CURRENT COGR PRIORITIES - BY COMMITTEE (7/10/17)
|
|
|
- Claire Wilson
- 6 years ago
- Views:
Transcription
1 CURRENT COGR PRIORITIES - BY COMMITTEE (7/10/17) No. 1 Student Financial Aid and "Securing Student Information" and Compliance Supplement New single audit requirement. Delayed until MEDIUM Pending Edu cause, NACUBO, NASFAA, AICPA, and NASACT OMB, Dept. of Ed. 2 2/2/2017, RRR HHS Policy Office engagement and Prior collaboration with 120-day close-out, Closeout advocating for changes to Grant Closeout report, encourage all OpDivs to join RTCs. Recent policies. outreach to new HHS POC MEDIUM Pending HHS 3 NIH Notice NOT-OD ; reminds recipients to submit timely closeouts. The Notice may be a precursor to more active enforcement of timely closeouts. MEDIUM Pending 4 RRR OMB Letter to Reduce Administrative Burden OMB request (in coordination with procurement issue) to provide actions to reduce burden LOW Complete OMB 5 Procurement Standards (2 CFR ) implementation, including MicroPurchaseThreshold (MPT). Most schools will have to implement 7/1/2018 (some 9/1/2018). MPT guidance (NDAA) may not be available, which will create uncertainty. AIRI 6 2/2/2017, RRR Work with Foundations to address low F&A rates. In light of proposed NIH 10% cap, and Foundations cited by Members of Congress, sense of urgency to better partner with the Foundations. Milken Institute, Faster Cures 7 RRR F&A and proposed NIH 10% cap. Possibly other agencies, too? Even if Trump budget is rejected, unilateral action by HHS/OMB is possible. AAU, APLU, AAMC, AIRI, NACUBO For COGR Membership Use Only - Do Not Forward or Distribute 1
2 8 2/2/2017,, RRR Uniform Guidance, 2 CFR Part 200, revisions. We were awaiting release of Fed Register (FR) Notice and a separate release of FAQs. However, per OMB, this initiative is no longer active. LOW Pending 9 COFAR is disbanded. Could this impact status of COFAR / UG FAQs? Consensus is "No", since these have been memorialized in the RTC, the Compliance Supplement, and possibly on the CFOC website. MEDIUM 10 2/2/2017 F&A Survey Final reports, negotiation experiences, and Exec Summary LOW Complete Off Campus Research Centers / Lease Costs Request to NIH to modify direct cost maximum to exclude lease costs, so as not to unfairly penalize off campus research centers. Considering "SPECIAL NOTES" per the F&A rate agreement solution MEDIUM 11 2/2/2017 HHS-CAS 2017 Best Practices Manual Internal Guidance for CAS rate negotiators to review F&A rate proposals. MEDIUM Complete HHS 12 2/2/ /2/ /2/2017 CAS Interpretation of Software Capital Threshold per the UG F&A concerns (CAS): DS-2, UCA, On- Off definition per F&A rate agreement CAS is using a strict $5,000 for all flavors of software purchased with federal funds. Requires HHS-CAS and/or OMB engagement. With OMB currently disengaged in UG issues, some of these issues are tabled, for now. MEDIUM Pending MEDIUM Pending 15 2/2/2017 F&A concerns (ONR): Interest expense, 4 year extensions Issues that required ONR and/or OMB engagement LOW Complete For COGR Membership Use Only - Do Not Forward or Distribute 2
3 Student Financial Aid and Dept of Ed. Requirement for Annual Audit (Compliance Supplement) Dept. of Education may require an annual audit of the SFA Cluster, though UG and other guidance does not support. MEDIUM NASACT, NACUBO, AICPA 16 2/2/2017 Interpretation of "Reimbursement" and "Advance Payment" (Compliance Supplement) Single auditors, under pressure from IGs, are starting to require "cancelled checks" as documentation showing "Advance billing" was not done. 17 2/2/2017 engagement in commenting on the 2018 Compliance Supplement. "Reimbursement" an "Advance Payment" issue is the most significant concern and possibly could be addressed in the 2018 CS. 18 Cost of Data/IT enterprise. Follow up to Thursday AM session at 6/8/17 COGR meeting and further engagement in related costing issues. MEDIUM Restriction in Health Coverage Benefits; NIH, DOL, other agencies Federal funds cannot be used for abortions. Longstanding policy, but new concern on how we demonstrate compliance. Our internal policies provide the needed documentation, but risk assessment may still be appropriate. LOW Complete NSF IG memo to OMB covering "observations" on the FDP payroll certification pilots. NSF IG cited monthly/bi-monthly reconciliations and full allocations for PI salaries. MEDIUM Complete 21 For COGR Membership Use Only - Do Not Forward or Distribute 3
