IDeA Post Award Management
|
|
|
- Amie Walton
- 5 years ago
- Views:
Transcription
1 IDeA Post Award Management Lisa Hildred Grants Management Specialist Grants Administration Branch National Institute of General Medical Sciences, NIH
2 Meet the Grants Management Team Christy Leake Sam Farrell Lisa Hildred Brian Iglesias Arina Kramer Courtney Tardd-Wright Emily Tran
3 IDeA Post Award Management IDeA Policy Reminders Cost Considerations Annual Performance Reporting Requirements Prior Approvals NIH Updates and Policy Reminders
4 IDeA Policy Reminders Funds awarded to an IDeA-eligible institution may not be transferred via subcontract to a consortium partner institution in a non-idea state. IDeA funds may be used in other IDeA and non-idea states for fee-forservice type of activities that include activities associated with collaborative projects, research education and training, sample and data analysis, workshops, etc. The PD(s)/PI(s) of IDeA awards may not receive research project support from any COBRE or INBRE program and may not use IDeA funds to supplement research activities within his/her laboratory. Junior investigators of COBRE or INBRE funded research projects or pilot projects may not receive simultaneous research support from more than one IDeA program or receive multiple research projects or pilot projects.
5 Selected items of Cost Honorarium unallowable when the primary intent is to confer distinction on, or to symbolize respect, esteem, or admiration for, the recipient of the honorarium; a payment for services rendered such as speakers fees is allowable Stipends only allowable on training grants; unallowable on IDeA grants; Compensation of Students is allowable Housing not allowable on NIH grants per diem for research participants as part of other expenses is allowable General Supplies Only costs directly related to the grant and/or project are allowable as direct costs; must be consistently treated and reasonable within organizational policy Meals/Food allowable in cases when they are an integral and necessary part of a meeting; must be consistently treated and reasonable within organizational policy Scholarships not allowable on NIH grants; Compensation for research experiences, Participation or Incentive costs is allowable
6 Post Award Requirements
7 Annual Performance Reporting Requirements Research Performance Progress Report (RPPR) Annual progress reports for non-snaps are due two months (60 days) prior to the anniversary date Failure to submit complete and timely progress reports may affect future funding to the organization Grantees may access a list of progress reports that are due using the Status page in era Commons, and selecting the Tab "List of Applications/Grants
8 Annual Performance Reporting Requirements Scientific Information Reporting System (SIRS) Inclusion Management System (IMS) data Inclusion Reports Submitted with the RPPR submission Progress Report Additional Materials (PRAM) Must be initiated by the Grants Management Specialist Allows additional information to be submitted via the era Commons If you need to submit an inclusion report after the RPPR is submitted, you must use the PRAM
9 Prior Approvals NIH Grants Policy Statement Sections 8.1 Changes in Project and Budget _changes_in_project_and_budget.htm Prior Approval Requirements _changes_in_project_and_budget.htm#prior Requests for Prior Approval _changes_in_project_and_budget.htm#requests
10 Requests for Prior Approval Requests for actions that require prior approval must be submitted by an Authorized Organization Representative (AOR) to the NIGMS Grants Management Specialist and Program Officer no later than 30 days before the proposed change NIGMS tries to respond to all prior approval requests within 30 days of receiving all necessary information from the grantee
11 Prior Approvals The following are some Prior Approvals frequently requested for IDeA grants: Carryover of Unobligated Balances Change of Key Personnel Change of Scope Addition of new research projects and changes to currently approved research projects Addition of new pilot projects and changes to currently approved pilot projects
12 Carryover of Unobligated Balances IDeA grants do NOT have automatic carryover Prior approval is required to use unobligated funds from a previous budget period during the current budget period
13 Carryover Requests A carryover request should include the following: Explanation of why there is an unobligated balance Justification of why carryover funds are needed in the current year If the request will generate recurring costs in future years, an explanation of how costs will be supported without future use of unobligated balances or new supplemental funding Budget and justification pages (PHS398 or SF424), including any subcontract sites Calculation of F&A costs (checklist page of PHS398 or SF424) Appropriate Just In Time (JIT) documents (e.g., biosketches for new key personnel, other support, human subjects education certification) Equipment quotes, if appropriate
14 Reasons Why Carryover Requests are Delayed or Rejected: The prior year Federal Financial Report (FFR) has not been received and approved by NIH The request was not signed by or sent through an Authorized Organization Representative (AOR) Requested costs duplicate costs in the current year s award Unlikely that carryover funds can be spent in the budget period they are requested Unallowable costs were included Use of the carryover funds are not within the currently approved scope of the grant Carryover request does not reflect immediate needs No plan to support recurring costs in future years
15 Change of Key Personnel Prior approval is required for the following changes regarding the PI and any Key Personnel named on the Notice of Award: Reduction in effort of 25% or more of the originally approved effort level Reduction in effort that would bring the person below the required minimum effort level Removal from the grant Replacement/Addition of Key Personnel
16 Change of Key Personnel A request for changes to key personnel must include: The reason for the request (leaving/joining/replacing) Justification for selection of the person Level of effort and duties to be performed Explanation of any changes to the budget or scope of the grant Biographical sketch for new person Updated other support for new person Human subjects education if person is involved in the design and conduct of human subjects work
17 Change of Scope A change of scope requires prior approval Potential indicators of a change in scope: Change in the specific aims approved at the time of award Change from the approved involvement of human subjects Change from the approved use of live vertebrate animals Substitution of one animal model for another Shifting research emphasis from one disease area to another Applying a new technology; i.e., changing assays from those approved to a different type of assay Transfer of performance of substantive programmatic work to a third party Change in other senior/key personnel not specifically named in the NoA Significant rebudgeting of 25% or more in a single direct cost budget category Purchase of a unit of equipment exceeding $25,000
18 IDeA Research and Pilot Projects Official approval from NIGMS is required prior to the commencement of any new research project or pilot project COBRE I and II : research and pilot projects COBRE III and IDeA-CTR : pilot projects only INBRE : research and pilot projects
19 Addition of Research Projects COBRE I and II Prior to the commencement of any new research project, the following documentation must be submitted to NIGMS staff: Research project proposal. Using PHS398 forms and instructions, only the following sections have to be submitted: Face Page Project Summary (Page 2) Research Strategy Section External Advisory Committee approval - communication from the EAC chair (at a minimum) indicating that the EAC concurs with supporting the research project Biographical Sketch of Research Project PI Updated other support of Research Project PI Detailed Budget with justification Checklist Format Page with F&A cost breakdown IRB approval, human subjects education certification, and Targeted/Planned Enrollment Table (if applicable) IACUC approval and Vertebrate Animal Section (if applicable) Terms of the NoA
20 Addition of Pilot Projects COBRE I, II, III, and IDeA-CTR Prior to the commencement of any pilot project, the following documentation must be submitted to NIGMS staff per NOT-GM ( Pilot project proposal. Using PHS398 forms and instructions, only the following sections have to be submitted: Face Page Project Summary (Page 2) Research Strategy Section External Advisory Committee approval - communication from the EAC chair (at a minimum) indicating that the EAC concurs with supporting the pilot project. IRB approval, human subjects education certification, and Targeted/Planned Enrollment Table (if applicable) IACUC approval and Vertebrate Animal Section (if applicable) Terms of the NoA
21 Addition of Research and Pilot Projects - INBRE Prior to the commencement of any developmental research project and pilot project, the following documentation must be submitted to NIGMS staff: Developmental Research Project/Pilot Project proposal. Using PHS398 forms and instructions, only the following sections have to be submitted: Face Page Project Summary (Page 2) Research Strategy Section External Advisory Committee approval - communication from the EAC chair (at a minimum) indicating that the EAC concurs with supporting the developmental research project/pilot project. IRB approval, human subjects education certification, and Targeted/Planned Enrollment Table (if applicable) IACUC approval and Vertebrate Animal Section (if applicable) Terms of the NoA
22 Changes of Human Subjects and Vertebrate Animals Involvement Changes of human and vertebrate animal involvement require prior approval For IDeA grants, changes to human subjects and vertebrate animals involvement are usually due to research and pilot projects The required prior approval items for both human subjects and vertebrate animals are submitted as part of the prior approval request for research and pilot projects
23 When Prior Approval is Not Needed NIH prior approval is not required for certain budget and project changes, unless the incurrence of costs is associated with or is considered to be a change in scope. Incur pre-award costs up to 90 days before the beginning date of the initial budget period of a new or competing continuation award. Recipients may incur pre-award costs before the beginning date of a non-competing continuation award without regard to the time parameters stated above. Initiate a one-time extension of the final budget period of a previously approved project period without additional funds (a.k.a First No-Cost Extension) Rebudget among budget categories Rebudget between direct and F&A costs
24 When Prior Approval is Not Needed Level of Effort Changes The personnel are not named on the Notice of Award The personnel are not required to maintain a minimum level of effort (e.g. Pilot Project PIs) The departure or addition of personnel does not result in a change of scope
25 If you aren t sure if prior approval is needed Check the NIH Grants Policy Statement on Prior Approvals for guidance Ask your Sponsored Programs Office / AOR Ask your Program Officer Ask your Grants Management Specialist
26 NIH Updates & Policy Reminders
27 Grant Closeout Requirements NIH GPS 8.6 Closeout: Recipients must submit all closeout documents within 120 calendar days of the end of the period of performance (project period) Final Research Performance Progress Report (F-RPPR) Final Invention Statement and Certification (FIS) Final Federal Financial Report (FFR) HHS policy requirement - Unilateral closeout of awards if the following final reports are not timely and acceptable. Other enforcement actions, such as withholding of further awards, can also be taken.
