I was asked to pass this along. Not too many of us use Petty Cash for our subjects, but for those who do, this might be helpful:
|
|
|
- Edith Robbins
- 6 years ago
- Views:
Transcription
1 January 2009 There has been some confusion noted lately about "safety" vs "efficacy" in clinical trials. In general: "safety tests" are tests that are done to monitor for side effects while "efficacy tests" are those that monitor how well the drug or device is working. Safety tests are things such as: lab tests, VS, EKGs, other 'safety' tests such as radiology, review of AEs/SAEs, etc. Efficacy tests would include: pharmacokinetics or scales that are done specifically to see if the drug is effective. For example: cognitive tests for Alzheimer's drugs or HAM-D scales for depression drugs. February 2009 I was asked to pass this along. Not too many of us use Petty Cash for our subjects, but for those who do, this might be helpful: Per UVa Policy, petty cash payments are limited to a maximum of $100 per payment and $599 per calendar year per individual. The web site address for this policy is The IRB-HSR has added this tip into their protocol template in order to make research personnel aware of this policy. March 2009 Personnel and Delegation of Duties forms When opening a new study, whether it is sponsored or Investigator-initiated (sometimes particularly if Investigator-initiated): It is recommended that you complete a Personnel and Delegation of Duties form. This is not a federally-required form, but it is exceptionally useful in identifying roles especially if you have a situation where staff rotates through, or no one person or group manages the whole study.
2 The form, in Word format, is attached. You may amend the duties as best suits your specific study. April 2009 We had an interesting issue come up recently: A trial involved a new procedure" which would be performed by UVA physicians. This was not a standard procedure being done in a new way, but a NEW PROCEDURE. The protocol required an extensive physician training program - with hands on skills and didactic training, etc. as well as proctored procedures here with the first subjects in the study. But, what it also required was that our physicians be "credentialed" here at UVA by the "Credentialing Committee". (see 2 attachments) There may be different situations in which you need to contact the Clinical Staff Office about credentialing. For instance: Your study requires a non-uva physician to perform a procedure here at UVA Your study requires that UVA physicians perform a new procedure, technique or treatment modality that has never been done at UVA before Your first step will be to call the Clinical Staff Office at to get help regarding applications and what paperwork you will need. In the event there are no approved criteria, the Medical Center shall first determine whether it will allow the new or additional Clinical Privilege. Applications for new, or additional Clinical Privileges, must be in writing and submitted by the Applicant as well as by the appropriate Department Chair. The Credentials Committee shall determine the conditions and requirements upon which any new or additional Clinical Privileges shall be granted including, but not limited to, how current competence will be demonstrated, any proctoring or other monitoring requirements. The Committee will recommend the requirements to the Clinical Staff Executive Committee (CSEC) for consideration. In turn, the CSEC shall make appropriate recommendations regarding new or additional Clinical Privileges to the Medical Center Operating Board for final determination. (The above taken from Clinical Staff Bylaws; section scroll down to Bylaws of the Clinical Staff)
3 May 2009 For study participants who may want to donate blood: If the study involves a drug that is not approved by the FDA, the American Red Cross guidelines state that the study participant may not donate blood for one year after they take their last dose of study drug. The following is allowed: Clinical trial participants who are in a non-study drug study (survey, blood draw only (depending on amount drawn for study), etc.) Clinical trial participants who have a device implanted that does not have a drug associated with it. Clinical trial participants who are participating in Stage IV studies where the drug is already approved by the FDA for another indication There are quite a few situations where they defer to their physician for his/her opinion as to whether or not the subjects can donate. If you or the study subject have any questions call the organization where they donate blood and talk to one of their nurses OR call the American Red Cross at GIVELIFE or and ask to speak to one of their nurses. June 2009 None submitted July 2009 Reminder regarding the very important topic of billing: All members of the study team need to have an understanding of the study contract's budget language in order to ensure that charges are billed correctly. No service or procedure that is paid for by a sponsor may be billed to insurance or Medicare, including what might otherwise be considered standard of care. The Informed Consent language in the Will being in this study cost you any money? section must be consistent with what is agreed to in the study contract. Preparing a Billing Coverage Analysis when beginning a study will identify each expected study charge and how it is to be billed. This must also reflect the study contract s budget language.
4 Please contact Kathy Richardson, School of Medicine Clinical Trials Office Budget and Billing coordinator, at or for assistance with preparing a Billing Coverage Analysis or with any clinical trials budget or billing issues. August 2009 There is still some confusion about device studies and the Office of Supply Chain Management, so we re re-sending the November TIP: (revised a bit) Ask yourself: Am I using a device, not currently used at UVA, in our research that is purchased, contracted, donated, or loaned for trial? IF the answer is YES: UVa (NOT IRB) policy requires that the device be evaluated by the Office of Supply Chain Management. The IRB does need a copy of the application for evaluation, but does not need a copy of the actual approval or an approval number. For your convenience, the information about obtaining Supply Chain Management evaluation is available on the IRB website ( and the application called the New Medical Device Request is present under Forms. IF your study is already approved by the IRB but you have not obtained Supply Chain Management review, please do so at this time. In order to obtain approval; complete the Form, fax a copy to the IRB at and then submit the form to Supply Chain Management. If there are questions on how to complete the form contact their office at For additional information see the following Hospital Policies: # 0076: Medical Devices Evaluation and Monitoring System # 0165: Safe Medical Devices Act Reporting October 2009 Tips for transporting samples by car Since some of you need to transport blood and/or urine specimens from your off-site clinic to the central lab at UVA, the following is information provided by Ericka Pearce during her Shipping of Hazardous Materials certification class for our continuing education series: Pack specimens, in a leak-proof primary container, with enough absorbent material to soak up any spillage
5 Put primary containers in a leak-proof package (possibly a leak-proof sealable shipping bag); but a leak-proof zip lock baggie will work Securely place everything in a separate closed box preferably in the trunk of the car If using dry ice be sure that the container is allowed to vent Have the attached courtesy letter with you whenever you transport specimens. **Please note that it would also be a good idea to always have a box of gloves and/or spill materials (a biohazard/red bag and Cavicide) with you. Call Ericka Pearce at if you have any questions Per Ericka, REMEMBER: this should be for patient specimens only. Category B cultures should be shipped by air or packaged as a Category B and transported using a trained and certified courier.
