Research Administration Updates. May 16, :00 am IOP Auditorium
|
|
|
- Brendan Hodges
- 5 years ago
- Views:
Transcription
1 Research Administration Updates May 16, :00 am IOP Auditorium
2 Topics Covered Using ClinCard (Greenphire) for Federally Sponsored Clinical Research Studies Ryan Mulligan, SCTR Institute OCR & ORSP Workflow for Federal and Industry Sponsored Clinical Studies Cullen McWhite, OCR Outgoing Sub-awards Becky Timpner, ORSP Cayuse Expansion Update Robbie Lee, ORSP Notices/Updates/Reminders ORSP
3 ClinCard for Federal Studies Ryan Mulligan, MRA, MS, CCRP, CRA ClinCard Program Manager, Office of Clinical Research
4 What Is the ClinCard? The ClinCard system is a web based portal that automates participant compensation and mileage reimbursement, for clinical research studies, utilizing a reloadable debit card to provide realtime payments. The goal of the system is to reduce administration, increase patient retention, promote protocol compliance, and overall improve the site and patient experience while centralizing reporting and reducing costs.
5 ClinCard Package Example
6 ClinCard is now available for federally sponsored clinical research studies
7 Budgeting for ClinCard Federal Studies Federally funded clinical research No setup fee $0 ClinCard* $0 Transaction/Load Fee* *The cost of the ClinCard and the transaction fees have been deemed an institutional F&A expense.
8 Budgeting for ClinCard Industry Studies Industry initiated/industry funded: $350 New Study Setup Fee $5 ClinCard (each) $1.15 Transaction/Load Fee (each)
9 Budgeting for ClinCard Other Studies Foundation/Internal/Investigator initiated/etc. No Setup Fee $5 ClinCard (each) $1.15 Transaction/Load Fee (each)
10 Requesting to use ClinCard Office of Clinical Research, PRA Intake Form Submit a request for a Study Setup (new) under Greenphire ClinCard
11
12 For Questions or More Information: Office of Clinical Research (843)
13 Billing Compliance Workflows Leila Forney, DNP, CCRP Associate Director, Prospective Reimbursement Analysis Office of Clinical Research Cullen McWhite, BS, CCRC Prospective Reimbursement Analysis Manager Office of Clinical Research
14 OCR and ORSP Workflows
15 Corporate Studies Study Team finalizes budget PRA Team reviews final budget/contract ORSP executes contract PRA Team/Study Team requests changes to budget ORSP finalizes contract Study Team submits PRA Study Team submits epds PRA team receives notification of epds submission PRA Team/ORSP requests changes contract ORSP negotiates contract language with Sponsor
16 Non-Corporate Studies Coming June 1 st 2018! PRA Team works with Study Team to resolve billing risks Study Team notifies ORSP when all institutional approvals have been obtained ORSP receives Just-In-Time request ORSP notifies Study Team and PRA Team of Just-In-Time notice Study Team submits PRA PRA Team assesses billing risks PRA Team notifies Study Team if billing risks are identified PRA Team provides notification to Study Team and ORSP
17 OCR and IRB Workflows
18 Corporate Studies (IRB III) Federal regulations require the ICF to detail any additional costs to the subject that may result from participation in the research * All studies submitted to IRB III will require a PRA review before the study may proceed with IRB review REGARDLESS of review type (exempt, expedited, full board) PRA Team will ensure the cost language in the ICF is consistent with the Study Billing Plan *45 CFR (b)(3) and 21 CFR 50.25(b)(3)
19 Corporate Studies (IRB III) Coming June 20 th 2018! Study Team submits PRA PRA Team creates DRAFT Study Billing Plan PRA Team generates PRA Summary Memo with approved Cost Language for ICF Study Team submits to IRB PRA Team will review cost language in ICF and eirb application PRA Team issues Ancillary Committee Approval and uploads PRA Summary Memo in eirb IRB reviews study and notifies PRA Team of any changes affecting cost language PRA Team will issue OCR approval once IRB approves and releases the study.
20 PRA Summary Memo Includes: Study Demographics Sponsor Regulatory Status of IP IND, IDE, etc. Qualifying Clinical Trial Status Will have potential benefit Payer Research, Participant, Mixed Approved ICF Cost Language
21 Corporate Studies (IRB III) Plan ahead and know your target IRB submission date PRA team needs time to review, approve, or request changes to the cost language IRB Exempt IRB Expedited or Full Board Submit to OCR 4 days prior to IRB submission Submit to OCR 3 weeks prior to IRB submission *This is the minimum time required for OCR review. Please submit as soon as possible.
22 THANK YOU Office of Clinical Research (OCR) Address: Phone Number: (843) Address: Roper Medical Office Building, Suite 170 Web Address:
23 Office of Research & Sponsored Programs Subawards: (MUSC as the PTE*) *Pass-Through Entity
24 What is needed so ORSP can issue an outgoing Subaward? Typical NIH requirements for PTE to collect from Subawardees at proposal stage: - Letter/Statement of Intent signed by subrecipient - Statement of Work - Detailed budget - Budget Justification - FCOI Certification / applicable forms - Subrecipient s federally negotiated F&A rate agreement (if applicable) - Biosketches/CVs as required by the funding agency - A completed PHS 398 Facepage is recommended, and contains most of the information necessary to prepare the subaward NOTE: Please upload these as a separate attachment from the full proposal file
25 PHS 398 Facepage
26 What considerations are required to establish a subaward/consortium once an award is made? Has the subaward been approved as part of the awarded budget and project? Is separate sponsor approval required by the award terms? Has the subaward budget and/or scope of work changed from the proposal? Likely, if the overall budget or work scope has changed. Does the subaward scope of work include human and/or animal subjects? If so, additional information may be needed to complete the subaward and complete our risk assessment. Once these details have been determined, ORSP will begin the subaward issuance process and fulfil all requirements of the Uniform Guidance (2 CFR ).
27 ORSP Subaward/Amendment Issuance Process - Verify Subrecipient SAM registration is active (must be renewed each year). - Requires Subrecipient s DUNS Number - Verify Subrecipient s FCOI policy is PHS compliant - Request additional documentation if needed - Participation in the FDP FCOI Clearinghouse - Review A-133 audit or Mini-audit form as applicable. - Subrecipient s Tax ID (EIN) number may be necessary - Participation in FDP Expanded Clearinghouse - Check to make sure Subrecipient and Subrecipient PI are not debarred or suspended - Assess risk of flowing funds to each potential Subrecipient - Risk Assessment Questionnaire (new subaward) - Continuing Assessment Tool (modification/amendment) - Federal Funding Accountability and Transparency Act (FFATA) - Monthly FFATA Reporting - NIH FFATA resources: Why?? 2 CFR : Uniform Guidance Subrecipient Monitoring Requirements
28 Subaward Amendment Requirements Amendment (modification) The following should be uploaded to the epds (with RPPR) - Updated Statement of Work (if applicable) - Detailed budget - Budget Justification - FCOI Certification / applicable forms NOTE: Please upload these as a separate attachment from the RPPR file
29 Outgoing Subaward References and Resources ORSP How Do I web page: Determine subawardee or contractor relationship? Prepare for an outgoing subaward as part of an MUSC proposal? FDP FCOI Clearinghouse FDP Expanded Clearinghouse Still have questions? Please contact ORSP s Becky Timpner timpner@musc.edu Phone:
30 Office of Research & Sponsored Programs Revised VA Statement of Work Process
31 Revised Process Lead Time: 6 weeks (or more) prior to desired effective date Single Point of Contact: Becky Timpner, timpner@musc.edu The NEW process: The requesting department will route an epds which will include the following: - Completed Statement of Work Template - Budget for the work (with salary and fringe separated) - Indicate that this is a VA Personnel Services Agreement (PSA) and include the funded person s name in the title of the project within the epds preceding the actual title of the VA Merit Award. Example: VA PSA Jane Doe: Actual Title of the Merit Award here. After the epds has been approved by all at the department level, it will route to ORSP for review. When ORSP reviews/approves the SOW, ORSP will send the SOW via to the VA for approval. Upon VA approval, ORSP will approve the epds proposal. When the Task Order is issued by the Pittsburgh VA contracting office, ORSP will award the epds associated with the SOW and Grants and Contracts Accounting (GCA) will assign a UDAK
32 Single Point of Contact ORSP is hopeful that a single point of contact for both MUSC and the VA will alleviate some confusion, streamline/simplify the overall process, and allow for more accurate tracking.
