Patti Manheim - Director February 13, 2014
|
|
|
- Dominic Evans
- 6 years ago
- Views:
Transcription
1 Patti Manheim - Director February 13, 2014
2 Today s Topics NIH Fiscal Operations for FY 2014 Updates from the January 2014 FDP Meeting Other OCGA Updates
3 NIH Fiscal Operations for FY2014 NIH Budget $30.15 billon Increase of $1 billion over FY2013 FY2014 Non-competing continuations Awards already issued at reduced levels, may be partially or fully restored, depending on the institute/center Awards to be issued may be reduced up to 3% below previously committed levels Out year commitments for FY2015 and beyond remain unchanged Institutes and Centers will be posting their fiscal policies See NOT-OD
4 NIH Fiscal Operations for FY2014 FY2014 NRSA Awards Stipend levels increased by 2% 2014 awards that were issued at 2012 level will be revised to adjust stipends to 2014 levels Appointments made at 2012 levels will need to be amended in xtrain once the award adjustment is made See NOT-OD and NOT-OD
5 Federal Demonstration Partnership (FDP) Updates Federal Agency Updates NIH Still operating under continuing resolution. Salary cap increase, effective 1/12/14 For FY14 (October 1, 2014 September 30, 2015) Salary earned: 10/1/13 1/11/14 capped at $179,700 Salary earned: 1/12/14 9/30/14 capped at $181,500 See NOT-OD
6 Federal Demonstration Partnership (FDP) Updates Federal Agency Updates NIH Payment Management System Subaccounts: FY14 Transition of all awards with new document numbers (Types 1, 2, 4, 6, 7, and 9) FY15 transition of continuing awards (Types 5 & 8) Administrative supplements will be awarded to the same type of account (subaccount or pooled) as the parent award Carryover authority will not change
7 Federal Demonstration Partnership (FDP) Updates Federal Agency Updates NIH Grants Policy Statement revised 10/01/13 Does not introduce any new material. Clarifies certain policies. See NOT-OD New HHS Closeout Requirements Proposed implementation date: 10/01/14 No change to 90 day requirement to submit closeout documents New Revised financial reports no later than 6 months from due date of original report 270 requirement for agency to complete entire closeout process Stay Tuned for Further Updates!
8 Federal Demonstration Partnership (FDP) Updates Federal Agency Updates NIH RPPR for non-snap Awards: Pilot ended February 1, 2014 Anticipate required use for all grantees August 1, 2014 RPPR for Multi-Year Funded Awards: Required for progress reports submitted on/after Janaury 31, 2014 See NOT-OD
9 Federal Demonstration Partnership (FDP) Updates Federal Agency Updates NIH Effective with due dates on/after January 25, 2014 F, K, T, and D applications require use of Adobe Forms C. If prepared in S2S Grants using Adobe Forms B, use Copy/Transform functionality
10 Federal Demonstration Partnership (FDP) Updates Federal Agency Updates NSF New Proposal and Awards Prolicies and Procedures Guide (PAPPG) Released November 2013 Effective for proposals submitted/due on/after February 24, 2014 Changes include: Project Summary Formatting Participant Support Guidance on Visa Costs Conferences, Symposia and Workshops Project Reporting Not Cumulative
11 Federal Demonstration Partnership (FDP) Updates Federal Agency Updates NASA New Proposer s Guidebook due for release January 31, 2014* debook/ *still waiting
12 Federal Demonstration Partnership (FDP) Updates Meeting Agendas and Presentations available at: _ UCLA FDP participants: Kim Duiker Sharon Lam Cindy Gilbert
13 EPASS Section 2
14 EPASS Section 6
15 NIH Modular Budgets OCGA will not ask for, look for, or review detailed budgets for NIH modular applications. While detailed budgets are not required by NIH they are important to have when calculating F&A cost base, writing justifications, and for audit purposes PIs/Departments are required to ensure that all costs charged are in accordance with applicable cost principles NIH Grants Policy Statement legislatively imposed restrictions (e.g., salary cap)
16 Why did OCGA Deny my RAS? Requests for Authorization to Spend (RAS) are processed in accordance with UCLA Policy 920 AVC Marcia Smith is currently reviewing the policy
17 Why did OCGA Deny my RAS? The same issues that prevent OCGA from accepting an award will prevent OCGA from approving a RAS Human/animal subjects without IRB/ARC approval COI disclosure and/or reporting requirements not met (EDGE, 740, 700) Sponsor imposed restrictions on spending prior to full execution of agreement No Coverage Analysis Missing/Incomplete EPASS Missing PI Exception Lack of Vital Interest Waiver
18 Staffing Updates Records Management Yushan Lin - Specialist Proposal Intake Jennifer Caldejon Analyst Gerald Gamble Specialist Magaly Chavez Specialist Award Intake Jaimie To Specialist Outgoing Subawards Elizabeth Serrano Work Study
19 Staffing Updates Grants and Cooperative Agreements EnJunaya Canton Senior Analyst Ghazaleh Nourparvar Specialist Jenelle Bossett Specialist Subawards and Contracts Kristin Lund Officer Johnny Kidd Officer (beginning March 3) Jeffrey Alonzo - Specialist
20
Today s Topics. OCGA Quick Updates Kim Duiker and Heather Winters Updates from NIH Regional Seminar Kim Duiker S2S Grants Updates Cindy Gilbert
 August 8, 2013 Today s Topics OCGA Quick Updates Kim Duiker and Heather Winters Updates from NIH Regional Seminar Kim Duiker S2S Grants Updates Cindy Gilbert Quick Updates OCGA Staffing Update Three retirements
August 8, 2013 Today s Topics OCGA Quick Updates Kim Duiker and Heather Winters Updates from NIH Regional Seminar Kim Duiker S2S Grants Updates Cindy Gilbert Quick Updates OCGA Staffing Update Three retirements