28 Interim and Final Progress Reports NOT-OD : The Final Research Performance Progress Report (F-RPPR) has replaced the Final Progress Report (FPR) for closeout The general format is the same as the interim/annual RPPR Recipients are required to report on Project Outcomes Due dates have not changed NOT-OD : Implementation of the Interim-RPPR while a Renewal Application is Under Consideration - Effective 02/09/2017 If the recipient organization has submitted a renewal application on or before the date by which a Final-RPPR would be required for the current competitive segment, then submission of an Interim-RPPR via era Commons is required no later than 120 calendar days from the period of performance end date If the renewal application is funded, NIH will treat the Interim-RPPR as the annual performance report for the final year of the previous competitive segment. If the renewal application is not funded, the Interim-RPPR will be treated by NIH staff as the institution's Final-RPPR.
29 NIH Forms NIH transitioned to FORMS-D Grant Application Forms in May 2016 NOT-OD includes a summary of the changes NOT-OD : New NIH "FORMS-E" Grant Application Forms and Instructions Coming for Due Dates On or After January 25, 2018 Consolidation of human subjects, inclusion enrollment, and clinical trial information previously collected across multiple agency forms Expansion and use of discrete form fields for clinical trial information Incorporation of recent Grants.gov changes to R&R Budget and SBIR/STTR Information forms Application guides for FORMS-E application packages will be posted to the How to Apply - Application Guide page no later than 10/25/2017 v COBRE Phase I applications due date: January 24, 2018 v INBRE due date applications due date: March 27, 2018
30 Clinical Trials NOT-OD : Expects all NIH-funded investigators and staff who are involved in the conduct, oversight, or management of clinical trials be trained in Good Clinical Practice (GCP) Effective 01/01/2017 NOT-OD : Expects all NIH-funded clinical trials are registered and that results are submitted to ClinicalTrials.gov whether or not subject to FDAAA Applies to grants, contracts, and intramural clinical trials submitted on or after 01/18/2017 NOT-OD : Single Institutional Review Board for Multi-Site Research. Applicants will be expected to include a plan for the use of an sirb in grant applications submitted to the NIH for due dates on or after 1/25/2018. NOT-OD : All grant applications with plans to conduct clinical trials must be submitted in response to an FOA which specifically states that clinical trials are allowed Effective 01/25/2018 New and reissued FOAs expected to be updated fall 2017
31 Stay Informed Through outreach & education (formal & informal) Through training within your own institution Utilize era Commons New & revised policies announced through NIH GUIDE Notices (sign-up for the listserv; NIGMS Feedback Loop, NIH Extramural Nexus, OER Blog posts) The Notice of Grant Award (reference terms and conditions of award) Through Your GMS! Communicate early and often
32 Questions? NIGMS GAB CRCB Team Christy Leake GAB CRCB Team Leader Specialists IDeA State Assignments Arina Kramer Kentucky, New Hampshire, Oklahoma, South Dakota Brian Iglesias Alaska, Arkansas, North Dakota, Rhode Island Courtney Tardd-Wright Courtney.Tardd- Nebraska, New Mexico, West Virginia, Wyoming Emily Tran Delaware, Kansas, Mississippi, Vermont Lisa Hildred Hawaii, Idaho, Nevada, South Carolina Samantha Farrell Maine, Montana, Louisiana, Puerto Rico
After the Award is Made THEN WHAT?
