,
Southern Illinois University Carbondale Cost Sharing Administrative Guidelines Summary Cost sharing refers to the resources contributed or allocated by the University to an externally sponsored project,
Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS
 Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar Chicago, IL October 2016 Diane Dean, Director
Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar Chicago, IL October 2016 Diane Dean, Director
Policy and Compliance: Working Together Like Hand in Glove
 Policy and Compliance: Working Together Like Hand in Glove Samuel Ashe, Director, Division of Grants Policy, OPERA, OER Diane W. Dean, Director, Division of Grants Compliance and Oversight, OPERA, OER
Policy and Compliance: Working Together Like Hand in Glove Samuel Ashe, Director, Division of Grants Policy, OPERA, OER Diane W. Dean, Director, Division of Grants Compliance and Oversight, OPERA, OER
UNIFORM GUIDANCE UPDATE
 UNIFORM GUIDANCE UPDATE CINDY KIEL Executive Associate Vice Chancellor Office of Research Michael Allred Associate Vice Chancellor for Finance/Controller What is the Uniform Guidance? Uniform Administrative
UNIFORM GUIDANCE UPDATE CINDY KIEL Executive Associate Vice Chancellor Office of Research Michael Allred Associate Vice Chancellor for Finance/Controller What is the Uniform Guidance? Uniform Administrative
2017 State of Connecticut Regenerative Medicine Research Fund (RMRF) General RFP
 2017 State of Connecticut Regenerative Medicine Research Fund (RMRF) General RFP (Seed, Established Investigator, Group) Full RFP, Forms and additional FAQs: http://ctinnovations.com/rmrf Deadline Friday,
2017 State of Connecticut Regenerative Medicine Research Fund (RMRF) General RFP (Seed, Established Investigator, Group) Full RFP, Forms and additional FAQs: http://ctinnovations.com/rmrf Deadline Friday,
Are You Ready for This? The New Uniform Guidance 2 CFR 200
 Are You Ready for This? The New Uniform Guidance 2 CFR 200 Increase in Federal Grants Activity The Catalog of Federal Domestic Assistance lists over 2,000 Federal grant programs $600B $200B $7B $24B $91B
Are You Ready for This? The New Uniform Guidance 2 CFR 200 Increase in Federal Grants Activity The Catalog of Federal Domestic Assistance lists over 2,000 Federal grant programs $600B $200B $7B $24B $91B
FY 2018 SNAP PROCESS AND TECHNOLOGY IMPROVEMENT GRANTS (PTIG)
 FY 2018 SNAP PROCESS AND TECHNOLOGY IMPROVEMENT GRANTS (PTIG) MAY 2018 THIS WEBINAR IS BEING RECORDED Presented by Alice McKenney and Sarah Goldberg, SNAP Program Analysts Webinar Agenda PTIG Basics Fiscal
FY 2018 SNAP PROCESS AND TECHNOLOGY IMPROVEMENT GRANTS (PTIG) MAY 2018 THIS WEBINAR IS BEING RECORDED Presented by Alice McKenney and Sarah Goldberg, SNAP Program Analysts Webinar Agenda PTIG Basics Fiscal
An Exercise in Effort
 3 rd Annual Symposium for Research Administrators An Exercise in Effort Brian Bertlshofer, Director, Cost Analysis and Compliance bertlsbj@email.unc.edu Aja Saylor, Central Effort Coordinator ajasaylor@unc.edu
3 rd Annual Symposium for Research Administrators An Exercise in Effort Brian Bertlshofer, Director, Cost Analysis and Compliance bertlsbj@email.unc.edu Aja Saylor, Central Effort Coordinator ajasaylor@unc.edu
Office of Sponsored Programs Budgetary and Cost Accounting Procedures
 Office of Sponsored Programs Budgetary and Cost Accounting Procedures Table of Contents 1. Purpose and Services 2. Definitions of Terms 3. Budget Items 4. Travel 5. Effort Certification Reporting 6. Costing
Office of Sponsored Programs Budgetary and Cost Accounting Procedures Table of Contents 1. Purpose and Services 2. Definitions of Terms 3. Budget Items 4. Travel 5. Effort Certification Reporting 6. Costing
Sponsored Programs New Developments and Important Reminders
 Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs January 23, 2018 1 NSF Updates NIH Updates Federal Updates Defense Base Act (DBA) Insurance OSP Updates
Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs January 23, 2018 1 NSF Updates NIH Updates Federal Updates Defense Base Act (DBA) Insurance OSP Updates
Uniform Guidance Subpart D Administrative Requirements
 Uniform Guidance Subpart D Administrative Requirements Purpose and Introduction Understanding the Uniform Guidance is essential to increase accountability of managing grant funds. The Administrative Requirements
Uniform Guidance Subpart D Administrative Requirements Purpose and Introduction Understanding the Uniform Guidance is essential to increase accountability of managing grant funds. The Administrative Requirements
Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32)
 Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32) Overview: This document is intended to help you complete a KC System to System (S2S) proposal submission
Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32) Overview: This document is intended to help you complete a KC System to System (S2S) proposal submission
office of research administration newsletter
 july/august 2010 vol. 5 no. 1 office of research administration newsletter Yale has a clear obligation to comply with all regulations pertaining to the administration of federal grants, and we will spare
july/august 2010 vol. 5 no. 1 office of research administration newsletter Yale has a clear obligation to comply with all regulations pertaining to the administration of federal grants, and we will spare
Grants - Uniform Guidance/Tracking & Reporting. Presented by: Marc Grace, CPA, Partner Stephen Emery, CPA, Manager
 Grants - Uniform Guidance/Tracking & Reporting Presented by: Marc Grace, CPA, Partner Stephen Emery, CPA, Manager Schedule of Expenditures of Federal Awards Major program determination Internal control
Grants - Uniform Guidance/Tracking & Reporting Presented by: Marc Grace, CPA, Partner Stephen Emery, CPA, Manager Schedule of Expenditures of Federal Awards Major program determination Internal control
Life Sciences Tax Incentive Program
 Life Sciences Tax Incentive Program Solicitation No. 2017 TAX-01 Program Manager: Cheryl Sadeli, Vice President of Finance Questions: Taxprogram@masslifesciences.com Solicitation Issued: December 4, 2017
Life Sciences Tax Incentive Program Solicitation No. 2017 TAX-01 Program Manager: Cheryl Sadeli, Vice President of Finance Questions: Taxprogram@masslifesciences.com Solicitation Issued: December 4, 2017
OSR Monthly Meeting. August 16, 2018 (Chicago) August 21, 2018 (Evanston)
 OSR Monthly Meeting August 16, 2018 (Chicago) August 21, 2018 (Evanston) Agenda OSR Announcements/Staffing Updates ESPR Replacement Project Update Year-end Procedures NSF Updates NIH Biosketch Reminder
OSR Monthly Meeting August 16, 2018 (Chicago) August 21, 2018 (Evanston) Agenda OSR Announcements/Staffing Updates ESPR Replacement Project Update Year-end Procedures NSF Updates NIH Biosketch Reminder
Shared Services for Research Administration
 Shared Services for Research Administration Design, Implementation and Lessons Learned Laura Kozma Director, Research Administration & Faculty Services Sponsored Program Services Office of the Vice President
Shared Services for Research Administration Design, Implementation and Lessons Learned Laura Kozma Director, Research Administration & Faculty Services Sponsored Program Services Office of the Vice President
University of Central Florida
 Guidance & Directive No: ORC-01 University of Central Florida Guidance & Directive Date of Adoption/Revision: February 2013 Subject 1.0 Statement and Purpose Federal and state agencies, private foundations,
Guidance & Directive No: ORC-01 University of Central Florida Guidance & Directive Date of Adoption/Revision: February 2013 Subject 1.0 Statement and Purpose Federal and state agencies, private foundations,
Uniform Guidance. Overview and Implementation Plan. November 21, 2014
 Uniform Guidance Overview and Implementation Plan November 21, 2014 Uniform Guidance. Change is coming! College or Department name here 1 Overarching goal of the reform is to: streamline the rules and
Uniform Guidance Overview and Implementation Plan November 21, 2014 Uniform Guidance. Change is coming! College or Department name here 1 Overarching goal of the reform is to: streamline the rules and
U.S. Department of Housing and Urban Development Office of Housing Counseling
 U.S. Department of Housing and Urban Development Office of Housing Counseling Facilitated by Booth Management Consulting 7230 Lee Deforest Drive, Suite 202 Columbia, MD 21046 Overview of Procurement September
U.S. Department of Housing and Urban Development Office of Housing Counseling Facilitated by Booth Management Consulting 7230 Lee Deforest Drive, Suite 202 Columbia, MD 21046 Overview of Procurement September
Grants Management Scenarios
 Grants Management Scenarios SCENARIO 1: Parker School District received a TIF grant in 2012. It followed the guidelines set forth in the application package and did not list specific vendors in its application.