Research Administrators Forum September 13, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum September 13, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith OCGA Initiatives Patti Manheim, Kim Duiker Proposal Intake
Research Administrators Forum September 13, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith OCGA Initiatives Patti Manheim, Kim Duiker Proposal Intake
Research Administration Forum March 9, Marcia Smith Associate Vice Chancellor for Research
 Research Administration Forum March 9, 2017 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements - Marcia Smith Immigration Update and Q & A Sam Nahidi, Assistant Director,
Research Administration Forum March 9, 2017 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements - Marcia Smith Immigration Update and Q & A Sam Nahidi, Assistant Director,
Federal Demonstration Partnership May 13, 2013
 Current News From NIH Michelle Bulls Director Office of Policy for Extramural Research Administration Federal Demonstration Partnership May 13, 2013 1 NIH Fiscal Policy for Grant FY 2013 Consolidated and
Current News From NIH Michelle Bulls Director Office of Policy for Extramural Research Administration Federal Demonstration Partnership May 13, 2013 1 NIH Fiscal Policy for Grant FY 2013 Consolidated and
Research Administrators Forum October 10th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum October 10th, 2013 Marcia Smith Associate Vice Chancellor for Research Agenda Meeting Agenda Welcome and Announcements Marcia Smith, Office of Research Administration Contract
Research Administrators Forum October 10th, 2013 Marcia Smith Associate Vice Chancellor for Research Agenda Meeting Agenda Welcome and Announcements Marcia Smith, Office of Research Administration Contract
Please fill out the survey forms 2/9/2012. Upcoming ORA Training Opportunities. Agenda
 Agenda Welcome and Announcements Marcia Smith Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Data Management Tool Sharon Farb, Associate University Librarian,
Agenda Welcome and Announcements Marcia Smith Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Data Management Tool Sharon Farb, Associate University Librarian,
Research Administrators Forum May 8th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum May 8th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Office of Research Administration OHRPP Retention of Research
Research Administrators Forum May 8th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Office of Research Administration OHRPP Retention of Research
Presentation. Title. Presented Name. Office of Policy for Extramural Research Administration, OER, NIH. Title. Office
 Presentation Title Michelle Presented G. Bulls, By: Director Name Office of Policy for Extramural Research Administration, OER, NIH Title Office Federal Demonstration Partnership, January 2015 NIH is currently
Presentation Title Michelle Presented G. Bulls, By: Director Name Office of Policy for Extramural Research Administration, OER, NIH Title Office Federal Demonstration Partnership, January 2015 NIH is currently
Presentation. Title. Presented Name. Office of Policy for Extramural Research Administration, OER, NIH. Title. Office
 Presentation Title Michelle Presented G. Bulls, By: Director Name Office of Policy for Extramural Research Administration, OER, NIH Title Office Federal Demonstration Partnership, May 2015 NIH is currently
Presentation Title Michelle Presented G. Bulls, By: Director Name Office of Policy for Extramural Research Administration, OER, NIH Title Office Federal Demonstration Partnership, May 2015 NIH is currently
Research Administrators Forum August 14th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum August 14th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Associate Vice Chancellor, Office of Research Administration
Research Administrators Forum August 14th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Associate Vice Chancellor, Office of Research Administration
Office of Sponsored Projects 5/31/ Volume 2, Issue 5
 The OSP News & Updates, published by the Office of Sponsored Projects, is a bi-weekly subscription-based newsletter that provides OSP and sponsor updates and reminders, quick facts, guidance and training
The OSP News & Updates, published by the Office of Sponsored Projects, is a bi-weekly subscription-based newsletter that provides OSP and sponsor updates and reminders, quick facts, guidance and training
NIH Update. Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH. FDP January 11, 2016
 NIH Update Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH FDP January 11, 2016 1 2 NIH FY 2016 Budget News NIH is currently funded under the Consolidated
NIH Update Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH FDP January 11, 2016 1 2 NIH FY 2016 Budget News NIH is currently funded under the Consolidated
Patti Manheim, Director April 11, 2013
 Patti Manheim, Director April 11, 2013 Today s Topics NIH Research Performance Project Report (RPPR) Update Patti Manheim NIH Public Access Policy Reporting Requirements Patti Manheim and Lisa Federer,
Patti Manheim, Director April 11, 2013 Today s Topics NIH Research Performance Project Report (RPPR) Update Patti Manheim NIH Public Access Policy Reporting Requirements Patti Manheim and Lisa Federer,
Marcia Smith Associate Vice Chancellor for Research
 12-11-2014 Marcia Smith Associate Vice Chancellor for Research Agenda Research Administrator Forum December 11, 2014 Welcome and Announcements - Marcia Smith UCLA Implementation of: Uniform Administrative