 After the Award is Made THEN WHAT? George Tucker Chief Grants Management Officer National Institute of Environmental Health Sciences October 2016 TOPICS OF DISCUSSION The ins & outs of the NoA Accessing
After the Award is Made THEN WHAT? George Tucker Chief Grants Management Officer National Institute of Environmental Health Sciences October 2016 TOPICS OF DISCUSSION The ins & outs of the NoA Accessing
Notice of Grant Award (NGA): STANDARD Terms and Conditions
 After the Award is Made Then What? Cheryl Chick Chief Grants Management Officer National Human Genome Research Institute Roger G. Sorensen, Ph.D., MPA Division of Neuroscience and Behavior National Institute
After the Award is Made Then What? Cheryl Chick Chief Grants Management Officer National Human Genome Research Institute Roger G. Sorensen, Ph.D., MPA Division of Neuroscience and Behavior National Institute
October electronic Research Administration (era) OER, OD, National Institutes of Health
 October 2016 electronic Research Administration (era) OER, OD, National Institutes of Health Scarlett Gibb era Customer Relationship Manager, era Commons Joe Schumaker era Communications and Outreach 2
October 2016 electronic Research Administration (era) OER, OD, National Institutes of Health Scarlett Gibb era Customer Relationship Manager, era Commons Joe Schumaker era Communications and Outreach 2
Kathy Hancock, Assistant Grants Compliance Officer, DGCO, OPERA, Office of Extramural Research, NIH, HHS
 Division of Grants Compliance and Oversight (DGCO) Office of Policy for Extramural Research Administration (OPERA), OER National Institutes of Health (NIH), DHHS 19 th Annual SBIR/STTR Conference Milwaukee,
Division of Grants Compliance and Oversight (DGCO) Office of Policy for Extramural Research Administration (OPERA), OER National Institutes of Health (NIH), DHHS 19 th Annual SBIR/STTR Conference Milwaukee,
What Require Prior Approval. Debbie Pettitt 2018 SRA Western/Northeast Section Meeting March 2018
 What Require Prior Approval Debbie Pettitt 2018 SRA Western/Northeast Section Meeting March 2018 1 Discussion Topics Definitions to know NIH Standard Terms of Award Expanded Authorities Prior Approval
What Require Prior Approval Debbie Pettitt 2018 SRA Western/Northeast Section Meeting March 2018 1 Discussion Topics Definitions to know NIH Standard Terms of Award Expanded Authorities Prior Approval
Diane Dean, Director Kathy Hancock, Assistant Grants Compliance Officer Joel Snyderman, Assistant Grants Compliance Officer
 Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar San Diego, CA October 2015 Diane Dean,
Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar San Diego, CA October 2015 Diane Dean,
 3+ 3+ N = 155, 442 3+ R 2 =.32 < < < 3+ N = 149, 685 3+ R 2 =.27 < < < 3+ N = 99, 752 3+ R 2 =.4 < < < 3+ N = 98, 887 3+ R 2 =.6 < < < 3+ N = 52, 624 3+ R 2 =.28 < < < 3+ N = 36, 281 3+ R 2 =.5 < < < 7+
3+ 3+ N = 155, 442 3+ R 2 =.32 < < < 3+ N = 149, 685 3+ R 2 =.27 < < < 3+ N = 99, 752 3+ R 2 =.4 < < < 3+ N = 98, 887 3+ R 2 =.6 < < < 3+ N = 52, 624 3+ R 2 =.28 < < < 3+ N = 36, 281 3+ R 2 =.5 < < < 7+
Presentation. Title. Presented Name. Office of Policy for Extramural Research Administration, OER, NIH. Title. Office
 Presentation Title Michelle Presented G. Bulls, By: Director Name Office of Policy for Extramural Research Administration, OER, NIH Title Office Federal Demonstration Partnership, May 2015 NIH is currently
Presentation Title Michelle Presented G. Bulls, By: Director Name Office of Policy for Extramural Research Administration, OER, NIH Title Office Federal Demonstration Partnership, May 2015 NIH is currently
Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS
 Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar Chicago, IL October 2016 Diane Dean, Director
Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar Chicago, IL October 2016 Diane Dean, Director
The American Legion NATIONAL MEMBERSHIP RECORD
 The American Legion NATIONAL MEMBERSHIP RECORD www.legion.org 2016 The American Legion NATIONAL MEMBERSHIP RECORD 1920-1929 Department 1920 1921 1922 1923 1924 1925 1926 1927 1928 1929 Alabama 4,474 3,246
The American Legion NATIONAL MEMBERSHIP RECORD www.legion.org 2016 The American Legion NATIONAL MEMBERSHIP RECORD 1920-1929 Department 1920 1921 1922 1923 1924 1925 1926 1927 1928 1929 Alabama 4,474 3,246
A Walk Through The SF424 (R&R) Marcia Hahn Office of Policy for Extramural Research Administration, OER, NIH January 11, 2006
 A Walk Through The SF424 (R&R) Marcia Hahn Office of Policy for Extramural Research Administration, OER, NIH January 11, 2006 1 Handouts for This Session A Walk Through The SF424 (R&R) (Powerpoint Presentation
A Walk Through The SF424 (R&R) Marcia Hahn Office of Policy for Extramural Research Administration, OER, NIH January 11, 2006 1 Handouts for This Session A Walk Through The SF424 (R&R) (Powerpoint Presentation
Presentation. Title. Presented Name. Office of Policy for Extramural Research Administration, OER, NIH. Title. Office
 Presentation Title Michelle Presented G. Bulls, By: Director Name Office of Policy for Extramural Research Administration, OER, NIH Title Office Federal Demonstration Partnership, January 2015 NIH is currently
Presentation Title Michelle Presented G. Bulls, By: Director Name Office of Policy for Extramural Research Administration, OER, NIH Title Office Federal Demonstration Partnership, January 2015 NIH is currently
SECTION 1: UPDATES ON 5 YEAR PLAN
 Office of Program Support, Administration on Intellectual and Developmental Disabilities SECTION 1: UPDATES ON 5 YEAR PLAN PROGRAMMATIC CHANGES TO THE UCEDD 5-YEAR PLAN There are no changes to the goals
Office of Program Support, Administration on Intellectual and Developmental Disabilities SECTION 1: UPDATES ON 5 YEAR PLAN PROGRAMMATIC CHANGES TO THE UCEDD 5-YEAR PLAN There are no changes to the goals
Agenda. NIH Update. Other Updates. Proposal & Progress Report Statistics Research Administration Training Topic:
 Agenda NIH Update Salary Cap Guidance NOT OD 17 037: NIH Implementation of the Interim RPPR While a Renewal Application is Under Consideration Prior Approval for No Cost Extensions via the era Commons
Agenda NIH Update Salary Cap Guidance NOT OD 17 037: NIH Implementation of the Interim RPPR While a Renewal Application is Under Consideration Prior Approval for No Cost Extensions via the era Commons
NIH Update. Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH. FDP January 11, 2016
 NIH Update Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH FDP January 11, 2016 1 2 NIH FY 2016 Budget News NIH is currently funded under the Consolidated
NIH Update Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH FDP January 11, 2016 1 2 NIH FY 2016 Budget News NIH is currently funded under the Consolidated
NIH Agency Specifics August 11, 2015
 NIH Agency Specifics August 11, 2015 Maria Skinner Office of Sponsored Programs Stacy Salisbury Contract & Grant Administration NIH Pre-Award Presented by: Maria Skinner Office of Sponsored Programs (OSP)
NIH Agency Specifics August 11, 2015 Maria Skinner Office of Sponsored Programs Stacy Salisbury Contract & Grant Administration NIH Pre-Award Presented by: Maria Skinner Office of Sponsored Programs (OSP)
Interstate Pay Differential
 Interstate Pay Differential APPENDIX IV Adjustments for differences in interstate pay in various locations are computed using the state average weekly pay. This appendix provides a table for the second