EMA Inspection Site perspective
 EMA Inspection Site perspective Hermien Gous Wits RHI Shandukani Research Centre 27.09.2016 Cape Town Why were we inspected times? Pharmaceutical company applied for registration of the study drug in a
EMA Inspection Site perspective Hermien Gous Wits RHI Shandukani Research Centre 27.09.2016 Cape Town Why were we inspected times? Pharmaceutical company applied for registration of the study drug in a
VCU Clinical Research Quality Assurance Assessment
 VCU Clinical Research Quality Assurance Assessment Principal Investigator Protocol Title Protocol IRB Number Name of Person Completing Assessment Date Assessment was Completed The goal of this assessment
VCU Clinical Research Quality Assurance Assessment Principal Investigator Protocol Title Protocol IRB Number Name of Person Completing Assessment Date Assessment was Completed The goal of this assessment
EMA & FDA Inspections: Site perspective. Shandukani Research Centre
 EMA & FDA Inspections: Site perspective Shandukani Research Centre Why were we inspected? Pharmaceutical company applied for registration of the study drug (Phase I/II dosing studies in paediatrics) Large
EMA & FDA Inspections: Site perspective Shandukani Research Centre Why were we inspected? Pharmaceutical company applied for registration of the study drug (Phase I/II dosing studies in paediatrics) Large
ARTICLE IV. MEDICAL STAFF CATEGORIES. The Active Staff shall consist of practitioners each of whom:
 ARTICLE IV. MEDICAL STAFF CATEGORIES A. ACTIVE STAFF. The Active Staff shall consist of practitioners each of whom: a. meets all the basic qualifications set forth in Article III; b. will be available
ARTICLE IV. MEDICAL STAFF CATEGORIES A. ACTIVE STAFF. The Active Staff shall consist of practitioners each of whom: a. meets all the basic qualifications set forth in Article III; b. will be available
Thank you, in advance, for being a partner in your care.
 477 Cooper Road, Suite 220 Westerville, OH 43081 614-818-0215 Your appointment with: Dr. David H. Brown Dr. Jed W. Henry Dr. Adam J. Clemens is scheduled for. Welcome to our practice. It is our desire
477 Cooper Road, Suite 220 Westerville, OH 43081 614-818-0215 Your appointment with: Dr. David H. Brown Dr. Jed W. Henry Dr. Adam J. Clemens is scheduled for. Welcome to our practice. It is our desire
Good Clinical Practice: A Ground Level View
 Good Clinical Practice: A Ground Level View Jeanna Julo, BA, BA, CCRP Assistant Director, Clinical Data Management & Quality Controls, Auditing & Training Clinical Research Administration Research Institute,
Good Clinical Practice: A Ground Level View Jeanna Julo, BA, BA, CCRP Assistant Director, Clinical Data Management & Quality Controls, Auditing & Training Clinical Research Administration Research Institute,
SPECIMEN REQUIREMENTS
 SPECIMEN REQUIREMENTS General Guidelines for Specimen Handling Specimen requirements generally include the requested volume, storage temperature, and any special handling notes. The requested volume provides
SPECIMEN REQUIREMENTS General Guidelines for Specimen Handling Specimen requirements generally include the requested volume, storage temperature, and any special handling notes. The requested volume provides
SARASOTA MEMORIAL HOSPITAL CANCER RESEARCH PROGRAM POLICY
 PS1006 SARASOTA MEMORIAL HOSPITAL CANCER RESEARCH PROGRAM POLICY TITLE: Satellite Site Management Plan Job Title of Reviewer: POLICY #: EFFECTIVE DATE: REVISED DATE: POLICY TYPE: Elizabeth Carr, R.N.,
PS1006 SARASOTA MEMORIAL HOSPITAL CANCER RESEARCH PROGRAM POLICY TITLE: Satellite Site Management Plan Job Title of Reviewer: POLICY #: EFFECTIVE DATE: REVISED DATE: POLICY TYPE: Elizabeth Carr, R.N.,
WARNING LETTER. an both of which were sponsored by. (formerly ). The products
 g5~5s c Public Health Service ' SLRV7CLS r r f+ ~1Mr~la DEPARTMENT OF HEALTH & HUMAN SERVICES DEC 2 1 2005 Food and Drug Administration 9200 Corporate Blvd. Rockville, Maryland 20850 WARNING LETTER Via
g5~5s c Public Health Service ' SLRV7CLS r r f+ ~1Mr~la DEPARTMENT OF HEALTH & HUMAN SERVICES DEC 2 1 2005 Food and Drug Administration 9200 Corporate Blvd. Rockville, Maryland 20850 WARNING LETTER Via
General Office and Patient Compliance Policies
 General Office and Patient Compliance Policies Thank you for choosing Innate Wellness & Medical Center as your medical provider. We are providing you this updated information to keep you informed of our
General Office and Patient Compliance Policies Thank you for choosing Innate Wellness & Medical Center as your medical provider. We are providing you this updated information to keep you informed of our
Documenting the Story of a Clinical Trial: Concept to CAPA. Lori T. Gilmartin Gilmartin Consulting LLC
 Documenting the Story of a Clinical Trial: Concept to CAPA Lori T. Gilmartin Gilmartin Consulting LLC The regulations represent the floor while ethical thinking is the sky. Dr. Thomas Moore Boston University
Documenting the Story of a Clinical Trial: Concept to CAPA Lori T. Gilmartin Gilmartin Consulting LLC The regulations represent the floor while ethical thinking is the sky. Dr. Thomas Moore Boston University
QUALITY ASSURANCE PROGRAM
 QUALITY ASSURANCE PROGRAM Elaine Armstrong, MS Quality Assurance Manager PURPOSE Verify accuracy of submitted data Verify compliance with protocol and regulatory requirements Provide educational support
QUALITY ASSURANCE PROGRAM Elaine Armstrong, MS Quality Assurance Manager PURPOSE Verify accuracy of submitted data Verify compliance with protocol and regulatory requirements Provide educational support
Clinical Trial Budgeting and Negotiation March 2018
 Clinical Trial Budgeting and Negotiation March 2018 Terry Stone Director, Clinical Trial Office Kati Cini Associate Director, Clinical Trial Office Workshop Objectives Basics of clinical trial budgeting
Clinical Trial Budgeting and Negotiation March 2018 Terry Stone Director, Clinical Trial Office Kati Cini Associate Director, Clinical Trial Office Workshop Objectives Basics of clinical trial budgeting
Step 1A: Before entering patient room, be sure you have all the material ready and available:
 RECOMMENDATIONS FOR SAFELY COLLECTION AND PROPERLY MANAGEMENT OF POTENTIALLY INFECTED SAMPLES WITH HIGHLY PATHOGENIC AGENTS 1 (Adapted from How to safely collect blood samples from persons suspected to
RECOMMENDATIONS FOR SAFELY COLLECTION AND PROPERLY MANAGEMENT OF POTENTIALLY INFECTED SAMPLES WITH HIGHLY PATHOGENIC AGENTS 1 (Adapted from How to safely collect blood samples from persons suspected to
TORRANCE MEMORIAL MEDICAL STAFF
 BYLAWS COMMITTEE: APPROVED WITH NO CHANGES 10/3/2017 Dates Approved: Medical Executive Committee 09/14/2010; 12/9/2014 PATIENT ATTRIBUTION PLAN: This Attribution Plan assures that all staff are able to
BYLAWS COMMITTEE: APPROVED WITH NO CHANGES 10/3/2017 Dates Approved: Medical Executive Committee 09/14/2010; 12/9/2014 PATIENT ATTRIBUTION PLAN: This Attribution Plan assures that all staff are able to
Dr. R. Sathianathan. Role & Responsibilities of Principal Investigators in Clinical Trials. 18 August 2015
 18 August 2015 Role & Responsibilities of Principal Dr. R. Sathianathan Professor of Psychiatry, SRMC, Porur & Former Director, Institute of Mental Health, Chennai Principal Investigators & GOOD CLINICAL
18 August 2015 Role & Responsibilities of Principal Dr. R. Sathianathan Professor of Psychiatry, SRMC, Porur & Former Director, Institute of Mental Health, Chennai Principal Investigators & GOOD CLINICAL
Policy on the collection, handling and transport of clinical specimens
 Policy on the collection, handling and transport of clinical specimens Page 1 of 6 Document Control Sheet Name of Document: Policy on the collection, handling and transport of clinical specimens Version:
Policy on the collection, handling and transport of clinical specimens Page 1 of 6 Document Control Sheet Name of Document: Policy on the collection, handling and transport of clinical specimens Version:
Mastering Clinical Research April 19, :30 am
 Mastering Clinical Research April 19, 2017 7:30 am New Question and Answer Response System Log In Directions Use the following link to access pre and post test questions: http://www.socrative.com/ Click
Mastering Clinical Research April 19, 2017 7:30 am New Question and Answer Response System Log In Directions Use the following link to access pre and post test questions: http://www.socrative.com/ Click
Radiology/Nuclear Medicine Section
 Huntington Hospital Radiology/Nuclear Medicine Section Rules and Regulations May 2013 HUNTINGTON MEMORIAL HOSPITAL RADIOLOGY/NUCLEAR MEDICINE SECTION RULES & REGULATIONS Table of Contents I. MEMBERSHIP...