12-11-2014 Marcia Smith Associate Vice Chancellor for Research Agenda Research Administrator Forum December 11, 2014 Welcome and Announcements - Marcia Smith UCLA Implementation of: Uniform Administrative
Research Administrators Forum August 8th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum August 8th, 2013 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Office of Research Administration Conflicts of Interest
Research Administrators Forum August 8th, 2013 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Office of Research Administration Conflicts of Interest
Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith Data Management Tool Sharon Farb, Associate University Librarian,
Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith Data Management Tool Sharon Farb, Associate University Librarian,
Cindy Gilbert Assistant Director January 14, 2016
 Cindy Gilbert Assistant Director cgilbert@research.ucla.edu January 14, 2016 OCGA era Help Email S2S Grants Help (s2sgrants@research.ucla.edu) retired Replaced by OCGA era Help (erahelp@research.ucla.edu)
Cindy Gilbert Assistant Director cgilbert@research.ucla.edu January 14, 2016 OCGA era Help Email S2S Grants Help (s2sgrants@research.ucla.edu) retired Replaced by OCGA era Help (erahelp@research.ucla.edu)
Sponsored Programs New Developments and Important Reminders
 Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs March 7, 2012 1 Training Session Overview National Institutes of Health (NIH) Update NSF Proposal
Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs March 7, 2012 1 Training Session Overview National Institutes of Health (NIH) Update NSF Proposal
Updates on American Recovery and Reinvestment Act (ARRA) Funding. Virginia Anders, Acting Director, OCGA Evelyn Balabis, Director, EFM
 Updates on American Recovery and Reinvestment Act (ARRA) Funding Virginia Anders, Acting Director, OCGA Evelyn Balabis, Director, EFM 15 OCGA ARRA Help Desk 10:00 a.m. 5:00 p.m., Monday Friday 310-794-0548
Updates on American Recovery and Reinvestment Act (ARRA) Funding Virginia Anders, Acting Director, OCGA Evelyn Balabis, Director, EFM 15 OCGA ARRA Help Desk 10:00 a.m. 5:00 p.m., Monday Friday 310-794-0548
Sponsored by Office of Research Administration
 Research Administrators Forum Sponsored by Office of Research Administration Marcia Smith Associate Vice Chancellor Research Administration January 14, 2010 Meeting Agenda Meeting Agenda Welcome and Announcements
Research Administrators Forum Sponsored by Office of Research Administration Marcia Smith Associate Vice Chancellor Research Administration January 14, 2010 Meeting Agenda Meeting Agenda Welcome and Announcements
Federal Demonstration Partnership, NIH Updates, & Grants/Cooperative Agreement Reminders November 13, 2015
 Federal Demonstration Partnership, NIH Updates, & Grants/Cooperative Agreement Reminders November 13, 2015 Federal Demonstration Partnership BACKGROUND 154 participating institutions Goal: Improve Government
Federal Demonstration Partnership, NIH Updates, & Grants/Cooperative Agreement Reminders November 13, 2015 Federal Demonstration Partnership BACKGROUND 154 participating institutions Goal: Improve Government
What You Need to Know When Submitting an NIH SF424 R&R Grant Application Through the UCLA Office of Contract and Grant Administration (OCGA)
 What You Need to Know When Submitting an NIH SF424 R&R Grant Application Through the UCLA Office of Contract and Grant Administration (OCGA) Kim Duiker, Assistant Director Mary Haskins, Contract & Grant
What You Need to Know When Submitting an NIH SF424 R&R Grant Application Through the UCLA Office of Contract and Grant Administration (OCGA) Kim Duiker, Assistant Director Mary Haskins, Contract & Grant
Presentation 2/26/2013. Director, TitleOPERA
 Presentation Title Michelle Presented G. Bulls By: Name Director, TitleOPERA Office OER AASCU Washington Conference February 21, 2013 2 NIH is operating under a continuing resolution. See NOT-OD- 13-018
Presentation Title Michelle Presented G. Bulls By: Name Director, TitleOPERA Office OER AASCU Washington Conference February 21, 2013 2 NIH is operating under a continuing resolution. See NOT-OD- 13-018
Patti Manheim, Director, OCGA
 Research Administrators Forum Patti Manheim, Director, OCGA Agenda Welcome - Patti Manheim OCGA Updates Patti Manheim * Consortium/contractual arrangements versus purchase of services * Master Calendar
Research Administrators Forum Patti Manheim, Director, OCGA Agenda Welcome - Patti Manheim OCGA Updates Patti Manheim * Consortium/contractual arrangements versus purchase of services * Master Calendar
Gina Billiot, CRA Grant/Contract Specialist 2 (ERA) Office of Sponsored Programs (OSP)
 Gina Billiot, CRA Grant/Contract Specialist 2 (ERA) Office of Sponsored Programs (OSP) SPS - System Features SPS - System Overview Workspace Proposal Creation Demonstration Resources LOUISIANA STATE UNIVERSITY
Gina Billiot, CRA Grant/Contract Specialist 2 (ERA) Office of Sponsored Programs (OSP) SPS - System Features SPS - System Overview Workspace Proposal Creation Demonstration Resources LOUISIANA STATE UNIVERSITY
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Preparing campus for what is changing and what is not What is Grants Reform and when is it effective? What
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Preparing campus for what is changing and what is not What is Grants Reform and when is it effective? What
OMB Uniform Guidance ( UG ) Briefing. ASRSP & OSR Brown Bag Tuesday, January 27 th
 OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
After the Award is Made THEN WHAT?