Grants Management Scenarios SCENARIO 1: Parker School District received a TIF grant in 2012. It followed the guidelines set forth in the application package and did not list specific vendors in its application.
Sponsored Program Administration
 Sponsored Program Administration March, 2018 research.uconn.edu AGENDA OVPR/SPS News and Information IPR Form FCOI SPS Procedures for Fund Set Up Banner NIH News and Information Federal Executive Pay Scale
Sponsored Program Administration March, 2018 research.uconn.edu AGENDA OVPR/SPS News and Information IPR Form FCOI SPS Procedures for Fund Set Up Banner NIH News and Information Federal Executive Pay Scale
The Lifecycle of a Sponsored Project
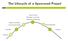 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
Johns Hopkins University Finance Document Library. Sponsored Projects - Effort Reporting Policies & Procedures. Table of Contents
 Johns Hopkins University Finance Document Library Sponsored Projects - Effort Reporting Policies & Procedures Table of Contents I. Policy Section SP-EFF-PL-01--Effort Reporting II. Procedure Section SP-EFF-PR-01--Effort
Johns Hopkins University Finance Document Library Sponsored Projects - Effort Reporting Policies & Procedures Table of Contents I. Policy Section SP-EFF-PL-01--Effort Reporting II. Procedure Section SP-EFF-PR-01--Effort
Texas Education Agency. Division of Federal Fiscal Monitoring
 Texas Education Agency Division of Federal Fiscal Monitoring The DO s and DON Ts of Administering Federal Grants Copyright 2016 by TEA DO s and DON'Ts of Administering Federal Grants Division of Federal
Texas Education Agency Division of Federal Fiscal Monitoring The DO s and DON Ts of Administering Federal Grants Copyright 2016 by TEA DO s and DON'Ts of Administering Federal Grants Division of Federal
National Academy of Sciences Committee on University IP Management
 National Academy of Sciences Committee on University IP Management June 30, 2008 Robert Hardy Director, Contracts and IP Management Council on Governmental Relations A Word About COGR Council on Governmental
National Academy of Sciences Committee on University IP Management June 30, 2008 Robert Hardy Director, Contracts and IP Management Council on Governmental Relations A Word About COGR Council on Governmental
PROPOSAL ROUTING FORM INSTRUCTIONS Dartmouth College/Dartmouth Hitchcock Medical Center
 PROPOSAL ROUTING FORM INSTRUCTIONS Dartmouth College/Dartmouth Hitchcock Medical Center INTRODUCTION This routing form must be completed for all proposals submitted to external funding entities. The form
PROPOSAL ROUTING FORM INSTRUCTIONS Dartmouth College/Dartmouth Hitchcock Medical Center INTRODUCTION This routing form must be completed for all proposals submitted to external funding entities. The form
Department of Human Services Division of Medical Assistance and Health Services Transportation Broker Services Contract Capitation Rates
 New Jersey State Legislature Office of Legislative Services Office of the State Auditor Department of Human Services Division of Medical Assistance and Health Services Transportation Broker Services Contract