4 Dept. of Ed. open licensing requirement (82 FR 7376; 1/19/17) Requires open licensing to the public of grant deliverables created with Department of Ed. competitive grant funds. 7/7/ /2/2017 Proposed Revised Bayh-Dole Regulations (81 FR 78090; 11/7/2016) Revises 37 CFR 401 Bayh-Dole implementing regulations. 10 concerns with rule in identified in joint association 12/9 comment letter.. Stalled AUTM, AAU, APLU, AAMC 2/2/ /2/ /2/ /2/2017 Invention reporting Controlled unclassified information (CUI) Increasingly visible compliance issue with escalating concerns about drug pricing. Recent march-in and Congressional requests to NIH have heightened the concerns. Significant resource implications with increased investments in compliance training and reporting. Development of new FAR clause to meet NARA/NIST requirements. Will heavily impact contracting and compliance staff at COGR member institutions. AUTM (NIH/NIST) NIH, NIST 6/22/2017 Pending FDP NARA 6/22/2017 Patent Reform Anticipated legislative attempts to address issues associated with patent trolls. Major potential impact on university patenting and licensing. Medium AAU,APLU, AAMC, AUTM 6/22/ /2/2017 Export controls Final ITAR revised definitions still pending. Would become high if issued with changes proposed in June, 2015 Medium Awaiting Federal Response AAU, AUECO Commerce, State 6/22/ /2/2017 For COGR Membership Use Only - Do Not Forward or Distribute 4
5 Cybersecurity FAR and DFARS requirements issued in May be superseded by NARA FAR clause. MEDIUM AAU DOD 6/22/ /2/ /2/2017 RRR IP Rights in NIH PMI Initiative NIH use of Other Transactions authority, with government ownership of inventions LOW Inactive AAU/AAMC NIH 6/22/ /2/2017 Technology Transfer Evolution Fundamental shifts in roles and responsibilities of tech transfer offices - but no particular time frame. AAU/APLU/ AUTM 6/22/2017 Public Access focus on storage/data types/compliance with agency PA Plans, with to accumulate data and understand cost impacts MEDIUM AAU/APLU 4/27/ /2/ /2/2017 Research Terms/National Policy Matrices Promote uniformity and adopt RTCs across all federal agencies; address any implementation issues that arise as new RTCs are adopted Pending 4/27/ /2/2017 Dual Use Research of Concern NAS held DURC workshop on 1/4/17. Awaiting committee report that assesses the various options presented in the commissioned papers and discussed during the workshop. LOW Pending 4/27/ /1/2017 FLSA Overtime Rule Monitor status to determine administration's next steps on adoption of these requirements LOW Pending ACE, CUPA-HR 4/27/ /2/2017 RRR,, DoD Research Terms and Conditions responded to first series of six (6) NPRMs on 2/6/17. MEDIUM Pending 6/29/ /2/2017 Subrecipient Monitoring to continue to work on issues related to subrecipient monitoring - continue to advance concept of safe harbor, clarify rules around former subrecipients, address standardized subrecipient monitoring, including foreign subrecipient monitoring. FDP 4/27/2017 For COGR Membership Use Only - Do Not Forward or Distribute 5
6 37 2/2/2017 Fixed Price Subawards Clarified in March 2017 NIH requirements for prior approval for fixed price sub awards; remaining issues are monitoring burden of acquiring approvals and (with FDP) investigating mechanisms to expedite approval) such as template documents MEDIUM FDP 6/29/ /3/2017 RRR Human Subjects Protections - Common Rule Anticipate additional related publications including revised subparts; privacy and security guidance; guidance on identifiable biospecimens and identifiable private information and techniques that might render them identifiable; a revised list of research eligible for expedited review; and a possible exempt decision tool. AAMC, AAU and APLU. 6/6/ /3/2017 RRR (costing aspects) NIH Single IRB Policy Addressing the scope and timing of the