Huntington Hospital Radiology/Nuclear Medicine Section Rules and Regulations May 2013 HUNTINGTON MEMORIAL HOSPITAL RADIOLOGY/NUCLEAR MEDICINE SECTION RULES & REGULATIONS Table of Contents I. MEMBERSHIP...
Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network of Clinical Research Professionals
 ORIENTATION FOR NEW CLINICAL RESEARCH COORDINATORS Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network of Clinical Research Professionals Overall Agenda for Orientation Module 1:
ORIENTATION FOR NEW CLINICAL RESEARCH COORDINATORS Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network of Clinical Research Professionals Overall Agenda for Orientation Module 1:
STANDARDIZED PROCEDURE FEMORAL VENOUS BLOOD DRAW (Adult, Peds)
 I. Definition The Femoral venous blood draw (FVBD) is the procedure of performing a needle stick into the femoral vein for the purpose of drawing blood work that will assist in lab monitoring. II. Background
I. Definition The Femoral venous blood draw (FVBD) is the procedure of performing a needle stick into the femoral vein for the purpose of drawing blood work that will assist in lab monitoring. II. Background
Preventing Medical Errors
 Presents Preventing Medical Errors Contact Hours: 2 First Published: March 31, 2017 This Course Expires on: March 31, 2019 Course Objectives Upon completion of this course, the nurse will be able to: 1.
Presents Preventing Medical Errors Contact Hours: 2 First Published: March 31, 2017 This Course Expires on: March 31, 2019 Course Objectives Upon completion of this course, the nurse will be able to: 1.
Project Expense Considerations. Study Budgeting Considerations Research Staff and Trainees Expense Projected Expense Estimated Cost & Notes
 These are items to consider when developing a budget for a clinical research study (includes site costs and cost to sponsor a study). The true estimates should be based upon the role (participating site
These are items to consider when developing a budget for a clinical research study (includes site costs and cost to sponsor a study). The true estimates should be based upon the role (participating site
REGISTERING A PATIENT
 REGISTERING A PATIENT Patient Eligibility It is important for the institution staff to review all eligibility criteria and follow-up requirements. A patient failing to meet all protocol eligibility requirements
REGISTERING A PATIENT Patient Eligibility It is important for the institution staff to review all eligibility criteria and follow-up requirements. A patient failing to meet all protocol eligibility requirements
SOP WP6-QUAL-04, Version 1.0, 23 February 2014 Page 1 of 8. SOP Title: Laboratory (GCLP) supervision visits
 SOP WP6-QUAL-04, Version 1.0, 23 February 2014 Page 1 of 8 SOP Title: Laboratory (GCLP) supervision visits Project/study: NIDIAG: this SOP applies to all NIDIAG clinical studies (WP2). 1. Scope and application
SOP WP6-QUAL-04, Version 1.0, 23 February 2014 Page 1 of 8 SOP Title: Laboratory (GCLP) supervision visits Project/study: NIDIAG: this SOP applies to all NIDIAG clinical studies (WP2). 1. Scope and application
HomeMed Information. for the UMHS Cancer Center
 HomeMed Information for the UMHS Cancer Center 1 In this manual you will find the following information: Your Health Care Team... HomeMed... 3 When to notify your team or HomeMed... 4 Infusion Pump Guide
HomeMed Information for the UMHS Cancer Center 1 In this manual you will find the following information: Your Health Care Team... HomeMed... 3 When to notify your team or HomeMed... 4 Infusion Pump Guide
WHAT IS AN IRB? WHAT IS AN IRB? 3/25/2015. Presentation Outline
 Education &Training WHAT IS AN IRB? Introduction to the UofL Institutional Review Boards & Human Subjects Protection Program IRB Review Process Post Approval Monitoring March 2015 1 Presentation Outline
Education &Training WHAT IS AN IRB? Introduction to the UofL Institutional Review Boards & Human Subjects Protection Program IRB Review Process Post Approval Monitoring March 2015 1 Presentation Outline
BIMO SITE AUDIT CHECKLIST
 Item AUTHORITY AND ADMINISTRATION FOR STUDIES INVOLVING HUMAN DRUGS, BIOLOGICS AND DEVICES 1. Compare the Investigator Agreement with the information provided by the assigning Center. Auditor will check
Item AUTHORITY AND ADMINISTRATION FOR STUDIES INVOLVING HUMAN DRUGS, BIOLOGICS AND DEVICES 1. Compare the Investigator Agreement with the information provided by the assigning Center. Auditor will check
C: Safety. Alberta Licensed Practical Nurses Competency Profile 23
 C: Alberta Licensed Practical Nurses Competency Profile 23 Competency: C-1 Fire Emergency C-1-1 C-1-2 C-1-3 C-1-4 C-1-5 C-1-6 Demonstrate ability to apply critical thinking and clinical judgment in response
C: Alberta Licensed Practical Nurses Competency Profile 23 Competency: C-1 Fire Emergency C-1-1 C-1-2 C-1-3 C-1-4 C-1-5 C-1-6 Demonstrate ability to apply critical thinking and clinical judgment in response
Name: Date of Birth: Phone: ( ) Gender: Mailing Address: City: State: Zip: Social Security Number:
 To apply for help in affording your Sunovion prescription, please mail or fax a completed application to: Sunovion Support Prescription Assistance Program ( Program ) PO Box 220285, Charlotte, NC 28222-0285
To apply for help in affording your Sunovion prescription, please mail or fax a completed application to: Sunovion Support Prescription Assistance Program ( Program ) PO Box 220285, Charlotte, NC 28222-0285
Audits/Inspections Be Prepared for Anything
 Audits/Inspections Be Prepared for Anything Practices, laboratories, institutions, and clinics that participate in clinical trials are subject to audits by a number of different entities. As a primary
Audits/Inspections Be Prepared for Anything Practices, laboratories, institutions, and clinics that participate in clinical trials are subject to audits by a number of different entities. As a primary
60 KNEES ROAD, PARK ORCHARDS, VICTORIA 3114
 60 KNEES ROAD, PARK ORCHARDS, VICTORIA 3114 POLICY: FIRST AID RATIONALE: At St Anne s we believe that the welfare of all people on the school site is a prime responsibility. In addition, all students and
60 KNEES ROAD, PARK ORCHARDS, VICTORIA 3114 POLICY: FIRST AID RATIONALE: At St Anne s we believe that the welfare of all people on the school site is a prime responsibility. In addition, all students and
Yes. Phone encounter? Part of screening process. Consent and enroll patient. Apply Treatment Plan. Verify link to Timeline. Conduct patient assessment
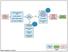 Referral - introduction to study Review consent and study Answer questions Review medical history Phone encounter? IRB approval Request required permission to schedule test (could change at screening)
Referral - introduction to study Review consent and study Answer questions Review medical history Phone encounter? IRB approval Request required permission to schedule test (could change at screening)
Essential Documents It s Not Just a Binder!
