Rebecca Trahan. Office of Sponsored Programs December 9, ORED Limited Submission Update
|
|
|
- Domenic Hicks
- 6 years ago
- Views:
Transcription
1 Rebecca Trahan Office of Sponsored Programs December 9, NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1
2 3 4 2
3 NSF 16-1 effective 1/25/2016 Effective for proposals submitted or due on or after 1/25/ Deadline of 5pm submitter s time will be strictly enforced by FastLane. FastLane will not accept proposals submitted at 5:00:01 or later. Only Authorized Representative can submit proposals, proposal file updates, revised budgets, and notifications and requests. Source: Jean Feldman s webinar, NSF Proposal & Award Policy Update, 10/29/
4 Biographical Sketch Collaborators section will no longer be a part of biosketch. Page limitation remains 2 pages. Can no longer upload biosketches as single PDF under PI. For compliance checking Special treatment of biosketches for Other Personnel and Equipment Users. Included under Other Supplementary Documents and clearly labeled Other Personnel Single Copy Documents Collaborators and Other Affiliates will now be uploaded here. Removed from Biosketch Used to help identity potential conflicts of bias in the selection of reviewers. Removes demographic Information about PIs and Co-PIs since this is part of PI profile in FastLane. Include address and institutional affiliation in List of Suggested Reviewers 7 Project Summary Stresses that only summaries that use special characters may be uploaded as a PDF under Supplementary Documents. Project Narrative Results from Prior NSF Support Clarifies to include NSF funding with a start date in the past five years. One each per PI and Co-PI Provides examples of types of awards to include. Clarifies to provide one award for each investigator. Additional instructions to address use of vertebrate animals. Current and Pending Support Can no longer upload Current and Pending Support as single PDF. For compliance checking Clarification that internal funds allocated towards a specific project must be listed. 8 4
5 Supplementary Documents The content of any letters of collaboration should be limited to the language included in this section. Additional changes Clarifies the information that must be included for proposals that include the use of vertebrate animals. Incorporates NSF s implementation of Dual Use Research of Concern. Reporting Final project report and Project Outcomes Report for awards issued under NSF 16-1 will be due 120 days after end date. 9 NSF-PAR (Public Access Repository) created to act as NSF publication repository Will be launched at end of PI s can voluntarily deposit publications into NSF-PAR Required for awards made from proposals submitted on or after January 25, 2016 First awards are expected June-July 2016 First publications requiring deposit into NSF-PAR will be in Fall See NSF Public Access FAQs. 10 5
6 Program Solicitation Specific Compliance Checks Warning messages will appear if the following are not included: References Cited Biosketches Budget Justification for Primary Organization Budget Justification for Subaward Organization Current and Pending Support Facilities, Equipment, and Other Resources 11 Effective October 24, 2015 Most NSF Notifications and Requests have moved to Research.gov Notifications and Requests that remain in FastLane are: Requests to add a Subaward PI Transfers Change PI and Add/Change Co-PI You can view status of all Notifications and Requests in both FastLane and Research.gov. Use NSF login credentials for Research.gov. 12 6
7 13 NOT-OD and NOT-OD Effective for due dates on or after January 25, Revised FORMS-C application guide (posted November 25, 2015) Updates to address Rigor and Transparency See NOT-OD Purpose is to enhance reproducibility of research findings through increased scientific rigor and transparency. Revised instructions to Significance and Approach sections of the Research Strategy attachment New Authentication of Key Biological and/or Chemical Resources attachment to be included in Other Attachments section of Other Project Information form. Limited to one page Not required if not part of the proposal. Reviewers will be asked to consider additional rigor and transparency questions. NIH Rigor and Reproducibility website: NIH Rigor and Reproducibility FAQ:
8 NOT-OD and NOT-OD Revised instructions for Vertebrate Animals sections. Redefining the age of child as 18 (vs. 21) for NIH inclusion of children policy related to human subject research. Changes to Research Training Plan for training grants to institute above. Revised Sample NIH Grants.gov Adobe package version C will soon be uploaded to OSP website. 15 NOT-OD Effective for due dates on or after May 25, New FORMS-D application guide Includes Institutional Training and Individual Fellowship Applications Revised instructions and program announcements expected by March 25, Authentication of Key Biological and/or Chemical Resources attachment moved to PHS398 Research Plan Not required if not part of the proposal. May see changes to Appendix Policy. Adding PHS Assignment Request Form to provide consistent way to request institute and study section preference, potential reviewer conflicts, and list of scientific expertise needed to review the application. (see NOT-OD ) New Font Guidelines (see NOT-OD ) Clarifications to Biosketch instructions 16 8
9 NOT-OD and NOT-OD Issued November 25, Applies to awards with budget periods beginning on or after October 1, 2015 Reminder that NIH may withdraw an application without review if non-compliant (Sec ) NIH will allow recipients to reduce effort during a no-cost extension without prior approval (Sec ) Inventions and Patents will need be reported electronically to NIH using iedison (Sec ) See NOT-OD
10 Incorporation of the Uniform Guidance. Draft posted to federal register on 10/14/ day public comment period. Posted on NSF s website. Would apply to research and research-related grants issued by: Dept. of Commerce/NOAA/NIST Dept. of Energy EPA NASA NSF DHHS/NIH USDA/NIFA Dept. of Transportation/FAA Dept. of Homeland Security Closeout Proposes to change due date for final financial, performance and other reports from 90 days to 120 days Fixed amount Subawards Proposes to waive prior approval requirement in UG provided amount is less than $150k paragraph (c) Direct Costs Proposes to waive the prior approval requirement in UG to direct charge salary of administrative and clerical staff provided conditions in 2 CFR are met Participant support costs (PSC) Proposes to waive the prior approval requirement in UG to add these costs to budget. Note: requires prior approval to rebudget funds budgeted for PSC to another category. Note: Agency Specific Requirements could differ from RTC. Stay tuned
11 2 CFR and 2 CFR As a Pass-Through Entity, LSU is responsible for oversight of the Federal awards issued to LSU. We must monitor activities under the Federal award to assure compliance with applicable Federal requirements and performance expectations. LSU s monitoring of subrecipients must include review of financial and programmatic reports required under the Subaward