rule. Identifying specific categories of research (i.e., areas of social and behavioral research) that should/could be exempt from sirb requirements. Make the case to NIH for categories for exemption rather than case-bycase exemption. focus is to ensure NIH guidance is clear on allowable costing methodologies. Evaluating the effectiveness of the policy via the assessment developed by RRR. AAMC, AAU, APLU 6/6/ /3/2017 RRR Engaging the new administration to advance regulatory reform efforts and congressional staff as needed. Organizing meetings with new leadership at OIRA, OMB, HHS and OSTP. AAU and APLU 6/6/2017 For COGR Membership Use Only - Do Not Forward or Distribute 6
7 41 2/3/2017 RRR,, as applicable Facilitating the implementation of recent regulatory reform legislation including 21 st Century Cures and the American Innovation and Competitiveness Act. Varies by provision. *See accompanying spreadsheet. AAU, AAMC, FASEB, FDP, APLU, AIRI 6/6/ RRR Reducing institutional administrative burden via the COGR checklist. Further analysis and next steps MEDIUM FASEB, AAMC 6/6/ RRR ClinicalTrials.gov Improving the interface and reducing administrative work. Potential to seek regulatory change. AAU, APLU and AAMC 6/6/ /3/2017 RRR Accreditation of Human Research Protection Programs Reducing the level of administrative work associated with the accreditation and re-accreditation process. MEDIUM 6/6/2017 For COGR Membership Use Only - Do Not Forward or Distribute 7
8 45 2/3/2017 RRR Office of Inspectors General audits. Continue to address and make members aware of OIG audits and reports. Consider a discussion on how to make more effective use of the Single Audit and reduce the number and duration of OIG audits. MEDIUM 6/6/ /3/2017 RRR Data Act Section 5 pilot. Efforts to reduce administrative burden. LOW FDP 6/6/2017 RRR NSF Intergovernmental Personnel Act Program OIG audits and 2017 pilot. LOW AAU 6/6/ /3/ /3/2017 RRR R35 Solicitations 49 4/23/2017 RRR Animal Research Regulatory Reform Continue to address inconsistencies in NIH solicitations with the Uniform Guidance and university processes and systems. Provide NIH, USDA and FDA with actionable recommendations for improving animal welfare and science while reducing regulatory burden associated with federally funded animal research in anticipation of a review that agencies are directed to conduct by the 21st Century Cures Act. MEDIUM Inactive 6/6/2017 6/6/ /6/2017 RRR Grant Support Index (GSI)/Biomedical Enterprise Efforts to stabilize the biomedical enterprise, including limiting the number of grants that an individual investigator can have. 6/6/2017 For COGR Membership Use Only - Do Not Forward or Distribute 8
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM. December 2017
 Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
2017 SRA International Annual Meeting October 14-18, Federal Demonstration Partnership (FDP)/Council on Governmental Relations (COGR) Update
 2017 SRA International Annual Meeting October 14-18, 2017 Federal Demonstration Partnership (FDP)/Council on Governmental Relations (COGR) Update Who are we? Anthony DeCrappeo, President, Council on Governmental
2017 SRA International Annual Meeting October 14-18, 2017 Federal Demonstration Partnership (FDP)/Council on Governmental Relations (COGR) Update Who are we? Anthony DeCrappeo, President, Council on Governmental
Costing and Procurement Updates
 Costing and Procurement Updates Jim Luther, Duke University Edwin Bemmel, University of Miami Doug Backman, University of South Florida Sara Bible, Stanford University January 8, 2018 Agenda Update (Costing
Costing and Procurement Updates Jim Luther, Duke University Edwin Bemmel, University of Miami Doug Backman, University of South Florida Sara Bible, Stanford University January 8, 2018 Agenda Update (Costing
UNIFORM ADMINISTRATIVE REQUIREMENTS, COST PRINCIPLES, AND AUDIT REQUIREMENTS FOR FEDERAL AWARDS - UPDATE FEBRUARY 2015
 UNIFORM ADMINISTRATIVE REQUIREMENTS, COST PRINCIPLES, AND AUDIT REQUIREMENTS FOR FEDERAL AWARDS - UPDATE FEBRUARY 2015 AOA Conference Pasadena, CA February 9, 2015 Agenda 1. Introduction / Disclaimer 2.