 Essential Documents It s Not Just a Binder! Kelly Unsworth, MS, CCRC, CIP Director of Research Education & Training Office for Human Subject Protection SCORE June 5, 2014 But What percentage of 2014 (to
Essential Documents It s Not Just a Binder! Kelly Unsworth, MS, CCRC, CIP Director of Research Education & Training Office for Human Subject Protection SCORE June 5, 2014 But What percentage of 2014 (to
Subject Screening, Recruitment, and Retention. Tiffany Morrison, MS, CCRP Director, Clinical Trials Rothman Institute
 Subject Screening, Recruitment, and Retention Tiffany Morrison, MS, CCRP Director, Clinical Trials Rothman Institute Learning objectives List two sources of potential subjects who can be screening for
Subject Screening, Recruitment, and Retention Tiffany Morrison, MS, CCRP Director, Clinical Trials Rothman Institute Learning objectives List two sources of potential subjects who can be screening for
CLOSE OUT VISIT REPORT (NO CRF TO MONITOR)
 Date: Page: 1 of 8 CLOSE OUT VISIT REPORT (NO CRF TO MONITOR) Protocol: PI Name: PI Address: Date of Visit: Monitor(s): Other Sponsor Personnel Present: Site Personnel Present at Visit (include names and
Date: Page: 1 of 8 CLOSE OUT VISIT REPORT (NO CRF TO MONITOR) Protocol: PI Name: PI Address: Date of Visit: Monitor(s): Other Sponsor Personnel Present: Site Personnel Present at Visit (include names and
STANDARDIZED PROCEDURE INFRARED COAGULATION THERAPY (Adults, Peds)
 I. Definition Infrared Coagulation (IRC) Therapy is a treatment for anogenital warts, low grade squamous intraepithelial neoplasia (LGAIN) or high grade squamous intraepithelial neoplasia (HGAIN). IRC
I. Definition Infrared Coagulation (IRC) Therapy is a treatment for anogenital warts, low grade squamous intraepithelial neoplasia (LGAIN) or high grade squamous intraepithelial neoplasia (HGAIN). IRC
STANDARDIZED PROCEDURE ARTERIAL CATHETER INSERTION (Adult)
 I. Definition: This protocol covers the task of arterial line insertion by an Advanced Health Practitioner. The purpose of this standardized procedure is to allow the Advanced Health Practitioner to safely
I. Definition: This protocol covers the task of arterial line insertion by an Advanced Health Practitioner. The purpose of this standardized procedure is to allow the Advanced Health Practitioner to safely
Loyola University Chicago Health Sciences Division Maywood, IL. Human Subject Research Project Start-Up Guide
 Loyola University Chicago Health Sciences Division Maywood, IL Human Subject Research Project Start-Up Guide This Start-Up Guide is intended to guide you through the process of designing a research project
Loyola University Chicago Health Sciences Division Maywood, IL Human Subject Research Project Start-Up Guide This Start-Up Guide is intended to guide you through the process of designing a research project
APEC Preliminary Workshop: Review of Drug Development in Clinical Trials
 APEC Preliminary Workshop: Review of Drug Development in Clinical Trials Session 9 B Clinical Trial Assessment Patient Protection Informed Consent Susan D Amico Vice President and Global Head Clinical
APEC Preliminary Workshop: Review of Drug Development in Clinical Trials Session 9 B Clinical Trial Assessment Patient Protection Informed Consent Susan D Amico Vice President and Global Head Clinical
Centralized Office of Research
 Centralized Office of Research The driving force for creating this model or type of clinical trials office (CTO) at JHS was noncompliance issues in billing. What we discovered was a general lack of education
Centralized Office of Research The driving force for creating this model or type of clinical trials office (CTO) at JHS was noncompliance issues in billing. What we discovered was a general lack of education
Pamela Richtmyer, MGH Research Compliance
 Pamela Richtmyer, MGH Research Compliance September 27 - Session 1 Basic overview October 4 - Session 2 Tracking patient care charges Invoices to sponsor Insight Patient Care Corrections October 11 - Session
Pamela Richtmyer, MGH Research Compliance September 27 - Session 1 Basic overview October 4 - Session 2 Tracking patient care charges Invoices to sponsor Insight Patient Care Corrections October 11 - Session
What will I do? Our HCSWs fall into three groups:
 As an HCSW, you will be a valued member of your team. Your focus will be on keeping patients at the centre of all we do, providing high quality care and living our Trust values: Together - Safe Kind Excellent.
As an HCSW, you will be a valued member of your team. Your focus will be on keeping patients at the centre of all we do, providing high quality care and living our Trust values: Together - Safe Kind Excellent.