12 New ORED Info Ready Portal for all limited submission competitions: lsu.infoready4.com The portal will also be used for ORED internal competitions Competitions will be announced via the ORED Newsletter and the Deans and Directors list serve notifications will come from research.lsu.edu or Letter of Intent vs. Competition All programs will initially be listed under the Letter of Intent heading. All faculty may submit a LOI for open programs. Once a program moves to the Competition stage, only those who have submitted a LOI may submit an internal preproposal. 23 Award Cycles and Deadlines Award Cycle Due Date Action Open Internal Submission Deadline Competition is open and accepting letters of intent. Available Agency Deadline The internal submission deadline has passed, but LSU has not reached the submission limit. Proposals will be accepted on a first-to-notify basis. If interested, submit a letter of intent. Pending Internal Preproposal Deadline/ Funding Agency Deadline The number of letters of intent exceeded the funding agency limit. LSU is conducting an internal competition, and additional LOI are no longer being accepted. Closed Funding Agency Deadline Proposals have been selected to apply for the program, and no additional applications will be accepted
13 25 Updated to address Uniform Guidance (UG) Definition of Institutional Base Salary added (I.C.8.i) Revised definition of Administrative/Clerical Support per UG (I.C.8.i) Clarified that fellowships do not receive tuition exemptions (I.C.8.iii) Added Fabricated Equipment (I.C.8.iv) Added Subrecipient vs. Contractor Determination from UG (I.C.8.vii) 26 13
14 Added 10% de minimis F&A rate for subrecipients with no negotiated rate from UG (I.C.8.vii) Added Participant Support Costs (I.C.8.xii) Clarified that this category does not include payments to participants in research studies. Included reminder for department administrators who assist faculty by creating and routing proposals in SPS to consult with investigators to answer the Special Approvals/Proposal Information questions in SPS. (II.B.2.b) Added export controls and financial interest to Special Approvals section to coincide with recently added SPS coversheet questions (II.B.2.b.i.h and II.B.2.b.i.h i.) 27 Did you know that OSP has a budget template modeled after the Grants.gov SF 424 (R&R) detailed budget template? Key features: Calculates individual fringe benefits Calculates tuition remission Ability to calculate F&A using different F&A base types (e.g. Salaries, Wages, and Fringe or Total Direct Costs) Includes a signature line on Cumulative Budget if using for a proposal when LSU is the Subrecipient. Reminder: Make sure to use current budget templates on OSP website that include signature line for Darya Courville
15 Welcome our new staff! Stephanie Scheffel, Operations Coordinator 29 National Council of University Research Administrators (NCURA) conferences in New Orleans Financial Research Administration (FRA) March 7-8, 2016 Optional workshops on March 6 th Pre-Award Research Administration (PRA) March 9-10, 2016 Optional workshops on March 8 th Early Bird Registration ends January 22nd 30 15
16 31 Location: 202 Himes Hall Website: Phone: Fax: Rebecca Trahan: 32 16
Sponsored Programs New Developments and Important Reminders
 Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs January 23, 2018 1 NSF Updates NIH Updates Federal Updates Defense Base Act (DBA) Insurance OSP Updates
Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs January 23, 2018 1 NSF Updates NIH Updates Federal Updates Defense Base Act (DBA) Insurance OSP Updates
Rebecca Trahan Office of Sponsored Programs January 10, 2017
 Rebecca Trahan Office of Sponsored Programs January 10, 2017 1 NSF Update NIH Update OSP Updates and Reminders Q & A 2 1 3 4 2 NSF 17-1 effective 1/30/2017 Effective for proposals submitted or due on or
Rebecca Trahan Office of Sponsored Programs January 10, 2017 1 NSF Update NIH Update OSP Updates and Reminders Q & A 2 1 3 4 2 NSF 17-1 effective 1/30/2017 Effective for proposals submitted or due on or
Sponsored Programs New Developments and Important Reminders
 Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs March 7, 2012 1 Training Session Overview National Institutes of Health (NIH) Update NSF Proposal
Sponsored Programs New Developments and Important Reminders Rebecca Trahan Office of Sponsored Programs March 7, 2012 1 Training Session Overview National Institutes of Health (NIH) Update NSF Proposal
Grant Application Package
 Generates errors for all applications Grant Application Package Opportunity Title: Research Project Grant (Parent R01) Offering Agency: National Institutes of Health Generates errors in certain situations
Generates errors for all applications Grant Application Package Opportunity Title: Research Project Grant (Parent R01) Offering Agency: National Institutes of Health Generates errors in certain situations
Grant Application Package
 Grant Application Package Generates errors for all applications Generates errors in certain situations Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition
Grant Application Package Generates errors for all applications Generates errors in certain situations Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition
Rebecca Trahan Office of Sponsored Programs January 22, 2014
 Rebecca Trahan Office of Sponsored Programs January 22, 2014 1 NSF Updates NIH Updates OMB Super Circular Responsible Conduct of Research OSP Updates Q & A 2 1 NSF 14-001 effective 2/24/2014 Effective
Rebecca Trahan Office of Sponsored Programs January 22, 2014 1 NSF Updates NIH Updates OMB Super Circular Responsible Conduct of Research OSP Updates Q & A 2 1 NSF 14-001 effective 2/24/2014 Effective
December 2015 Research Administration Working Group WELCOME OFFICE OF THE VICE PRESIDENT FOR RESEARCH
 December 2015 Research Administration Working Group WELCOME Open Mike Fringe Rates FY17 proposed Budget v. Charging Distribution of Indirects PI award letter out & accounts funded Departments- this month
December 2015 Research Administration Working Group WELCOME Open Mike Fringe Rates FY17 proposed Budget v. Charging Distribution of Indirects PI award letter out & accounts funded Departments- this month
National Science Foundation Update. SRA Annual Meeting October 20, 2015
 National Science Foundation Update SRA Annual Meeting October 20, 2015 Speakers Jean Feldman Head, Policy Office Division of Institution & Award Support Office of Budget, Finance & Award Management 703.292.8243;
National Science Foundation Update SRA Annual Meeting October 20, 2015 Speakers Jean Feldman Head, Policy Office Division of Institution & Award Support Office of Budget, Finance & Award Management 703.292.8243;
Page 1 of 10 1/31/2008
 Page 1 of 10 1/31/2008 Step by Step Tips to Avoid Errors and Warnings o Full instructions can be found at: http://grants1.nih.gov/grants/funding/424/index.htm. o This document is intended to assist you
Page 1 of 10 1/31/2008 Step by Step Tips to Avoid Errors and Warnings o Full instructions can be found at: http://grants1.nih.gov/grants/funding/424/index.htm. o This document is intended to assist you
Research Grant Resources & Information for New Investigators
 Research Grant Resources & Information for New Investigators FEDERAL RESEARCH FUNDING 2018-2019 University of Nevada, Reno College of Engineering 1 P a g e Table of Contents A. Federal Funding Agencies.