33 Takeaways & Tools SOW Template VAMC Worksheet When Filling out the epds for a SOW (one SOW per epds) be sure to: - Include VA PSA (Insert Name of Person Here): (Title of merit award) in the title field - The Current Budget Dates: The proposed funding dates for the SOW - Total Project Dates: Merit Award Beginning and End dates - The IDC rate on these is 0 at this time - Make sure the Primary Unit is correct - Questions 1-18 should be marked as No
34 Office of Research and Sponsored Programs Cayuse Expansion Project
35 Cayuse Sponsored Projects MUSC has purchased the Cayuse Sponsored Projects Module Integrates with Cayuse 424 proposal preparation Consolidates Documents, Data and Communications Provides role-based dashboards for all users Grants on ERMA (epds) and Coeus will be retired Project Kick-Off is next Monday, May 21 st Expected implementation 6 to 8 months Relevant Focus and Work Groups will be formed as needed during the implementation process Send me an if you are interested in participating Robbie Lee - leer@musc.edu
36 Office of Research & Sponsored Programs News and Updates
37 FY2019 Fringe Benefit Rates - REMINDER Employee Benefits Category Fringe Benefits Rate Staff 38.4% Post-Doctoral Fellows 29.8% Temp Faculty/Temp non- Faculty 31.7% Students/Other 6.9% Applicable Employment Types Faculty 9-Month Faculty Unclassified Non-Faculty Classified, Research Post-Docs Residents Temporary Faculty Temporary Non-Faculty Student Ph.D. Students Research Grant Employees- No Benefits
38 Recent Notices NOT-OD Career Award (K) Policy Update: Temporary Adjustments to Percent Effort or Part- Time Institutional Appointment K awardees may request to reduce their professional effort to less than 75% (equivalent to 9 person-months) for up to 12 continuous months. See: NOT-OD and NIH Grants Policy Statement ( ) Clarification: During the period of reduced effort, NIH will adjust the total salary amount committed to the K award consistent with the adjusted level of effort. NIH will continue to provide full research costs in other budget categories. The K awardee may request to extend the duration of the award to account for the reduced effort.
39 Recent Notices NOT-OD Career Award (K) Policy Update: Concurrent Support from a Mentored K Award and a Research Grant Updates NIH policy NOT-OD concerning concurrent support from mentored career development (K) award and a research grant, specifically to: expand the categories of concurrent support for which mentored K awardees may request reduction of effort to include PI/PD of peer-reviewed research grants of at least $100,000 in direct costs, obtained from non-federal sources; e.g., foundations and professional societies clarify that during the period of reduced effort NIH will continue to provide full research development support costs i.e., Other Personnel, Equipment, Travel, Participant/Trainee Support, and Other Direct Costs budget categories
40 Recent Notices NOT-OD Financial Conflict of Interest: Investigator Disclosures of Foreign Financial Interests Reminder of the requirements of 42 CFR Part 50, Subpart F, Objectivity of Research Also known as the Financial Conflict of Interest (FCOI) regulation Clarifies Investigator disclosures with respect to foreign financial interests Investigators, including subrecipient Investigators, must disclose all financial interests received from a foreign Institution of higher education or the government of another country (which includes local, provincial, or equivalent governments of another country).
41 Recent Notices NOT-OD Ruth L. Kirschstein National Research Service Award (NRSA) Stipends, Tuition/Fees and Other Budgetary Levels Effective for Fiscal Year 2018 Budget levels in this Notice apply only to Kirschstein-NRSA awards made with FY 2018 funds All FY 2018 awards previously issued using NOT-OD and NOT-OD will be revised to adjust funding to the FY 2018 levels. Appointments to institutional training grants that have already been awarded in FY 2018 must be amended to reflect the FY 2018 stipend levels once the training grant award has been adjusted by the NIH. Amended appointments must be submitted through xtrain in the era Commons. Retroactive adjustments or supplementation of stipends or other budgetary categories with Kirschstein-NRSA funds for an award made prior to October 1, 2017 are not permitted
42 Recent Notices NOT-OD Transition from Inclusion Management System to New Human Subjects System (HSS) as of June 9, 2018 NIH has developed a new Human Subjects System (HSS), which consolidates human subjects and clinical trial information in once place Replaces the Inclusion Management System The Inclusion link will no longer appear on the Commons Status page as of June 9, 2018 Information captured in HSS is generally submitted on the PHS Human Subjects and Clinical Trials Information form Post-submission updates to human subjects and clinical trial-related information (including human subjects protections, participant and enrollment information, and Clinicaltrials.gov registration and reporting information) must be made in HSS via the era Commons Status page after June 9, 2018
43 Recent Notices NOT-OD (continued) Key Changes NIH will migrate enrollment records currently in IMS to HSS. Updates to enrollment records must be submitted to NIH no later than June 8, 2018 or entered in HSS. Updates not submitted by June 8, 2018 will not be available in HSS and must be reentered. NIH recipients completing an RPPR (Research Progress Performance Report) will be prompted to access HSS to update inclusion enrollment reports. Recipients may access the system through the Human Subjects link in the RPPR or the era Commons Status page.
44 Recent Notices NOT-OD (continued) Key Changes Section 6: Clinical Trial Milestone Plan is intended for use in progress reports for competing applications submitted on or after January 25, 2018 and is not currently required unless otherwise noted in the Funding Opportunity Announcement or terms and conditions of award. Recipients should refer to the RPPR Instruction Guide for guidance. The HSS system includes a new interface and workflow. When submitting studies to NIH, Signing Officials will submit all study records associated with an application at one time rather than separately. Participant-level sex/gender, race, ethnicity and age data may be submitted in a CSV file to populate the Inclusion Enrollment Report. Participant level data will be required for applications submitted January 25, 2019 or later. See NOT-OD-116 for additional information.
45 Recent Notices NOT-OD (continued) Key Changes Investigators and signing officials may make study updates or corrections (including just-in-time or off-cycle updates) by accessing HSS through the Human Subjects link in the era Commons Status page. Some changes, including those involving increased risk to human participants, may require prior approval by NIH. Users are currently unable to delegate authority for HSS updates and/or submissions to another user. Delegation authority is expected to be available in a future enhancement of HSS.