Medicare Coverage Analysis. BWH qualifying study training and discussion
 Medicare Coverage Analysis BWH qualifying study training and discussion Research Compliance at BWH ØAt BWH, Research Compliance ensures study teams and institution follow governing authorities interprets
Medicare Coverage Analysis BWH qualifying study training and discussion Research Compliance at BWH ØAt BWH, Research Compliance ensures study teams and institution follow governing authorities interprets
ONESOURCE FRINGE BENEFITS TAX ONESOURCE FBT INSTALLATION GUIDE 2017 STAND-ALONE INSTALLATION AND UPGRADE GUIDE. Thomson Reuters ONESOURCE Support
 ONESOURCE FRINGE BENEFITS TAX ONESOURCE FBT INSTALLATION GUIDE 2017 STAND-ALONE INSTALLATION AND UPGRADE GUIDE Thomson Reuters ONESOURCE Support Date of issue: 03 Feb 2017 Getting started: Decision tree
ONESOURCE FRINGE BENEFITS TAX ONESOURCE FBT INSTALLATION GUIDE 2017 STAND-ALONE INSTALLATION AND UPGRADE GUIDE Thomson Reuters ONESOURCE Support Date of issue: 03 Feb 2017 Getting started: Decision tree
DO I NEED TO SUBMIT FOR THIS?... & OTHER FREQUENTLY ASKED QUESTIONS. March 2015 IRB Forum
 DO I NEED TO SUBMIT FOR THIS?... & OTHER FREQUENTLY ASKED QUESTIONS March 2015 IRB Forum Topics Quality Assurance/Quality Improvement Projects Informed Consent- when is a waiver appropriate? Retrospective/Prospective
DO I NEED TO SUBMIT FOR THIS?... & OTHER FREQUENTLY ASKED QUESTIONS March 2015 IRB Forum Topics Quality Assurance/Quality Improvement Projects Informed Consent- when is a waiver appropriate? Retrospective/Prospective
Utilizing Proctors for Competency Evaluations
 Utilizing Proctors for Competency Evaluations WHITE PAPER Editor s note: In this white paper, Michael Callahan, Esq., partner at Katten Muchin Rosenman, LLP, in Chicago; and Christine Mobley, CPMSM, CPCS,
Utilizing Proctors for Competency Evaluations WHITE PAPER Editor s note: In this white paper, Michael Callahan, Esq., partner at Katten Muchin Rosenman, LLP, in Chicago; and Christine Mobley, CPMSM, CPCS,
CMA GUIDELINES FOR MEDICAL STAFF PROCTORING. Approved by the CMA Board of Trustees, April 26, 2012
 Last Revised: //0 0 0 0 0 CMA GUIDELINES FOR MEDICAL STAFF PROCTORING Approved by the CMA Board of Trustees, April, 0 These guidelines are intended to assist medical staffs with the establishment of a
Last Revised: //0 0 0 0 0 CMA GUIDELINES FOR MEDICAL STAFF PROCTORING Approved by the CMA Board of Trustees, April, 0 These guidelines are intended to assist medical staffs with the establishment of a
DEPARTMENT OF HEALTH & HUMAN SERVICES WARNING LETTER. (b) (4) clinical investigation (Protocol entitled A Phase II, Multicenter,
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville, MD 20857 WARNING LETTER CERTIFIED MAIL RETURN RECEIPT REQUESTED Ronald Bukowski, M.D. 28099 Gates Mills
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville, MD 20857 WARNING LETTER CERTIFIED MAIL RETURN RECEIPT REQUESTED Ronald Bukowski, M.D. 28099 Gates Mills
Your Wellness Visit Guide
 Your Wellness Visit Guide Prepare for your Annual Wellness Visit or Welcome to Medicare Visit. Let s make the most of your appointment. Annual Wellness Visit Provider Toolkit Caring for Seniors HIGHMARK.COM
Your Wellness Visit Guide Prepare for your Annual Wellness Visit or Welcome to Medicare Visit. Let s make the most of your appointment. Annual Wellness Visit Provider Toolkit Caring for Seniors HIGHMARK.COM
STANDARDIZED PROCEDURE INTRAVENTRICULAR CHEMOTHERAPY VIA OMMAYA RESERVOIR (Adult, Peds)
 I. Definition The administration of chemotherapy via Ommaya Reservoir into cerebrospinal fluid (CSF) for treatment of previously diagnosed central nervous system (CNS) involvement by leukemia and lymphoma
I. Definition The administration of chemotherapy via Ommaya Reservoir into cerebrospinal fluid (CSF) for treatment of previously diagnosed central nervous system (CNS) involvement by leukemia and lymphoma
Martin Health System Stuart, Florida Laboratory Services. Laboratory Services and Policies
 Martin Health System Stuart, Florida Laboratory Services Laboratory Services and Policies Service Commitment: It is the goal of the Martin Health System s Clinical Laboratory to provide the medical community
Martin Health System Stuart, Florida Laboratory Services Laboratory Services and Policies Service Commitment: It is the goal of the Martin Health System s Clinical Laboratory to provide the medical community
MEDICAL LABORATORY SCIENCE MANDATORIES INFORMATION
 MEDICAL LABORATORY SCIENCE MANDATORIES INFORMATION FIRST YEAR MANDATORIES HIPAA/OSHA Training You will complete your training through the Evolve e Learning Solutions website. You will receive an email
MEDICAL LABORATORY SCIENCE MANDATORIES INFORMATION FIRST YEAR MANDATORIES HIPAA/OSHA Training You will complete your training through the Evolve e Learning Solutions website. You will receive an email
Clinical Trial Readiness Checklist October 2014
 The clinical trial readiness checklist is a tool which can help new and experienced researchers prepare for upcoming clinical studies. It is designed to be a quick reference to ensure that you have the
The clinical trial readiness checklist is a tool which can help new and experienced researchers prepare for upcoming clinical studies. It is designed to be a quick reference to ensure that you have the
The presenter has owns Kelly Willenberg, LLC in relation to this educational activity.