 After the Award is Made THEN WHAT? George Tucker Chief Grants Management Officer National Institute of Environmental Health Sciences October 2016 TOPICS OF DISCUSSION The ins & outs of the NoA Accessing
After the Award is Made THEN WHAT? George Tucker Chief Grants Management Officer National Institute of Environmental Health Sciences October 2016 TOPICS OF DISCUSSION The ins & outs of the NoA Accessing
Research Proposals & Awards Fiscal Year 2015 July 1, 2014 to June 30, 2015
 Research Proposals & Awards Fiscal Year 2015 July 1, 2014 to June 30, 2015 Report Produced December 2015 Additional ORA Resources are available at http://portal.research.ucla.edu/ Nebula Messier 78. Courtesy
Research Proposals & Awards Fiscal Year 2015 July 1, 2014 to June 30, 2015 Report Produced December 2015 Additional ORA Resources are available at http://portal.research.ucla.edu/ Nebula Messier 78. Courtesy
Sponsored Programs New Developments and Important Reminders
 Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs January 23, 2018 1 NSF Updates NIH Updates Federal Updates Defense Base Act (DBA) Insurance OSP Updates
Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs January 23, 2018 1 NSF Updates NIH Updates Federal Updates Defense Base Act (DBA) Insurance OSP Updates
Uniform Guidance. Overview and Implementation Plan. November 21, 2014
 Uniform Guidance Overview and Implementation Plan November 21, 2014 Uniform Guidance. Change is coming! College or Department name here 1 Overarching goal of the reform is to: streamline the rules and
Uniform Guidance Overview and Implementation Plan November 21, 2014 Uniform Guidance. Change is coming! College or Department name here 1 Overarching goal of the reform is to: streamline the rules and
Rebecca Trahan Office of Sponsored Programs January 22, 2014
 Rebecca Trahan Office of Sponsored Programs January 22, 2014 1 NSF Updates NIH Updates OMB Super Circular Responsible Conduct of Research OSP Updates Q & A 2 1 NSF 14-001 effective 2/24/2014 Effective
Rebecca Trahan Office of Sponsored Programs January 22, 2014 1 NSF Updates NIH Updates OMB Super Circular Responsible Conduct of Research OSP Updates Q & A 2 1 NSF 14-001 effective 2/24/2014 Effective
Outgoing Subaward Basics - Subawards Issued by UCLA under Extramurally Funded Grants/Cooperative Agreements
 Outgoing Subaward Basics - Subawards Issued by UCLA under Extramurally Funded Grants/Cooperative Agreements PREVIEW: Master Training (Mary Haskins, OCGA Subaward Officer) When: Wednesday, April 18, 2018
Outgoing Subaward Basics - Subawards Issued by UCLA under Extramurally Funded Grants/Cooperative Agreements PREVIEW: Master Training (Mary Haskins, OCGA Subaward Officer) When: Wednesday, April 18, 2018
Research Administration Updates. May 16, :00 am IOP Auditorium
 Research Administration Updates May 16, 2018 11:00 am IOP Auditorium Topics Covered Using ClinCard (Greenphire) for Federally Sponsored Clinical Research Studies Ryan Mulligan, SCTR Institute OCR & ORSP
Research Administration Updates May 16, 2018 11:00 am IOP Auditorium Topics Covered Using ClinCard (Greenphire) for Federally Sponsored Clinical Research Studies Ryan Mulligan, SCTR Institute OCR & ORSP
NIH Update FDP September 2009 Office of Policy for Extramural Research Administration
 NIH Update FDP September 2009 Office of Policy for Extramural Research Administration OPERA New NIH Director Francis S. Collins, M.D., Ph.D., was officially sworn in on Monday, August 17, 2009 as the 16th
NIH Update FDP September 2009 Office of Policy for Extramural Research Administration OPERA New NIH Director Francis S. Collins, M.D., Ph.D., was officially sworn in on Monday, August 17, 2009 as the 16th
AURA Meeting. July 23, 2014
 AURA Meeting July 23, 2014 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored
AURA Meeting July 23, 2014 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored
Diane Dean, Director Kathy Hancock, Assistant Grants Compliance Officer Joel Snyderman, Assistant Grants Compliance Officer
 Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar San Diego, CA October 2015 Diane Dean,
Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar San Diego, CA October 2015 Diane Dean,
Agenda. NIH Update. Other Updates. Proposal & Progress Report Statistics Research Administration Training Topic:
 Agenda NIH Update Salary Cap Guidance NOT OD 17 037: NIH Implementation of the Interim RPPR While a Renewal Application is Under Consideration Prior Approval for No Cost Extensions via the era Commons
Agenda NIH Update Salary Cap Guidance NOT OD 17 037: NIH Implementation of the Interim RPPR While a Renewal Application is Under Consideration Prior Approval for No Cost Extensions via the era Commons
THINKING OF MOVING: A GUIDE FOR TRANSFERRING AN NIH GRANT TO A NEW INSTITUTION
 THINKING OF MOVING: A GUIDE FOR TRANSFERRING AN NIH GRANT TO A NEW INSTITUTION It is not unusual for NIH funded Principal Investigators (PIs) to move from one institution to another. We fully expect that
THINKING OF MOVING: A GUIDE FOR TRANSFERRING AN NIH GRANT TO A NEW INSTITUTION It is not unusual for NIH funded Principal Investigators (PIs) to move from one institution to another. We fully expect that
NIH Agency Specifics August 11, 2015
 NIH Agency Specifics August 11, 2015 Maria Skinner Office of Sponsored Programs Stacy Salisbury Contract & Grant Administration NIH Pre-Award Presented by: Maria Skinner Office of Sponsored Programs (OSP)
NIH Agency Specifics August 11, 2015 Maria Skinner Office of Sponsored Programs Stacy Salisbury Contract & Grant Administration NIH Pre-Award Presented by: Maria Skinner Office of Sponsored Programs (OSP)
Interacting Electronically with NIH
 Interacting Electronically with NIH Sheri Cummins NIH era Customer Relationship Manager Electronic Submission of Grant Applications Scarlett Gibb NIH era Customer Relationship Manager era Commons Learn