UNIFORM ADMINISTRATIVE REQUIREMENTS, COST PRINCIPLES, AND AUDIT REQUIREMENTS FOR FEDERAL AWARDS - UPDATE FEBRUARY 2015 AOA Conference Pasadena, CA February 9, 2015 Agenda 1. Introduction / Disclaimer 2.
Reducing Investigators Administrative Workload for Federally-Funded Research
 Reducing Investigators Administrative Workload for Federally-Funded Research National Science Board National Research Council Committee on Federal Research Regulations and Reporting Requirements February
Reducing Investigators Administrative Workload for Federally-Funded Research National Science Board National Research Council Committee on Federal Research Regulations and Reporting Requirements February
OMB Uniform Guidance ( UG ) Briefing. ASRSP & OSR Brown Bag Tuesday, January 27 th
 OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
Recent Legislative Actions Taken to Reduce Research Regulatory Burden. 21st Century Cures (Passed House and Senate. Signed into law Dec.
 . Signed into law Dec. 13) Link to PDF Link to PDF Link to PDF Research Policy Board - A public-private entity recommended by the National Academies "to foster more effective conception, development and
. Signed into law Dec. 13) Link to PDF Link to PDF Link to PDF Research Policy Board - A public-private entity recommended by the National Academies "to foster more effective conception, development and
UNIFORM GUIDANCE IMPLEMENTATION
 UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Preparing campus for what is changing and what is not What is Grants Reform and when is it effective? What
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Preparing campus for what is changing and what is not What is Grants Reform and when is it effective? What
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards (New Uniform Guidance)
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards (New Uniform Guidance) Spring Research Administrators Series March 19, 2015 1 Agenda Background UW-Madison
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards (New Uniform Guidance) Spring Research Administrators Series March 19, 2015 1 Agenda Background UW-Madison
The Lifecycle of a Sponsored Project
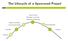 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE
 THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE NECA Conference, August 21, 2012 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE NECA Conference, August 21, 2012 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research
Sponsored Program Administration. October 10, 2017
 Sponsored Program Administration October 10, 2017 Agenda Procurement Services FDP Updates Jaggaer Pre-Award Federal Agency Updates Grants.gov Transition to Workspace NIH FORMS-E Post-Award GONE Act Certificate
Sponsored Program Administration October 10, 2017 Agenda Procurement Services FDP Updates Jaggaer Pre-Award Federal Agency Updates Grants.gov Transition to Workspace NIH FORMS-E Post-Award GONE Act Certificate
Are You Ready for This? The New Uniform Guidance 2 CFR 200
 Are You Ready for This? The New Uniform Guidance 2 CFR 200 Increase in Federal Grants Activity The Catalog of Federal Domestic Assistance lists over 2,000 Federal grant programs $600B $200B $7B $24B $91B
Are You Ready for This? The New Uniform Guidance 2 CFR 200 Increase in Federal Grants Activity The Catalog of Federal Domestic Assistance lists over 2,000 Federal grant programs $600B $200B $7B $24B $91B
OMB Uniform Guidance: Cost Principles, Audit, and Administrative Requirements for Federal Awards
 OMB Uniform Guidance: Cost Principles, Audit, and Administrative Requirements for Federal Awards Chad Person May 1, 2013 Presented By: Devesh Kamal, CPA Shareholder deveshk@cshco.com Jesse Young, CPA Principal
OMB Uniform Guidance: Cost Principles, Audit, and Administrative Requirements for Federal Awards Chad Person May 1, 2013 Presented By: Devesh Kamal, CPA Shareholder deveshk@cshco.com Jesse Young, CPA Principal
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE
 THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research organizations aimed at streamlining
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE 1 What is the FDP? The FDP is a cooperative effort among federal research agencies, universities and other research organizations aimed at streamlining
The Uniform Guidance 2 CFR 200 A Guide to Risk-Based Grants Management