 Kelly M Willenberg, MBA, BSN, CCRP, CHC, CHRC 1 The presenter has owns Kelly Willenberg, LLC in relation to this educational activity. 2 1 Medical Necessity when you submit claims Coding for qualifying
Kelly M Willenberg, MBA, BSN, CCRP, CHC, CHRC 1 The presenter has owns Kelly Willenberg, LLC in relation to this educational activity. 2 1 Medical Necessity when you submit claims Coding for qualifying
The Clinical Investigation Policy and Procedure Manual Document: CIPP
 Clinical Research Credential Policy Children s Hospital is committed to assuring that all individuals who perform research are appropriately qualified to perform the roles assigned. It is also recognized
Clinical Research Credential Policy Children s Hospital is committed to assuring that all individuals who perform research are appropriately qualified to perform the roles assigned. It is also recognized
Comprehensive Protocol Feasibility Questionnaire
 Protocol Title: Potential Principal Investigator: Regulatory Coordinators: Department Chair: PROJECT FEASIBILITY PI and Study Team: YOUR RESPONSES TO THIS SURVEY CONSTITUTE A BEST ESTIMATE OF RESOURCES
Protocol Title: Potential Principal Investigator: Regulatory Coordinators: Department Chair: PROJECT FEASIBILITY PI and Study Team: YOUR RESPONSES TO THIS SURVEY CONSTITUTE A BEST ESTIMATE OF RESOURCES
Immunizations Criminal Background check Infection Control HIPPA Health Insurance Portability and Accountability Act
 Reedsburg Area Senior Life Center Welcome to Reedsburg Area Senior Life Center for your clinical! We hope you will have a positive and rewarding learning experience. If you have any questions during your
Reedsburg Area Senior Life Center Welcome to Reedsburg Area Senior Life Center for your clinical! We hope you will have a positive and rewarding learning experience. If you have any questions during your
PO Box , Charlotte, NC Phone: (877) Fax: (877)
 To apply for help in affording your prescription for Latuda (lurasidone HCl) tablets, please mail or fax a completed application to Sunovion Support Prescription Assistance Program ( Program ), PO Box
To apply for help in affording your prescription for Latuda (lurasidone HCl) tablets, please mail or fax a completed application to Sunovion Support Prescription Assistance Program ( Program ), PO Box
STUDY INFORMATION POST-IRB APPROVAL FDA DEVICE (IDE) SPONSOR AND INVESTIGATOR RESPONSIBILITY (21 CFR 812)
 POST-IRB APPROVAL FDA DEVICE (IDE) SPONSOR AND INVESTIGATOR RESPONSIBILITY (21 CFR 812) Purpose: Investigators who initiate and submit an IDE application to the FDA assume the responsibilities of both
POST-IRB APPROVAL FDA DEVICE (IDE) SPONSOR AND INVESTIGATOR RESPONSIBILITY (21 CFR 812) Purpose: Investigators who initiate and submit an IDE application to the FDA assume the responsibilities of both
Criteria for granting privileges:
 SPECIALTY OF NURSE PRACTITIONER Provider-based Clinic (PBC) Delineation of Clinical Privileges (DOP) Criteria for granting privileges: Current national board certification in the appropriate advanced practice
SPECIALTY OF NURSE PRACTITIONER Provider-based Clinic (PBC) Delineation of Clinical Privileges (DOP) Criteria for granting privileges: Current national board certification in the appropriate advanced practice
The GCP Perspective on Study Monitoring
 The GCP Perspective on Study Monitoring Heidi Judge, CCRP Sr. Clinical Trials Project Manager Clinical Trials Network and Institute Massachusetts General Hospital 1 Overview Monitoring Basics Who, What,
The GCP Perspective on Study Monitoring Heidi Judge, CCRP Sr. Clinical Trials Project Manager Clinical Trials Network and Institute Massachusetts General Hospital 1 Overview Monitoring Basics Who, What,
Clinical Research Coordinator - Researcher Startup Tool 1 of 7
 Clinical Research Coordinator - Researcher Startup Tool 1 of 7 Mount Sinai Hospital Employees: ITHelpDesk@mountsinai.org 212-241-4357 Sinai Central Account Sinai Central is the system used to manage HR
Clinical Research Coordinator - Researcher Startup Tool 1 of 7 Mount Sinai Hospital Employees: ITHelpDesk@mountsinai.org 212-241-4357 Sinai Central Account Sinai Central is the system used to manage HR
STANDARDIZED PROCEDURE LUMBAR DRAIN INSERTION (Adults, Peds)
 I. Definition The purpose of this standardized procedure is for the Advanced Health Practitioner to safely place a lumbar drain. II. Background Information A. Setting: The setting (inpatient vs outpatient)
I. Definition The purpose of this standardized procedure is for the Advanced Health Practitioner to safely place a lumbar drain. II. Background Information A. Setting: The setting (inpatient vs outpatient)
Principal Investigator/Co-Investigator - Researcher Startup Tool 1 of 9
 Principal Investigator/Co-Investigator - Researcher Startup Tool 1 of 9 Mount Sinai Hospital Employees: ITHelpDesk@mountsinai.org 212-241-4357 Sinai Central Account Sinai Central is the system used to
Principal Investigator/Co-Investigator - Researcher Startup Tool 1 of 9 Mount Sinai Hospital Employees: ITHelpDesk@mountsinai.org 212-241-4357 Sinai Central Account Sinai Central is the system used to
DEPN AND GRADUATE NURSING MANDATORIES INFORMATION
 DEPN AND GRADUATE NURSING MANDATORIES INFORMATION INITIAL MANDATORIES DUE AUGUST 15, 2018 Pre Clinical Mandatories Form If you have a first time positive PPD, include a radiology report If you have a history
DEPN AND GRADUATE NURSING MANDATORIES INFORMATION INITIAL MANDATORIES DUE AUGUST 15, 2018 Pre Clinical Mandatories Form If you have a first time positive PPD, include a radiology report If you have a history
Roles of Investigators in the Managements of Clinical Trials
 Roles of Investigators in the Managements of Clinical Trials Chii-Min Hwu, M.D. Section of General Medicine Department of Medicine Taipei Veterans General Hospital Learning Objectives PI Outlines How to
Roles of Investigators in the Managements of Clinical Trials Chii-Min Hwu, M.D. Section of General Medicine Department of Medicine Taipei Veterans General Hospital Learning Objectives PI Outlines How to
Medical Staff Rules & Regulations Last Updated: October University Hospital Medical Staff. Rules & Regulations
 University Hospital Medical Staff Rules & Regulations 1 UNIVERSITY HOSPITAL MEDICAL STAFF RULES AND REGULATIONS The Medical Staff shall adopt Rules and Regulations as may be necessary to implement the
University Hospital Medical Staff Rules & Regulations 1 UNIVERSITY HOSPITAL MEDICAL STAFF RULES AND REGULATIONS The Medical Staff shall adopt Rules and Regulations as may be necessary to implement the
Laboratory System Improvement Program (L SIP)
 Colorado Department of Public Health and Environment Laboratory Services Division Laboratory System Improvement Program (L SIP) EXECUTIVE ASSESSMENT SUMMARY 2013 Colorado Department of Public Health and
Colorado Department of Public Health and Environment Laboratory Services Division Laboratory System Improvement Program (L SIP) EXECUTIVE ASSESSMENT SUMMARY 2013 Colorado Department of Public Health and
STANDARDIZED PROCEDURE HEPATIC ARTERY INFUSION OF CHEMOTHERAPY (Adults, Peds)