Interstate Pay Differential APPENDIX IV Adjustments for differences in interstate pay in various locations are computed using the state average weekly pay. This appendix provides a table for the second
International Researchers: Where to Start
 Fogarty International Center International Researchers: Where to Start Christine Jessup, PhD Program Officer Division of International Training and Research Presentation at 2016 NIH Regional Seminar October
Fogarty International Center International Researchers: Where to Start Christine Jessup, PhD Program Officer Division of International Training and Research Presentation at 2016 NIH Regional Seminar October
National Institutes of Health
 National Institutes of Health NIH NIH s mission is to seek fundamental knowledge about the nature and behavior of living systems and the application of that knowledge to enhance health, lengthen life,
National Institutes of Health NIH NIH s mission is to seek fundamental knowledge about the nature and behavior of living systems and the application of that knowledge to enhance health, lengthen life,
Presentation 2/26/2013. Director, TitleOPERA
 Presentation Title Michelle Presented G. Bulls By: Name Director, TitleOPERA Office OER AASCU Washington Conference February 21, 2013 2 NIH is operating under a continuing resolution. See NOT-OD- 13-018
Presentation Title Michelle Presented G. Bulls By: Name Director, TitleOPERA Office OER AASCU Washington Conference February 21, 2013 2 NIH is operating under a continuing resolution. See NOT-OD- 13-018
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by February 2018 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 Hawaii 2.1 19 Alabama 3.7 33 Ohio 4.5 2 New Hampshire 2.6 19 Missouri 3.7 33 Rhode Island 4.5
States Ranked by February 2018 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 Hawaii 2.1 19 Alabama 3.7 33 Ohio 4.5 2 New Hampshire 2.6 19 Missouri 3.7 33 Rhode Island 4.5
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by November 2015 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.7 19 Indiana 4.4 37 Georgia 5.6 2 Nebraska 2.9 20 Ohio 4.5 37 Tennessee 5.6
States Ranked by November 2015 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.7 19 Indiana 4.4 37 Georgia 5.6 2 Nebraska 2.9 20 Ohio 4.5 37 Tennessee 5.6
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by April 2017 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 Colorado 2.3 17 Virginia 3.8 37 California 4.8 2 Hawaii 2.7 20 Massachusetts 3.9 37 West Virginia
States Ranked by April 2017 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 Colorado 2.3 17 Virginia 3.8 37 California 4.8 2 Hawaii 2.7 20 Massachusetts 3.9 37 West Virginia
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by August 2017 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.3 18 Maryland 3.9 36 New York 4.8 2 Colorado 2.4 18 Michigan 3.9 38 Delaware 4.9
States Ranked by August 2017 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.3 18 Maryland 3.9 36 New York 4.8 2 Colorado 2.4 18 Michigan 3.9 38 Delaware 4.9
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by March 2016 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 South Dakota 2.5 19 Delaware 4.4 37 Georgia 5.5 2 New Hampshire 2.6 19 Massachusetts 4.4 37 North
States Ranked by March 2016 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 South Dakota 2.5 19 Delaware 4.4 37 Georgia 5.5 2 New Hampshire 2.6 19 Massachusetts 4.4 37 North
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by September 2017 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.4 17 Indiana 3.8 36 New Jersey 4.7 2 Colorado 2.5 17 Kansas 3.8 38 Pennsylvania
States Ranked by September 2017 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.4 17 Indiana 3.8 36 New Jersey 4.7 2 Colorado 2.5 17 Kansas 3.8 38 Pennsylvania
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by December 2017 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 Hawaii 2.0 16 South Dakota 3.5 37 Connecticut 4.6 2 New Hampshire 2.6 20 Arkansas 3.7 37 Delaware
States Ranked by December 2017 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 Hawaii 2.0 16 South Dakota 3.5 37 Connecticut 4.6 2 New Hampshire 2.6 20 Arkansas 3.7 37 Delaware
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by September 2015 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.8 17 Oklahoma 4.4 37 South Carolina 5.7 2 Nebraska 2.9 20 Indiana 4.5 37 Tennessee
States Ranked by September 2015 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.8 17 Oklahoma 4.4 37 South Carolina 5.7 2 Nebraska 2.9 20 Indiana 4.5 37 Tennessee
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by November 2014 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.7 19 Pennsylvania 5.1 35 New Mexico 6.4 2 Nebraska 3.1 20 Wisconsin 5.2 38 Connecticut
States Ranked by November 2014 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.7 19 Pennsylvania 5.1 35 New Mexico 6.4 2 Nebraska 3.1 20 Wisconsin 5.2 38 Connecticut
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by July 2018 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 Hawaii 2.1 19 Massachusetts 3.6 37 Kentucky 4.3 2 Iowa 2.6 19 South Carolina 3.6 37 Maryland 4.3
States Ranked by July 2018 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 Hawaii 2.1 19 Massachusetts 3.6 37 Kentucky 4.3 2 Iowa 2.6 19 South Carolina 3.6 37 Maryland 4.3
Department of Defense INSTRUCTION
 Department of Defense INSTRUCTION NUMBER 4715.02 August 28, 2009 Incorporating Change 2, August 31, 2018 USD(A&S) SUBJECT: Regional Environmental Coordination References: (a) DoD Instruction 4715.2, DoD
Department of Defense INSTRUCTION NUMBER 4715.02 August 28, 2009 Incorporating Change 2, August 31, 2018 USD(A&S) SUBJECT: Regional Environmental Coordination References: (a) DoD Instruction 4715.2, DoD
TABLE 3c: Congressional Districts with Number and Percent of Hispanics* Living in Hard-to-Count (HTC) Census Tracts**
 living Alaska 00 47,808 21,213 44.4 Alabama 01 20,661 3,288 15.9 Alabama 02 23,949 6,614 27.6 Alabama 03 20,225 3,247 16.1 Alabama 04 41,412 7,933 19.2 Alabama 05 34,388 11,863 34.5 Alabama 06 34,849 4,074
living Alaska 00 47,808 21,213 44.4 Alabama 01 20,661 3,288 15.9 Alabama 02 23,949 6,614 27.6 Alabama 03 20,225 3,247 16.1 Alabama 04 41,412 7,933 19.2 Alabama 05 34,388 11,863 34.5 Alabama 06 34,849 4,074
Table 6 Medicaid Eligibility Systems for Children, Pregnant Women, Parents, and Expansion Adults, January Share of Determinations
 Table 6 Medicaid Eligibility Systems for Children, Pregnant Women, Parents, and Expansion Adults, January 2017 Able to Make Share of Determinations System determines eligibility for: 2 State Real-Time
Table 6 Medicaid Eligibility Systems for Children, Pregnant Women, Parents, and Expansion Adults, January 2017 Able to Make Share of Determinations System determines eligibility for: 2 State Real-Time
TABLE 3b: Congressional Districts Ranked by Percent of Hispanics* Living in Hard-to- Count (HTC) Census Tracts**
 Rank State District Count (HTC) 1 New York 05 150,499 141,567 94.1 2 New York 08 133,453 109,629 82.1 3 Massachusetts 07 158,518 120,827 76.2 4 Michigan 13 47,921 36,145 75.4 5 Illinois 04 508,677 379,527
Rank State District Count (HTC) 1 New York 05 150,499 141,567 94.1 2 New York 08 133,453 109,629 82.1 3 Massachusetts 07 158,518 120,827 76.2 4 Michigan 13 47,921 36,145 75.4 5 Illinois 04 508,677 379,527
Prior Approval Request What is taking so long?