Research Grant Resources & Information for New Investigators FEDERAL RESEARCH FUNDING 2018-2019 University of Nevada, Reno College of Engineering 1 P a g e Table of Contents A. Federal Funding Agencies.
2016 NIH Update. Presented by Stephanie Smith, Stacey Wade, and Jennifer Webster
 2016 NIH Update Presented by Stephanie Smith, Stacey Wade, and Jennifer Webster PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs Jennifer
2016 NIH Update Presented by Stephanie Smith, Stacey Wade, and Jennifer Webster PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs Jennifer
Navigating Grant Resources. 10/3/2018 Dr. Susan Fell
 Navigating Grant Resources 10/3/2018 Dr. Susan Fell sfell@fsu.edu 850-645-2172 Sponsored Research Administration https://www.research.fsu.edu/ Applying for a Grant Opportunity Does this require sign-off
Navigating Grant Resources 10/3/2018 Dr. Susan Fell sfell@fsu.edu 850-645-2172 Sponsored Research Administration https://www.research.fsu.edu/ Applying for a Grant Opportunity Does this require sign-off
NSF Update: 17-1 Proposal & Award Policies & Procedures Guide (PAPPG)
 NSF Update: 17-1 Proposal & Award Policies & Procedures Guide (PAPPG) Daniel Lewis Sponsored Programs Administrator dlewis15@utk.edu 865-974-8873 Raymond Norris Sponsored Programs Administrator rnorris6@utk.edu
NSF Update: 17-1 Proposal & Award Policies & Procedures Guide (PAPPG) Daniel Lewis Sponsored Programs Administrator dlewis15@utk.edu 865-974-8873 Raymond Norris Sponsored Programs Administrator rnorris6@utk.edu
Grant/Sponsor Related Systems. Department and OSP Perspectives on ERA
 Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
Grant/Sponsor Related Systems Department and OSP Perspectives on ERA Grant/Sponsor Related Systems General Proposal Submission Information ERA & Sponsor Systems National Institutes of Health BC internal
SYSTEM REQUIREMENTS AND USEFUL INFORMATION LOGGING INTO THE PERIS PORTAL
 SYSTEM REQUIREMENTS AND USEFUL INFORMATION ------------------------------------------------- LOGGING INTO THE PERIS PORTAL -------------------------------------------------------------------------- CREATING
SYSTEM REQUIREMENTS AND USEFUL INFORMATION ------------------------------------------------- LOGGING INTO THE PERIS PORTAL -------------------------------------------------------------------------- CREATING
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM. December 2017
 Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Gina Billiot, CRA Grant/Contract Specialist 2 (ERA) Office of Sponsored Programs (OSP)
 Gina Billiot, CRA Grant/Contract Specialist 2 (ERA) Office of Sponsored Programs (OSP) SPS - System Features SPS - System Overview Workspace Proposal Creation Demonstration Resources LOUISIANA STATE UNIVERSITY
Gina Billiot, CRA Grant/Contract Specialist 2 (ERA) Office of Sponsored Programs (OSP) SPS - System Features SPS - System Overview Workspace Proposal Creation Demonstration Resources LOUISIANA STATE UNIVERSITY
Sonia Esperança Program Director; Directorate for Geosciences; Division of Earth Sciences
 Proposal Preparation Panelists David Campbell Acting Division Director and Program Director; Directorate for Education & Human Resources; Division of Research on Learning in Formal & Informal Settings
Proposal Preparation Panelists David Campbell Acting Division Director and Program Director; Directorate for Education & Human Resources; Division of Research on Learning in Formal & Informal Settings
OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015
 OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015 INTRODUCTION The University of Wisconsin-Milwaukee (UWM) is the prime recipient on a wide range of sponsored awards
OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015 INTRODUCTION The University of Wisconsin-Milwaukee (UWM) is the prime recipient on a wide range of sponsored awards
Federal Demonstration Partnership, NIH Updates, & Grants/Cooperative Agreement Reminders November 13, 2015
 Federal Demonstration Partnership, NIH Updates, & Grants/Cooperative Agreement Reminders November 13, 2015 Federal Demonstration Partnership BACKGROUND 154 participating institutions Goal: Improve Government
Federal Demonstration Partnership, NIH Updates, & Grants/Cooperative Agreement Reminders November 13, 2015 Federal Demonstration Partnership BACKGROUND 154 participating institutions Goal: Improve Government
FAQ S FOR UNIFORM GUIDANCE
 FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
Research Administration Frequently Asked Questions
 Research Administration Frequently Asked Questions I am submitting a proposal to NIH, but I can t download the grants.gov package, how do I submit? A. Use an old grants.gov package from a previous submission
Research Administration Frequently Asked Questions I am submitting a proposal to NIH, but I can t download the grants.gov package, how do I submit? A. Use an old grants.gov package from a previous submission
Narration: Welcome to the Anatomy of an Administrative Shell mini course.