46 Office of Research & Sponsored Programs Reminders and Updates
47 REMINDERS 3 Business Day Deadline What Exactly is REQUIRED in ORSP by the 3 Business Day Deadline? Answer: the complete proposal package which includes: budget, budget justification, project narrative (science), all subaward documentation, FCOI forms, VA MOUs, Grant Certification forms, and any other items required by the sponsor in the proposal package
48 REMINDERS Confirm (PI or PI support staff) Correct Funding Opportunity Announcement (FOA) is Used & the Intended Institute is Participating in the FOA Note: Verify the NIH institute (e.g. NIMH, NIDA, NHLBI, etc.) is participating! New Human Subjects Section Requirements (longer to complete, so plan accordingly allowing adequate preparation time, let s not be surprised) Note: Forms E etc. are still relatively new, so if a PI has not experienced them yet, please alert them to allow enough time to address the new requirements. We understand the information requested is very time consuming to compile.
49 REMINDERS Utilize the Cayuse Validation Feature BEFORE ORSP Review 1.) Select (click-on) lightning bolt in top bar above proposal 2.) Which opens a window called Electronic Submission 3.) Then select (click-on) Validate Proposal to see validations 4.) System generated errors and warnings will listed for action to be taken 5.) Proposal will NOT get submitted by using the validation feature
50 REMINDERS ACH Payments Claim forms for ACH payments need to be done immediately upon notice of payment by completing a Remittance Advice. If the payment has already hit the unclaimed cash report the Unidentified Receipts Claim will need to be used. These forms can be found at: Unclaimed Cash report should be checked by the department/fiscal unit at least monthly.
51 Final Thoughts Any Announcements? Questions or Comments? PLEASE complete a survey & THANK YOU Next ORSP Information Session: June 20, 11:00 IOP Auditorium
Subrecipient Monitoring Procedures
 Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
Subrecipient Profile Questionnaire
 Subrecipient Profile Questionnaire How to use: The questionnaire is used to help determine a subrecipient organization s financial and management strength, which helps assess risk and dictates the monitoring
Subrecipient Profile Questionnaire How to use: The questionnaire is used to help determine a subrecipient organization s financial and management strength, which helps assess risk and dictates the monitoring
FINANCE-315 7/1/2017 SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a proposal to UACES. It provides a checklist of documents and certifications required by sponsors, as well as an
SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a proposal to UACES. It provides a checklist of documents and certifications required by sponsors, as well as an
Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed:
 COEUS NIH-GRANTS.GOV PROPOSAL REVIEW CHECKLIST REVIEWER: Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed: Has the Program Announcement been reviewed? What is
COEUS NIH-GRANTS.GOV PROPOSAL REVIEW CHECKLIST REVIEWER: Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed: Has the Program Announcement been reviewed? What is
Grant/Sponsor Related Systems. Department and OSP Perspectives on ERA
 Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP)
 Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Research Proposal Application Process NUMBER: RAS SOP 1002 VERSION: 4.0 LAST REVISED: PREPARED BY: Office
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Research Proposal Application Process NUMBER: RAS SOP 1002 VERSION: 4.0 LAST REVISED: PREPARED BY: Office
Subaward Policies and Procedures Manual
 JOHNS HOPKINS UNIVERISTY Krieger School of Arts and Sciences Business and Research Administration Subaward Policies and Procedures Manual 2016 Contents Introduction...3 Differentiating types of relationships...3
JOHNS HOPKINS UNIVERISTY Krieger School of Arts and Sciences Business and Research Administration Subaward Policies and Procedures Manual 2016 Contents Introduction...3 Differentiating types of relationships...3
Office of Sponsored Projects 5/31/ Volume 2, Issue 5
 The OSP News & Updates, published by the Office of Sponsored Projects, is a bi-weekly subscription-based newsletter that provides OSP and sponsor updates and reminders, quick facts, guidance and training
The OSP News & Updates, published by the Office of Sponsored Projects, is a bi-weekly subscription-based newsletter that provides OSP and sponsor updates and reminders, quick facts, guidance and training
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates!
 FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
ORA Quarterly Meeting April
 ORA Quarterly Meeting April 19 2017 Agenda 1. Workday Grants Implementation Items General Information Blackout Period Sub K s - Set-up and Invoicing Advance Accounts 2. Jackson/ UM Partnership Master Agreement
ORA Quarterly Meeting April 19 2017 Agenda 1. Workday Grants Implementation Items General Information Blackout Period Sub K s - Set-up and Invoicing Advance Accounts 2. Jackson/ UM Partnership Master Agreement
PEER (PAPERLESS ENVIRONMENT FOR ELECTRONIC REVIEW)
 PEER (PAPERLESS ENVIRONMENT FOR ELECTRONIC REVIEW) 1 What is PEER? PEER is a web portal maintained by Vanderbilt University which handles internal processing of grant related requests and contract submission
PEER (PAPERLESS ENVIRONMENT FOR ELECTRONIC REVIEW) 1 What is PEER? PEER is a web portal maintained by Vanderbilt University which handles internal processing of grant related requests and contract submission
The Metis Foundation Office of Grant and Contract Administration SUBRECIPIENT INFORMATION AND COMPLIANCE FORM
 The Metis Foundation Office of Grant and Contract Administration SUBRECIPIENT INFORMATION AND COMPLIANCE FORM All subrecipients must complete this form when submitting a proposal to the Metis Foundation
The Metis Foundation Office of Grant and Contract Administration SUBRECIPIENT INFORMATION AND COMPLIANCE FORM All subrecipients must complete this form when submitting a proposal to the Metis Foundation
SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients must complete this form when submitting a proposal to KSU, along with documents and certifications required by sponsors. This form must be endorsed by the
SUBRECIPIENT COMMITMENT FORM All subrecipients must complete this form when submitting a proposal to KSU, along with documents and certifications required by sponsors. This form must be endorsed by the
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM. December 2017
 Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Sponsored Programs Roles & Responsibilities
 The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
Outgoing Subaward Guide and F-A-Qs
 1. What is a subaward? Outgoing Subaward Guide and F-A-Qs 2. After an award is made to Rutgers, how is a subaward established? 3. What is the outgoing subaward process? 4. How can a subaward be changed/amended?
1. What is a subaward? Outgoing Subaward Guide and F-A-Qs 2. After an award is made to Rutgers, how is a subaward established? 3. What is the outgoing subaward process? 4. How can a subaward be changed/amended?
OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015
 OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015 INTRODUCTION The University of Wisconsin-Milwaukee (UWM) is the prime recipient on a wide range of sponsored awards
OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015 INTRODUCTION The University of Wisconsin-Milwaukee (UWM) is the prime recipient on a wide range of sponsored awards
Proposal Submission Guidelines
 March 28, 2016 Proposal Submission Guidelines Elliott School faculty have led a rapid increase in sponsored research over the past few years and the Dean s Office has increased its administrative capacity
March 28, 2016 Proposal Submission Guidelines Elliott School faculty have led a rapid increase in sponsored research over the past few years and the Dean s Office has increased its administrative capacity
Accelerated Translational Incubator Pilot (ATIP) Program. Frequently Asked Questions. ICTR Research Navigators January 19, 2017 Version 7.
