Subject Screening, Recruitment, and Retention. Tiffany Morrison, MS, CCRP Director, Clinical Trials Rothman Institute
|
|
|
- Joel Barnett
- 6 years ago
- Views:
Transcription
1 Subject Screening, Recruitment, and Retention Tiffany Morrison, MS, CCRP Director, Clinical Trials Rothman Institute
2 Learning objectives List two sources of potential subjects who can be screening for enrollment Explain why it would be important for the IRB to review and approve advertising for clinical trials Provide examples of ways in which patients can be engaged during a clinical trial in an effort to retain them in the study
3 Patient Recruitment Paramount in conducting a successful trial Recruitment tips: Before committing, evaluate the enrollment criteria to see if they are realist for your site Develop a recruitment plan Be sure the entire team is committed Instill confidence in research subjects
4 Sources of subjects Chart review: pulling patients from the existing schedule of patients: normal patient population Database: using a diagnosis code or billing code to pull all patients that match within a certain time frame Referrals: some physicians can send out formal letters to PCP or other physicians that might see patients that match inclusion/exclusion Advertising: must be approved by IRB: can be google ads, newspapers, radio, etc.
5 Advertising Because it s part of the informed consent and subject selection process, all direct advertising must: Not imply certainty of favorable outcome Not over-promote compensation or benefits Not be misleading Not include claims of safety, efficacy, equivalence or superiority Include investigational Can t be termed new drug, new device
6 Subject screening The steps listed below are the industry standard for screening potential subjects: Determine subjects appear to match I/E to review: Medical history Medications Test/ procedure results Lab results Physical exam results Diagnostic exam results Review screening checklist: withdraw from meds, if necessary Never perform study-specific procedures before consent!
7 Screening visit and logs Most clinical trials require updated screening logs to be completed and sent on a weekly basis to the Sponsor Screening generally refers to patients whom have sign the informed consent form but have not met all inclusion/ exclusion criteria for enrollment
8 Example of a Screening log
9 Screening visit and logs Screening of patients is usually a fluid activity and can encompass up to 30 days prior to official enrollment into a trial This is due to numerous lab values, radiographic or imaging analysis, or surgical clearance Patients who sign a consent form but are not ultimately enrolled are termed a screen failure. Many studies limit the amount of screen failures that can occur at each site for budgetary and statistical reasons
10 Scheduling subject visits Good idea to create a subject tracker in excel or calendar. This usually has the allowable windows included so it s easier to make appointments within visit windows Make sure MRIs, infusion appointments, or anything else is scheduled with a reasonable amount of time between visits Reminder s/ phone calls, letters always help keep up compliance
11 Enrollment and Patient Retention Nearly 80% of clinical trials fail to meet trial timelines Kremidas, Jim. Recruitment Roles. Applied Clinical Trials Online. Sept.1, 2011 This includes recruitment and retention of qualified subjects
12 Patient enrollment and the importance of education Medications Medical conditions/ status Diaries/ drug or device compliance Study procedures Visit requirements
13 Patient Retention A CenterWatch report stated that dropout rates of 15%-40% are not uncommon, though the average is about 25% Common Reasons for Drop Out: Difficulty complying with the protocol dosages, timelines, or procedures AE/SAE Loss of motivation Peer pressure Financial constraints Disease improvement or lack of improvement
14 Retention Strategies 1. Maintain Communication 2. Listen 3. Be Convenient 4. Maintain a Positive Attitude 5. Know the Protocol
15 Tips for Patient Retention 6. Patient Stipend 7. Reminder s, letters, calls, text messages 8. Transportation Options
Protocol Feasibility and Operationalization Framework. Beth Harper, BS, MBA. President, Clinical Performance Partners, Inc.
 Protocol Feasibility and Beth Harper, BS, MBA President, Clinical Performance Partners, Inc. Overview This will be a highly interactive session We will introduce you to new ways of thinking about the study
Protocol Feasibility and Beth Harper, BS, MBA President, Clinical Performance Partners, Inc. Overview This will be a highly interactive session We will introduce you to new ways of thinking about the study
Section 11. Recruitment of Study Subjects (Revised 7/1/10)
 Section 11 Recruitment of Study Subjects (Revised 7/1/10) The IRB shall review and approve, prior to utilization, all documents and activities that affect the rights and welfare of research subjects, including
Section 11 Recruitment of Study Subjects (Revised 7/1/10) The IRB shall review and approve, prior to utilization, all documents and activities that affect the rights and welfare of research subjects, including
Primary Care Specialist Physician Compact
 I. Purpose To provide optimal health care for our patients. To provide a framework for better communication and safe transition of care between primary care and specialty care providers. II. Principles
I. Purpose To provide optimal health care for our patients. To provide a framework for better communication and safe transition of care between primary care and specialty care providers. II. Principles
The GCP Perspective on Study Monitoring
 The GCP Perspective on Study Monitoring Heidi Judge, CCRP Sr. Clinical Trials Project Manager Clinical Trials Network and Institute Massachusetts General Hospital 1 Overview Monitoring Basics Who, What,