Interacting Electronically with NIH Sheri Cummins NIH era Customer Relationship Manager Electronic Submission of Grant Applications Scarlett Gibb NIH era Customer Relationship Manager era Commons Learn
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards (New Uniform Guidance)
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards (New Uniform Guidance) Spring Research Administrators Series March 19, 2015 1 Agenda Background UW-Madison
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards (New Uniform Guidance) Spring Research Administrators Series March 19, 2015 1 Agenda Background UW-Madison
Federal Grants Compliance Training
 Federal Grants Compliance Training Agenda Overview of cost principles Allocating expenses between grants Timely reimbursement of expenses Participant support costs Summer salary calculations Fly America
Federal Grants Compliance Training Agenda Overview of cost principles Allocating expenses between grants Timely reimbursement of expenses Participant support costs Summer salary calculations Fly America
OSR Monthly Meeting. August 16, 2018 (Chicago) August 21, 2018 (Evanston)
 OSR Monthly Meeting August 16, 2018 (Chicago) August 21, 2018 (Evanston) Agenda OSR Announcements/Staffing Updates ESPR Replacement Project Update Year-end Procedures NSF Updates NIH Biosketch Reminder
OSR Monthly Meeting August 16, 2018 (Chicago) August 21, 2018 (Evanston) Agenda OSR Announcements/Staffing Updates ESPR Replacement Project Update Year-end Procedures NSF Updates NIH Biosketch Reminder
National Science Foundation (NSF) Update --- Spring Federal Demonstration Partnership Meeting May 13, 2013
 National Science Foundation (NSF) Update --- Spring 2013 Federal Demonstration Partnership Meeting May 13, 2013 Ask Early, Ask Often Jean Feldman Head, Policy Office Division of Institution & Award Support
National Science Foundation (NSF) Update --- Spring 2013 Federal Demonstration Partnership Meeting May 13, 2013 Ask Early, Ask Often Jean Feldman Head, Policy Office Division of Institution & Award Support
NCURA Region II. Hot Topics in Research Administration 1. Denise Clark Associate Vice-President for Research, University of Maryland College Park
 Hot Topics in Research Administration Denise Clark Associate Vice-President for Research, University of Maryland College Park Ann Holmes Assistant Dean of Finance & Administration, College of Behavioral
Hot Topics in Research Administration Denise Clark Associate Vice-President for Research, University of Maryland College Park Ann Holmes Assistant Dean of Finance & Administration, College of Behavioral
Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed:
 COEUS NIH-GRANTS.GOV PROPOSAL REVIEW CHECKLIST REVIEWER: Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed: Has the Program Announcement been reviewed? What is
COEUS NIH-GRANTS.GOV PROPOSAL REVIEW CHECKLIST REVIEWER: Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed: Has the Program Announcement been reviewed? What is
Principal Investigator
 Principal Investigator HEADER TEXT Award Management Roles & Responsibilities November 4, 2016 Susan Mundt, Chris Barnhill, & Jing Liu Agenda PI Definition and Eligibility PreAward Roles & Responsibilities
Principal Investigator HEADER TEXT Award Management Roles & Responsibilities November 4, 2016 Susan Mundt, Chris Barnhill, & Jing Liu Agenda PI Definition and Eligibility PreAward Roles & Responsibilities
HS SPPO Contacts Meeting December 8, 2016
 HS SPPO Contacts Meeting December 8, 2016 Agenda HS SPPO Updates NIH Updates & Reminders OCGA Updates HS SPPO Updates 2016 Holiday Closure HS SPPO, along with the Vice Chancellor Health Sciences/Dean s
HS SPPO Contacts Meeting December 8, 2016 Agenda HS SPPO Updates NIH Updates & Reminders OCGA Updates HS SPPO Updates 2016 Holiday Closure HS SPPO, along with the Vice Chancellor Health Sciences/Dean s
FEDERAL FUND CLOSEOUT. RAF August 13, 2015 Yoon Lee
 FEDERAL FUND CLOSEOUT RAF August 13, 2015 Yoon Lee Agenda Communication update Applicability and population Next steps Resources 2 Federal Fund Closeout Procedure: Communication Update April 2015 Presented
FEDERAL FUND CLOSEOUT RAF August 13, 2015 Yoon Lee Agenda Communication update Applicability and population Next steps Resources 2 Federal Fund Closeout Procedure: Communication Update April 2015 Presented
Policy and Compliance: Working Together Like Hand in Glove
 Policy and Compliance: Working Together Like Hand in Glove Samuel Ashe, Director, Division of Grants Policy, OPERA, OER Diane W. Dean, Director, Division of Grants Compliance and Oversight, OPERA, OER
Policy and Compliance: Working Together Like Hand in Glove Samuel Ashe, Director, Division of Grants Policy, OPERA, OER Diane W. Dean, Director, Division of Grants Compliance and Oversight, OPERA, OER
+ Pre and Post Award Grants Management for Mailman School Researchers
 + Pre and Post Award Grants Management for Mailman School Researchers Rosa Rivera, Associate Dean, Grants & Contracts and Director of MSPH Operations, Office of Sponsored Projects Administration Katherine
+ Pre and Post Award Grants Management for Mailman School Researchers Rosa Rivera, Associate Dean, Grants & Contracts and Director of MSPH Operations, Office of Sponsored Projects Administration Katherine
Research Administrator Meeting Tuesday, January 23, :00-12:00 Illinois Room (348), IMU
 Research Administrator Meeting Tuesday, January 23, 2018 10:00-12:00 Illinois Room (348), IMU Agenda: 1. GAO Announcements 2. Costing Considerations 3. Quick Reference Guide Overview 4. DSP General Announcements
Research Administrator Meeting Tuesday, January 23, 2018 10:00-12:00 Illinois Room (348), IMU Agenda: 1. GAO Announcements 2. Costing Considerations 3. Quick Reference Guide Overview 4. DSP General Announcements
Research Administrators Forum November 8, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum November 8, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith RAPID Update PAMS Jessica Carney, Allison Philabaum Post