 Prior Approval Request What is taking so long? Mark Langer National Institute of Arthritis Musculoskeletal and Skin Diseases SOCIETY OF RESEARCH ADMINISTRATORS INTERNATIONAL ANNUAL MEETING VANCOUVER, CANADA
Prior Approval Request What is taking so long? Mark Langer National Institute of Arthritis Musculoskeletal and Skin Diseases SOCIETY OF RESEARCH ADMINISTRATORS INTERNATIONAL ANNUAL MEETING VANCOUVER, CANADA
2017 NIH Update. Presented by Stephanie Smith and Stacey Wade
 2017 NIH Update Presented by Stephanie Smith and Stacey Wade PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs NIH INSTITUTES National
2017 NIH Update Presented by Stephanie Smith and Stacey Wade PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs NIH INSTITUTES National
THINKING OF MOVING: A GUIDE FOR TRANSFERRING AN NIH GRANT TO A NEW INSTITUTION
 THINKING OF MOVING: A GUIDE FOR TRANSFERRING AN NIH GRANT TO A NEW INSTITUTION It is not unusual for NIH funded Principal Investigators (PIs) to move from one institution to another. We fully expect that
THINKING OF MOVING: A GUIDE FOR TRANSFERRING AN NIH GRANT TO A NEW INSTITUTION It is not unusual for NIH funded Principal Investigators (PIs) to move from one institution to another. We fully expect that
5 x 7 Notecards $1.50 with Envelopes - MOQ - 12
 5 x 7 Notecards $1.50 with Envelopes - MOQ - 12 Magnets 2½ 3½ Magnet $1.75 - MOQ - 5 - Add $0.25 for packaging Die Cut Acrylic Magnet $2.00 - MOQ - 24 - Add $0.25 for packaging 2535-22225 California AM-22225
5 x 7 Notecards $1.50 with Envelopes - MOQ - 12 Magnets 2½ 3½ Magnet $1.75 - MOQ - 5 - Add $0.25 for packaging Die Cut Acrylic Magnet $2.00 - MOQ - 24 - Add $0.25 for packaging 2535-22225 California AM-22225
Senior American Access to Care Grant
 Senior American Access to Care Grant Grant Guidelines SENIOR AMERICAN (age 62 plus) ACCESS TO CARE GRANT GUIDELINES: The (ADAF) is committed to supporting U.S. based organizations exempt from taxation
Senior American Access to Care Grant Grant Guidelines SENIOR AMERICAN (age 62 plus) ACCESS TO CARE GRANT GUIDELINES: The (ADAF) is committed to supporting U.S. based organizations exempt from taxation
2016 INCOME EARNED BY STATE INFORMATION
 BY STATE INFORMATION This information is being provided to assist in your 2016 tax preparations. The information is also mailed to applicable Columbia fund non-corporate shareholders with their year-end
BY STATE INFORMATION This information is being provided to assist in your 2016 tax preparations. The information is also mailed to applicable Columbia fund non-corporate shareholders with their year-end
NAFCC Accreditation Annual Update
 NAFCC Accreditation Annual Update 1st year 2nd year First MI Last Co-provider (if applicable) Address on License, Registration or Certificate Phone Fax Mailing Address Email City State Zip County Country
NAFCC Accreditation Annual Update 1st year 2nd year First MI Last Co-provider (if applicable) Address on License, Registration or Certificate Phone Fax Mailing Address Email City State Zip County Country
Current Medicare Advantage Enrollment Penetration: State and County-Level Tabulations
 Current Advantage Enrollment : State and County-Level Tabulations 5 Slide Series, Volume 40 September 2016 Summary of Tabulations and Findings As of September 2016, 17.9 million of the nation s 56.1 million
Current Advantage Enrollment : State and County-Level Tabulations 5 Slide Series, Volume 40 September 2016 Summary of Tabulations and Findings As of September 2016, 17.9 million of the nation s 56.1 million
FY 2014 Per Capita Federal Spending on Major Grant Programs Curtis Smith, Nick Jacobs, and Trinity Tomsic
 Special Analysis 15-03, June 18, 2015 FY 2014 Per Capita Federal Spending on Major Grant Programs Curtis Smith, Nick Jacobs, and Trinity Tomsic 202-624-8577 ttomsic@ffis.org Summary Per capita federal
Special Analysis 15-03, June 18, 2015 FY 2014 Per Capita Federal Spending on Major Grant Programs Curtis Smith, Nick Jacobs, and Trinity Tomsic 202-624-8577 ttomsic@ffis.org Summary Per capita federal
Interacting Electronically with NIH
 Interacting Electronically with NIH Sheri Cummins NIH era Customer Relationship Manager Electronic Submission of Grant Applications Scarlett Gibb NIH era Customer Relationship Manager era Commons Learn
Interacting Electronically with NIH Sheri Cummins NIH era Customer Relationship Manager Electronic Submission of Grant Applications Scarlett Gibb NIH era Customer Relationship Manager era Commons Learn
Federal Funding for Health Insurance Exchanges
 Federal Funding for Health Insurance Exchanges Annie L. Mach Analyst in Health Care Financing C. Stephen Redhead Specialist in Health Policy June 11, 2014 Congressional Research Service 7-5700 www.crs.gov
Federal Funding for Health Insurance Exchanges Annie L. Mach Analyst in Health Care Financing C. Stephen Redhead Specialist in Health Policy June 11, 2014 Congressional Research Service 7-5700 www.crs.gov
Sentinel Event Data. General Information Copyright, The Joint Commission
 Sentinel Event Data General Information 1995 2015 Data Limitations The reporting of most sentinel events to The Joint Commission is voluntary and represents only a small proportion of actual events. Therefore,
Sentinel Event Data General Information 1995 2015 Data Limitations The reporting of most sentinel events to The Joint Commission is voluntary and represents only a small proportion of actual events. Therefore,
Federal Demonstration Partnership May 13, 2013
 Current News From NIH Michelle Bulls Director Office of Policy for Extramural Research Administration Federal Demonstration Partnership May 13, 2013 1 NIH Fiscal Policy for Grant FY 2013 Consolidated and
Current News From NIH Michelle Bulls Director Office of Policy for Extramural Research Administration Federal Demonstration Partnership May 13, 2013 1 NIH Fiscal Policy for Grant FY 2013 Consolidated and
Interstate Turbine Advisory Council (CESA-ITAC)
 Interstate Turbine Advisory Council (CESA-ITAC) Mark Mayhew NYSERDA for Val Stori Clean Energy States Alliance SWAT 4/25/12 Today CESA ITAC, LLC - What, who and why The Unified List - What, why, how and
Interstate Turbine Advisory Council (CESA-ITAC) Mark Mayhew NYSERDA for Val Stori Clean Energy States Alliance SWAT 4/25/12 Today CESA ITAC, LLC - What, who and why The Unified List - What, why, how and
Grant Application Package
 Generates errors for all applications Grant Application Package Opportunity Title: Research Project Grant (Parent R01) Offering Agency: National Institutes of Health Generates errors in certain situations
Generates errors for all applications Grant Application Package Opportunity Title: Research Project Grant (Parent R01) Offering Agency: National Institutes of Health Generates errors in certain situations
Sentinel Event Data. General Information Q Copyright, The Joint Commission
 Sentinel Event Data General Information 1995 2Q 2014 Data Limitations The reporting of most sentinel events to The Joint Commission is voluntary and represents only a small proportion of actual events.
Sentinel Event Data General Information 1995 2Q 2014 Data Limitations The reporting of most sentinel events to The Joint Commission is voluntary and represents only a small proportion of actual events.