 Welcome to the Anatomy of an Administrative Shell mini course. 1 If you have previously joined us for other sponsored project mini courses, you will be familiar with the Sponsored Project Life Cycle. In
Welcome to the Anatomy of an Administrative Shell mini course. 1 If you have previously joined us for other sponsored project mini courses, you will be familiar with the Sponsored Project Life Cycle. In
The Mechanics of Proposal Submission
 The Mechanics of Proposal Submission Jeanette Bernard- Snyder Senior Research Administrator Office of Sponsored Programs Julie Longo Technical Writer College of Engineering Table of Contents Resources
The Mechanics of Proposal Submission Jeanette Bernard- Snyder Senior Research Administrator Office of Sponsored Programs Julie Longo Technical Writer College of Engineering Table of Contents Resources
NSF 17-1 January 30, Significant Changes and Clarifications to the PAPPG. Overall Document
 NSF 17-1 January 30, 2017 Significant Changes and Clarifications to the PAPPG Overall Document The PAPPG has been modified in its entirety, to remove all references to the Grant Proposal Guide (GPG) and
NSF 17-1 January 30, 2017 Significant Changes and Clarifications to the PAPPG Overall Document The PAPPG has been modified in its entirety, to remove all references to the Grant Proposal Guide (GPG) and
NSF Grants Conference NSF Policies and Procedures Update
 Slide 1 NSF Grants Conference NSF Policies and Procedures Update March 11-12, 2013 Hosted by Howard University, Arlington, Virginia Slide 2 Panelist Jean Feldman Head, Policy Office, Division of Institution
Slide 1 NSF Grants Conference NSF Policies and Procedures Update March 11-12, 2013 Hosted by Howard University, Arlington, Virginia Slide 2 Panelist Jean Feldman Head, Policy Office, Division of Institution
RESEARCH ADMINISTRATION SERIES: SESSION 2 BUDGET PREPARATION WORKSHOP Amber Jensen Sarah Marcotte November 17 th, 2016
 RESEARCH ADMINISTRATION SERIES: SESSION 2 BUDGET PREPARATION WORKSHOP Amber Jensen Sarah Marcotte November 17 th, 2016 What we are covering today: Proposal Announcement Proposal Docs Budget & Budget Justification
RESEARCH ADMINISTRATION SERIES: SESSION 2 BUDGET PREPARATION WORKSHOP Amber Jensen Sarah Marcotte November 17 th, 2016 What we are covering today: Proposal Announcement Proposal Docs Budget & Budget Justification
Effective January 25, 2016
 Effective January 25, 2016 Pam Baker-Webber, Senior Director of Research Administration, SEAS Jennifer Lech, Sponsored Research Officer, FAS/RAS Marrybell Ramos, Manager, Grants & Contracts Operations,
Effective January 25, 2016 Pam Baker-Webber, Senior Director of Research Administration, SEAS Jennifer Lech, Sponsored Research Officer, FAS/RAS Marrybell Ramos, Manager, Grants & Contracts Operations,
FY 2015 Continuation of Solicitation for the Office of Science Financial Assistance Program Funding Opportunity Number: DE-FOA
 Department of Energy/ Office of Science FY 2015 Continuation of Solicitation for the Office of Science Financial Assistance Program Funding Opportunity Number: DE-FOA-0001204 This announcement will remain
Department of Energy/ Office of Science FY 2015 Continuation of Solicitation for the Office of Science Financial Assistance Program Funding Opportunity Number: DE-FOA-0001204 This announcement will remain
Sponsored Program Administration
 Sponsored Program Administration March, 2018 research.uconn.edu AGENDA OVPR/SPS News and Information IPR Form FCOI SPS Procedures for Fund Set Up Banner NIH News and Information Federal Executive Pay Scale
Sponsored Program Administration March, 2018 research.uconn.edu AGENDA OVPR/SPS News and Information IPR Form FCOI SPS Procedures for Fund Set Up Banner NIH News and Information Federal Executive Pay Scale
The Metis Foundation Office of Grant and Contract Administration SUBRECIPIENT INFORMATION AND COMPLIANCE FORM
 The Metis Foundation Office of Grant and Contract Administration SUBRECIPIENT INFORMATION AND COMPLIANCE FORM All subrecipients must complete this form when submitting a proposal to the Metis Foundation
The Metis Foundation Office of Grant and Contract Administration SUBRECIPIENT INFORMATION AND COMPLIANCE FORM All subrecipients must complete this form when submitting a proposal to the Metis Foundation
Jean Feldman Head, Policy Office, Office of Budget, Finance & Award Management; Division of Institution & Award Support
 NSF Grants Conference Proposal Preparation October 6-7, 2014 Hosted by The George Washington University, Arlington, Virginia Panelists Jean Feldman Head, Policy Office, Office of Budget, Finance & Award
NSF Grants Conference Proposal Preparation October 6-7, 2014 Hosted by The George Washington University, Arlington, Virginia Panelists Jean Feldman Head, Policy Office, Office of Budget, Finance & Award
A Walk Through The SF424 (R&R) Marcia Hahn Office of Policy for Extramural Research Administration, OER, NIH January 11, 2006
 A Walk Through The SF424 (R&R) Marcia Hahn Office of Policy for Extramural Research Administration, OER, NIH January 11, 2006 1 Handouts for This Session A Walk Through The SF424 (R&R) (Powerpoint Presentation
A Walk Through The SF424 (R&R) Marcia Hahn Office of Policy for Extramural Research Administration, OER, NIH January 11, 2006 1 Handouts for This Session A Walk Through The SF424 (R&R) (Powerpoint Presentation
National Science Foundation (NSF) Update --- Spring Federal Demonstration Partnership Meeting May 13, 2013
 National Science Foundation (NSF) Update --- Spring 2013 Federal Demonstration Partnership Meeting May 13, 2013 Ask Early, Ask Often Jean Feldman Head, Policy Office Division of Institution & Award Support
National Science Foundation (NSF) Update --- Spring 2013 Federal Demonstration Partnership Meeting May 13, 2013 Ask Early, Ask Often Jean Feldman Head, Policy Office Division of Institution & Award Support
NIH R-Mechanism Mini Guide for Completing SF424 (R&R) Applications Using Grants.gov *Adobe Forms Version B2*
 NIH R-Mechanism Mini Guide for Completing SF424 (R&R) Applications Using Grants.gov *Adobe Forms Version B2* Please note that this mini guide is a tool for assisting in correctly preparing a grant application
NIH R-Mechanism Mini Guide for Completing SF424 (R&R) Applications Using Grants.gov *Adobe Forms Version B2* Please note that this mini guide is a tool for assisting in correctly preparing a grant application
RESEARCH ADMINISTRATION NEWSLETTER
 RESEARCH ADMINISTRATION NEWSLETTER http://www.brown.edu/research/research-administration-newsletters SEPTEMBER 2016 The monthly newsletter provides timely information on agency updates; sponsor and University
RESEARCH ADMINISTRATION NEWSLETTER http://www.brown.edu/research/research-administration-newsletters SEPTEMBER 2016 The monthly newsletter provides timely information on agency updates; sponsor and University