New Jersey State Legislature Office of Legislative Services Office of the State Auditor Department of Human Services Division of Medical Assistance and Health Services Transportation Broker Services Contract
Presentation 2/26/2013. Director, TitleOPERA
 Presentation Title Michelle Presented G. Bulls By: Name Director, TitleOPERA Office OER AASCU Washington Conference February 21, 2013 2 NIH is operating under a continuing resolution. See NOT-OD- 13-018
Presentation Title Michelle Presented G. Bulls By: Name Director, TitleOPERA Office OER AASCU Washington Conference February 21, 2013 2 NIH is operating under a continuing resolution. See NOT-OD- 13-018
How to Manage Externally Funded Grants PROJECTS - FUND CODE 501 or 502
 How to Manage Externally Funded Grants PROJECTS - FUND CODE 501 or 502 What is a Grant? Funding that is coming from a source external to Wesleyan Project conceived by the Investigator Response to a solicitation
How to Manage Externally Funded Grants PROJECTS - FUND CODE 501 or 502 What is a Grant? Funding that is coming from a source external to Wesleyan Project conceived by the Investigator Response to a solicitation
Sponsored Projects Manual
 Sponsored Projects Manual University Policies and Procedures for Requesting, Accepting, and Administering Grants, Contracts, and Cooperative Agreements February 2, 2005 CONTENTS 1. Overview... 4 1.1. Purpose
Sponsored Projects Manual University Policies and Procedures for Requesting, Accepting, and Administering Grants, Contracts, and Cooperative Agreements February 2, 2005 CONTENTS 1. Overview... 4 1.1. Purpose
Effort Certifications
 Effort Certifications Presented by Terry Shoebotham 1/12/09 prepared by Terry Shoebotham Business Management Specialist With assistance from: Contracts and Grants Accounting Main Campus and HSC TABLE OF
Effort Certifications Presented by Terry Shoebotham 1/12/09 prepared by Terry Shoebotham Business Management Specialist With assistance from: Contracts and Grants Accounting Main Campus and HSC TABLE OF
Outgoing Subagreements: Subawards and Subcontracts
 Effective Date: March 1, 2014 Version: 1 Page: 1 of 6 Overview Sponsored programs may include partnerships with other organizations or institutions whereby a portion of the project is conducted by investigators
Effective Date: March 1, 2014 Version: 1 Page: 1 of 6 Overview Sponsored programs may include partnerships with other organizations or institutions whereby a portion of the project is conducted by investigators
Cayuse 424. Office of Sponsored Programs (OSP) in collaboration with RAS team
 Cayuse 424 Office of Sponsored Programs (OSP) in collaboration with RAS team Agenda Overview of Research Administration Program (RAP) What is Cayuse 424? Benefits of Cayuse 424 When to use Cayuse 424 How
Cayuse 424 Office of Sponsored Programs (OSP) in collaboration with RAS team Agenda Overview of Research Administration Program (RAP) What is Cayuse 424? Benefits of Cayuse 424 When to use Cayuse 424 How
Administrative and Indirect Costs. What s the difference?