Grant/Sponsor Related Systems. Department and OSP Perspectives on ERA
 Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
Table 8 Online and Telephone Medicaid Applications for Children, Pregnant Women, Parents, and Expansion Adults, January 2017
 Table 8 Online and Telephone Medicaid Applications for Children, Pregnant Women, Parents, and Expansion Adults, January 2017 State Applications Can be Submitted Online at the State Level 1 < 25% 25% -
Table 8 Online and Telephone Medicaid Applications for Children, Pregnant Women, Parents, and Expansion Adults, January 2017 State Applications Can be Submitted Online at the State Level 1 < 25% 25% -
Index of religiosity, by state
 Index of religiosity, by state Low Medium High Total United States 19 26 55=100 Alabama 7 16 77 Alaska 28 27 45 Arizona 21 26 53 Arkansas 12 19 70 California 24 27 49 Colorado 24 29 47 Connecticut 25 32
Index of religiosity, by state Low Medium High Total United States 19 26 55=100 Alabama 7 16 77 Alaska 28 27 45 Arizona 21 26 53 Arkansas 12 19 70 California 24 27 49 Colorado 24 29 47 Connecticut 25 32
Preparing for a RPPR Submission
 Preparing for a RPPR Submission Review current NoA Review most current budget and expenditure information. Determine unobligated balance in order to respond to 2590 questions. Review last esnap if applicable
Preparing for a RPPR Submission Review current NoA Review most current budget and expenditure information. Determine unobligated balance in order to respond to 2590 questions. Review last esnap if applicable
FORTIETH TRIENNIAL ASSEMBLY
 FORTIETH TRIENNIAL ASSEMBLY MOST PUISSANT GENERAL GRAND MASTER GENERAL GRAND COUNCIL OF CRYPTIC MASONS INTERNATIONAL 1996-1999 -
FORTIETH TRIENNIAL ASSEMBLY MOST PUISSANT GENERAL GRAND MASTER GENERAL GRAND COUNCIL OF CRYPTIC MASONS INTERNATIONAL 1996-1999 -
NIH Transition to the SF424 (R&R) Application & Electronic Submission Plus A Walk Through The SF424 (R&R)
 NIH Transition to the SF424 (R&R) Application & Electronic Submission Plus A Walk Through The SF424 (R&R) Nancy Saunders, PhD National Institute of Allergy and Infectious Diseases National Institutes of
NIH Transition to the SF424 (R&R) Application & Electronic Submission Plus A Walk Through The SF424 (R&R) Nancy Saunders, PhD National Institute of Allergy and Infectious Diseases National Institutes of
2015 State Hospice Report 2013 Medicare Information 1/1/15
 2015 State Hospice Report 2013 Medicare Information 1/1/15 www.hospiceanalytics.com 2 2013 Demographics & Hospice Utilization National Population 316,022,508 Total Deaths 2,529,792 Medicare Beneficiaries
2015 State Hospice Report 2013 Medicare Information 1/1/15 www.hospiceanalytics.com 2 2013 Demographics & Hospice Utilization National Population 316,022,508 Total Deaths 2,529,792 Medicare Beneficiaries
MAP 1: Seriously Delinquent Rate by State for Q3, 2008
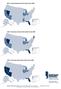 MAP 1: Seriously Delinquent Rate by State for Q3, 2008 Seriously Delinquent Rate Greater than 6.93% 5.18% 6.93% 0 5.17% Source: MBA s National Deliquency Survey MAP 2: Foreclosure Inventory Rate by State
MAP 1: Seriously Delinquent Rate by State for Q3, 2008 Seriously Delinquent Rate Greater than 6.93% 5.18% 6.93% 0 5.17% Source: MBA s National Deliquency Survey MAP 2: Foreclosure Inventory Rate by State
Voter Registration and Absentee Ballot Deadlines by State 2018 General Election: Tuesday, November 6. Saturday, Oct 27 (postal ballot)
 Voter Registration and Absentee Ballot Deadlines by State 2018 General Election: All dates in 2018 unless otherwise noted STATE REG DEADLINE ABSENTEE BALLOT REQUEST DEADLINE Alabama November 1 ABSENTEE
Voter Registration and Absentee Ballot Deadlines by State 2018 General Election: All dates in 2018 unless otherwise noted STATE REG DEADLINE ABSENTEE BALLOT REQUEST DEADLINE Alabama November 1 ABSENTEE
Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed:
 COEUS NIH-GRANTS.GOV PROPOSAL REVIEW CHECKLIST REVIEWER: Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed: Has the Program Announcement been reviewed? What is
COEUS NIH-GRANTS.GOV PROPOSAL REVIEW CHECKLIST REVIEWER: Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed: Has the Program Announcement been reviewed? What is
Rutgers Revenue Sources
 Rutgers Revenue Sources 31.2% Tuition and Fees 27.3% State Appropriations with Fringes 1.0% Endowment and Investments.5% Federal Appropriations 17.8% Federal, State, and Municipal Grants and Contracts
Rutgers Revenue Sources 31.2% Tuition and Fees 27.3% State Appropriations with Fringes 1.0% Endowment and Investments.5% Federal Appropriations 17.8% Federal, State, and Municipal Grants and Contracts
Estimated Economic Impacts of the Small Business Jobs and Tax Relief Act National Report
 Regional Economic Models, Inc. Estimated Economic Impacts of the Small Business Jobs and Tax Relief Act National Report Prepared by Frederick Treyz, CEO June 2012 The following is a summary of the Estimated
Regional Economic Models, Inc. Estimated Economic Impacts of the Small Business Jobs and Tax Relief Act National Report Prepared by Frederick Treyz, CEO June 2012 The following is a summary of the Estimated
National Collegiate Soils Contest Rules
 National Collegiate Soils Contest Rules Students of Agronomy, Soils, and Environmental Sciences (SASES) Revised September 30, 2008 I. NAME The contest shall be known as the National Collegiate Soils Contest
National Collegiate Soils Contest Rules Students of Agronomy, Soils, and Environmental Sciences (SASES) Revised September 30, 2008 I. NAME The contest shall be known as the National Collegiate Soils Contest
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018
 CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
Supplemental Nutrition Assistance Program. STATE ACTIVITY REPORT Fiscal Year 2016
 Supplemental Nutrition Assistance Program ACTIVITY REPORT Fiscal Year 2016 Food and Nutrition Service Supplemental Nutrition Assistance Program Program Accountability and Administration Division September
Supplemental Nutrition Assistance Program ACTIVITY REPORT Fiscal Year 2016 Food and Nutrition Service Supplemental Nutrition Assistance Program Program Accountability and Administration Division September
Grants 101: An Introduction to Federal Grants for State and Local Governments
 Grants 101: An Introduction to Federal Grants for State and Local Governments Introduction FFIS has been in the federal grant reporting business for a long time about 30 years. The main thing we ve learned
Grants 101: An Introduction to Federal Grants for State and Local Governments Introduction FFIS has been in the federal grant reporting business for a long time about 30 years. The main thing we ve learned
STATE INDUSTRY ASSOCIATIONS $ - LISTED NEXT PAGE. TOTAL $ 88,000 * for each contribution of $500 for Board Meeting sponsorship
 Exhibit D -- TRIP 2017 FUNDING SOURCES -- February 3, 2017 CORPORATE $ 12,000 Construction Companies $ 5,500 Consulting Engineers Equipment Distributors Manufacturer/Supplier/Producer 6,500 Surety Bond
Exhibit D -- TRIP 2017 FUNDING SOURCES -- February 3, 2017 CORPORATE $ 12,000 Construction Companies $ 5,500 Consulting Engineers Equipment Distributors Manufacturer/Supplier/Producer 6,500 Surety Bond
UTHSCSA LAB TRANSFER INFORMATION
 UTHSCSA LAB TRANSFER INFORMATION The information provided below is a list of offices and contacts that are needed for various approvals to move a lab to UTHSCSA. Animal Transfer to UTHSCSA http://research.uthscsa.edu/lar/
UTHSCSA LAB TRANSFER INFORMATION The information provided below is a list of offices and contacts that are needed for various approvals to move a lab to UTHSCSA. Animal Transfer to UTHSCSA http://research.uthscsa.edu/lar/
Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32)
 Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32) Overview: This document is intended to help you complete a KC System to System (S2S) proposal submission
Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32) Overview: This document is intended to help you complete a KC System to System (S2S) proposal submission
National Institute of Health (NIH)
 National Institute of Health (NIH) Funding Presented by: Samantha J. Taylor Senior Research Officer Wyss Institute for Biologically Inspired Engineering, Harvard University What is NIH s primary mission?