Principal Investigator User Guide
 INFOED ELECTRONIC RESEARCH ADMINISTRATION infoed.clemson.edu Principal Investigator User Guide November 2014 Version 1.7 i Document Revisions Date Version Number Document Changes 12/06/2013 1.0 Initial
INFOED ELECTRONIC RESEARCH ADMINISTRATION infoed.clemson.edu Principal Investigator User Guide November 2014 Version 1.7 i Document Revisions Date Version Number Document Changes 12/06/2013 1.0 Initial
Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32)
 Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32) Overview: This document is intended to help you complete a KC System to System (S2S) proposal submission
Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32) Overview: This document is intended to help you complete a KC System to System (S2S) proposal submission
Subrecipient Monitoring Procedures
 Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
Federal Rules for Sponsored Programs. Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards 2 CFR 200
 Federal Rules for Sponsored Programs Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards 2 CFR 200 Uniform Guidance (UG) The Basics Presented by Dan Evon Director
Federal Rules for Sponsored Programs Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards 2 CFR 200 Uniform Guidance (UG) The Basics Presented by Dan Evon Director
OSP Checklist - NIH Pathway to Independence Award (Parent K99/R00). Page 1 of 5
 OSP Checklist - NIH Pathway to Independence Award (Parent K99/R00). Page 1 of 5 ü Read the solicitation: https://researchtraining.nih.gov/programs/career-development ü In all cases the SF 424 and instructions
OSP Checklist - NIH Pathway to Independence Award (Parent K99/R00). Page 1 of 5 ü Read the solicitation: https://researchtraining.nih.gov/programs/career-development ü In all cases the SF 424 and instructions
Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed:
 COEUS NIH-GRANTS.GOV PROPOSAL REVIEW CHECKLIST REVIEWER: Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed: Has the Program Announcement been reviewed? What is
COEUS NIH-GRANTS.GOV PROPOSAL REVIEW CHECKLIST REVIEWER: Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed: Has the Program Announcement been reviewed? What is
Instructions for Application Submission National MS Society-American Brain Foundation (ABF) Clinician Scientist Development Award
 Instructions for Application Submission National MS Society-American Brain Foundation (ABF) Clinician Scientist Development Award INTRODUCTION Please read these instructions and follow them carefully.
Instructions for Application Submission National MS Society-American Brain Foundation (ABF) Clinician Scientist Development Award INTRODUCTION Please read these instructions and follow them carefully.
Instructions for Application Submission Sylvia Lawry Physician Fellowship
 Instructions for Application Submission Sylvia Lawry Physician Fellowship INTRODUCTION Please read these instructions and follow them carefully. Applications that are incomplete exceed the page limitations,
Instructions for Application Submission Sylvia Lawry Physician Fellowship INTRODUCTION Please read these instructions and follow them carefully. Applications that are incomplete exceed the page limitations,
Minimum Proposal Requirements for Routing
 Minimum Proposal Requirements for Routing This QuickCard contains the minimum requirements for routing a proposal for approval in Coeus. Review the sponsor s solicitation for specific requirements and
Minimum Proposal Requirements for Routing This QuickCard contains the minimum requirements for routing a proposal for approval in Coeus. Review the sponsor s solicitation for specific requirements and
NSF FUNDAMENTALS WORKSHOP. Thomas Jefferson University December 2017
 NSF FUNDAMENTALS WORKSHOP Thomas Jefferson University December 2017 SPEAKERS Jean Feldman Head, Policy Office Division of Institution and Award Support Office of Budget, Finance and Award Management 703.292.8243;
NSF FUNDAMENTALS WORKSHOP Thomas Jefferson University December 2017 SPEAKERS Jean Feldman Head, Policy Office Division of Institution and Award Support Office of Budget, Finance and Award Management 703.292.8243;
UNIFORM GUIDANCE IMPLEMENTATION
 UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
Grant Application Package
 Grant Application Package Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Methodology
Grant Application Package Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Methodology
Grant Application Package
 Grant Application Package Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Research
Grant Application Package Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Research
The Lifecycle of a Sponsored Project
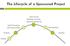 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where does the
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where does the
Topics Today. Revised Research Terms and Conditions (RTC) NSF Pilot Identification of Collaborators at Proposal. Encore Presentation
 Topics Today Revised Research Terms and Conditions (RTC) NSF Pilot Identification of Collaborators at Proposal Encore Presentation Account Set-Up Overview 1 Federal-Wide Research Terms and Conditions April
Topics Today Revised Research Terms and Conditions (RTC) NSF Pilot Identification of Collaborators at Proposal Encore Presentation Account Set-Up Overview 1 Federal-Wide Research Terms and Conditions April
2017 State of Connecticut Regenerative Medicine Research Fund (RMRF) General RFP
 2017 State of Connecticut Regenerative Medicine Research Fund (RMRF) General RFP (Seed, Established Investigator, Group) Full RFP, Forms and additional FAQs: http://ctinnovations.com/rmrf Deadline Friday,
2017 State of Connecticut Regenerative Medicine Research Fund (RMRF) General RFP (Seed, Established Investigator, Group) Full RFP, Forms and additional FAQs: http://ctinnovations.com/rmrf Deadline Friday,
National Science Foundation Fall Grants Conference Pittsburgh, PA - November 14 & 15 - Carnegie Mellon University
 Proposal Preparation National Science Foundation Fall Grants Conference Pittsburgh, PA - November 14 & 15 - Carnegie Mellon University Panelists Sankar Basu Program Director, Directorate for Computer &
Proposal Preparation National Science Foundation Fall Grants Conference Pittsburgh, PA - November 14 & 15 - Carnegie Mellon University Panelists Sankar Basu Program Director, Directorate for Computer &
NIH ASSIST Multi-Component NIH Submissions Updated: August 7, 2018
 NIH ASSIST Multi-Component NIH Submissions Updated: August 7, 2018 Training Outline Class Overview Multi-Component NIH Submission Definition Submitting a Multi-Component Application to NIH Resources Learning