Research Administrators Forum November 8, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith RAPID Update PAMS Jessica Carney, Allison Philabaum Post
The Uniform Guidance (2 CFR, Part 200)
 WCMC Implementation of The Uniform Guidance (2 CFR, Part 200) Tuesday, September 22, 2015 & Wednesday, September 23, 2015 UG Workshop Michelle A. Lewis, M.S. Director of Research Administration Interim
WCMC Implementation of The Uniform Guidance (2 CFR, Part 200) Tuesday, September 22, 2015 & Wednesday, September 23, 2015 UG Workshop Michelle A. Lewis, M.S. Director of Research Administration Interim
2016 NIH Update. Presented by Stephanie Smith, Stacey Wade, and Jennifer Webster
 2016 NIH Update Presented by Stephanie Smith, Stacey Wade, and Jennifer Webster PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs Jennifer
2016 NIH Update Presented by Stephanie Smith, Stacey Wade, and Jennifer Webster PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs Jennifer
The Lifecycle of a Sponsored Project
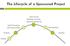 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where does the
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where does the
Grant Accounting Announcements New Electronic Grant Financial Reports - Demo/Implementation Plan New NIH Procedures for Change of Grantee
 December 13, 2012 Grant Accounting Announcements Grant Financial Reports - Demo/Implementation Plan New NIH Procedures for Change of Grantee Organization (Sue Holtkamp & Paul Below)Relinquishment Form
December 13, 2012 Grant Accounting Announcements Grant Financial Reports - Demo/Implementation Plan New NIH Procedures for Change of Grantee Organization (Sue Holtkamp & Paul Below)Relinquishment Form
Rebecca Trahan. Office of Sponsored Programs December 9, ORED Limited Submission Update
 Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
Sponsored by Office of Research Administration. Marcia Smith Associate Vice Chancellor Research Administration. July 9, 2009
 Research Administrators Forum Sponsored by Office of Research Administration Marcia Smith Associate Vice Chancellor Research Administration July 9, 2009 Agenda 2 Updates from the Animal Research Committee
Research Administrators Forum Sponsored by Office of Research Administration Marcia Smith Associate Vice Chancellor Research Administration July 9, 2009 Agenda 2 Updates from the Animal Research Committee
Grant/Sponsor Related Systems. Department and OSP Perspectives on ERA
 Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP)
 Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Complex Award Management Program Projects/Multi-Fund Awards- Post Award NUMBER: RAS SOP 2001 VERSION: 4.0
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Complex Award Management Program Projects/Multi-Fund Awards- Post Award NUMBER: RAS SOP 2001 VERSION: 4.0
Research Administrators Round Table
 Research Administrators Round Table Recap Set framework for going forward Review new Uniform Guidance driven WVU policy changes Sponsored Agreement Closeouts (WVU-FS-23) http://adminfin.wvu.edu/policies/finance/financial-services/wvu-fs-23-policy
Research Administrators Round Table Recap Set framework for going forward Review new Uniform Guidance driven WVU policy changes Sponsored Agreement Closeouts (WVU-FS-23) http://adminfin.wvu.edu/policies/finance/financial-services/wvu-fs-23-policy
Sponsored Projects Manual
 Sponsored Projects Manual University Policies and Procedures for Requesting, Accepting, and Administering Grants, Contracts, and Cooperative Agreements February 2, 2005 CONTENTS 1. Overview... 4 1.1. Purpose
Sponsored Projects Manual University Policies and Procedures for Requesting, Accepting, and Administering Grants, Contracts, and Cooperative Agreements February 2, 2005 CONTENTS 1. Overview... 4 1.1. Purpose
ORA Quarterly Meeting October 26, 2016 (Medical) October 28, 2016 (Coral Gables)
 ORA Quarterly Meeting October 26, 2016 (Medical) October 28, 2016 (Coral Gables) Agenda Quarterly Updates New Reporting Functionality in RRS (PI Portal) Workday Update ORA Website Presentation PCRF-Updates
ORA Quarterly Meeting October 26, 2016 (Medical) October 28, 2016 (Coral Gables) Agenda Quarterly Updates New Reporting Functionality in RRS (PI Portal) Workday Update ORA Website Presentation PCRF-Updates
UNIFORM GUIDANCE IMPLEMENTATION
 UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
Sponsored Programs Roles & Responsibilities
 The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
Participant Support Costs Guidance
 Participant Support Costs Guidance Updated June 11, 2018 This guidance addresses fundamental concepts related to participant support costs and offers some examples. Please contact RSP with questions. Participant
Participant Support Costs Guidance Updated June 11, 2018 This guidance addresses fundamental concepts related to participant support costs and offers some examples. Please contact RSP with questions. Participant
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM. December 2017
 Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
UNIFORM GUIDANCE UPDATE
 UNIFORM GUIDANCE UPDATE CINDY KIEL Executive Associate Vice Chancellor Office of Research Michael Allred Associate Vice Chancellor for Finance/Controller What is the Uniform Guidance? Uniform Administrative
UNIFORM GUIDANCE UPDATE CINDY KIEL Executive Associate Vice Chancellor Office of Research Michael Allred Associate Vice Chancellor for Finance/Controller What is the Uniform Guidance? Uniform Administrative
Preparing for a RPPR Submission
 Preparing for a RPPR Submission Review current NoA Review most current budget and expenditure information. Determine unobligated balance in order to respond to 2590 questions. Review last esnap if applicable
Preparing for a RPPR Submission Review current NoA Review most current budget and expenditure information. Determine unobligated balance in order to respond to 2590 questions. Review last esnap if applicable