National Institute of Health (NIH) Funding Presented by: Samantha J. Taylor Senior Research Officer Wyss Institute for Biologically Inspired Engineering, Harvard University What is NIH s primary mission?
national assembly of state arts agencies
 STATE ARTS AGENCY GRANT MAKING AND FUNDING Each of America's 50 states and six jurisdictions has a government that works to make the cultural, civic, economic and educational benefits of the available
STATE ARTS AGENCY GRANT MAKING AND FUNDING Each of America's 50 states and six jurisdictions has a government that works to make the cultural, civic, economic and educational benefits of the available
HOME HEALTH AIDE TRAINING REQUIREMENTS, DECEMBER 2016
 BACKGROUND HOME HEALTH AIDE TRAINING REQUIREMENTS, DECEMBER 2016 Federal legislation (42 CFR 484.36) requires that Medicare-certified home health agencies employ home health aides who are trained and evaluated
BACKGROUND HOME HEALTH AIDE TRAINING REQUIREMENTS, DECEMBER 2016 Federal legislation (42 CFR 484.36) requires that Medicare-certified home health agencies employ home health aides who are trained and evaluated
NMLS Mortgage Industry Report 2016 Q1 Update
 NMLS Mortgage Industry Report 2016 Q1 Update Released June 10, 2016 Conference of State Bank Supervisors 1129 20 th Street, NW, 9 th Floor Washington, D.C. 20036-4307 NMLS Mortgage Industry Report: 2016Q1
NMLS Mortgage Industry Report 2016 Q1 Update Released June 10, 2016 Conference of State Bank Supervisors 1129 20 th Street, NW, 9 th Floor Washington, D.C. 20036-4307 NMLS Mortgage Industry Report: 2016Q1
U.S. Army Civilian Personnel Evaluation Agency
 Army Regulation 10 89 Organizations and Functions U.S. Army Civilian Personnel Evaluation Agency Headquarters Department of the Army Washington, DC 15 December 1989 Unclassified SUMMARY of CHANGE AR 10
Army Regulation 10 89 Organizations and Functions U.S. Army Civilian Personnel Evaluation Agency Headquarters Department of the Army Washington, DC 15 December 1989 Unclassified SUMMARY of CHANGE AR 10
NMLS Mortgage Industry Report 2017Q2 Update
 NMLS Mortgage Industry Report 2017Q2 Update Released September 18, 2017 Conference of State Bank Supervisors 1129 20 th Street, NW, 9 th Floor Washington, D.C. 20036-4307 NMLS Mortgage Industry Report:
NMLS Mortgage Industry Report 2017Q2 Update Released September 18, 2017 Conference of State Bank Supervisors 1129 20 th Street, NW, 9 th Floor Washington, D.C. 20036-4307 NMLS Mortgage Industry Report:
Utilizing Grants to Achieve Your Farm Objectives
 Utilizing Grants to Achieve Your Farm Objectives Ohio Ecological Food and Farm Association Annual Conference- Granville, OH February 13, 2010 Mike Hogan Extension Educator & Associate Professor Sustainable
Utilizing Grants to Achieve Your Farm Objectives Ohio Ecological Food and Farm Association Annual Conference- Granville, OH February 13, 2010 Mike Hogan Extension Educator & Associate Professor Sustainable
NMLS Mortgage Industry Report 2017Q4 Update
 NMLS Mortgage Industry Report 2017Q4 Update Released March 9, 2018 Conference of State Bank Supervisors 1129 20 th Street, NW, 9 th Floor Washington, D.C. 20036-4307 NMLS Mortgage Industry Report: 2017Q4
NMLS Mortgage Industry Report 2017Q4 Update Released March 9, 2018 Conference of State Bank Supervisors 1129 20 th Street, NW, 9 th Floor Washington, D.C. 20036-4307 NMLS Mortgage Industry Report: 2017Q4
NMLS Mortgage Industry Report 2018Q1 Update
 NMLS Mortgage Industry Report 2018Q1 Update Released July 5, 2018 Conference of State Bank Supervisors 1129 20 th Street, NW, 9 th Floor Washington, D.C. 20036-4307 NMLS Mortgage Industry Report: 2018Q1
NMLS Mortgage Industry Report 2018Q1 Update Released July 5, 2018 Conference of State Bank Supervisors 1129 20 th Street, NW, 9 th Floor Washington, D.C. 20036-4307 NMLS Mortgage Industry Report: 2018Q1
States Ranked by Annual Nonagricultural Employment Change October 2017, Seasonally Adjusted
 States Ranked by Annual Nonagricultural Employment Change Change (Jobs) Change (Jobs) Change (Jobs) 1 Texas 316,100 19 Nevada 36,600 37 Hawaii 7,100 2 California 256,800 20 Tennessee 34,800 38 Mississippi
States Ranked by Annual Nonagricultural Employment Change Change (Jobs) Change (Jobs) Change (Jobs) 1 Texas 316,100 19 Nevada 36,600 37 Hawaii 7,100 2 California 256,800 20 Tennessee 34,800 38 Mississippi
Statutory change to name availability standard. Jurisdiction. Date: April 8, [Statutory change to name availability standard] [April 8, 2015]
![Statutory change to name availability standard. Jurisdiction. Date: April 8, [Statutory change to name availability standard] [April 8, 2015] Statutory change to name availability standard. Jurisdiction. Date: April 8, [Statutory change to name availability standard] [April 8, 2015]](/thumbs/83/87461560.jpg) Topic: Question by: : Statutory change to name availability standard Michael Powell Texas Date: April 8, 2015 Manitoba Corporations Canada Alabama Alaska Arizona Arkansas California Colorado Connecticut
Topic: Question by: : Statutory change to name availability standard Michael Powell Texas Date: April 8, 2015 Manitoba Corporations Canada Alabama Alaska Arizona Arkansas California Colorado Connecticut
2016 NIH Update. Presented by Stephanie Smith, Stacey Wade, and Jennifer Webster
 2016 NIH Update Presented by Stephanie Smith, Stacey Wade, and Jennifer Webster PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs Jennifer
2016 NIH Update Presented by Stephanie Smith, Stacey Wade, and Jennifer Webster PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs Jennifer
November 24, First Street NE, Suite 510 Washington, DC 20002
 820 First Street NE, Suite 510 Washington, DC 20002 Tel: 202-408-1080 Fax: 202-408-1056 center@cbpp.org www.cbpp.org November 24, 2008 TANF BENEFITS ARE LOW AND HAVE NOT KEPT PACE WITH INFLATION But Most
820 First Street NE, Suite 510 Washington, DC 20002 Tel: 202-408-1080 Fax: 202-408-1056 center@cbpp.org www.cbpp.org November 24, 2008 TANF BENEFITS ARE LOW AND HAVE NOT KEPT PACE WITH INFLATION But Most
STATUTORY/REGULATORY NURSE ANESTHETIST RECOGNITION
 Alabama NPA and SBON R&R CRNAs are a type of advanced practice nurse. Advanced practice nurses are "certified by the Board of Nursing to engage in the practice of advanced practice nursing." [Alabama Nurse
Alabama NPA and SBON R&R CRNAs are a type of advanced practice nurse. Advanced practice nurses are "certified by the Board of Nursing to engage in the practice of advanced practice nursing." [Alabama Nurse
Pipeline Safety Regulations and the Effects on Operator Qualification Programs. March 28, 2017
 Pipeline Safety Regulations and the Effects on Operator Qualification Programs March 28, 2017 Community Assistance and Technical Services (CATS) Name Change Community Liaison (CL) Effective: January 1,
Pipeline Safety Regulations and the Effects on Operator Qualification Programs March 28, 2017 Community Assistance and Technical Services (CATS) Name Change Community Liaison (CL) Effective: January 1,
TENNESSEE TEXAS UTAH VERMONT VIRGINIA WASHINGTON WEST VIRGINIA WISCONSIN WYOMING ALABAMA ALASKA ARIZONA ARKANSAS
 ALABAMA ALASKA ARIZONA ARKANSAS CALIFORNIA COLORADO CONNECTICUT DELAWARE DISTRICT OF COLUMBIA FLORIDA GEORGIA GUAM MISSOURI MONTANA NEBRASKA NEVADA NEW HAMPSHIRE NEW JERSEY NEW MEXICO NEW YORK NORTH CAROLINA