The GCP Perspective on Study Monitoring Heidi Judge, CCRP Sr. Clinical Trials Project Manager Clinical Trials Network and Institute Massachusetts General Hospital 1 Overview Monitoring Basics Who, What,
Study Start-Up SS STANDARD OPERATING PROCEDURE FOR PRE-STUDY SITE VISIT (PSSV)
 Replaces previous version 203.01: 01 July 2014 Study Start-Up SS 203.01 STANDARD OPERATING PROCEDURE FOR PRE-STUDY SITE VISIT (PSSV) Approval: Nancy Paris, MS, FACHE President and CEO 24 May 2017 (Signature
Replaces previous version 203.01: 01 July 2014 Study Start-Up SS 203.01 STANDARD OPERATING PROCEDURE FOR PRE-STUDY SITE VISIT (PSSV) Approval: Nancy Paris, MS, FACHE President and CEO 24 May 2017 (Signature
DO I NEED TO SUBMIT FOR THIS?... & OTHER FREQUENTLY ASKED QUESTIONS. March 2015 IRB Forum
 DO I NEED TO SUBMIT FOR THIS?... & OTHER FREQUENTLY ASKED QUESTIONS March 2015 IRB Forum Topics Quality Assurance/Quality Improvement Projects Informed Consent- when is a waiver appropriate? Retrospective/Prospective
DO I NEED TO SUBMIT FOR THIS?... & OTHER FREQUENTLY ASKED QUESTIONS March 2015 IRB Forum Topics Quality Assurance/Quality Improvement Projects Informed Consent- when is a waiver appropriate? Retrospective/Prospective
20 STEPS FROM STUDY IDEA INCEPTION TO PUBLISHING RESEARCH/ Evidence-Based Practice
 20 STEPS FROM STUDY IDEA INCEPTION TO PUBLISHING RESEARCH/ Evidence-Based Practice Nursing Research/ Evidence-Based Practice Checklist (Version 31 January 2012) Specify the date in the left column when
20 STEPS FROM STUDY IDEA INCEPTION TO PUBLISHING RESEARCH/ Evidence-Based Practice Nursing Research/ Evidence-Based Practice Checklist (Version 31 January 2012) Specify the date in the left column when
Comprehensive Protocol Feasibility Questionnaire
 Protocol Title: Potential Principal Investigator: Regulatory Coordinators: Department Chair: PROJECT FEASIBILITY PI and Study Team: YOUR RESPONSES TO THIS SURVEY CONSTITUTE A BEST ESTIMATE OF RESOURCES
Protocol Title: Potential Principal Investigator: Regulatory Coordinators: Department Chair: PROJECT FEASIBILITY PI and Study Team: YOUR RESPONSES TO THIS SURVEY CONSTITUTE A BEST ESTIMATE OF RESOURCES
BIMO SITE AUDIT CHECKLIST
 Item AUTHORITY AND ADMINISTRATION FOR STUDIES INVOLVING HUMAN DRUGS, BIOLOGICS AND DEVICES 1. Compare the Investigator Agreement with the information provided by the assigning Center. Auditor will check
Item AUTHORITY AND ADMINISTRATION FOR STUDIES INVOLVING HUMAN DRUGS, BIOLOGICS AND DEVICES 1. Compare the Investigator Agreement with the information provided by the assigning Center. Auditor will check
VISIT NOTES QUIZ. C. Individually select each system, then select the negative box for each item
 VISIT NOTES QUIZ 1. In the Examination section of the visit note template, how would you quickly mark all sections of the exam as normal? A. Select (-) at the top of the template B. Select the negative
VISIT NOTES QUIZ 1. In the Examination section of the visit note template, how would you quickly mark all sections of the exam as normal? A. Select (-) at the top of the template B. Select the negative
Roles of Investigators in the Managements of Clinical Trials
 Roles of Investigators in the Managements of Clinical Trials Chii-Min Hwu, M.D. Section of General Medicine Department of Medicine Taipei Veterans General Hospital Learning Objectives PI Outlines How to
Roles of Investigators in the Managements of Clinical Trials Chii-Min Hwu, M.D. Section of General Medicine Department of Medicine Taipei Veterans General Hospital Learning Objectives PI Outlines How to
TRACK-TBI: CLINICAL PROTOCOL CHANGE LOG
 TRACK-TBI: CLINICAL PROTOCOL CHANGE LOG CHANGE LOG V13 to V14 (July 6, 2016) New text in red 5.1 SUBJECT GROUPS The Controls will be adult orthopedic trauma patients who meet the following criteria: 1.
TRACK-TBI: CLINICAL PROTOCOL CHANGE LOG CHANGE LOG V13 to V14 (July 6, 2016) New text in red 5.1 SUBJECT GROUPS The Controls will be adult orthopedic trauma patients who meet the following criteria: 1.
GCP INSPECTION CHECKLIST
 (This list is not all inclusive; item may be added &/or deleted as per the Study/Site/Sponsor/Lab) I. General. Name and address of the clinical trial site Tel. No. & e- mail:. Date of Inspection. Inspection
(This list is not all inclusive; item may be added &/or deleted as per the Study/Site/Sponsor/Lab) I. General. Name and address of the clinical trial site Tel. No. & e- mail:. Date of Inspection. Inspection
Join the Conquest What s New? Information and Feedback Session
 Join the Conquest What s New? Information and Feedback Session North Carolina Translational and Clinical Sciences (NC TraCS) Institute Carol Breland, Research Recruitment Director December 13, 2017 Thanks
Join the Conquest What s New? Information and Feedback Session North Carolina Translational and Clinical Sciences (NC TraCS) Institute Carol Breland, Research Recruitment Director December 13, 2017 Thanks
SAMPLE WORKFLOW. DAY OF CONSULT - Patient Site (Pease refer to the flow chart for event timing and site participation requirement)
 WORKFLOW Workflow varies from organization to organization. The following pages illustrate how a typical telemedicine clinic operates, and are intended to be used as a starting point in developing your
WORKFLOW Workflow varies from organization to organization. The following pages illustrate how a typical telemedicine clinic operates, and are intended to be used as a starting point in developing your
The presenter has owns Kelly Willenberg, LLC in relation to this educational activity.
 Kelly M Willenberg, MBA, BSN, CCRP, CHC, CHRC 1 The presenter has owns Kelly Willenberg, LLC in relation to this educational activity. 2 1 Medical Necessity when you submit claims Coding for qualifying
Kelly M Willenberg, MBA, BSN, CCRP, CHC, CHRC 1 The presenter has owns Kelly Willenberg, LLC in relation to this educational activity. 2 1 Medical Necessity when you submit claims Coding for qualifying
Acurian on. Patient Attrition in Clinical Trial Enrollment. Is There an Awareness Issue?