 Administrative and Indirect Costs What s the difference? Learning Objectives Define administrative costs. Describe the difference between direct and indirect costs. Describe what an indirect cost rate
Administrative and Indirect Costs What s the difference? Learning Objectives Define administrative costs. Describe the difference between direct and indirect costs. Describe what an indirect cost rate
UPDATES WELCOME BACK! OFFICE OF SPONSORED
 UPDATES INSIDE THIS ISSUE: Meet the Proposal Deadline RU s New Endorsement Form Rutgers Budget Template NIH Changes to Form Packages NIH Changes for Reports NSF MRI Program Limited Submission Pre-proposal
UPDATES INSIDE THIS ISSUE: Meet the Proposal Deadline RU s New Endorsement Form Rutgers Budget Template NIH Changes to Form Packages NIH Changes for Reports NSF MRI Program Limited Submission Pre-proposal
NIH Update FDP September 2009 Office of Policy for Extramural Research Administration
 NIH Update FDP September 2009 Office of Policy for Extramural Research Administration OPERA New NIH Director Francis S. Collins, M.D., Ph.D., was officially sworn in on Monday, August 17, 2009 as the 16th
NIH Update FDP September 2009 Office of Policy for Extramural Research Administration OPERA New NIH Director Francis S. Collins, M.D., Ph.D., was officially sworn in on Monday, August 17, 2009 as the 16th
RSP 101: Introduction to Research and Sponsored Programs Policies and Procedures. Marcus W. Shute, P.E., Ph.D.
 RSP 101: Introduction to Research and Sponsored Programs Policies and Procedures Marcus W. Shute, P.E., Ph.D. mwshute@cau.edu, x6990 2008-2009 Shute & Clark Atlanta University. All Rights Reserved Introduction
RSP 101: Introduction to Research and Sponsored Programs Policies and Procedures Marcus W. Shute, P.E., Ph.D. mwshute@cau.edu, x6990 2008-2009 Shute & Clark Atlanta University. All Rights Reserved Introduction
NSF OIG Audit Update NORTHEAST CONFERENCE ON COLLEGE COST ACCOUNTING
 NSF OIG Audit Update 1 NORTHEAST CONFERENCE ON COLLEGE COST ACCOUNTING S e p t e m b e r 2 3, 2 0 1 4 Overview 2 Overview of NSF OIG Office of Audit Overview of Federal financial assistance in the U.S.
NSF OIG Audit Update 1 NORTHEAST CONFERENCE ON COLLEGE COST ACCOUNTING S e p t e m b e r 2 3, 2 0 1 4 Overview 2 Overview of NSF OIG Office of Audit Overview of Federal financial assistance in the U.S.
PMS Project Closeout. Dan Evon, Michigan State University Nate Martinez Wayman, Duke University
 PMS Project Closeout Dan Evon, Michigan State University Nate Martinez Wayman, Duke University 1 Session Outline GAO Audit Reports Closeout Parameters The FCTR Closeout History Sample Numbers Details Late
PMS Project Closeout Dan Evon, Michigan State University Nate Martinez Wayman, Duke University 1 Session Outline GAO Audit Reports Closeout Parameters The FCTR Closeout History Sample Numbers Details Late
NSF 2 Month Handbook. Effective for Reviews Performed as of 07/01/17. NSF Account Management. Updated 07/24/17
 Updated 07/24/17 NSF 2 Month Handbook Effective for Reviews Performed as of 07/01/17 NSF Account Management Table of Contents Contents 2 Month Rule Policy... 2 NSF Definition... 2 Sponsor/University Guidance:...
Updated 07/24/17 NSF 2 Month Handbook Effective for Reviews Performed as of 07/01/17 NSF Account Management Table of Contents Contents 2 Month Rule Policy... 2 NSF Definition... 2 Sponsor/University Guidance:...
Trainer(s): Dr. Beverly A. Browning ecivis, Inc.