 This image cannot currently be displayed. The Uniform Guidance 2 CFR 200 A Guide to Risk-Based Grants Management 2015 This image cannot currently be displayed. Increase in Federal Grants Activity The Catalog
This image cannot currently be displayed. The Uniform Guidance 2 CFR 200 A Guide to Risk-Based Grants Management 2015 This image cannot currently be displayed. Increase in Federal Grants Activity The Catalog
NECA Update The New Uniform Guidance 2 CFR 200
 NECA Update The New Uniform Guidance 2 CFR 200 September 23, 2014 Increase in Federal Grants Activity The Catalog of Federal Domestic Assistance lists over 2,000 Federal grant programs $600B $200B $7B
NECA Update The New Uniform Guidance 2 CFR 200 September 23, 2014 Increase in Federal Grants Activity The Catalog of Federal Domestic Assistance lists over 2,000 Federal grant programs $600B $200B $7B
Implementing the OMB s Super Circular (aka UGG) Presented by: Anne Fritz, Finance Director, City of St. Petersburg, Florida
 Implementing the OMB s Super Circular (aka UGG) Presented by: Anne Fritz, Finance Director, City of St. Petersburg, Florida Acknowledgement to Heather Acker, Partner, Baker Tilly, Virchow Krause, LLP for
Implementing the OMB s Super Circular (aka UGG) Presented by: Anne Fritz, Finance Director, City of St. Petersburg, Florida Acknowledgement to Heather Acker, Partner, Baker Tilly, Virchow Krause, LLP for
Rebecca Trahan. Office of Sponsored Programs December 9, ORED Limited Submission Update
 Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
National Academy of Sciences Committee on University IP Management
 National Academy of Sciences Committee on University IP Management June 30, 2008 Robert Hardy Director, Contracts and IP Management Council on Governmental Relations A Word About COGR Council on Governmental
National Academy of Sciences Committee on University IP Management June 30, 2008 Robert Hardy Director, Contracts and IP Management Council on Governmental Relations A Word About COGR Council on Governmental
The Uniform Guidance (2 CFR, Part 200)
 WCMC Implementation of The Uniform Guidance (2 CFR, Part 200) Tuesday, September 22, 2015 & Wednesday, September 23, 2015 UG Workshop Michelle A. Lewis, M.S. Director of Research Administration Interim
WCMC Implementation of The Uniform Guidance (2 CFR, Part 200) Tuesday, September 22, 2015 & Wednesday, September 23, 2015 UG Workshop Michelle A. Lewis, M.S. Director of Research Administration Interim
OMB Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards
 OMB Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Research Advisory Board What PIs Need to Know January 2015 1 Background o When and Why? o Overview Important
OMB Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Research Advisory Board What PIs Need to Know January 2015 1 Background o When and Why? o Overview Important
Navigating the New Uniform Grant Guidance. Jack Reagan, Audit Partner Grant Thornton LLP. Grant Thornton. All rights reserved.
 Navigating the New Uniform Grant Guidance Jack Reagan, Audit Partner Grant Thornton LLP Objectives What s New with OMB: Uniform Administrative Requirements, Cost Principles, and Audit requirements for
Navigating the New Uniform Grant Guidance Jack Reagan, Audit Partner Grant Thornton LLP Objectives What s New with OMB: Uniform Administrative Requirements, Cost Principles, and Audit requirements for
Something for Everyone: Adjusting to the OMB s Super Circular May 25, :30 10:10 am 2 CPE
 Something for Everyone: Adjusting to the OMB s Super Circular May 25, 2016 8:30 10:10 am 2 CPE Heather Acker, Partner, Baker Tilly Virchow Krause, LLP Anne Fritz, Finance Director, City of St. Petersburg,
Something for Everyone: Adjusting to the OMB s Super Circular May 25, 2016 8:30 10:10 am 2 CPE Heather Acker, Partner, Baker Tilly Virchow Krause, LLP Anne Fritz, Finance Director, City of St. Petersburg,
Document Downloaded: Monday July 27, Federal Research Policy - Recommendations to the NRC, January Author: David Kennedy
 Document Downloaded: Monday July 27, 2015 Federal Research Policy - Recommendations to the NRC, January 2011 Author: David Kennedy Published Date: 06/23/2011 AAU Association of American Universities APLU
Document Downloaded: Monday July 27, 2015 Federal Research Policy - Recommendations to the NRC, January 2011 Author: David Kennedy Published Date: 06/23/2011 AAU Association of American Universities APLU
UNIFORM GUIDANCE UPDATE