 Acurian on Patient Attrition in Clinical Trial Enrollment Why does it seem so hard to recruit patients for a trial outside of my selected clinical site practices? This is a question that Patient Recruitment
Acurian on Patient Attrition in Clinical Trial Enrollment Why does it seem so hard to recruit patients for a trial outside of my selected clinical site practices? This is a question that Patient Recruitment
Ryan White Part A. Quality Management
 Quality Management Medical Case Management 2014 Broward County/Fort Lauderdale Eligible Metropolitan Area (EMA) The creation of this public document is fully funded by a federal Ryan White CARE Act Part
Quality Management Medical Case Management 2014 Broward County/Fort Lauderdale Eligible Metropolitan Area (EMA) The creation of this public document is fully funded by a federal Ryan White CARE Act Part
Guidance for Investigators Subject Recruitment & Retention
 Guidance for Investigators Subject Recruitment & Retention Meeting of Investigators supported by the NHLBI Clinical Trials Planning Studies for Rare Thrombotic and Hemostatic Disorders (U34) program Sreelatha
Guidance for Investigators Subject Recruitment & Retention Meeting of Investigators supported by the NHLBI Clinical Trials Planning Studies for Rare Thrombotic and Hemostatic Disorders (U34) program Sreelatha
HIC Standard Operating Procedure. For-Cause Audits of Human Research Studies
 HIC Standard Operating Procedure For-Cause Audits of Human Research Studies Background As part of the Wayne State University (WSU) Human Investigation Committee s (HIC) Human Research Protection Program,
HIC Standard Operating Procedure For-Cause Audits of Human Research Studies Background As part of the Wayne State University (WSU) Human Investigation Committee s (HIC) Human Research Protection Program,
RESIDENT JOB DESCRIPTION
 RESIDENT JOB DESCRIPTION Summary: All residents in the UMKC Family Medicine Residency Program are employees of UMKC and must abide by their policies. At a minimum, resident must hold a temporary license
RESIDENT JOB DESCRIPTION Summary: All residents in the UMKC Family Medicine Residency Program are employees of UMKC and must abide by their policies. At a minimum, resident must hold a temporary license
IMPORTANT! Some sections of this article require you have appropriate security clearance to things like the System Manger.
 Author: Joel Kristenson Last Updated: 2015-09-04 Overview This article is primarily for our nonprofit customers, but does contain useful information related to log notes and pivot reports for political
Author: Joel Kristenson Last Updated: 2015-09-04 Overview This article is primarily for our nonprofit customers, but does contain useful information related to log notes and pivot reports for political
Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network for Research Professionals
 ORIENTATION FOR NEW CLINICAL RESEARCH COORDINATORS Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network for Research Professionals Overall Agenda for Orientation Module 1: Introduction
ORIENTATION FOR NEW CLINICAL RESEARCH COORDINATORS Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network for Research Professionals Overall Agenda for Orientation Module 1: Introduction
CHAPTER 1. Documentation is a vital part of nursing practice.
 CHAPTER 1 PURPOSE OF DOCUMENTATION CHAPTER OBJECTIVE After completing this chapter, the reader will be able to identify the importance and purpose of complete documentation in the medical record. LEARNING
CHAPTER 1 PURPOSE OF DOCUMENTATION CHAPTER OBJECTIVE After completing this chapter, the reader will be able to identify the importance and purpose of complete documentation in the medical record. LEARNING
Efficacy of Tympanostomy Tubes for Children with Recurrent Acute Otitis Media Randomization Phase
 CONSENT FOR A CHILD TO BE A SUBJECT IN MEDICAL RESEARCH AND AUTHORIZATION TO PERMIT THE USE AND SHARING OF IDENTIFIABLE MEDICAL INFORMATION FOR RESEARCH PURPOSES TITLE Efficacy of Tympanostomy Tubes for
CONSENT FOR A CHILD TO BE A SUBJECT IN MEDICAL RESEARCH AND AUTHORIZATION TO PERMIT THE USE AND SHARING OF IDENTIFIABLE MEDICAL INFORMATION FOR RESEARCH PURPOSES TITLE Efficacy of Tympanostomy Tubes for
UNIVERSITY MEDICAL CENTER NEW ORLEANS RESEARCH REVIEW APPLICATION
 UNIVERSITY MEDICAL CENTER NEW ORLEANS RESEARCH REVIEW APPLICATION Submission and Approval Process 1. Submit complete RRC application to Office of Research as early as possible in study process. 2. Quality
UNIVERSITY MEDICAL CENTER NEW ORLEANS RESEARCH REVIEW APPLICATION Submission and Approval Process 1. Submit complete RRC application to Office of Research as early as possible in study process. 2. Quality
REGISTERING A PATIENT
 REGISTERING A PATIENT Patient Eligibility It is important for the institution staff to review all eligibility criteria and follow-up requirements. A patient failing to meet all protocol eligibility requirements
REGISTERING A PATIENT Patient Eligibility It is important for the institution staff to review all eligibility criteria and follow-up requirements. A patient failing to meet all protocol eligibility requirements
Subject Research Records. Essential Regulatory and Source Documents. Subject Research Records. Regulatory Files
 Essential Regulatory and Source Documents Phyllis Carello, BS, CCRC, CTRC Research Study Coordinator Manager Bridget Adams, MSHS, CCRA, Manager Clinical Trials Office & Investigator Support and Integration
Essential Regulatory and Source Documents Phyllis Carello, BS, CCRC, CTRC Research Study Coordinator Manager Bridget Adams, MSHS, CCRA, Manager Clinical Trials Office & Investigator Support and Integration
Ryan White Part A. Quality Management
 Quality Management Central Intake and Eligibility Determination (CIED) 2014 Broward County/Fort Lauderdale Eligible Metropolitan Area (EMA) The creation of this public document is fully funded by a federal
Quality Management Central Intake and Eligibility Determination (CIED) 2014 Broward County/Fort Lauderdale Eligible Metropolitan Area (EMA) The creation of this public document is fully funded by a federal
Your Plan Explained. MetLife. UnitedHealthcare Group Medicare Advantage (PPO) Group Number: 12359
 2016 Your Plan Explained MetLife UnitedHealthcare Group Medicare Advantage (PPO) Effective: January 1, 2016 through December 31, 2016 Group Number: 12359 Benefit highlights MetLife 12359 Effective January
2016 Your Plan Explained MetLife UnitedHealthcare Group Medicare Advantage (PPO) Effective: January 1, 2016 through December 31, 2016 Group Number: 12359 Benefit highlights MetLife 12359 Effective January
Clinical Trial Readiness Checklist October 2014
 The clinical trial readiness checklist is a tool which can help new and experienced researchers prepare for upcoming clinical studies. It is designed to be a quick reference to ensure that you have the
The clinical trial readiness checklist is a tool which can help new and experienced researchers prepare for upcoming clinical studies. It is designed to be a quick reference to ensure that you have the
Family Practice Clinic
 Family Practice Clinic FNP Job Description (Hospital Privileges) General: The Family Nurse Practitioner (FNP) assesses, plans and provides comprehensive patient care independently or in autonomous collaboration
Family Practice Clinic FNP Job Description (Hospital Privileges) General: The Family Nurse Practitioner (FNP) assesses, plans and provides comprehensive patient care independently or in autonomous collaboration
WIRBinar. How to Survive an FDA Inspection. Upcoming Trainings: Contact Us: (360)
 WIRBinar How to Survive an FDA Inspection 10-26-2011 Brought to you by WIRB Education and Consulting Services. Improve your ability to maintain compliance and protect human subjects with guidance from
WIRBinar How to Survive an FDA Inspection 10-26-2011 Brought to you by WIRB Education and Consulting Services. Improve your ability to maintain compliance and protect human subjects with guidance from
Theradex Audit 2013: Findings & Corrective Action
 Theradex Audit 2013: Findings & Corrective Action Overview Discuss Findings and CAP for: Informed Consent Content IRB Informed Consent Eligibility Treatment Serious Adverse Events Response General Data
Theradex Audit 2013: Findings & Corrective Action Overview Discuss Findings and CAP for: Informed Consent Content IRB Informed Consent Eligibility Treatment Serious Adverse Events Response General Data
Designated Record Set Health Record The health information described below may be maintained in any medium (paper, electronic, digital, etc)
 Designated Record Set Health Record The health information described below may be maintained in any medium (paper, electronic, digital, etc) DEFINITION Designated Record Set A group of records (recorded
Designated Record Set Health Record The health information described below may be maintained in any medium (paper, electronic, digital, etc) DEFINITION Designated Record Set A group of records (recorded
Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network of Clinical Research Professionals
 ORIENTATION FOR NEW CLINICAL RESEARCH COORDINATORS Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network of Clinical Research Professionals Overall Agenda for Orientation Module 1:
ORIENTATION FOR NEW CLINICAL RESEARCH COORDINATORS Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network of Clinical Research Professionals Overall Agenda for Orientation Module 1:
How Much Money Do you Ask for When Submitting Grant Budgets?