 Accelerated Translational Incubator Pilot (ATIP) Program Frequently Asked Questions ICTR Research Navigators January 19, 2017 Version 7.0 TABLE OF CONTENTS Section # Title Page 1. ABOUT THE ATIP PROGRAM...
Accelerated Translational Incubator Pilot (ATIP) Program Frequently Asked Questions ICTR Research Navigators January 19, 2017 Version 7.0 TABLE OF CONTENTS Section # Title Page 1. ABOUT THE ATIP PROGRAM...
Sponsored Programs Roles & Responsibilities
 The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
What You Need to Know When Submitting an NIH SF424 R&R Grant Application Through the UCLA Office of Contract and Grant Administration (OCGA)
 What You Need to Know When Submitting an NIH SF424 R&R Grant Application Through the UCLA Office of Contract and Grant Administration (OCGA) Kim Duiker, Assistant Director Mary Haskins, Contract & Grant
What You Need to Know When Submitting an NIH SF424 R&R Grant Application Through the UCLA Office of Contract and Grant Administration (OCGA) Kim Duiker, Assistant Director Mary Haskins, Contract & Grant
Are You Ready for This? The New Uniform Grant Guidance 2 CFR 200
 Are You Ready for This? The New Uniform Grant Guidance 2 CFR 200 Increase in Federal Grants Activity The Catalog of Federal Domestic Assistance lists over 2,000 Federal grant programs $600B $200B $7B $24B
Are You Ready for This? The New Uniform Grant Guidance 2 CFR 200 Increase in Federal Grants Activity The Catalog of Federal Domestic Assistance lists over 2,000 Federal grant programs $600B $200B $7B $24B
NIH Update. Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH. FDP January 11, 2016
 NIH Update Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH FDP January 11, 2016 1 2 NIH FY 2016 Budget News NIH is currently funded under the Consolidated
NIH Update Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH FDP January 11, 2016 1 2 NIH FY 2016 Budget News NIH is currently funded under the Consolidated
Navigating Grant Resources. 10/3/2018 Dr. Susan Fell
 Navigating Grant Resources 10/3/2018 Dr. Susan Fell sfell@fsu.edu 850-645-2172 Sponsored Research Administration https://www.research.fsu.edu/ Applying for a Grant Opportunity Does this require sign-off
Navigating Grant Resources 10/3/2018 Dr. Susan Fell sfell@fsu.edu 850-645-2172 Sponsored Research Administration https://www.research.fsu.edu/ Applying for a Grant Opportunity Does this require sign-off
The Uniform Guidance 2 CFR 200 A Guide to Risk-Based Grants Management
 This image cannot currently be displayed. The Uniform Guidance 2 CFR 200 A Guide to Risk-Based Grants Management 2015 This image cannot currently be displayed. Increase in Federal Grants Activity The Catalog
This image cannot currently be displayed. The Uniform Guidance 2 CFR 200 A Guide to Risk-Based Grants Management 2015 This image cannot currently be displayed. Increase in Federal Grants Activity The Catalog
Grants Financial Procedures (Post-Award) v. 2.0
 Grants Financial Procedures (Post-Award) v. 2.0 1 Grants Financial Procedures (Post Award) Version Number: 2.0 Procedures Identifier: Superseded Procedure(s): BU-PR0001 N/A Date Approved: 9/1/2013 Effective
Grants Financial Procedures (Post-Award) v. 2.0 1 Grants Financial Procedures (Post Award) Version Number: 2.0 Procedures Identifier: Superseded Procedure(s): BU-PR0001 N/A Date Approved: 9/1/2013 Effective
Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32)
 Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32) Overview: This document is intended to help you complete a KC System to System (S2S) proposal submission
Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32) Overview: This document is intended to help you complete a KC System to System (S2S) proposal submission
Agenda. NIH Update. Other Updates. Proposal & Progress Report Statistics Research Administration Training Topic:
 Agenda NIH Update Salary Cap Guidance NOT OD 17 037: NIH Implementation of the Interim RPPR While a Renewal Application is Under Consideration Prior Approval for No Cost Extensions via the era Commons
Agenda NIH Update Salary Cap Guidance NOT OD 17 037: NIH Implementation of the Interim RPPR While a Renewal Application is Under Consideration Prior Approval for No Cost Extensions via the era Commons
How to Manage Externally Funded Grants PROJECTS - FUND CODE 501 or 502
 How to Manage Externally Funded Grants PROJECTS - FUND CODE 501 or 502 What is a Grant? Funding that is coming from a source external to Wesleyan Project conceived by the Investigator Response to a solicitation
How to Manage Externally Funded Grants PROJECTS - FUND CODE 501 or 502 What is a Grant? Funding that is coming from a source external to Wesleyan Project conceived by the Investigator Response to a solicitation
Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6
 Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6 KC-PRE28: Prepare Summary Proposal KC-AWD22: Research Gifts November 13, 2013 Goals and Outcomes Goals Obtain feedback on the proposed
Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6 KC-PRE28: Prepare Summary Proposal KC-AWD22: Research Gifts November 13, 2013 Goals and Outcomes Goals Obtain feedback on the proposed
Are You Ready for This? The New Uniform Guidance 2 CFR 200
 Are You Ready for This? The New Uniform Guidance 2 CFR 200 Increase in Federal Grants Activity The Catalog of Federal Domestic Assistance lists over 2,000 Federal grant programs $600B $200B $7B $24B $91B
Are You Ready for This? The New Uniform Guidance 2 CFR 200 Increase in Federal Grants Activity The Catalog of Federal Domestic Assistance lists over 2,000 Federal grant programs $600B $200B $7B $24B $91B
Subrecipient Monitoring Under 2 CFR 200. Kris Rhodes, MS Director, Higher Education Practice
 Subrecipient Monitoring Under 2 CFR 200 Kris Rhodes, MS Director, Higher Education Practice krisrhodes@maximus.com 1 MAXIMUS Higher Education Practice Headquartered in Northbrook, IL Satellite Offices
Subrecipient Monitoring Under 2 CFR 200 Kris Rhodes, MS Director, Higher Education Practice krisrhodes@maximus.com 1 MAXIMUS Higher Education Practice Headquartered in Northbrook, IL Satellite Offices
University of Pittsburgh SPONSORED PROJECT FINANCIAL GUIDELINE Subject: SUBRECIPIENT MONITORING
 I. Scope Subrecipient monitoring is the process by which the University selects, monitors, controls, and accounts for University subcontractors or subrecipients utilized on sponsored projects in accordance
I. Scope Subrecipient monitoring is the process by which the University selects, monitors, controls, and accounts for University subcontractors or subrecipients utilized on sponsored projects in accordance
Rebecca Trahan. Office of Sponsored Programs December 9, ORED Limited Submission Update
 Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
Grant Accounting Announcements New Electronic Grant Financial Reports - Demo/Implementation Plan New NIH Procedures for Change of Grantee
 December 13, 2012 Grant Accounting Announcements Grant Financial Reports - Demo/Implementation Plan New NIH Procedures for Change of Grantee Organization (Sue Holtkamp & Paul Below)Relinquishment Form
December 13, 2012 Grant Accounting Announcements Grant Financial Reports - Demo/Implementation Plan New NIH Procedures for Change of Grantee Organization (Sue Holtkamp & Paul Below)Relinquishment Form
OSP Checklist - NIH Pathway to Independence Award (Parent K99/R00). Page 1 of 5
 OSP Checklist - NIH Pathway to Independence Award (Parent K99/R00). Page 1 of 5 ü Read the solicitation: https://researchtraining.nih.gov/programs/career-development ü In all cases the SF 424 and instructions