 01: Securing and Managing Federal Grants Trainer(s): Dr. Beverly A. Browning ecivis, Inc. Securing and Managing Federal Grants Dr. Beverly A. Browning Vice President Grants Professional Services (GPS)
01: Securing and Managing Federal Grants Trainer(s): Dr. Beverly A. Browning ecivis, Inc. Securing and Managing Federal Grants Dr. Beverly A. Browning Vice President Grants Professional Services (GPS)
The Rollout of OMB A-81 and its Effect on UH
 The Rollout of OMB A-81 and its Effect on UH Cris Milligan, Interim Assistant Vice President for Research Administration Beverly Rymer, Executive Director, Office of Contracts and GrantsOffice of Contracts
The Rollout of OMB A-81 and its Effect on UH Cris Milligan, Interim Assistant Vice President for Research Administration Beverly Rymer, Executive Director, Office of Contracts and GrantsOffice of Contracts
Uniform Guidance Sponsored Projects Services
 Arizona s First University. Uniform Guidance Sponsored Projects Services 520-626-6000 sponsor@email.arizona.edu Agenda What is Uniform Guidance (UG)? Effective dates Structure of the Uniform Guidance Significant
Arizona s First University. Uniform Guidance Sponsored Projects Services 520-626-6000 sponsor@email.arizona.edu Agenda What is Uniform Guidance (UG)? Effective dates Structure of the Uniform Guidance Significant
Federal Funding Opportunity Page 1 of 13. Fiscal Year 2019 National Sea Grant College Program Dean John A. Knauss Marine Policy Fellowship
 Federal Funding Opportunity Page 1 of 13 Fiscal Year 2019 National Sea Grant College Program Dean John A. Knauss Marine Policy Fellowship TABLE OF CONTENTS I. Funding Opportunity Description 3 A. Program
Federal Funding Opportunity Page 1 of 13 Fiscal Year 2019 National Sea Grant College Program Dean John A. Knauss Marine Policy Fellowship TABLE OF CONTENTS I. Funding Opportunity Description 3 A. Program
Developing Proposal Budgets
 Grant Writing & Developing Proposal Budgets Sandra H. Harpole Associate VP for Research Sandy Williamson ORED Fiscal Officer January 8, 2008 Idea First or Opportunity First? Idea Have an idea for a project
Grant Writing & Developing Proposal Budgets Sandra H. Harpole Associate VP for Research Sandy Williamson ORED Fiscal Officer January 8, 2008 Idea First or Opportunity First? Idea Have an idea for a project
M E M O R A N D U M PAYROLL OFFICE
 THE UNIVERSITYofTENNESSEE M E M O R A N D U M Office of Vice Chancellor for Finance and Administration 407 Andy Holt Tower Knoxville, TN 37996-0141 (865) 974-4204 FAX (865) 974-8131 TO: FROM: Campus Fiscal
THE UNIVERSITYofTENNESSEE M E M O R A N D U M Office of Vice Chancellor for Finance and Administration 407 Andy Holt Tower Knoxville, TN 37996-0141 (865) 974-4204 FAX (865) 974-8131 TO: FROM: Campus Fiscal
Here Come the Feds! What a Sponsor Audit is Looking for and How to Prepare Your Institution
 Here Come the Feds! What a Sponsor Audit is Looking for and How to Prepare Your Institution Presented by: Richard Cordova, University of Washington Kevin Robinson, Auburn University Kim Ginn, Baker Tilly
Here Come the Feds! What a Sponsor Audit is Looking for and How to Prepare Your Institution Presented by: Richard Cordova, University of Washington Kevin Robinson, Auburn University Kim Ginn, Baker Tilly
OSR s Annual Symposium for Research Administrators: Conflict of Interest The Kitchen Sink Version
 OSR s Annual Symposium for Research Administrators: Conflict of Interest The Kitchen Sink Version Version 07.23.2015 Joy Bryde, MSW Conflict of Interest Officer Conflict of Interest Program Office of the
OSR s Annual Symposium for Research Administrators: Conflict of Interest The Kitchen Sink Version Version 07.23.2015 Joy Bryde, MSW Conflict of Interest Officer Conflict of Interest Program Office of the
Uniform Guidance Update. Ruth Boardman, Associate Director Office of Grants and Contracts March 2015
 Uniform Guidance Update Ruth Boardman, Associate Director Office of Grants and Contracts March 2015 Objectives Latest Updates Internal Controls Procurement Status of Policies and Procedures Research Terms
Uniform Guidance Update Ruth Boardman, Associate Director Office of Grants and Contracts March 2015 Objectives Latest Updates Internal Controls Procurement Status of Policies and Procedures Research Terms