 How Much Money Do you Ask for When Submitting Grant Budgets? Partner s Office for Women s Careers at MGH Kay Ryan, Director for Clinical Research Operations at MGH Loralie Brennen, Manager for Coordinator
How Much Money Do you Ask for When Submitting Grant Budgets? Partner s Office for Women s Careers at MGH Kay Ryan, Director for Clinical Research Operations at MGH Loralie Brennen, Manager for Coordinator
Good Clinical Practice: A Ground Level View
 Good Clinical Practice: A Ground Level View Jeanna Julo, BA, BA, CCRP Assistant Director, Clinical Data Management & Quality Controls, Auditing & Training Clinical Research Administration Research Institute,
Good Clinical Practice: A Ground Level View Jeanna Julo, BA, BA, CCRP Assistant Director, Clinical Data Management & Quality Controls, Auditing & Training Clinical Research Administration Research Institute,
Transitional Care Management Services: New Codes, New Requirements
 Transitional Care Management Services: New Codes, New Requirements hospital 99496 99495 99496 family practice o n Jan. 1, 2013, the much anticipated transitional care management (TCM) Two new codes will
Transitional Care Management Services: New Codes, New Requirements hospital 99496 99495 99496 family practice o n Jan. 1, 2013, the much anticipated transitional care management (TCM) Two new codes will
PATIENT PORTAL USERS GUIDE
 PATIENT PORTAL USERS GUIDE V 5.0 December 2012 eclinicalworks, 2012. All rights reserved Login and Pre-Registration Patients enter a valid Username and secure Password, then click the Sign In button to
PATIENT PORTAL USERS GUIDE V 5.0 December 2012 eclinicalworks, 2012. All rights reserved Login and Pre-Registration Patients enter a valid Username and secure Password, then click the Sign In button to
Exclusively for Health Advocate Members. All-in-1 Benefit. Benefits Gateway Personal Dashboard Healthcare Help Wellness Support EAP+Work/Life
 Exclusively for Health Advocate Members All-in-1 Benefit Benefits Gateway Benefits Gateway Connect to the right benefit Welcome to HealthAdvocate Health Advocate is a service provided by your employer
Exclusively for Health Advocate Members All-in-1 Benefit Benefits Gateway Benefits Gateway Connect to the right benefit Welcome to HealthAdvocate Health Advocate is a service provided by your employer
Forward-Thinking Strategies for Marketing Employed Physicians
 Forward-Thinking Strategies for Marketing Employed Physicians by Carolyn Merriman If you asked people 15 years ago to describe the medical profession, they might have labeled it a cottage industry because
Forward-Thinking Strategies for Marketing Employed Physicians by Carolyn Merriman If you asked people 15 years ago to describe the medical profession, they might have labeled it a cottage industry because
Online Tools and Resources
 Online Tools and Resources Log on to Blue Access for Members SM Go to bcbstx.com via web or mobile Or click Register Now for New Users To register you will need your ID number (1) on the back your ID card,
Online Tools and Resources Log on to Blue Access for Members SM Go to bcbstx.com via web or mobile Or click Register Now for New Users To register you will need your ID number (1) on the back your ID card,
CLINICAL RESEARCH BILLING 101
 CLINICAL RESEARCH BILLING 101 HCCA Research Compliance Conference October 31, 2007 Ann G. Mathias, JD, MHSA Ann E. Mitch-Resignalo, RN, MNEd Prepared September 2007 Updated October 2007 UPMC l University
CLINICAL RESEARCH BILLING 101 HCCA Research Compliance Conference October 31, 2007 Ann G. Mathias, JD, MHSA Ann E. Mitch-Resignalo, RN, MNEd Prepared September 2007 Updated October 2007 UPMC l University
Vascular Surgery Academic Coordinating Center (VSACC) & Peripheral Vascular Core Lab (PVCL)
 Vascular Surgery Academic Coordinating Center (VSACC) & Peripheral Vascular Core Lab (PVCL) What are the VSACC and the PVCL? The Vascular Surgery Academic Coordinating Center (VSACC) is an academic research
Vascular Surgery Academic Coordinating Center (VSACC) & Peripheral Vascular Core Lab (PVCL) What are the VSACC and the PVCL? The Vascular Surgery Academic Coordinating Center (VSACC) is an academic research
Record Keeping - Legal and Ethical Core CPD
 Record Keeping - Legal and Ethical Core CPD Aims: This article provides information about record keeping and the legal aspects relating to record keeping; details about CQC requirements for record keeping;
Record Keeping - Legal and Ethical Core CPD Aims: This article provides information about record keeping and the legal aspects relating to record keeping; details about CQC requirements for record keeping;
Yes. Phone encounter? Part of screening process. Consent and enroll patient. Apply Treatment Plan. Verify link to Timeline. Conduct patient assessment
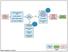 Referral - introduction to study Review consent and study Answer questions Review medical history Phone encounter? IRB approval Request required permission to schedule test (could change at screening)
Referral - introduction to study Review consent and study Answer questions Review medical history Phone encounter? IRB approval Request required permission to schedule test (could change at screening)
Subject: Coordination and Continuity of Care for enrollees with Special Healthcare Needs Services for DMAP Members (Page 1 of 5)
 (Page 1 of 5) Objective: To ensure that Health Share/ Tuality Health Alliance (THA) members with special needs are identified and provided individual attention directed to meeting their special health
(Page 1 of 5) Objective: To ensure that Health Share/ Tuality Health Alliance (THA) members with special needs are identified and provided individual attention directed to meeting their special health
Once the feasibility assessment has been conducted the study team will be notified via (Appendix 3) of the outcome and whether the study is;
 1. INTRODUCTION 2. SCOPE Feasibility assessments will ascertain any operational concerns about a research study which may delay NHS Permission or cause issues with study delivery. By conducting an assessment
1. INTRODUCTION 2. SCOPE Feasibility assessments will ascertain any operational concerns about a research study which may delay NHS Permission or cause issues with study delivery. By conducting an assessment
11/15/2012. Course Overview. Course Overview. What are your challenges? What is stressing you out? DENTAL PRACTICE SOLUTIONS