OSP Checklist - NIH Pathway to Independence Award (Parent K99/R00). Page 1 of 5 ü Read the solicitation: https://researchtraining.nih.gov/programs/career-development ü In all cases the SF 424 and instructions
Research Administrators Forum September 13, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum September 13, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith OCGA Initiatives Patti Manheim, Kim Duiker Proposal Intake
Research Administrators Forum September 13, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith OCGA Initiatives Patti Manheim, Kim Duiker Proposal Intake
Division of Research
 Division of Research Quarterly Research Administration Meeting October 30, 2012 Page 1 1 Division of Research Quarterly Research Administration Meeting AGENDA Welcome and Introductions Pre Award Update
Division of Research Quarterly Research Administration Meeting October 30, 2012 Page 1 1 Division of Research Quarterly Research Administration Meeting AGENDA Welcome and Introductions Pre Award Update
SUBRECIPIENT COMMITMENT FORM
 SECTION A SUBRECIPIENT INFORMATION SUBRECIPIENT COMMITMENT FORM Phone: Email: SUBRECIPIENT: CU PI: PROPOSAL TITLE: TOTAL FUNDING REQUESTED: ORIGINATING SPONSOR, IF A PASS THRU APPLICATION: PRIME SPONSOR:
SECTION A SUBRECIPIENT INFORMATION SUBRECIPIENT COMMITMENT FORM Phone: Email: SUBRECIPIENT: CU PI: PROPOSAL TITLE: TOTAL FUNDING REQUESTED: ORIGINATING SPONSOR, IF A PASS THRU APPLICATION: PRIME SPONSOR:
Pre Award Table of Contents
 Pre Award Table of Contents ORA Internal Procedures for Account Establishment 2 Conflict of Interest: Thomas Jefferson University Conflict of Interest (COI) Policy 4 Cost Sharing and Matching Funds 6 Developing
Pre Award Table of Contents ORA Internal Procedures for Account Establishment 2 Conflict of Interest: Thomas Jefferson University Conflict of Interest (COI) Policy 4 Cost Sharing and Matching Funds 6 Developing
Welcome to RAIN! Research Administration Information Network
 Welcome to RAIN! Research Administration Information Network DATE: March 17, 2016 HOSTED BY: Andrew Watson RAIN Agenda with Andrew Watson Introduction Updates and timely information Special guest: CFS
Welcome to RAIN! Research Administration Information Network DATE: March 17, 2016 HOSTED BY: Andrew Watson RAIN Agenda with Andrew Watson Introduction Updates and timely information Special guest: CFS
Subrecipient Risk Assessment and Monitoring of Northeastern University Issued Subawards
 Subrecipient Risk Assessment and Monitoring of Northeastern University Issued Subawards What is a Subaward? A Subaward is a contractual agreement between Northeastern University and a third party organization
Subrecipient Risk Assessment and Monitoring of Northeastern University Issued Subawards What is a Subaward? A Subaward is a contractual agreement between Northeastern University and a third party organization
Sponsored Programs New Developments and Important Reminders
 Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs March 7, 2012 1 Training Session Overview National Institutes of Health (NIH) Update NSF Proposal
Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs March 7, 2012 1 Training Session Overview National Institutes of Health (NIH) Update NSF Proposal
MUSC Center for Global Health Request for Applications (RFA) for Faculty Pilot Project Grants
 MUSC Center for Global Health Request for Applications (RFA) for Faculty Pilot Project Grants RFA Release Date: Monday, February 1, 2016 Full Proposal (5 page limit) Deadline: March 16, 2016, 12:00 p.m.
MUSC Center for Global Health Request for Applications (RFA) for Faculty Pilot Project Grants RFA Release Date: Monday, February 1, 2016 Full Proposal (5 page limit) Deadline: March 16, 2016, 12:00 p.m.
NSF 2 Month Handbook. Effective for Reviews Performed as of 07/01/17. NSF Account Management. Updated 07/24/17
 Updated 07/24/17 NSF 2 Month Handbook Effective for Reviews Performed as of 07/01/17 NSF Account Management Table of Contents Contents 2 Month Rule Policy... 2 NSF Definition... 2 Sponsor/University Guidance:...
Updated 07/24/17 NSF 2 Month Handbook Effective for Reviews Performed as of 07/01/17 NSF Account Management Table of Contents Contents 2 Month Rule Policy... 2 NSF Definition... 2 Sponsor/University Guidance:...
AURA Meeting. July 23, 2014
 AURA Meeting July 23, 2014 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored
AURA Meeting July 23, 2014 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored
Outgoing Subagreements: Subawards and Subcontracts
 Effective Date: March 1, 2014 Version: 1 Page: 1 of 6 Overview Sponsored programs may include partnerships with other organizations or institutions whereby a portion of the project is conducted by investigators
Effective Date: March 1, 2014 Version: 1 Page: 1 of 6 Overview Sponsored programs may include partnerships with other organizations or institutions whereby a portion of the project is conducted by investigators
Research Administration Forum March 9, Marcia Smith Associate Vice Chancellor for Research
 Research Administration Forum March 9, 2017 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements - Marcia Smith Immigration Update and Q & A Sam Nahidi, Assistant Director,
Research Administration Forum March 9, 2017 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements - Marcia Smith Immigration Update and Q & A Sam Nahidi, Assistant Director,
October electronic Research Administration (era) OER, OD, National Institutes of Health
 October 2016 electronic Research Administration (era) OER, OD, National Institutes of Health Scarlett Gibb era Customer Relationship Manager, era Commons Joe Schumaker era Communications and Outreach 2
October 2016 electronic Research Administration (era) OER, OD, National Institutes of Health Scarlett Gibb era Customer Relationship Manager, era Commons Joe Schumaker era Communications and Outreach 2
Sponsored Programs and Research Compliance SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a subaward proposal to University of North Carolina Wilmington (UNCW). Please complete this form and send all required
SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a subaward proposal to University of North Carolina Wilmington (UNCW). Please complete this form and send all required
Uniform Guidance Subpart D Administrative Requirements. Why This Session Is Needed. Lesson Overview & Module Objectives
 Uniform Guidance Subpart D Administrative Requirements 1 Why This Session Is Needed Changes impact grant operations and policies Changes to recipient and subrecipient responsibilities 2 Lesson Overview
Uniform Guidance Subpart D Administrative Requirements 1 Why This Session Is Needed Changes impact grant operations and policies Changes to recipient and subrecipient responsibilities 2 Lesson Overview
Office of Research Administration Quarterly Research Administrators Meeting 10/27/2016
 Office of Research Administration Quarterly Research Administrators Meeting 10/27/2016 Agenda RCC Update Julie Cole HR Training Requirements Betsy Hames My Research Home Rebecca Brouwer Duke Apps Beth
Office of Research Administration Quarterly Research Administrators Meeting 10/27/2016 Agenda RCC Update Julie Cole HR Training Requirements Betsy Hames My Research Home Rebecca Brouwer Duke Apps Beth
University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures
 Summary 1. Subaward Definitions A. Subaward B. Subrecipient University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures C. Office of Contracts and Grants (OCG) 2. Distinguishing
Summary 1. Subaward Definitions A. Subaward B. Subrecipient University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures C. Office of Contracts and Grants (OCG) 2. Distinguishing
PeopleSoft Grants 3/3/ /1/2017 Phase 1 cutover initiated.