Presentation. Title. Presented By: Name. Title
 Presentation Title Presented By: Name Title Lisa Moeller, CRA, Grants Administration OfficeBranch, National Institute of General Medical Sciences, National Institutes of Health Why? Protected time for
Presentation Title Presented By: Name Title Lisa Moeller, CRA, Grants Administration OfficeBranch, National Institute of General Medical Sciences, National Institutes of Health Why? Protected time for
Financial Administrator Development Program: Indirect Costs/Facilities & Administrative/Overhead
 Financial Administrator Development Program: Indirect Costs/Facilities & Administrative/Overhead February 6, 2013 Dan Evon, Director Lindsey Grulke, Manager Contract & Grant Administration Agenda Introduction
Financial Administrator Development Program: Indirect Costs/Facilities & Administrative/Overhead February 6, 2013 Dan Evon, Director Lindsey Grulke, Manager Contract & Grant Administration Agenda Introduction
Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32)
 Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32) Overview: This document is intended to help you complete a KC System to System (S2S) proposal submission
Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32) Overview: This document is intended to help you complete a KC System to System (S2S) proposal submission
Sponsored Programs Roles & Responsibilities
 The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
APLU Analysis of the Administration s FY2018 Budget Request
 APLU Analysis of the Administration s FY2018 Budget Request [May 24, 2017] This document represents an initial analysis of the administration s FY2018 Budget Request for the various agencies and programs
APLU Analysis of the Administration s FY2018 Budget Request [May 24, 2017] This document represents an initial analysis of the administration s FY2018 Budget Request for the various agencies and programs
2017 NIH Update. Presented by Stephanie Smith and Stacey Wade
 2017 NIH Update Presented by Stephanie Smith and Stacey Wade PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs NIH INSTITUTES National
2017 NIH Update Presented by Stephanie Smith and Stacey Wade PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs NIH INSTITUTES National
Notice of Grant Award (NGA): STANDARD Terms and Conditions
 After the Award is Made Then What? Cheryl Chick Chief Grants Management Officer National Human Genome Research Institute Roger G. Sorensen, Ph.D., MPA Division of Neuroscience and Behavior National Institute
After the Award is Made Then What? Cheryl Chick Chief Grants Management Officer National Human Genome Research Institute Roger G. Sorensen, Ph.D., MPA Division of Neuroscience and Behavior National Institute
SJSU Research Foundation
 SJSU Research Foundation The Nuts & Bolts of Proposal Preparation and Award Management Sponsored Programs Workshop February 16, 2017 Agenda Discussion of Common Issues and Solutions Terms and Conditions
SJSU Research Foundation The Nuts & Bolts of Proposal Preparation and Award Management Sponsored Programs Workshop February 16, 2017 Agenda Discussion of Common Issues and Solutions Terms and Conditions
October electronic Research Administration (era) OER, OD, National Institutes of Health
 October 2016 electronic Research Administration (era) OER, OD, National Institutes of Health Scarlett Gibb era Customer Relationship Manager, era Commons Joe Schumaker era Communications and Outreach 2
October 2016 electronic Research Administration (era) OER, OD, National Institutes of Health Scarlett Gibb era Customer Relationship Manager, era Commons Joe Schumaker era Communications and Outreach 2
Subrecipient Monitoring Procedures
 Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
GOVERNMENT SHUTDOWN FREQUENTLY ASKED QUESTIONS
 GOVERNMENT SHUTDOWN FREQUENTLY ASKED QUESTIONS C&G Contact Info: 919-515-8008 or mailto:cnghelpdesk@ncsu.edu SPARCS Contact Info: 919-515-2444 or mailto:sps@ncsu.edu Q: Is my award/account still open for
GOVERNMENT SHUTDOWN FREQUENTLY ASKED QUESTIONS C&G Contact Info: 919-515-8008 or mailto:cnghelpdesk@ncsu.edu SPARCS Contact Info: 919-515-2444 or mailto:sps@ncsu.edu Q: Is my award/account still open for
FDP / No Cost Extension Form Instructions
 Instructions Overview: The FDP / No Cost Extension form is used to request additional time to complete the original scope of a project, utilizing existing funds available on a grant award issued by a federal
Instructions Overview: The FDP / No Cost Extension form is used to request additional time to complete the original scope of a project, utilizing existing funds available on a grant award issued by a federal
Award Transfer Guidelines
 Award Transfer Guidelines The process of transferring awards from one institution to another is not a simple task and requires negotiation between the sponsoring agency, the old institution and the new
Award Transfer Guidelines The process of transferring awards from one institution to another is not a simple task and requires negotiation between the sponsoring agency, the old institution and the new
Sponsored Projects: Curriculum Plan Johns Hopkins University Panel Members:
 Sponsored Projects: Curriculum Plan Johns Hopkins University Panel Members: Mike Alexander, University Administration Jim Aumiller, University Administration Holly Benze, Engineering Don Cornely, University
Sponsored Projects: Curriculum Plan Johns Hopkins University Panel Members: Mike Alexander, University Administration Jim Aumiller, University Administration Holly Benze, Engineering Don Cornely, University
Base. Base Determination and Cost Sharing. Bases represent the direct cost activities of an institution. Generally they consist of: 2/10/2014
 Determination and Cost Sharing s represent the direct cost activities of an institution. Generally they consist of:» Instruction & departmental research» Organized research» Other sponsored activity (public