 DENTAL PRACTICE SOLUTIONS It s More Than Just a Prophy: The Missing,,, One of Dentistry Today s Top Consultants Debra Seidel Bittke, RDH, BS What are your challenges? What is stressing you out? Course
DENTAL PRACTICE SOLUTIONS It s More Than Just a Prophy: The Missing,,, One of Dentistry Today s Top Consultants Debra Seidel Bittke, RDH, BS What are your challenges? What is stressing you out? Course
Targeted technology and data management solutions for observational studies
 Targeted technology and data management solutions for observational studies August 18th 2016 Zia Haque Arshad Mohammed Copyright 2016 Quintiles Your Presenters Zia Haque Senior Director of Data Management,
Targeted technology and data management solutions for observational studies August 18th 2016 Zia Haque Arshad Mohammed Copyright 2016 Quintiles Your Presenters Zia Haque Senior Director of Data Management,
MASTER COURSE SYLLABUS
 MASTER COURSE SYLLABUS PNR 130 ~ MEDICAL SURGICAL NURSING II Course Number PNR 130 Course Title Medical Surgical Nursing II Credit Hours 5 Prerequisites 80 in PNR 120 Medical Surgical Nursing I, 80 in
MASTER COURSE SYLLABUS PNR 130 ~ MEDICAL SURGICAL NURSING II Course Number PNR 130 Course Title Medical Surgical Nursing II Credit Hours 5 Prerequisites 80 in PNR 120 Medical Surgical Nursing I, 80 in
Care Management Policies
 POLICY: Category: Care Management Policies Care Management 2.1 Patient Tracking and Registry Functions Effective Date: Est. 12/1/2010 Revised Date: Purpose: To ensure management and monitoring of patient
POLICY: Category: Care Management Policies Care Management 2.1 Patient Tracking and Registry Functions Effective Date: Est. 12/1/2010 Revised Date: Purpose: To ensure management and monitoring of patient
The Care Compact. 11 PCPI All rights reserved.
 The Care Compact There are several change package ideas provided in this tool kit and none were more important than the Care Compact during the pilot project. It will be your starting point. So, what is
The Care Compact There are several change package ideas provided in this tool kit and none were more important than the Care Compact during the pilot project. It will be your starting point. So, what is
I. Description. Triage Counseling is an individual level intervention that establishes a direct link between primary. Rural
 Rural triage Counseling 2 Triage Counseling is an individual level intervention that establishes a direct link between primary medical care and mental health services for patients living with HIV. The
Rural triage Counseling 2 Triage Counseling is an individual level intervention that establishes a direct link between primary medical care and mental health services for patients living with HIV. The
Choice PPO Retired Employees Health Program Non-Medicare Eligible Retired Members
 Choice PPO Retired Employees Health Program Non-Medicare Eligible Retired Members DEDUCTIBLE (per calendar year) Annual in-network deductible must be paid first for the following services: Imaging, hospital
Choice PPO Retired Employees Health Program Non-Medicare Eligible Retired Members DEDUCTIBLE (per calendar year) Annual in-network deductible must be paid first for the following services: Imaging, hospital
LifeBridge Health HIPAA Policy 4. Uses of Protected Health Information for Research
 LifeBridge Health HIPAA Policy 4 Uses of Protected Health Information for Research This Policy contains the following Sections: I. Policy II. III. IV. Definitions Applicability Procedures A. Individual
LifeBridge Health HIPAA Policy 4 Uses of Protected Health Information for Research This Policy contains the following Sections: I. Policy II. III. IV. Definitions Applicability Procedures A. Individual
AN OVERVIEW OF CLINICAL STUDY TASKS AND ACTIVITIES
 1 AN OVERVIEW OF CLINICAL STUDY TASKS AND ACTIVITIES Key Clinical Study Tasks and Activities 2 Discussion of Key Tasks and Activities 3 Development of the Clinical Protocol and Study Materials 3 Qualification
1 AN OVERVIEW OF CLINICAL STUDY TASKS AND ACTIVITIES Key Clinical Study Tasks and Activities 2 Discussion of Key Tasks and Activities 3 Development of the Clinical Protocol and Study Materials 3 Qualification
INSPIRing Changes to the IRB Process: New templates and more
 INSPIRing Changes to the IRB Process: New templates and more John F. Ennever, MD, PhD, CIP Director, Human Research Protection Program Office of Human Research Affairs Boston Medical Center and Boston
INSPIRing Changes to the IRB Process: New templates and more John F. Ennever, MD, PhD, CIP Director, Human Research Protection Program Office of Human Research Affairs Boston Medical Center and Boston
NURSING Nursing Practice V COURSE OUTLINE. Fall 2004
 UNIVERSITY OF ALBERTA COLLABORATIVE BACCALAUREATE NURSING PROGRAM Grande Prairie Regional College Grant MacEwan College Keyano College Red Deer College University of Alberta NURSING 3910 Nursing Practice
UNIVERSITY OF ALBERTA COLLABORATIVE BACCALAUREATE NURSING PROGRAM Grande Prairie Regional College Grant MacEwan College Keyano College Red Deer College University of Alberta NURSING 3910 Nursing Practice
eqsuite User Guide for Electronic Review Request Acute Inpatient Medical/Surgical DRG Reimbursed
 eqsuite User Guide for Electronic Review Request Acute Inpatient Medical/Surgical DRG Reimbursed CONTENTS OVERVIEW OF SYSTEM FEATURES... 3 ACCESSING THE SYSTEM... 4 USER LOG IN - GETTING STARTED... 5 SUBMITTING
eqsuite User Guide for Electronic Review Request Acute Inpatient Medical/Surgical DRG Reimbursed CONTENTS OVERVIEW OF SYSTEM FEATURES... 3 ACCESSING THE SYSTEM... 4 USER LOG IN - GETTING STARTED... 5 SUBMITTING
USING PSYCKES TO SUPPORT CARE COORDINATION IN NEW YORK STATE
 USING PSYCKES TO SUPPORT CARE COORDINATION IN NEW YORK STATE NYS Office of Mental Health Edith Kealey, PhD Deputy Director, PSYCKES OVERVIEW Introduction to PSYCKES: The Psychiatric Services and Clinical
USING PSYCKES TO SUPPORT CARE COORDINATION IN NEW YORK STATE NYS Office of Mental Health Edith Kealey, PhD Deputy Director, PSYCKES OVERVIEW Introduction to PSYCKES: The Psychiatric Services and Clinical
Office of Clinical Research. CTMS Reference Guide Patient Entry & Visit Tracking
 Se Office of Clinical Research CTMS Reference Guide Patient Entry & Visit Tracking Table of Contents Logging into CTMS... 3 Search and Recruitment / Quick Search... 4 How to Configure Quick Search Fields...