 PeopleSoft Grants 3/3/2017 3/1/2017 Phase 1 cutover initiated. 3/6/2017 OCG stops creating sponsored project cost centers and/or modifications in PS finance 3/13/2017 UH IT will setup award profiles in
PeopleSoft Grants 3/3/2017 3/1/2017 Phase 1 cutover initiated. 3/6/2017 OCG stops creating sponsored project cost centers and/or modifications in PS finance 3/13/2017 UH IT will setup award profiles in
Research Administration Services Roles & Responsibilities For Grants and Contracts (Excludes Clinical Trials) Version 3.1
 Research Administration Services Roles & Responsibilities For Grants and Contracts (Excludes Clinical Trials) Version 3.1 Category PI/PI Staff Department* RAS Unit School/Unit ORA Offices Relevant SOP
Research Administration Services Roles & Responsibilities For Grants and Contracts (Excludes Clinical Trials) Version 3.1 Category PI/PI Staff Department* RAS Unit School/Unit ORA Offices Relevant SOP
IRES Proposal Tracking (PT) Presented by: Kathi Goodfriend Office of Sponsored Projects Revised 03/15/2018 PRN: 5/14/ :19 PM
 IRES Proposal Tracking (PT) Presented by: Kathi Goodfriend Office of Sponsored Projects Revised 03/15/2018 PRN: 5/14/2018 12:19 PM Topics Covered Class Objective:. To become more familiar with the IRES
IRES Proposal Tracking (PT) Presented by: Kathi Goodfriend Office of Sponsored Projects Revised 03/15/2018 PRN: 5/14/2018 12:19 PM Topics Covered Class Objective:. To become more familiar with the IRES
Financial Conflict of Interest Training
 Financial Conflict of Interest Training This module is required for all Vanderbilt Faculty and Staff who receive support through the Public Health Service (PHS). Training provided by: VU Office of Conflict
Financial Conflict of Interest Training This module is required for all Vanderbilt Faculty and Staff who receive support through the Public Health Service (PHS). Training provided by: VU Office of Conflict
MUSC Center for Global Health Request for Applications (RFA) for Faculty Pilot Project Grants
 MUSC Center for Global Health Request for Applications (RFA) for Faculty Pilot Project Grants RFA Release Date: Monday, April 10, 2017 Full Proposal (6 page limit) Deadline: May 16, 2017 12 p.m. (noon)
MUSC Center for Global Health Request for Applications (RFA) for Faculty Pilot Project Grants RFA Release Date: Monday, April 10, 2017 Full Proposal (6 page limit) Deadline: May 16, 2017 12 p.m. (noon)
Grants Management Workshop. Proposal and Award Management Support
 Grants Management Workshop Proposal and Award Management Support Agenda Sponsored Project Administration UVM and RSENR Support Systems Provide an overview of the grant life cycle For each stage of the
Grants Management Workshop Proposal and Award Management Support Agenda Sponsored Project Administration UVM and RSENR Support Systems Provide an overview of the grant life cycle For each stage of the
Sponsored Projects Manual
 Sponsored Projects Manual University Policies and Procedures for Requesting, Accepting, and Administering Grants, Contracts, and Cooperative Agreements February 2, 2005 CONTENTS 1. Overview... 4 1.1. Purpose
Sponsored Projects Manual University Policies and Procedures for Requesting, Accepting, and Administering Grants, Contracts, and Cooperative Agreements February 2, 2005 CONTENTS 1. Overview... 4 1.1. Purpose
Principal Investigator User Guide
 INFOED ELECTRONIC RESEARCH ADMINISTRATION infoed.clemson.edu Principal Investigator User Guide November 2014 Version 1.7 i Document Revisions Date Version Number Document Changes 12/06/2013 1.0 Initial
INFOED ELECTRONIC RESEARCH ADMINISTRATION infoed.clemson.edu Principal Investigator User Guide November 2014 Version 1.7 i Document Revisions Date Version Number Document Changes 12/06/2013 1.0 Initial
2017 NIH Update. Presented by Stephanie Smith and Stacey Wade
 2017 NIH Update Presented by Stephanie Smith and Stacey Wade PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs NIH INSTITUTES National
2017 NIH Update Presented by Stephanie Smith and Stacey Wade PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs NIH INSTITUTES National
SYSTEM REQUIREMENTS AND USEFUL INFORMATION LOGGING INTO THE PERIS PORTAL
 SYSTEM REQUIREMENTS AND USEFUL INFORMATION ------------------------------------------------- LOGGING INTO THE PERIS PORTAL -------------------------------------------------------------------------- CREATING
SYSTEM REQUIREMENTS AND USEFUL INFORMATION ------------------------------------------------- LOGGING INTO THE PERIS PORTAL -------------------------------------------------------------------------- CREATING
FAQ S FOR UNIFORM GUIDANCE
 FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
Enter a Subcontract in erpm
 This procedure details the process of adding a subcontract (SUBK) to a project. I. PAF: indicating that a project will include a subcontract II. Subcontract Worksheet: Adding a subcontract III. PAF Workspace:
This procedure details the process of adding a subcontract (SUBK) to a project. I. PAF: indicating that a project will include a subcontract II. Subcontract Worksheet: Adding a subcontract III. PAF Workspace:
2016 NIH Update. Presented by Stephanie Smith, Stacey Wade, and Jennifer Webster
 2016 NIH Update Presented by Stephanie Smith, Stacey Wade, and Jennifer Webster PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs Jennifer
2016 NIH Update Presented by Stephanie Smith, Stacey Wade, and Jennifer Webster PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs Jennifer
D. PROPOSAL DETAILS CREATE A NEW PROPOSAL GENERAL INFO ORGANIZATION ADD INVESTIGATORS AND KEY PERSONS CREDIT SPLIT SPECIAL REVIEW D.3.
 D. PROPOSAL DETAILS D. D. D.3. D.4. D.5. D.6. D.7. D.8. D.9. D.10. D.1 D.1 CREATE A NEW PROPOSAL GENERAL INFO ORGANIZATION ADD INVESTIGATORS AND KEY PERSONS CREDIT SPLIT SPECIAL REVIEW ABSTRACT OTHER YNQ
D. PROPOSAL DETAILS D. D. D.3. D.4. D.5. D.6. D.7. D.8. D.9. D.10. D.1 D.1 CREATE A NEW PROPOSAL GENERAL INFO ORGANIZATION ADD INVESTIGATORS AND KEY PERSONS CREDIT SPLIT SPECIAL REVIEW ABSTRACT OTHER YNQ
NIH Agency Specifics August 11, 2015
 NIH Agency Specifics August 11, 2015 Maria Skinner Office of Sponsored Programs Stacy Salisbury Contract & Grant Administration NIH Pre-Award Presented by: Maria Skinner Office of Sponsored Programs (OSP)
NIH Agency Specifics August 11, 2015 Maria Skinner Office of Sponsored Programs Stacy Salisbury Contract & Grant Administration NIH Pre-Award Presented by: Maria Skinner Office of Sponsored Programs (OSP)
Trigger / Timing / Frequency: When a new award is received by the University and OSP determines that the award can be accepted.