Determination and Cost Sharing s represent the direct cost activities of an institution. Generally they consist of:» Instruction & departmental research» Organized research» Other sponsored activity (public
IDeA Post Award Management
 IDeA Post Award Management Lisa Hildred Grants Management Specialist Grants Administration Branch National Institute of General Medical Sciences, NIH Meet the Grants Management Team Christy Leake Sam Farrell
IDeA Post Award Management Lisa Hildred Grants Management Specialist Grants Administration Branch National Institute of General Medical Sciences, NIH Meet the Grants Management Team Christy Leake Sam Farrell
American Reinvestment and Recovery Act of 2009 Office of Policy for Extramural Research Administration
 American Reinvestment and Recovery Act of 2009 Office of Policy for Extramural Research Administration OPERA NIH Recovery Web site www.nih.gov/recovery ARRA Grant Funding Opps and Info at Grants.nih.gov/Recovery
American Reinvestment and Recovery Act of 2009 Office of Policy for Extramural Research Administration OPERA NIH Recovery Web site www.nih.gov/recovery ARRA Grant Funding Opps and Info at Grants.nih.gov/Recovery
Research Administrators Group (RAG) Meeting July 2015
 Research Administrators Group (RAG) Meeting July 2015 Office of Sponsored Projects 1 Agenda Office of Sponsored Projects 2 Introduction of New Staff Proposal/PD Update Proposal Metrics RPPR Transition
Research Administrators Group (RAG) Meeting July 2015 Office of Sponsored Projects 1 Agenda Office of Sponsored Projects 2 Introduction of New Staff Proposal/PD Update Proposal Metrics RPPR Transition
Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS
 Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar Chicago, IL October 2016 Diane Dean, Director
Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar Chicago, IL October 2016 Diane Dean, Director
Grant Application Package
 Generates errors for all applications Grant Application Package Opportunity Title: Research Project Grant (Parent R01) Offering Agency: National Institutes of Health Generates errors in certain situations
Generates errors for all applications Grant Application Package Opportunity Title: Research Project Grant (Parent R01) Offering Agency: National Institutes of Health Generates errors in certain situations
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates!
 FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
FDP Phase VI Kicks Off at Recent Meeting; New Members Are Excited to Participate
 October 2014 3 4 7 In This Issue UCLA Challenges Majority Of Questioned Costs in NSF Audit HHS OIG Issues Reports Reviewing OHRP, NIH, SUPPORT Controversy Agency Developments If you re not accessing the
October 2014 3 4 7 In This Issue UCLA Challenges Majority Of Questioned Costs in NSF Audit HHS OIG Issues Reports Reviewing OHRP, NIH, SUPPORT Controversy Agency Developments If you re not accessing the
The Metis Foundation Office of Grant and Contract Administration SUBRECIPIENT INFORMATION AND COMPLIANCE FORM
 The Metis Foundation Office of Grant and Contract Administration SUBRECIPIENT INFORMATION AND COMPLIANCE FORM All subrecipients must complete this form when submitting a proposal to the Metis Foundation
The Metis Foundation Office of Grant and Contract Administration SUBRECIPIENT INFORMATION AND COMPLIANCE FORM All subrecipients must complete this form when submitting a proposal to the Metis Foundation
Current Issues in Sponsored Projects Administration. March 11 10:30-11:45 3:15 4:30
 Current Issues in Sponsored Projects Administration March 11 10:30-11:45 3:15 4:30 Workshop Presenters Name: Linda Learned Title: Associate Director/Interim Director, Office of Sponsored Programs and Research
Current Issues in Sponsored Projects Administration March 11 10:30-11:45 3:15 4:30 Workshop Presenters Name: Linda Learned Title: Associate Director/Interim Director, Office of Sponsored Programs and Research
Sponsored Program Administration. October 10, 2017
 Sponsored Program Administration October 10, 2017 Agenda Procurement Services FDP Updates Jaggaer Pre-Award Federal Agency Updates Grants.gov Transition to Workspace NIH FORMS-E Post-Award GONE Act Certificate
Sponsored Program Administration October 10, 2017 Agenda Procurement Services FDP Updates Jaggaer Pre-Award Federal Agency Updates Grants.gov Transition to Workspace NIH FORMS-E Post-Award GONE Act Certificate
PMS Project Closeout. Dan Evon, Michigan State University Nate Martinez Wayman, Duke University
 PMS Project Closeout Dan Evon, Michigan State University Nate Martinez Wayman, Duke University 1 Session Outline GAO Audit Reports Closeout Parameters The FCTR Closeout History Sample Numbers Details Late
PMS Project Closeout Dan Evon, Michigan State University Nate Martinez Wayman, Duke University 1 Session Outline GAO Audit Reports Closeout Parameters The FCTR Closeout History Sample Numbers Details Late
Uniform Guidance Update. Ruth Boardman, Associate Director Office of Grants and Contracts March 2015
 Uniform Guidance Update Ruth Boardman, Associate Director Office of Grants and Contracts March 2015 Objectives Latest Updates Internal Controls Procurement Status of Policies and Procedures Research Terms
Uniform Guidance Update Ruth Boardman, Associate Director Office of Grants and Contracts March 2015 Objectives Latest Updates Internal Controls Procurement Status of Policies and Procedures Research Terms
Indirect Costs/Facilities & Administrative/Overhead
 Indirect Costs/Facilities & Administrative/Overhead Presenters: Andrew Cascaddan, Manager, Contract and Grant Administration Ryan Kelemen, Administrator/F&A Analyst, Contract and Grant Administration Agenda
Indirect Costs/Facilities & Administrative/Overhead Presenters: Andrew Cascaddan, Manager, Contract and Grant Administration Ryan Kelemen, Administrator/F&A Analyst, Contract and Grant Administration Agenda
FAQ S FOR UNIFORM GUIDANCE
 FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