NIH ASSIST Multi-Component NIH Submissions Updated: August 7, 2018 Training Outline Class Overview Multi-Component NIH Submission Definition Submitting a Multi-Component Application to NIH Resources Learning
The Lifecycle of a Sponsored Project
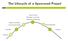 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
SUBRECIPIENT COMMITMENT FORM
 SECTION A SUBRECIPIENT INFORMATION SUBRECIPIENT COMMITMENT FORM Phone: Email: SUBRECIPIENT: CU PI: PROPOSAL TITLE: TOTAL FUNDING REQUESTED: ORIGINATING SPONSOR, IF A PASS THRU APPLICATION: PRIME SPONSOR:
SECTION A SUBRECIPIENT INFORMATION SUBRECIPIENT COMMITMENT FORM Phone: Email: SUBRECIPIENT: CU PI: PROPOSAL TITLE: TOTAL FUNDING REQUESTED: ORIGINATING SPONSOR, IF A PASS THRU APPLICATION: PRIME SPONSOR:
Sponsored Programs Roles & Responsibilities
 The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Preparing campus for what is changing and what is not What is Grants Reform and when is it effective? What
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Preparing campus for what is changing and what is not What is Grants Reform and when is it effective? What
Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6
 Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6 KC-PRE28: Prepare Summary Proposal KC-AWD22: Research Gifts November 13, 2013 Goals and Outcomes Goals Obtain feedback on the proposed
Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6 KC-PRE28: Prepare Summary Proposal KC-AWD22: Research Gifts November 13, 2013 Goals and Outcomes Goals Obtain feedback on the proposed
Subrecipient vs. Contractor: Guidance on Appropriate Classification of Legal Relationship
 HARVARD UNIVERSITY Responsible Office: Office for Sponsored Programs Date First Effective: October 18, 2011 Revision Date: December 26, 2014 Subrecipient vs. Contractor: Guidance on Appropriate Classification
HARVARD UNIVERSITY Responsible Office: Office for Sponsored Programs Date First Effective: October 18, 2011 Revision Date: December 26, 2014 Subrecipient vs. Contractor: Guidance on Appropriate Classification
Sponsored Programs Roles & Responsibilities
 The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
OMB Uniform Guidance ( UG ) Briefing. ASRSP & OSR Brown Bag Tuesday, January 27 th
 OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
Proposal Preparation and Submission February 2018
 Proposal Preparation and Submission February 2018 Introductions We are. Chris Dye-Hixenbaugh, C&G Officer, Proposals Brooke Herevia, C&G Analyst, Proposals Shanna Nation Jose, C&G Analyst, Proposals Kassie
Proposal Preparation and Submission February 2018 Introductions We are. Chris Dye-Hixenbaugh, C&G Officer, Proposals Brooke Herevia, C&G Analyst, Proposals Shanna Nation Jose, C&G Analyst, Proposals Kassie
Agenda. NIH Update. Other Updates. Proposal & Progress Report Statistics Research Administration Training Topic:
 Agenda NIH Update Salary Cap Guidance NOT OD 17 037: NIH Implementation of the Interim RPPR While a Renewal Application is Under Consideration Prior Approval for No Cost Extensions via the era Commons
Agenda NIH Update Salary Cap Guidance NOT OD 17 037: NIH Implementation of the Interim RPPR While a Renewal Application is Under Consideration Prior Approval for No Cost Extensions via the era Commons
Research Administration Demonstration Training Series
 Research Administration Demonstration Training Series UTSW Proposal Development & Submission Jamie F. Wilson Supervisor, Pre-Award Administration (Proposals) Lifecycle of an Award Identify Funding Opportunity
Research Administration Demonstration Training Series UTSW Proposal Development & Submission Jamie F. Wilson Supervisor, Pre-Award Administration (Proposals) Lifecycle of an Award Identify Funding Opportunity
FINANCE-315 7/1/2017 SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a proposal to UACES. It provides a checklist of documents and certifications required by sponsors, as well as an
SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a proposal to UACES. It provides a checklist of documents and certifications required by sponsors, as well as an
TABLE OF CONTENTS Guidelines About the Leukemia & Lymphoma Society Description of Awards Who Can Apply General Eligibility Criteria
 - TABLE OF CONTENTS Guidelines About the Leukemia & Lymphoma Society Description of Awards Who Can Apply General Eligibility Criteria Citizenship and Degree Leadership and Staffing Application Process
- TABLE OF CONTENTS Guidelines About the Leukemia & Lymphoma Society Description of Awards Who Can Apply General Eligibility Criteria Citizenship and Degree Leadership and Staffing Application Process
Principal Investigator
 Appendix A: Roles and Responsibilities Details Principal Investigator 1. Proposal Preparation 1.1 Has an overall responsibility to assemble a submission ready proposal meaning a complete, accurate proposal
Appendix A: Roles and Responsibilities Details Principal Investigator 1. Proposal Preparation 1.1 Has an overall responsibility to assemble a submission ready proposal meaning a complete, accurate proposal
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards (New Uniform Guidance)
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards (New Uniform Guidance) Spring Research Administrators Series March 19, 2015 1 Agenda Background UW-Madison
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards (New Uniform Guidance) Spring Research Administrators Series March 19, 2015 1 Agenda Background UW-Madison
NSF Checklist for Coeus
 NSF Checklist for Coeus General NSF and Coeus Requirements Due Date: Coeus # PI Name Allowable fonts Type Density 10 pt min: Arial, Courier New, Palatino Linotype; 11 pt min: Times New Roman, Computer
NSF Checklist for Coeus General NSF and Coeus Requirements Due Date: Coeus # PI Name Allowable fonts Type Density 10 pt min: Arial, Courier New, Palatino Linotype; 11 pt min: Times New Roman, Computer
Proposal Submission Timeline
 Proposal Submission Timeline Anytime 10 business days 5 business days 2 business day Identify Funding Notify GSP of planned proposal via PARS Proposal and Budget created in Cayuse SP Internal Routing and
Proposal Submission Timeline Anytime 10 business days 5 business days 2 business day Identify Funding Notify GSP of planned proposal via PARS Proposal and Budget created in Cayuse SP Internal Routing and
UPDATES. Meet the Proposal Deadline. NIH: Public Access to Research Results OFFICE OF SPONSORED INSIDE THIS ISSUE:
 UPDATES INSIDE THIS ISSUE: Meet the Proposal Deadline Responsible Conduct of Research New Federal Regulations on Conflict of Interest NSF s New Proposal Guidelines and Project Reporting Changes Rutgers