Sponsored Programs New Developments and Important Reminders
 Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs March 7, 2012 1 Training Session Overview National Institutes of Health (NIH) Update NSF Proposal
Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs March 7, 2012 1 Training Session Overview National Institutes of Health (NIH) Update NSF Proposal
Advancing Accountability
 Advancing Accountability BEST PRACTICES IN CONTRACT AND GRANT MANAGEMENT FLORIDA DEPARTMENT OF FINANCIAL SERVICES 1 Intro & Housekeeping Introduction of instructors Housekeeping Class history Class material
Advancing Accountability BEST PRACTICES IN CONTRACT AND GRANT MANAGEMENT FLORIDA DEPARTMENT OF FINANCIAL SERVICES 1 Intro & Housekeeping Introduction of instructors Housekeeping Class history Class material
Implementing the OMB s Super Circular (aka UGG) Presented by: Anne Fritz, Finance Director, City of St. Petersburg, Florida
 Implementing the OMB s Super Circular (aka UGG) Presented by: Anne Fritz, Finance Director, City of St. Petersburg, Florida Acknowledgement to Heather Acker, Partner, Baker Tilly, Virchow Krause, LLP for
Implementing the OMB s Super Circular (aka UGG) Presented by: Anne Fritz, Finance Director, City of St. Petersburg, Florida Acknowledgement to Heather Acker, Partner, Baker Tilly, Virchow Krause, LLP for
QUICK REFERENCE CARD (QRC) University of Oregon Fringe Benefits or OPE (Other Personnel Expense)
 QUICK REFERENCE CARD (QRC) EPCS records must be FINALIZED at least 3 business days in advance of the due date. The following information should be used as the basis for preparing sponsored project proposal
QUICK REFERENCE CARD (QRC) EPCS records must be FINALIZED at least 3 business days in advance of the due date. The following information should be used as the basis for preparing sponsored project proposal
Ask the Experts Panel
 Ask the Experts Panel Compliance in Research Colleen Fritsche, Assistant Director of Office of Animal Care and Use Cassie Myers, Deputy Director of Office of Human Research Ethics Chris Nelson, Director
Ask the Experts Panel Compliance in Research Colleen Fritsche, Assistant Director of Office of Animal Care and Use Cassie Myers, Deputy Director of Office of Human Research Ethics Chris Nelson, Director
Sponsored Programs and Research Compliance SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a subaward proposal to University of North Carolina Wilmington (UNCW). Please complete this form and send all required
SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a subaward proposal to University of North Carolina Wilmington (UNCW). Please complete this form and send all required
QUICK REFERENCE CARD (QRC) University of Oregon Fringe Benefits or OPE (Other Personnel Expense)
 QUICK REFERENCE CARD (QRC) EPCS records must be FINALIZED at least 3 business days in advance of the due date. The following information should be used as the basis for preparing sponsored project proposal
QUICK REFERENCE CARD (QRC) EPCS records must be FINALIZED at least 3 business days in advance of the due date. The following information should be used as the basis for preparing sponsored project proposal
The AOFAS Research Grants Program Description, Policies, and Guidelines for Applicants and Institutional Representatives
 The AOFAS Research Grants Program Description, Policies, and Guidelines for Applicants and Institutional Representatives Updated October 2016 for the 2017 Grants Cycle Table of Contents ABOUT THE AOFAS
The AOFAS Research Grants Program Description, Policies, and Guidelines for Applicants and Institutional Representatives Updated October 2016 for the 2017 Grants Cycle Table of Contents ABOUT THE AOFAS
UNIFORM GUIDANCE OVERVIEW. Budget Officers Meeting January 28, 2015
 UNIFORM GUIDANCE OVERVIEW Budget Officers Meeting January 28, 2015 OMB Circulars Before and After Eight circulars condensed to one new comprehensive set of requirements for recipients of federal awards
UNIFORM GUIDANCE OVERVIEW Budget Officers Meeting January 28, 2015 OMB Circulars Before and After Eight circulars condensed to one new comprehensive set of requirements for recipients of federal awards