 UNIFORM GUIDANCE UPDATE CINDY KIEL Executive Associate Vice Chancellor Office of Research Michael Allred Associate Vice Chancellor for Finance/Controller What is the Uniform Guidance? Uniform Administrative
UNIFORM GUIDANCE UPDATE CINDY KIEL Executive Associate Vice Chancellor Office of Research Michael Allred Associate Vice Chancellor for Finance/Controller What is the Uniform Guidance? Uniform Administrative
FAQ S FOR UNIFORM GUIDANCE
 FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
PART 3 COMPLIANCE REQUIREMENTS
 PART 3 COMPLIANCE REQUIREMENTS INTRODUCTION Overview The objectives of most compliance requirements for Federal programs administered by States, local governments, Indian tribes, institutions of higher
PART 3 COMPLIANCE REQUIREMENTS INTRODUCTION Overview The objectives of most compliance requirements for Federal programs administered by States, local governments, Indian tribes, institutions of higher
Federal Rules for Sponsored Programs. Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards 2 CFR 200
 Federal Rules for Sponsored Programs Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards 2 CFR 200 Uniform Guidance (UG) The Basics Presented by Dan Evon Director
Federal Rules for Sponsored Programs Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards 2 CFR 200 Uniform Guidance (UG) The Basics Presented by Dan Evon Director
Are You Ready for This? The New Uniform Grant Guidance 2 CFR 200
 Are You Ready for This? The New Uniform Grant Guidance 2 CFR 200 Increase in Federal Grants Activity The Catalog of Federal Domestic Assistance lists over 2,000 Federal grant programs $600B $200B $7B $24B
Are You Ready for This? The New Uniform Grant Guidance 2 CFR 200 Increase in Federal Grants Activity The Catalog of Federal Domestic Assistance lists over 2,000 Federal grant programs $600B $200B $7B $24B
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates!
 FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
Policy and Compliance: Working Together Like Hand in Glove
 Policy and Compliance: Working Together Like Hand in Glove Samuel Ashe, Director, Division of Grants Policy, OPERA, OER Diane W. Dean, Director, Division of Grants Compliance and Oversight, OPERA, OER
Policy and Compliance: Working Together Like Hand in Glove Samuel Ashe, Director, Division of Grants Policy, OPERA, OER Diane W. Dean, Director, Division of Grants Compliance and Oversight, OPERA, OER
RESEARCH POLICY MANUAL
 POLICY MANUAL RESEARCH Number 588 Subject: Research Data Covered Employees: USU Employees and Students Date of Origin: May 5, 2017 588.1 INTRODUCTION Research data are an essential component of any research
POLICY MANUAL RESEARCH Number 588 Subject: Research Data Covered Employees: USU Employees and Students Date of Origin: May 5, 2017 588.1 INTRODUCTION Research data are an essential component of any research
APPENDIX VII OTHER AUDIT ADVISORIES
 APPENDIX VII OTHER AUDIT ADVISORIES I. Effect of Changes to Generally Applicable Compliance Requirements in the 2015 Supplement In the 2015 Supplement, OMB has removed several of the compliance requirements
APPENDIX VII OTHER AUDIT ADVISORIES I. Effect of Changes to Generally Applicable Compliance Requirements in the 2015 Supplement In the 2015 Supplement, OMB has removed several of the compliance requirements
Open DFARS Cases as of 5/10/2018 2:29:59PM
 Open DFARS Cases as of 2:29:59PM 2018-D032 215 (R) Repeal of DFARS clause "Pricing Adjustments" 2018-D031 231 (R) Repeal of DFARS clause "Supplemental Cost Principles" 2018-D030 216 (R) Repeal of DFARS
Open DFARS Cases as of 2:29:59PM 2018-D032 215 (R) Repeal of DFARS clause "Pricing Adjustments" 2018-D031 231 (R) Repeal of DFARS clause "Supplemental Cost Principles" 2018-D030 216 (R) Repeal of DFARS
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE
 THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE Alexandra A. McKeown, Associate Dean Johns Hopkins University May 2, 2011 1 What is the FDP? The FDP is a cooperative effort among federal research
THE FEDERAL DEMONSTRATION PARTNERSHIP PHASE V UPDATE Alexandra A. McKeown, Associate Dean Johns Hopkins University May 2, 2011 1 What is the FDP? The FDP is a cooperative effort among federal research
NSF OIG Audit Update NORTHEAST CONFERENCE ON COLLEGE COST ACCOUNTING
 NSF OIG Audit Update 1 NORTHEAST CONFERENCE ON COLLEGE COST ACCOUNTING S e p t e m b e r 2 3, 2 0 1 4 Overview 2 Overview of NSF OIG Office of Audit Overview of Federal financial assistance in the U.S.