Se Office of Clinical Research CTMS Reference Guide Patient Entry & Visit Tracking Table of Contents Logging into CTMS... 3 Search and Recruitment / Quick Search... 4 How to Configure Quick Search Fields...
Implementing Health Coaching
 Implementing Health Coaching Presented by: Amireh Ghorob, MPH Adriana Najmabadi Camille Prado UCSF Center for Excellence in Primary Care IHI Summit 2014, Washington DC March 10, 2014 Session: L9 These
Implementing Health Coaching Presented by: Amireh Ghorob, MPH Adriana Najmabadi Camille Prado UCSF Center for Excellence in Primary Care IHI Summit 2014, Washington DC March 10, 2014 Session: L9 These
Steps when Subjects are Scheduled at CTRC
 CTRC Statement of Understanding Scheduling Subjects in the CTRC (5/6/14) As part of our move to the new location on Hatfield 10D, our scheduling processes have changed. The CTRC has begun scheduling all
CTRC Statement of Understanding Scheduling Subjects in the CTRC (5/6/14) As part of our move to the new location on Hatfield 10D, our scheduling processes have changed. The CTRC has begun scheduling all
Physician Assistant Jurisprudence Examination
 Physician ssistant Jurisprudence xamination The examination you take will be composed of 50 questions, randomly selected from the questions listed below. You will have 90 minutes to take the exam. For
Physician ssistant Jurisprudence xamination The examination you take will be composed of 50 questions, randomly selected from the questions listed below. You will have 90 minutes to take the exam. For
Informed Consent/Refusal Yes No Comments
 Dental Office Self-assessment Tool This checklist is designed to help dentists evaluate their risk management readiness and take measures to reduce exposure. While it focuses on the topics explored in
Dental Office Self-assessment Tool This checklist is designed to help dentists evaluate their risk management readiness and take measures to reduce exposure. While it focuses on the topics explored in
Physician Referral: Laws, Rules, and Ethics
 Physician Referral: Laws, Rules, and Ethics Nabil El Sanadi, MD, MBA, FACEP Chairman, Council on Ethical and Judicial Affairs, Florida Medical Association Chief of Emergency Medicine, Broward Health Clinical
Physician Referral: Laws, Rules, and Ethics Nabil El Sanadi, MD, MBA, FACEP Chairman, Council on Ethical and Judicial Affairs, Florida Medical Association Chief of Emergency Medicine, Broward Health Clinical
Preparing for your visit to the Gattuso Rapid Diagnostic Centre
 Preparing for your visit to the Gattuso Rapid Diagnostic Centre Princess Margaret For patients receiving testing for breast changes Read this resource to learn: What the Rapid Diagnostic Centre does Where
Preparing for your visit to the Gattuso Rapid Diagnostic Centre Princess Margaret For patients receiving testing for breast changes Read this resource to learn: What the Rapid Diagnostic Centre does Where
USF HRPP Updates October 2017
 USF HRPP Updates October 2017 Julie Moore, J.D., M.S. PA, CIP Associate Director, Research Integrity & Compliance Research Integrity and Compliance Revised Common Rule 45 CFR 46 Effective date and compliance
USF HRPP Updates October 2017 Julie Moore, J.D., M.S. PA, CIP Associate Director, Research Integrity & Compliance Research Integrity and Compliance Revised Common Rule 45 CFR 46 Effective date and compliance
IN ACCORDANCE WITH THIS INTERNATIONAL DEVICE STANDARD
 COMPLIANCE WITH ISO 14155:2011 GUIDANCE FOR ENSURING YOUR CLINICAL STUDY IS BEING DESIGNED, EXECUTED, AND MONITORED IN ACCORDANCE WITH THIS INTERNATIONAL DEVICE STANDARD Introduction: An increasing trend
COMPLIANCE WITH ISO 14155:2011 GUIDANCE FOR ENSURING YOUR CLINICAL STUDY IS BEING DESIGNED, EXECUTED, AND MONITORED IN ACCORDANCE WITH THIS INTERNATIONAL DEVICE STANDARD Introduction: An increasing trend
Payment Policy: High Complexity Medical Decision-Making Reference Number: CC.PP.051 Product Types: ALL
 Payment Policy: High Complexity Medical Decision-Making Reference Number: CC.PP.051 Product Types: ALL Effective Date: 6/2017 Last Review Date: See Important Reminder at the end of this policy for important
Payment Policy: High Complexity Medical Decision-Making Reference Number: CC.PP.051 Product Types: ALL Effective Date: 6/2017 Last Review Date: See Important Reminder at the end of this policy for important
Patient Case Records Review
 Patient Case Records Review Debra Herzan, RN, BSN, OCN, CCRP Alliance for Clinical Trials in Oncology Audit Prep Workshop - Alliance Group Meeting November, 2014 6 Categories l Informed Consent l Eligibility
Patient Case Records Review Debra Herzan, RN, BSN, OCN, CCRP Alliance for Clinical Trials in Oncology Audit Prep Workshop - Alliance Group Meeting November, 2014 6 Categories l Informed Consent l Eligibility
Saint Francis University. Health and Wellness Program
 2015 Saint Francis University www.francis.edu/disepio Health and Wellness Program ABOUT THE WELLNESS PROGRAM Saint Francis University is committed to being a University where employee health and wellness
2015 Saint Francis University www.francis.edu/disepio Health and Wellness Program ABOUT THE WELLNESS PROGRAM Saint Francis University is committed to being a University where employee health and wellness