 Kuali Research User Guide: Create a New Parent Award Version October 06 Purpose: To create a new parent award record in the system. Trigger / Timing / Frequency: When a new award is received by the University
Kuali Research User Guide: Create a New Parent Award Version October 06 Purpose: To create a new parent award record in the system. Trigger / Timing / Frequency: When a new award is received by the University
Preparing for a RPPR Submission
 Preparing for a RPPR Submission Review current NoA Review most current budget and expenditure information. Determine unobligated balance in order to respond to 2590 questions. Review last esnap if applicable
Preparing for a RPPR Submission Review current NoA Review most current budget and expenditure information. Determine unobligated balance in order to respond to 2590 questions. Review last esnap if applicable
THE SCHOOL OF NURSING FACULTY GRANT WRITER S HANDBOOK
 THE SCHOOL OF NURSING FACULTY GRANT WRITER S HANDBOOK September 2017 1 TABLE OF CONTENTS Table of Common Acronyms 3 Office of Grant Support Functions and Key Research Office Staff 4 Overview of Pre-and
THE SCHOOL OF NURSING FACULTY GRANT WRITER S HANDBOOK September 2017 1 TABLE OF CONTENTS Table of Common Acronyms 3 Office of Grant Support Functions and Key Research Office Staff 4 Overview of Pre-and
Agenda. Workday Training Session for Grants. Tuesday, December 15, :30 am 12:00pm. Office Depot Conference Room OD 101. Time Topic Presenter
 Agenda Workday Training Session for Grants Tuesday, December 15, 2015 9:30 am 12:00pm Office Depot Conference Room OD 101 Time Topic Presenter 9:30 9:40 am Welcome and Introductions Heather Saunders 9:40
Agenda Workday Training Session for Grants Tuesday, December 15, 2015 9:30 am 12:00pm Office Depot Conference Room OD 101 Time Topic Presenter 9:30 9:40 am Welcome and Introductions Heather Saunders 9:40
Research Administrator Meeting Tuesday, January 23, :00-12:00 Illinois Room (348), IMU
 Research Administrator Meeting Tuesday, January 23, 2018 10:00-12:00 Illinois Room (348), IMU Agenda: 1. GAO Announcements 2. Costing Considerations 3. Quick Reference Guide Overview 4. DSP General Announcements
Research Administrator Meeting Tuesday, January 23, 2018 10:00-12:00 Illinois Room (348), IMU Agenda: 1. GAO Announcements 2. Costing Considerations 3. Quick Reference Guide Overview 4. DSP General Announcements
Sponsored Programs New Developments and Important Reminders
 Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs January 23, 2018 1 NSF Updates NIH Updates Federal Updates Defense Base Act (DBA) Insurance OSP Updates
Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs January 23, 2018 1 NSF Updates NIH Updates Federal Updates Defense Base Act (DBA) Insurance OSP Updates
Federal Rules for Sponsored Programs. Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards 2 CFR 200
 Federal Rules for Sponsored Programs Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards 2 CFR 200 Uniform Guidance (UG) The Basics Presented by Dan Evon Director
Federal Rules for Sponsored Programs Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards 2 CFR 200 Uniform Guidance (UG) The Basics Presented by Dan Evon Director
The Lifecycle of a Sponsored Project
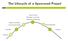 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
December 2015 Research Administration Working Group WELCOME OFFICE OF THE VICE PRESIDENT FOR RESEARCH
 December 2015 Research Administration Working Group WELCOME Open Mike Fringe Rates FY17 proposed Budget v. Charging Distribution of Indirects PI award letter out & accounts funded Departments- this month
December 2015 Research Administration Working Group WELCOME Open Mike Fringe Rates FY17 proposed Budget v. Charging Distribution of Indirects PI award letter out & accounts funded Departments- this month
UC Davis Policy and Procedure Manual
 UC Davis Policy and Procedure Manual Chapter 230, Sponsored Programs Section 07, Public Health Service Regulations on Objectivity in Research Date: Supersedes: 8/24/12 Responsible Department: Office of
UC Davis Policy and Procedure Manual Chapter 230, Sponsored Programs Section 07, Public Health Service Regulations on Objectivity in Research Date: Supersedes: 8/24/12 Responsible Department: Office of
Policy on Cost Allocation, Cost Recovery, and Cost Sharing
 President Page 1 of 11 PURPOSE: Provide guidance and structure when allocating and documenting costs (direct and indirect) for extramurally funded awards. Serves to provide direction for budgeting, allocating
President Page 1 of 11 PURPOSE: Provide guidance and structure when allocating and documenting costs (direct and indirect) for extramurally funded awards. Serves to provide direction for budgeting, allocating
Gina Billiot, CRA Grant/Contract Specialist 2 (ERA) Office of Sponsored Programs (OSP)
 Gina Billiot, CRA Grant/Contract Specialist 2 (ERA) Office of Sponsored Programs (OSP) SPS - System Features SPS - System Overview Workspace Proposal Creation Demonstration Resources LOUISIANA STATE UNIVERSITY
Gina Billiot, CRA Grant/Contract Specialist 2 (ERA) Office of Sponsored Programs (OSP) SPS - System Features SPS - System Overview Workspace Proposal Creation Demonstration Resources LOUISIANA STATE UNIVERSITY
Grant Application Package
 Generates errors for all applications Grant Application Package Opportunity Title: Research Project Grant (Parent R01) Offering Agency: National Institutes of Health Generates errors in certain situations
Generates errors for all applications Grant Application Package Opportunity Title: Research Project Grant (Parent R01) Offering Agency: National Institutes of Health Generates errors in certain situations
Procedure for Addressing PHS Animal Protocol-Proposal Congruency Requirements at the UMass Medical School
 Section I. Introduction A. In recent years, the NIH Office of Laboratory Animal Welfare has increasingly emphasized the importance of NIH-funded institutions proactively ensuring that their Investigators
Section I. Introduction A. In recent years, the NIH Office of Laboratory Animal Welfare has increasingly emphasized the importance of NIH-funded institutions proactively ensuring that their Investigators
Grant Application Package
 Grant Application Package Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Research
Grant Application Package Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Research
office of research administration newsletter
 july/august 2010 vol. 5 no. 1 office of research administration newsletter Yale has a clear obligation to comply with all regulations pertaining to the administration of federal grants, and we will spare
july/august 2010 vol. 5 no. 1 office of research administration newsletter Yale has a clear obligation to comply with all regulations pertaining to the administration of federal grants, and we will spare
Policy and Compliance: Working Together Like Hand in Glove
 Policy and Compliance: Working Together Like Hand in Glove Samuel Ashe, Director, Division of Grants Policy, OPERA, OER Diane W. Dean, Director, Division of Grants Compliance and Oversight, OPERA, OER
Policy and Compliance: Working Together Like Hand in Glove Samuel Ashe, Director, Division of Grants Policy, OPERA, OER Diane W. Dean, Director, Division of Grants Compliance and Oversight, OPERA, OER