ALABAMA ALASKA ARIZONA ARKANSAS CALIFORNIA COLORADO CONNECTICUT DELAWARE DISTRICT OF COLUMBIA FLORIDA GEORGIA GUAM MISSOURI MONTANA NEBRASKA NEVADA NEW HAMPSHIRE NEW JERSEY NEW MEXICO NEW YORK NORTH CAROLINA
Child & Adult Care Food Program: Participation Trends 2017
 Child & Adult Care Food Program: Participation Trends 2017 February 2018 About FRAC The Food Research and Action Center (FRAC) is the leading national organization working for more effective public and
Child & Adult Care Food Program: Participation Trends 2017 February 2018 About FRAC The Food Research and Action Center (FRAC) is the leading national organization working for more effective public and
Child & Adult Care Food Program: Participation Trends 2016
 Child & Adult Care Food Program: Participation Trends 2016 March 2017 About FRAC The Food Research and Action Center (FRAC) is the leading national organization working for more effective public and private
Child & Adult Care Food Program: Participation Trends 2016 March 2017 About FRAC The Food Research and Action Center (FRAC) is the leading national organization working for more effective public and private
*ALWAYS KEEP A COPY OF THE CERTIFICATE OF ATTENDANCE FOR YOUR RECORDS IN CASE OF AUDIT
 State Alabama Alaska Arizona Arkansas California INSTRUCTIONS FOR CLE ATTENDANCE REPORTING AT IADC 2012 TRIAL ACADEMY Attorney Reporting Method After the CLE activity, fill out the Certificate of Attendance
State Alabama Alaska Arizona Arkansas California INSTRUCTIONS FOR CLE ATTENDANCE REPORTING AT IADC 2012 TRIAL ACADEMY Attorney Reporting Method After the CLE activity, fill out the Certificate of Attendance
OPT OPTIONAL PRACTICAL TRAINING
 OPT OPTIONAL PRACTICAL TRAINING GUIDELINES FOR STUDENT COMPLETION PROCEDURE MAILING INFORMATION ATTACHED: I-765 FORM OPT APPLICATION CHECKLIST Check off items as you complete them. OPT application packet
OPT OPTIONAL PRACTICAL TRAINING GUIDELINES FOR STUDENT COMPLETION PROCEDURE MAILING INFORMATION ATTACHED: I-765 FORM OPT APPLICATION CHECKLIST Check off items as you complete them. OPT application packet
STATE ARTS AGENCY GRANT MAKING AND FUNDING
 STATE ARTS AGENCY GRANT MAKING AND FUNDING Each of America's 50 states and six jurisdictions has a government that works to make the cultural, civic, economic and educational benefits of the available
STATE ARTS AGENCY GRANT MAKING AND FUNDING Each of America's 50 states and six jurisdictions has a government that works to make the cultural, civic, economic and educational benefits of the available
NIH Proposal Outline Twelve Page Limit For Activity Codes R01, R10, R15, R18, R21/R33, R24, R33, R34, DP3, G08, G11, G13, SC1, X01
 NIH Proposal Outline Twelve Page Limit For Activity Codes R01, R10, R15, R18, R21/R33, R24, R33, R34, DP3, G08, G11, G13, SC1, X01 1. Introduction to Application (for Resubmission or Revision applications
NIH Proposal Outline Twelve Page Limit For Activity Codes R01, R10, R15, R18, R21/R33, R24, R33, R34, DP3, G08, G11, G13, SC1, X01 1. Introduction to Application (for Resubmission or Revision applications
Fiscal Year 1999 Comparisons. State by State Rankings of Revenues and Spending. Includes Fiscal Year 2000 Rankings for State Taxes Only
 Fiscal Year 1999 Comparisons State by State Rankings of Revenues and Spending Includes Fiscal Year 2000 Rankings for State Taxes Only January 2002 1 2 published annually by: The Minnesota Taxpayers Association
Fiscal Year 1999 Comparisons State by State Rankings of Revenues and Spending Includes Fiscal Year 2000 Rankings for State Taxes Only January 2002 1 2 published annually by: The Minnesota Taxpayers Association
Introduction. Current Law Distribution of Funds. MEMORANDUM May 8, Subject:
 MEMORANDUM May 8, 2018 Subject: TANF Family Assistance Grant Allocations Under the Ways and Means Committee (Majority) Proposal From: Gene Falk, Specialist in Social Policy, gfalk@crs.loc.gov, 7-7344 Jameson
MEMORANDUM May 8, 2018 Subject: TANF Family Assistance Grant Allocations Under the Ways and Means Committee (Majority) Proposal From: Gene Falk, Specialist in Social Policy, gfalk@crs.loc.gov, 7-7344 Jameson
NIH Update FDP September 2009 Office of Policy for Extramural Research Administration
 NIH Update FDP September 2009 Office of Policy for Extramural Research Administration OPERA New NIH Director Francis S. Collins, M.D., Ph.D., was officially sworn in on Monday, August 17, 2009 as the 16th
NIH Update FDP September 2009 Office of Policy for Extramural Research Administration OPERA New NIH Director Francis S. Collins, M.D., Ph.D., was officially sworn in on Monday, August 17, 2009 as the 16th
PRESS RELEASE Media Contact: Joseph Stefko, Director of Public Finance, ;
 PRESS RELEASE Media Contact: Joseph Stefko, Director of Public Finance, 585.327.7075; jstefko@cgr.org Highest Paid State Workers in New Jersey & New York in 2010; Lowest Paid in Dakotas and West Virginia
PRESS RELEASE Media Contact: Joseph Stefko, Director of Public Finance, 585.327.7075; jstefko@cgr.org Highest Paid State Workers in New Jersey & New York in 2010; Lowest Paid in Dakotas and West Virginia
Policy and Compliance: Working Together Like Hand in Glove
 Policy and Compliance: Working Together Like Hand in Glove Samuel Ashe, Director, Division of Grants Policy, OPERA, OER Diane W. Dean, Director, Division of Grants Compliance and Oversight, OPERA, OER
Policy and Compliance: Working Together Like Hand in Glove Samuel Ashe, Director, Division of Grants Policy, OPERA, OER Diane W. Dean, Director, Division of Grants Compliance and Oversight, OPERA, OER
Grant Application Package
 Grant Application Package Generates errors for all applications Generates errors in certain situations Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition
Grant Application Package Generates errors for all applications Generates errors in certain situations Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition
Page 1 of 10 1/31/2008
 Page 1 of 10 1/31/2008 Step by Step Tips to Avoid Errors and Warnings o Full instructions can be found at: http://grants1.nih.gov/grants/funding/424/index.htm. o This document is intended to assist you
Page 1 of 10 1/31/2008 Step by Step Tips to Avoid Errors and Warnings o Full instructions can be found at: http://grants1.nih.gov/grants/funding/424/index.htm. o This document is intended to assist you
Critical Access Hospitals and HCAHPS
 Critical Access Hospitals and HCAHPS Michelle Casey, MS Senior Research Fellow and Deputy Director University of Minnesota Rural Health Research Center June 12, 2012 Overview of Presentation Why is HCAHPS
Critical Access Hospitals and HCAHPS Michelle Casey, MS Senior Research Fellow and Deputy Director University of Minnesota Rural Health Research Center June 12, 2012 Overview of Presentation Why is HCAHPS
Rankings of the States 2017 and Estimates of School Statistics 2018
 Rankings of the States 2017 and Estimates of School Statistics 2018 NEA RESEARCH April 2018 Reproduction: No part of this report may be reproduced in any form without permission from NEA Research, except
Rankings of the States 2017 and Estimates of School Statistics 2018 NEA RESEARCH April 2018 Reproduction: No part of this report may be reproduced in any form without permission from NEA Research, except