Process and methods Published: 23 January 2017 nice.org.uk/process/pmg31
 Evidence summaries: process guide Process and methods Published: 23 January 2017 nice.org.uk/process/pmg31 NICE 2018. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Evidence summaries: process guide Process and methods Published: 23 January 2017 nice.org.uk/process/pmg31 NICE 2018. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Primum Computer-based Case Simulations (CCS) Frequently Asked Questions (FAQs)
 Primum Computer-based Case Simulations (CCS) Frequently Asked Questions (FAQs) 1. What are my responsibilities? You should function as a primary care physician and maintain responsibility for the patient
Primum Computer-based Case Simulations (CCS) Frequently Asked Questions (FAQs) 1. What are my responsibilities? You should function as a primary care physician and maintain responsibility for the patient
Healthcare Referral Program Mutual Patient Care/API
 Healthcare Referral Program Mutual Patient Care/API A. Program Goals I. To create the most comprehensive and formal referral program for dental rehabilitation in Northern Arizona II. Create, then expand,
Healthcare Referral Program Mutual Patient Care/API A. Program Goals I. To create the most comprehensive and formal referral program for dental rehabilitation in Northern Arizona II. Create, then expand,
Quanum Electronic Health Record Frequently Asked Questions
 Quanum Electronic Health Record Frequently Asked Questions Table of Contents... 4 What is Quanum EHR?... 4 What are the current capabilities of Quanum EHR?... 4 Is Quanum EHR an EMR?... 5 Can I have Quanum
Quanum Electronic Health Record Frequently Asked Questions Table of Contents... 4 What is Quanum EHR?... 4 What are the current capabilities of Quanum EHR?... 4 Is Quanum EHR an EMR?... 5 Can I have Quanum
Retail Clinics in Healthcare: Overcoming Complex Legal Challenges
 Presenting a live 90-minute webinar with interactive Q&A Retail Clinics in Healthcare: Overcoming Complex Legal Challenges Complying With Corporate Practice of Medicine, Licensure, and Scope of Practice
Presenting a live 90-minute webinar with interactive Q&A Retail Clinics in Healthcare: Overcoming Complex Legal Challenges Complying With Corporate Practice of Medicine, Licensure, and Scope of Practice
Research & Development. Case Report Form SOP. J H Pacynko and J Illingworth. Research, pharmacy and R&D staff
 Department Title of SOP Research & Development Case Report Form SOP SOP reference no: R&D GCP SOP 03 Authors: Current version number and date: J H Pacynko and J Illingworth Version 2, 01.02.18 Next review
Department Title of SOP Research & Development Case Report Form SOP SOP reference no: R&D GCP SOP 03 Authors: Current version number and date: J H Pacynko and J Illingworth Version 2, 01.02.18 Next review
ONE STOP CAREER CENTERS CUSTOMER HANDBOOK
 Workforce Alliance CT Department of Labor Human Resource Agency ONE STOP CAREER CENTERS CUSTOMER HANDBOOK 3 Convenient Locations CT Works New Haven 560 Ella T. Grasso Blvd. New Haven, CT 06519 (203)-624-1493
Workforce Alliance CT Department of Labor Human Resource Agency ONE STOP CAREER CENTERS CUSTOMER HANDBOOK 3 Convenient Locations CT Works New Haven 560 Ella T. Grasso Blvd. New Haven, CT 06519 (203)-624-1493
https://register.clinicaltrials.gov/prs/app/template/help%2chelpprotocolmo...
 Page 1 of 12 Help: Protocol Modules > All Help: Protocol Modules - All Protocol module help pages contain an introduction, examples, data entry tips and a review checklist for each module in the protocol
Page 1 of 12 Help: Protocol Modules > All Help: Protocol Modules - All Protocol module help pages contain an introduction, examples, data entry tips and a review checklist for each module in the protocol
OVERVIEW OF ESSENTIAL CHARTING ELEMENTS FOR THE EMERGENCY DEPARTMENT
 OVERVIEW OF ESSENTIAL CHARTING ELEMENTS FOR THE EMERGENCY DEPARTMENT ALL CHARTING NEEDS TO BE FINISHED AT THE END OF YOUR SHIFT PRIOR TO LEAVING THE ED IF YOU HAVE ANY QUESTIONS, ASK FOR HELP! All of the
OVERVIEW OF ESSENTIAL CHARTING ELEMENTS FOR THE EMERGENCY DEPARTMENT ALL CHARTING NEEDS TO BE FINISHED AT THE END OF YOUR SHIFT PRIOR TO LEAVING THE ED IF YOU HAVE ANY QUESTIONS, ASK FOR HELP! All of the
DUKE GENERAL MEDICINE SENIOR RESIDENT ORIENTATION
 Department of Medicine Hospital Medicine Program 2012-2013 DUKE GENERAL MEDICINE SENIOR RESIDENT ORIENTATION Your responsibilities and goals as the supervising resident on the Duke General Medicine Service
Department of Medicine Hospital Medicine Program 2012-2013 DUKE GENERAL MEDICINE SENIOR RESIDENT ORIENTATION Your responsibilities and goals as the supervising resident on the Duke General Medicine Service
NextGen Preventative Exam Template
 NextGen Preventative Exam Template Summary This guide describes the use of the Preventive Exam HPI template to document both the initial Welcome to Medicare Exam and subsequent Annual Wellness Visits.
NextGen Preventative Exam Template Summary This guide describes the use of the Preventive Exam HPI template to document both the initial Welcome to Medicare Exam and subsequent Annual Wellness Visits.