NSF OIG Audit Update 1 NORTHEAST CONFERENCE ON COLLEGE COST ACCOUNTING S e p t e m b e r 2 3, 2 0 1 4 Overview 2 Overview of NSF OIG Office of Audit Overview of Federal financial assistance in the U.S.
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards. AU SPAN Martha Taylor Larry Hankins
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards AU SPAN 6 23 2014 Martha Taylor Larry Hankins Grants Reform February 2011 President directed OMB to reduce
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards AU SPAN 6 23 2014 Martha Taylor Larry Hankins Grants Reform February 2011 President directed OMB to reduce
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards. AUSPAN Martha Taylor
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards AUSPAN 2 16 2015 Martha Taylor Grants Reform February 2011 President directed OMB to reduce unnecessary regulatory
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards AUSPAN 2 16 2015 Martha Taylor Grants Reform February 2011 President directed OMB to reduce unnecessary regulatory
The Metis Foundation Office of Grant and Contract Administration SUBRECIPIENT INFORMATION AND COMPLIANCE FORM
 The Metis Foundation Office of Grant and Contract Administration SUBRECIPIENT INFORMATION AND COMPLIANCE FORM All subrecipients must complete this form when submitting a proposal to the Metis Foundation
The Metis Foundation Office of Grant and Contract Administration SUBRECIPIENT INFORMATION AND COMPLIANCE FORM All subrecipients must complete this form when submitting a proposal to the Metis Foundation
FINANCE-315 7/1/2017 SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a proposal to UACES. It provides a checklist of documents and certifications required by sponsors, as well as an
SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a proposal to UACES. It provides a checklist of documents and certifications required by sponsors, as well as an
SUBRECIPIENT COMMITMENT FORM
 SECTION A SUBRECIPIENT INFORMATION SUBRECIPIENT COMMITMENT FORM Phone: Email: SUBRECIPIENT: CU PI: PROPOSAL TITLE: TOTAL FUNDING REQUESTED: ORIGINATING SPONSOR, IF A PASS THRU APPLICATION: PRIME SPONSOR:
SECTION A SUBRECIPIENT INFORMATION SUBRECIPIENT COMMITMENT FORM Phone: Email: SUBRECIPIENT: CU PI: PROPOSAL TITLE: TOTAL FUNDING REQUESTED: ORIGINATING SPONSOR, IF A PASS THRU APPLICATION: PRIME SPONSOR:
Uniform Guidance Update. Ruth Boardman, Associate Director Office of Grants and Contracts March 2015
 Uniform Guidance Update Ruth Boardman, Associate Director Office of Grants and Contracts March 2015 Objectives Latest Updates Internal Controls Procurement Status of Policies and Procedures Research Terms
Uniform Guidance Update Ruth Boardman, Associate Director Office of Grants and Contracts March 2015 Objectives Latest Updates Internal Controls Procurement Status of Policies and Procedures Research Terms
OMB Uniform Guidance 2 CFR Part 200
 OMB Uniform Guidance 2 CFR Part 200 What has changed - What hasn t What works What doesn t March 24, 2015 This session will focus on Brief outline of changes under the new OMB Uniform Guidance 2 CFR Part
OMB Uniform Guidance 2 CFR Part 200 What has changed - What hasn t What works What doesn t March 24, 2015 This session will focus on Brief outline of changes under the new OMB Uniform Guidance 2 CFR Part
Presenter. Changes to Federal Programs & Single Audits (A-87, A-21, A-122, A-102, A-110, A-89, A-133 & A-50) The New OMB Uniform Guidance
 Changes to Federal Programs & Single Audits (A-87, A-21, A-122, A-102, A-110, A-89, A-133 & A-50) The New OMB Uniform Guidance Presenter Richard Cunningham Quality Assurance & Technical Specialist Center