UPDATES INSIDE THIS ISSUE: Meet the Proposal Deadline Responsible Conduct of Research New Federal Regulations on Conflict of Interest NSF s New Proposal Guidelines and Project Reporting Changes Rutgers
NIH. Electronic Submissions
 NIH Electronic Submissions 1 HOW DOES IT WORK? PI finds FOA (Funding Opportunity Announcement) through Grants.gov and prints it out. PI double-checks that he/she has the correct Adobe software on his/her
NIH Electronic Submissions 1 HOW DOES IT WORK? PI finds FOA (Funding Opportunity Announcement) through Grants.gov and prints it out. PI double-checks that he/she has the correct Adobe software on his/her
Award Transfer Guidelines
 Award Transfer Guidelines The process of transferring awards from one institution to another is not a simple task and requires negotiation between the sponsoring agency, the old institution and the new
Award Transfer Guidelines The process of transferring awards from one institution to another is not a simple task and requires negotiation between the sponsoring agency, the old institution and the new
Grant Application Package
 Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Grant Application Package Reducing
Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Grant Application Package Reducing
Subawards and Subrecipient Monitoring
 Subawards and Subrecipient Monitoring H O L LY S O M M E R S D I R E C T O R, O S P G R A N T S J O S H R O S E N B E R G A C T I N G D I R E C T O R, O G C A Objectives 2 Distinguish Between: Procurement
Subawards and Subrecipient Monitoring H O L LY S O M M E R S D I R E C T O R, O S P G R A N T S J O S H R O S E N B E R G A C T I N G D I R E C T O R, O G C A Objectives 2 Distinguish Between: Procurement
Successful Submissions
 Successful Submissions Prepare Complete-Submit-Track-View Created by: Geeta Chhabra Electronic Submission of NIH Grant Applications Find and download announcement and application package Prepares the application
Successful Submissions Prepare Complete-Submit-Track-View Created by: Geeta Chhabra Electronic Submission of NIH Grant Applications Find and download announcement and application package Prepares the application
The LOI is only to allow organization of the review process, and all projects submitting an LOI will be permitted to submit a full application.
 A Complete Grant Application will include the following: 1) LOI Form submitted by May 15, 2017 An electronic letter of intent (LOI) submission form is required 30 days before the deadline for full application
A Complete Grant Application will include the following: 1) LOI Form submitted by May 15, 2017 An electronic letter of intent (LOI) submission form is required 30 days before the deadline for full application
Grant Application Package
 Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Grant Application Package NIH Research
Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Grant Application Package NIH Research
Grants Module. Create and Submit a Funding Proposal for Non System to System Submissions
 Grants Module Create and Submit a Funding Proposal for Non System to System Submissions Summary of Major Changes The following directions are a combination of all steps in the Funding Proposal submission
Grants Module Create and Submit a Funding Proposal for Non System to System Submissions Summary of Major Changes The following directions are a combination of all steps in the Funding Proposal submission
Introduction to Cayuse424
 Introduction to Cayuse424 In this training you will learn: How to: Navigate Cayuse424 Personalize your password Create Professional Profiles Create Budgets Create subcontract proposal, budget Initiate
Introduction to Cayuse424 In this training you will learn: How to: Navigate Cayuse424 Personalize your password Create Professional Profiles Create Budgets Create subcontract proposal, budget Initiate
University of the District of Columbia Handbook for Principal Investigators Prepared by the Office of Sponsored Programs Revision V: April 2017
 University of the District of Columbia Handbook for Principal Investigators Prepared by the Office of Sponsored Programs Revision V: April 2017 With Financial Support from Title III U.S. Department of
University of the District of Columbia Handbook for Principal Investigators Prepared by the Office of Sponsored Programs Revision V: April 2017 With Financial Support from Title III U.S. Department of
Slide 1. NSF Grants Conference. Proposal Preparation. March 11-12, 2013 Hosted by Howard University, Arlington, Virginia
 Slide 1 NSF Grants Conference Proposal Preparation March 11-12, 2013 Hosted by Howard University, Arlington, Virginia Slide 2 Panelists Sonia Esperança Program Director, Directorate for Geosciences, Division
Slide 1 NSF Grants Conference Proposal Preparation March 11-12, 2013 Hosted by Howard University, Arlington, Virginia Slide 2 Panelists Sonia Esperança Program Director, Directorate for Geosciences, Division
Principal Investigator
 Principal Investigator HEADER TEXT Award Management Roles & Responsibilities November 4, 2016 Susan Mundt, Chris Barnhill, & Jing Liu Agenda PI Definition and Eligibility PreAward Roles & Responsibilities
Principal Investigator HEADER TEXT Award Management Roles & Responsibilities November 4, 2016 Susan Mundt, Chris Barnhill, & Jing Liu Agenda PI Definition and Eligibility PreAward Roles & Responsibilities
Instructions for Submission: Pilot Grant Applications National Multiple Sclerosis Society 2018
 Instructions for Submission: Pilot Grant Applications National Multiple Sclerosis Society 2018 INTRODUCTION Please read these instructions and follow them carefully. Applications that are incomplete, exceed
Instructions for Submission: Pilot Grant Applications National Multiple Sclerosis Society 2018 INTRODUCTION Please read these instructions and follow them carefully. Applications that are incomplete, exceed
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE
 THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research organizations aimed at streamlining
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research organizations aimed at streamlining
NSF Proposal and Award Policies & Procedures Guide (PAPPG) Update. Office of Grants and Contracts Administration December 23, 2014
 NSF Proposal and Award Policies & Procedures Guide (PAPPG) Update Office of Grants and Contracts Administration December 23, 2014 New PAPPG (NSF 15-1) Takes effect for proposals submitted or due on or
NSF Proposal and Award Policies & Procedures Guide (PAPPG) Update Office of Grants and Contracts Administration December 23, 2014 New PAPPG (NSF 15-1) Takes effect for proposals submitted or due on or
2. DATE SUBMITTED 3. DATE RECEIVED BY STATE. 4. Federal Identifier 7. * TYPE OF APPLICANT: Other (Specify): 9. * NAME OF FEDERAL AGENCY: 10.