Transition of Care Guide
 Transition of Care Guide If you received treatment for a mental health condition while in high school or before, there are several things you can do to continue to stay healthy while transitioning to and
Transition of Care Guide If you received treatment for a mental health condition while in high school or before, there are several things you can do to continue to stay healthy while transitioning to and
Frequently Asked Questions. The CIRB is located at 168 Jalan Bukit Merah #06-08 Tower 3 Connection One Singapore
 1. Where is the location of the CIRB? The CIRB is located at 168 Jalan Bukit Merah #06-08 Tower 3 Connection One Singapore 150168. 2. When is the submission deadline? The CIRB meets once a month. The submission
1. Where is the location of the CIRB? The CIRB is located at 168 Jalan Bukit Merah #06-08 Tower 3 Connection One Singapore 150168. 2. When is the submission deadline? The CIRB meets once a month. The submission
Qualified/registered nursing workforce survey
 Qualified/registered nursing workforce survey Guidelines for completion Please use this document as a guide to complete the online survey at www.nhsemployers.org/nursingworkforcesurvey. Each NHS provider
Qualified/registered nursing workforce survey Guidelines for completion Please use this document as a guide to complete the online survey at www.nhsemployers.org/nursingworkforcesurvey. Each NHS provider
The Medical Neighborhood: Ensuring Continuity of Care with Hospital and Specialist Neighborhoods
 The Medical Neighborhood: Ensuring Continuity of Care with Hospital and Specialist Neighborhoods R. Scott Hammond MD, FAAFP Chair, CAFP PCMH Task Force Medical Director, SOC-PCMH Initiative, Colorado Associate
The Medical Neighborhood: Ensuring Continuity of Care with Hospital and Specialist Neighborhoods R. Scott Hammond MD, FAAFP Chair, CAFP PCMH Task Force Medical Director, SOC-PCMH Initiative, Colorado Associate
Referred Patient Alerts & Online Recruitment Manager for Sites Instructions
 Referred Patient Alerts & Online Recruitment Manager for Sites Instructions The following training documentation will explain how you will obtain Referred Patient Alerts (RPAs), update a patient s enrollment
Referred Patient Alerts & Online Recruitment Manager for Sites Instructions The following training documentation will explain how you will obtain Referred Patient Alerts (RPAs), update a patient s enrollment
Dimension: I. Care Facilitation Specific Skills. Skill Rating Fail Pass
 T RI- S E R V I C E BHCF CORE C O M P E T E N C Y T OOL BHCF: Date: Trainer: A certified BHCF Trainer rates the BHCF trainee skill level based on their observations of trainee performance of each dimension.
T RI- S E R V I C E BHCF CORE C O M P E T E N C Y T OOL BHCF: Date: Trainer: A certified BHCF Trainer rates the BHCF trainee skill level based on their observations of trainee performance of each dimension.
Meets advising session registration requirement for the NURSA 1105 course for FALL 2018 through SUMMER 2019 at College of DuPage
 Meets advising session registration requirement for the NURSA 1105 course for FALL 2018 through SUMMER 2019 at College of DuPage Before you begin Navigate to the CNA Home page: http://www.cod.edu/programs/bna/
Meets advising session registration requirement for the NURSA 1105 course for FALL 2018 through SUMMER 2019 at College of DuPage Before you begin Navigate to the CNA Home page: http://www.cod.edu/programs/bna/
FDA Medical Device Regulations vs. ISO 14155
 Vol. 11, No. 9, September 2015 Happy Trials to You FDA Medical Device Regulations vs. ISO 14155 By Shawn Kennedy Medical device clinical trials must comply with 21 CFR Parts 11 (Electronic Records), 50
Vol. 11, No. 9, September 2015 Happy Trials to You FDA Medical Device Regulations vs. ISO 14155 By Shawn Kennedy Medical device clinical trials must comply with 21 CFR Parts 11 (Electronic Records), 50
Instructions for Completing a Human Research Billing Analysis Form
 Instructions for Completing a Human Research Billing Analysis Form Principal Investigators are required to submit one Human Research Billing Analysis Form per research protocol at the time of the IRB submission
Instructions for Completing a Human Research Billing Analysis Form Principal Investigators are required to submit one Human Research Billing Analysis Form per research protocol at the time of the IRB submission
MASTER COURSE SYLLABUS
 MASTER COURSE SYLLABUS PNR 155 ~ MATERNAL/INFANT/CHILD NURSING Course Number PNR 155 Course Title Maternal/Infant/Child Nursing Credit Hours 7 Prerequisites 80 in PNR 140 Medical Surgical Nursing III,
MASTER COURSE SYLLABUS PNR 155 ~ MATERNAL/INFANT/CHILD NURSING Course Number PNR 155 Course Title Maternal/Infant/Child Nursing Credit Hours 7 Prerequisites 80 in PNR 140 Medical Surgical Nursing III,
Medicare s Impact on Cardiology Drugs and Devices During Clinical Research
 Medicare s Impact on Cardiology Drugs and Devices During Clinical Research Ryan Meade, JD Meade & Roach, LLP July 15, 2008 Baltimore, Maryland University of Maryland School of Medicine 1 Overview Theme:
Medicare s Impact on Cardiology Drugs and Devices During Clinical Research Ryan Meade, JD Meade & Roach, LLP July 15, 2008 Baltimore, Maryland University of Maryland School of Medicine 1 Overview Theme:
Radiography Program. Caldwell Community College & Technical Institute
 Radiography Program Caldwell Community College & Technical Institute Purpose To provide Radiography applicants with a program overview Application and admission requirements Curriculum and program requirements
Radiography Program Caldwell Community College & Technical Institute Purpose To provide Radiography applicants with a program overview Application and admission requirements Curriculum and program requirements
Good Documentation Practices. Human Subject Research. for
 Good Documentation Practices for Human Subject Research Bridget M. Psicihulis, RHIA, CCRC Quality Improvement Unit Coordinator Human Research Protection Program Wheaton Franciscan Healthcare (last updated
Good Documentation Practices for Human Subject Research Bridget M. Psicihulis, RHIA, CCRC Quality Improvement Unit Coordinator Human Research Protection Program Wheaton Franciscan Healthcare (last updated
STUDY TEAM RESPONSIBILITIES ORIENTATION FOR NEW CLINICAL RESEARCH PERSONNEL MODULE 2
 ORIENTATION FOR NEW CLINICAL RESEARCH PERSONNEL MODULE 2 Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network for Research Professionals Overall Agenda for Orientation Module 1: Introduction
ORIENTATION FOR NEW CLINICAL RESEARCH PERSONNEL MODULE 2 Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network for Research Professionals Overall Agenda for Orientation Module 1: Introduction