 I. Definition Hepatic arterial infusion (HAI) of chemotherapy is accomplished by a small drug delivery system or pump that is implanted in a subcutaneous pocket in the lower abdomen. The pump reservoir
I. Definition Hepatic arterial infusion (HAI) of chemotherapy is accomplished by a small drug delivery system or pump that is implanted in a subcutaneous pocket in the lower abdomen. The pump reservoir
Objectives Top Ten Cited Deficiencies for Acute Care Facilities April 21, 2015
 2014 Top Ten Cited Deficiencies for Acute Care Facilities April 21, 2015 Michele Kala, MS, RN Director of Accreditation and Certification Objectives Understanding of the top scored deficient HFAP standards
2014 Top Ten Cited Deficiencies for Acute Care Facilities April 21, 2015 Michele Kala, MS, RN Director of Accreditation and Certification Objectives Understanding of the top scored deficient HFAP standards
Department of Transitional Assistance Transitional Aid to Families with Dependent Children Disability Supplement
 Department of Transitional Assistance Transitional Aid to Families with Dependent Children Disability Supplement Do you need help to fill out the attached form? Call DTA at 1-877-382-2363. DTA can help
Department of Transitional Assistance Transitional Aid to Families with Dependent Children Disability Supplement Do you need help to fill out the attached form? Call DTA at 1-877-382-2363. DTA can help
Section 9. Study Product Considerations for Non- Pharmacy Staff
 Section 9. Study Product Considerations for Non- Pharmacy Staff Table of Contents 9.1 Dispensing Study Product 9.1.1 Chain of Custody 9.1.2 Initial Vaginal Ring Dispensing(s)- Prescription Overview 9.2
Section 9. Study Product Considerations for Non- Pharmacy Staff Table of Contents 9.1 Dispensing Study Product 9.1.1 Chain of Custody 9.1.2 Initial Vaginal Ring Dispensing(s)- Prescription Overview 9.2
Good Documentation Practices. Human Subject Research. for
 Good Documentation Practices for Human Subject Research Bridget M. Psicihulis, RHIA, CCRC Quality Improvement Unit Coordinator Human Research Protection Program Wheaton Franciscan Healthcare (last updated
Good Documentation Practices for Human Subject Research Bridget M. Psicihulis, RHIA, CCRC Quality Improvement Unit Coordinator Human Research Protection Program Wheaton Franciscan Healthcare (last updated
Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network for Research Professionals
 ORIENTATION FOR NEW CLINICAL RESEARCH COORDINATORS Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network for Research Professionals Overall Agenda for Orientation Module 1: Introduction
ORIENTATION FOR NEW CLINICAL RESEARCH COORDINATORS Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network for Research Professionals Overall Agenda for Orientation Module 1: Introduction
MMC-Einstein Research IT Forum. November 6, 2017
 MMC-Einstein Research IT Forum November 6, 2017 Agenda Updated Training Workflow Reminders Updated Research Billing Policy Research Billing Executive Steering Software updates Studies requiring activation
MMC-Einstein Research IT Forum November 6, 2017 Agenda Updated Training Workflow Reminders Updated Research Billing Policy Research Billing Executive Steering Software updates Studies requiring activation
Drug Testing Program Prevention and Education
 Army Substance Abuse Program Drug Testing Program Prevention and Education UNCLASSIFIED Headquarters U.S. Army Cadet Command 204 1 st Cavalry Regiment Road Fort Knox, KY 40121 25 February 2013 HEADQUARTERS
Army Substance Abuse Program Drug Testing Program Prevention and Education UNCLASSIFIED Headquarters U.S. Army Cadet Command 204 1 st Cavalry Regiment Road Fort Knox, KY 40121 25 February 2013 HEADQUARTERS
Roles, Responsibilities and Patient Care Activities of Fellows UW SLEEP MEDICINE FELLOWSHIP
 Roles, Responsibilities and Patient Care Activities of Fellows UW SLEEP MEDICINE FELLOWSHIP Harborview Medical Center University of Washington Medical Center Seattle Children s Hospital Virginia Mason
Roles, Responsibilities and Patient Care Activities of Fellows UW SLEEP MEDICINE FELLOWSHIP Harborview Medical Center University of Washington Medical Center Seattle Children s Hospital Virginia Mason
Preventing Infection in Care
 Infection Prevention and Control: Older Person Care Homes & Home Environment Learning Programme Workbook NHS Education for Scotland 2011. You can copy or reproduce the information in this document for
Infection Prevention and Control: Older Person Care Homes & Home Environment Learning Programme Workbook NHS Education for Scotland 2011. You can copy or reproduce the information in this document for
STANDARDIZED PROCEDURE SKIN BIOPSY (Adult, Peds)
 I. Definition Skin biopsy is the removal of a small piece of tissue, under local anesthetic, from a lesion suspected of malignancy, other dermatitis, or for clinical research purposes. The technique to
I. Definition Skin biopsy is the removal of a small piece of tissue, under local anesthetic, from a lesion suspected of malignancy, other dermatitis, or for clinical research purposes. The technique to
AN OVERVIEW OF CLINICAL STUDY TASKS AND ACTIVITIES
 1 AN OVERVIEW OF CLINICAL STUDY TASKS AND ACTIVITIES Key Clinical Study Tasks and Activities 2 Discussion of Key Tasks and Activities 3 Development of the Clinical Protocol and Study Materials 3 Qualification
1 AN OVERVIEW OF CLINICAL STUDY TASKS AND ACTIVITIES Key Clinical Study Tasks and Activities 2 Discussion of Key Tasks and Activities 3 Development of the Clinical Protocol and Study Materials 3 Qualification
Nursing Documentation Changes and Reminders. CCTC Nursing Documentation
 Nursing Documentation Changes and Reminders CCTC Nursing Documentation Change #1 Standard ph range changed to match new RRT documentation Change #2 Clarification for documentation standards for IV solutions.
Nursing Documentation Changes and Reminders CCTC Nursing Documentation Change #1 Standard ph range changed to match new RRT documentation Change #2 Clarification for documentation standards for IV solutions.
YALE UNIVERSITY THE RESEARCHERS GUIDE TO HIPAA. Health Insurance Portability and Accountability Act of 1996
 YALE UNIVERSITY THE RESEARCHERS GUIDE TO HIPAA Health Insurance Portability and Accountability Act of 1996 Handbook Table of Contents I. Introduction What is HIPAA? What is PHI? What is a Covered Entity
YALE UNIVERSITY THE RESEARCHERS GUIDE TO HIPAA Health Insurance Portability and Accountability Act of 1996 Handbook Table of Contents I. Introduction What is HIPAA? What is PHI? What is a Covered Entity
Office Safety Policy & Procedure Manual. Section B
 Office Safety Policy & Manual 2011 Section B (Click on the sub-sections to jump to the specific section) OS-B100 OS-B101 OS-B102 OS-B103 OS-B104 OS-B105 OS-B106 Clinical Services Laboratory Services Medication
Office Safety Policy & Manual 2011 Section B (Click on the sub-sections to jump to the specific section) OS-B100 OS-B101 OS-B102 OS-B103 OS-B104 OS-B105 OS-B106 Clinical Services Laboratory Services Medication
Worksheet: Friend, Foe or Both?