 DATE SUBMITTED Applicant Identifier RSP will enter the assigned PALS # here DATE RECEIVED BY STATE State Application Identifier APPLICATION FOR FEDERAL ASSISTANCE SF 424 (R&R) Read agency specific guidelines
DATE SUBMITTED Applicant Identifier RSP will enter the assigned PALS # here DATE RECEIVED BY STATE State Application Identifier APPLICATION FOR FEDERAL ASSISTANCE SF 424 (R&R) Read agency specific guidelines
Purpose... 1 Scope... 1 Definition... 2 Procedure... 2 Responsibilities... 5 Reference... 6
 Title: Guidelines for the Department of Medicine (DoM) Career Development Award (CDA) K Bridge Funding Program Effective Date: 01/01/2017 Table of Contents Purpose... 1 Scope... 1 Definition... 2 Procedure...
Title: Guidelines for the Department of Medicine (DoM) Career Development Award (CDA) K Bridge Funding Program Effective Date: 01/01/2017 Table of Contents Purpose... 1 Scope... 1 Definition... 2 Procedure...
OSR Monthly Meeting. August 16, 2018 (Chicago) August 21, 2018 (Evanston)
 OSR Monthly Meeting August 16, 2018 (Chicago) August 21, 2018 (Evanston) Agenda OSR Announcements/Staffing Updates ESPR Replacement Project Update Year-end Procedures NSF Updates NIH Biosketch Reminder
OSR Monthly Meeting August 16, 2018 (Chicago) August 21, 2018 (Evanston) Agenda OSR Announcements/Staffing Updates ESPR Replacement Project Update Year-end Procedures NSF Updates NIH Biosketch Reminder
Form Approved Through 03/31/2020 OMB No
 Form Approved Through 03/31/2020 OMB No. 0925-0001 Department of Health and Human Services Public Health Services Grant Application Do not exceed character length restrictions indicated. 1. TITLE OF PROJECT
Form Approved Through 03/31/2020 OMB No. 0925-0001 Department of Health and Human Services Public Health Services Grant Application Do not exceed character length restrictions indicated. 1. TITLE OF PROJECT
OSP Forum: The New Federal Research Terms and Conditions Prior Approval Matrix. August 7,
 OSP Forum: The New Federal Research Terms and Conditions Prior Approval Matrix August 7, 2017-1 Agenda Recent changes in Federal terms OSP resources and how to use them Similarities & differences among
OSP Forum: The New Federal Research Terms and Conditions Prior Approval Matrix August 7, 2017-1 Agenda Recent changes in Federal terms OSP resources and how to use them Similarities & differences among
Sponsored Programs and Research Compliance SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a subaward proposal to University of North Carolina Wilmington (UNCW). Please complete this form and send all required
SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a subaward proposal to University of North Carolina Wilmington (UNCW). Please complete this form and send all required
Proposal Development Guide
 Proposal Development Guide Contents The Proposal Tab... 3 Document Overview Panel... 3 Required Fields for Saving Document Panel... 3 Institutional Fields (Conditionally Required)... 8 Sponsor & Program
Proposal Development Guide Contents The Proposal Tab... 3 Document Overview Panel... 3 Required Fields for Saving Document Panel... 3 Institutional Fields (Conditionally Required)... 8 Sponsor & Program
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018
 CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
Grant Application Package
 Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Research Project Grant (Parent R01)
Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Research Project Grant (Parent R01)
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP)
 Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Research Proposal Application Process NUMBER: RAS SOP 1002 VERSION: 4.0 LAST REVISED: PREPARED BY: Office
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Research Proposal Application Process NUMBER: RAS SOP 1002 VERSION: 4.0 LAST REVISED: PREPARED BY: Office
Spartan RAN. Research Administrators Network Biannual Meeting April 23 rd, Before we get started, please follow the instructions on your table!
 Spartan RAN Research Administrators Network Biannual Meeting April 23 rd, 2015 Before we get started, please follow the instructions on your table! We re Going Green in 2015, so paper handouts will not
Spartan RAN Research Administrators Network Biannual Meeting April 23 rd, 2015 Before we get started, please follow the instructions on your table! We re Going Green in 2015, so paper handouts will not
2018 Innovation Grant. Application Guidelines. Due April 2, 2018
 2018 Innovation Grant Application Guidelines Due April 2, 2018 In this packet you will find: Grant Guidelines & Dates - pages 2 to 3 Application Outline and Formatting - pages 3 to 5 Applications will
2018 Innovation Grant Application Guidelines Due April 2, 2018 In this packet you will find: Grant Guidelines & Dates - pages 2 to 3 Application Outline and Formatting - pages 3 to 5 Applications will
Grant Application Package
 Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Health Promotion Among Racial and Ethnic
Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Health Promotion Among Racial and Ethnic