Roles & Responsibilities UR Office of Research and Project Administration
 Roles and Responsibilities - UR ORPA Roles & Responsibilities UR Office of Research and Project Administration The Office of Research and Projects Administration (ORPA) is the administrative office legally
Roles and Responsibilities - UR ORPA Roles & Responsibilities UR Office of Research and Project Administration The Office of Research and Projects Administration (ORPA) is the administrative office legally
SERVICE LEVEL AGREEMENT: University of Arkansas for Medical Sciences OFFICE OF SPONSORED PROGRAMS ADMINISTRATIVE NETWORK (OSPAN)
 SERVICE LEVEL AGREEMENT: University of Arkansas for Medical Sciences OFFICE OF SPONSORED PROGRAMS ADMINISTRATIVE NETWORK (OSPAN) Table of Contents Complete later Section I General Information and Objectives
SERVICE LEVEL AGREEMENT: University of Arkansas for Medical Sciences OFFICE OF SPONSORED PROGRAMS ADMINISTRATIVE NETWORK (OSPAN) Table of Contents Complete later Section I General Information and Objectives
Sponsored Program Administration. October 10, 2017
 Sponsored Program Administration October 10, 2017 Agenda Procurement Services FDP Updates Jaggaer Pre-Award Federal Agency Updates Grants.gov Transition to Workspace NIH FORMS-E Post-Award GONE Act Certificate
Sponsored Program Administration October 10, 2017 Agenda Procurement Services FDP Updates Jaggaer Pre-Award Federal Agency Updates Grants.gov Transition to Workspace NIH FORMS-E Post-Award GONE Act Certificate
MAXIMUS Higher Education Practice
 Federal Compliance Webinar Series 2 CFR 200 Uniform Guidance Kris Rhodes, Director, MAXIMUS Jason Guilbeault, Senior Consultant, MAXIMUS 1 MAXIMUS Higher Education Practice Headquartered in Northbrook,
Federal Compliance Webinar Series 2 CFR 200 Uniform Guidance Kris Rhodes, Director, MAXIMUS Jason Guilbeault, Senior Consultant, MAXIMUS 1 MAXIMUS Higher Education Practice Headquartered in Northbrook,
Research Administrators Group (RAG) Meeting July 2015
 Research Administrators Group (RAG) Meeting July 2015 Office of Sponsored Projects 1 Agenda Office of Sponsored Projects 2 Introduction of New Staff Proposal/PD Update Proposal Metrics RPPR Transition
Research Administrators Group (RAG) Meeting July 2015 Office of Sponsored Projects 1 Agenda Office of Sponsored Projects 2 Introduction of New Staff Proposal/PD Update Proposal Metrics RPPR Transition
Capacity Building Grant Program (Section 4 and RCB) DRGR Guidance DRGR Action Plan Module Guide
 Capacity Building Grant Program (Section 4 and RCB) DRGR Guidance DRGR Action Plan Module Guide Background Starting in Fiscal Year 2015 (FY15), Section 4 and Rural Capacity Building Program Grantees (
Capacity Building Grant Program (Section 4 and RCB) DRGR Guidance DRGR Action Plan Module Guide Background Starting in Fiscal Year 2015 (FY15), Section 4 and Rural Capacity Building Program Grantees (
Opportunity Finance Network Guide to CDFI Program (Financial Assistance and Technical Assistance) FY2015 v.3 October 23, 2014
 Opportunity Finance Network Guide to CDFI Program (Financial Assistance and Technical Assistance) FY2015 v.3 October 23, 2014 This guide reflects an analysis of the FY2015 Notice of Funds Available (NOFA),
Opportunity Finance Network Guide to CDFI Program (Financial Assistance and Technical Assistance) FY2015 v.3 October 23, 2014 This guide reflects an analysis of the FY2015 Notice of Funds Available (NOFA),
Uniform Guidance Subpart D Administrative Requirements
 Uniform Guidance Subpart D Administrative Requirements Purpose and Introduction Understanding the Uniform Guidance is essential to increase accountability of managing grant funds. The Administrative Requirements
Uniform Guidance Subpart D Administrative Requirements Purpose and Introduction Understanding the Uniform Guidance is essential to increase accountability of managing grant funds. The Administrative Requirements
Office of Academic Grants and Sponsored Research Financial Conflict of Interest Disclosure, Review, and Management Procedures
 Office of Academic Grants and Sponsored Research Financial Conflict of Interest Disclosure, Review, and Management Procedures I. Financial Conflict of Interest Disclosures A. Mandatory Investigator Disclosures
Office of Academic Grants and Sponsored Research Financial Conflict of Interest Disclosure, Review, and Management Procedures I. Financial Conflict of Interest Disclosures A. Mandatory Investigator Disclosures
Principal Investigator
 Appendix A: Roles and Responsibilities Details Principal Investigator 1. Proposal Preparation 1.1 Has an overall responsibility to assemble a submission ready proposal meaning a complete, accurate proposal
Appendix A: Roles and Responsibilities Details Principal Investigator 1. Proposal Preparation 1.1 Has an overall responsibility to assemble a submission ready proposal meaning a complete, accurate proposal
IRB Approval of Research Using Survey Procedures Kerry Agnitsch, Office for Responsible Research October 1, 2009
 IRB Approval of Research Using Survey Procedures Kerry Agnitsch, Office for Responsible Research October 1, 2009 Key Definitions (per 45 CFR 46.102(d) & (f): Research a systematic investigation, including
IRB Approval of Research Using Survey Procedures Kerry Agnitsch, Office for Responsible Research October 1, 2009 Key Definitions (per 45 CFR 46.102(d) & (f): Research a systematic investigation, including
Quickguide to Creating a Proposal in Kuali Coeus
 Proposal Tab v.1 Revised 7/18/2014 Panel Field Action Notes Proposal Development Guide Reference Page(s) Enter a description of the proposal in this format: Document Overview Description Due Date xx/xx/xx_pi
Proposal Tab v.1 Revised 7/18/2014 Panel Field Action Notes Proposal Development Guide Reference Page(s) Enter a description of the proposal in this format: Document Overview Description Due Date xx/xx/xx_pi
Commodity Credit Corporation and Foreign Agricultural Service. Notice of Funding Availability: Inviting Applications for the Emerging Markets
 This document is scheduled to be published in the Federal Register on 05/09/2018 and available online at https://federalregister.gov/d/2018-09866, and on FDsys.gov Billing Code 3410 10 DEPARTMENT OF AGRICULTURE
This document is scheduled to be published in the Federal Register on 05/09/2018 and available online at https://federalregister.gov/d/2018-09866, and on FDsys.gov Billing Code 3410 10 DEPARTMENT OF AGRICULTURE
Overview of the NIH Career Development Programs
 NIH Office of Extramural Research EB2010/FASEB April 2010 Overview of the NIH Career Development Programs Henry Khachaturian, Ph.D. NIH Extramural Program Policy Officer Office of Extramural Research,
NIH Office of Extramural Research EB2010/FASEB April 2010 Overview of the NIH Career Development Programs Henry Khachaturian, Ph.D. NIH Extramural Program Policy Officer Office of Extramural Research,
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE
 THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research organizations aimed at streamlining
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research organizations aimed at streamlining