 Medicare s ASC Infection Control Worksheet: Friend, Foe or Both? Tammeria Tyler, RN CIC Infection Preventionist Learning Objectives To understand outlined Conditions for Coverage in the ASC Infection Control
Medicare s ASC Infection Control Worksheet: Friend, Foe or Both? Tammeria Tyler, RN CIC Infection Preventionist Learning Objectives To understand outlined Conditions for Coverage in the ASC Infection Control
Prescriptive Authority for Pharmacists. Frequently Asked Questions for Pharmacists
 Prescriptive Authority for Pharmacists Frequently Asked Questions for Pharmacists Disclaimer: When in doubt, the text of the official bylaws should be consulted. They are available at: http://napra.ca/content_files/files/saskatchewan/proposedprescribingbylawsawaitingtheministerofhealt
Prescriptive Authority for Pharmacists Frequently Asked Questions for Pharmacists Disclaimer: When in doubt, the text of the official bylaws should be consulted. They are available at: http://napra.ca/content_files/files/saskatchewan/proposedprescribingbylawsawaitingtheministerofhealt
QUALITY TIPS FOR CLINICAL SITES. Athena Thomas-Visel. Clinical Quality Consultant QUALITY TIPS FOR CLINICAL SITES
 QUALITY TIPS FOR CLINICAL SITES Athena Thomas-Visel Clinical Quality Consultant QUALITY TIPS FOR CLINICAL SITES Purpose of presentation: Share best practices seen from 150+ sites visited Spark conversation
QUALITY TIPS FOR CLINICAL SITES Athena Thomas-Visel Clinical Quality Consultant QUALITY TIPS FOR CLINICAL SITES Purpose of presentation: Share best practices seen from 150+ sites visited Spark conversation
Nursing Home C. difficile Initiative Three-Part Educational Webinar Series
 Nursing Home C. difficile Initiative Three-Part Educational Webinar Series Webinar #2 Enrollment in NHSN for LTC Facilities August 23, 2016 Webinar #2 Enrollment in NHSN for LTC Facilities Nursing Home
Nursing Home C. difficile Initiative Three-Part Educational Webinar Series Webinar #2 Enrollment in NHSN for LTC Facilities August 23, 2016 Webinar #2 Enrollment in NHSN for LTC Facilities Nursing Home
First Aid Policy. Appletree Treatment Centre
 First Aid Policy Appletree Treatment Centre This document has been prepared to provide guidance on the policy and procedures for dealing with First Aid emergences at Appletree Treatment Centre. As a company
First Aid Policy Appletree Treatment Centre This document has been prepared to provide guidance on the policy and procedures for dealing with First Aid emergences at Appletree Treatment Centre. As a company
FREQUENTLY ASKED QUESTIONS
 FREQUENTLY ASKED QUESTIONS What does ispecimen do? Privately held and headquartered in Lexington, MA, ispecimen is a trusted, one-stop source of customized human biospecimen collections. Every day at hospitals
FREQUENTLY ASKED QUESTIONS What does ispecimen do? Privately held and headquartered in Lexington, MA, ispecimen is a trusted, one-stop source of customized human biospecimen collections. Every day at hospitals
Standards. Successfully Preparing for Your Next AAAHC Accreditation Survey Annual Conference
 Successfully Preparing for Your Next AAAHC Accreditation Survey 2012 Annual Conference Guest Speaker Ray Grundman, MSN, MPA, CASC AAAHC Senior Director External Relations AAAHC Surveyor AAAHC - Past President
Successfully Preparing for Your Next AAAHC Accreditation Survey 2012 Annual Conference Guest Speaker Ray Grundman, MSN, MPA, CASC AAAHC Senior Director External Relations AAAHC Surveyor AAAHC - Past President
The hospital s anesthesia services must be integrated into the hospital-wide QAPI program.
 A-0416 482.52 Condition of Participation: Anesthesia Services If the hospital furnishes anesthesia services, they must be provided in a well-organized manner under the direction of a qualified doctor of
A-0416 482.52 Condition of Participation: Anesthesia Services If the hospital furnishes anesthesia services, they must be provided in a well-organized manner under the direction of a qualified doctor of
St. Vincent s East Page 1 of 5
 St. Vincent s East Page 1 of 5 TITLE: PATIENT CARE PRACTICE GUIDELINE CARE OF PATIENTS BLOOD AND BLOOD COMPONENTS - ADMINISTRATION FACILITY: FUNCTION: ORIGINATING DEPT: St. Vincent s East HOSPITAL SHARED
St. Vincent s East Page 1 of 5 TITLE: PATIENT CARE PRACTICE GUIDELINE CARE OF PATIENTS BLOOD AND BLOOD COMPONENTS - ADMINISTRATION FACILITY: FUNCTION: ORIGINATING DEPT: St. Vincent s East HOSPITAL SHARED
SUCCESS FEE FOR SERVICE PROGRAMS
 SUCCESS GCRC Research CENTER Environment SUCCESS FEE FOR SERVICE PROGRAMS Clare Tyson, MA, CCRA Leila Forney, BSN, RN, CCRP Brigette White MA, CCRP Stephen Skelton, MA SUCCESS CENTER FEE FOR SERIVCE PROGRAMS
SUCCESS GCRC Research CENTER Environment SUCCESS FEE FOR SERVICE PROGRAMS Clare Tyson, MA, CCRA Leila Forney, BSN, RN, CCRP Brigette White MA, CCRP Stephen Skelton, MA SUCCESS CENTER FEE FOR SERIVCE PROGRAMS
Caldwell Medical Center Departments
 Caldwell Medical Center Departments Surgery Medical / Surgery Same Day Surgery Lab Education Administration Special Care Unit Women s Center Admission Emergency Services Radiology Cardiac Rehab Admission
Caldwell Medical Center Departments Surgery Medical / Surgery Same Day Surgery Lab Education Administration Special Care Unit Women s Center Admission Emergency Services Radiology Cardiac Rehab Admission
WARNING LETTER CERTIFIED MAIL RETURN RECEIPT REQUESTED
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville, MD 20857 WARNING LETTER CERTIFIED MAIL RETURN RECEIPT REQUESTED Charles J. Coté, M.D. Ref: 09-HFD-45-02-04
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville, MD 20857 WARNING LETTER CERTIFIED MAIL RETURN RECEIPT REQUESTED Charles J. Coté, M.D. Ref: 09-HFD-45-02-04
Procedure Code Job Aid
 Procedure Code 99211 Job Aid Definition for 99211: Office or other outpatient visit for the evaluation and management of an established patient that may not require the presence of a physician. Usually,
Procedure Code 99211 Job Aid Definition for 99211: Office or other outpatient visit for the evaluation and management of an established patient that may not require the presence of a physician. Usually,
Understanding Health Care in America An introduction for immigrant patients
 Patient Education Understanding Health Care in America An introduction for immigrant patients The health care system in the United States is complex. Some parts of the system are different in different
Patient Education Understanding Health Care in America An introduction for immigrant patients The health care system in the United States is complex. Some parts of the system are different in different
Welcome to BCHC Your Medical Home
 START HERE 1 Welcome to BCHC Your Medical Home Thank you for choosing Berks Community Health Center (BCHC) as your medical home. This booklet gives you information about being a patient at BCHC and what
START HERE 1 Welcome to BCHC Your Medical Home Thank you for choosing Berks Community Health Center (BCHC) as your medical home. This booklet gives you information about being a patient at BCHC and what
EASTERN ARIZONA COLLEGE Pharmacy Practice for Technician
 EASTERN ARIZONA COLLEGE Pharmacy Practice for Technician Course Design 2014-2015 Course Information Division Allied Health Course Number HCE 176 Title Pharmacy Practice for Technician Credits 2 Developed
EASTERN ARIZONA COLLEGE Pharmacy Practice for Technician Course Design 2014-2015 Course Information Division Allied Health Course Number HCE 176 Title Pharmacy Practice for Technician Credits 2 Developed