Changes to Federal Programs & Single Audits (A-87, A-21, A-122, A-102, A-110, A-89, A-133 & A-50) The New OMB Uniform Guidance Presenter Richard Cunningham Quality Assurance & Technical Specialist Center
Post Uniform Grant Guidance implementation from an auditor perspective
 Post Uniform Grant Guidance implementation from an auditor perspective Jeff Zeichner Patrick Smith Agenda Uniform Grant Guidance review Challenges with UGG implementation SEFA Internal controls and compliance
Post Uniform Grant Guidance implementation from an auditor perspective Jeff Zeichner Patrick Smith Agenda Uniform Grant Guidance review Challenges with UGG implementation SEFA Internal controls and compliance
Research Administrators Round Table
 Research Administrators Round Table Recap Set framework for going forward Review new Uniform Guidance driven WVU policy changes Sponsored Agreement Closeouts (WVU-FS-23) http://adminfin.wvu.edu/policies/finance/financial-services/wvu-fs-23-policy
Research Administrators Round Table Recap Set framework for going forward Review new Uniform Guidance driven WVU policy changes Sponsored Agreement Closeouts (WVU-FS-23) http://adminfin.wvu.edu/policies/finance/financial-services/wvu-fs-23-policy
Reducing Regulatory and Institutional Burden Associated with Animal Research. June 8, 2017
 Reducing Regulatory and Institutional Burden Associated with Animal Research June 8, 2017 Matt Bailey, President, National Association for Biomedical Research Molly Greene, IACUC Advisor, Michigan State
Reducing Regulatory and Institutional Burden Associated with Animal Research June 8, 2017 Matt Bailey, President, National Association for Biomedical Research Molly Greene, IACUC Advisor, Michigan State
MAXIMUS Higher Education Practice
 Federal Compliance Webinar Series 2 CFR 200 Uniform Guidance Kris Rhodes, Director, MAXIMUS Jason Guilbeault, Senior Consultant, MAXIMUS 1 MAXIMUS Higher Education Practice Headquartered in Northbrook,
Federal Compliance Webinar Series 2 CFR 200 Uniform Guidance Kris Rhodes, Director, MAXIMUS Jason Guilbeault, Senior Consultant, MAXIMUS 1 MAXIMUS Higher Education Practice Headquartered in Northbrook,
Financial Research Compliance. April 2013
 Financial Research Compliance April 2013 Overview I. What is a Sponsored Award? II. Sponsored Awards at WFU III. Compliance IV. Hot Topics V. Audits VI. WFU Updates VII. Questions VIII. Contacts What is
Financial Research Compliance April 2013 Overview I. What is a Sponsored Award? II. Sponsored Awards at WFU III. Compliance IV. Hot Topics V. Audits VI. WFU Updates VII. Questions VIII. Contacts What is
OVERVIEW OF OMB SUPERCIRCULAR... 1 OBJECTIVES OF THE REFORM... 1 OMB A-21 (COST PRINCIPLES FOR EDUCATIONAL INSTITUTIONS) TO 2 CFR 200 (UNIFORM ADMIN
 Table of Contents OVERVIEW OF OMB SUPERCIRCULAR... 1 OBJECTIVES OF THE REFORM... 1 OMB A-21 (COST PRINCIPLES FOR EDUCATIONAL INSTITUTIONS) TO 2 CFR 200 (UNIFORM ADMIN REQUIREMENTS, COST PRINCIPLES, AND
Table of Contents OVERVIEW OF OMB SUPERCIRCULAR... 1 OBJECTIVES OF THE REFORM... 1 OMB A-21 (COST PRINCIPLES FOR EDUCATIONAL INSTITUTIONS) TO 2 CFR 200 (UNIFORM ADMIN REQUIREMENTS, COST PRINCIPLES, AND
UNIFORM GUIDANCE OVERVIEW. Budget Officers Meeting January 28, 2015
 UNIFORM GUIDANCE OVERVIEW Budget Officers Meeting January 28, 2015 OMB Circulars Before and After Eight circulars condensed to one new comprehensive set of requirements for recipients of federal awards
UNIFORM GUIDANCE OVERVIEW Budget Officers Meeting January 28, 2015 OMB Circulars Before and After Eight circulars condensed to one new comprehensive set of requirements for recipients of federal awards
EPA s Implementation of OMB Uniform Grants Guidance FEDERAL DEMONSTRATION PARTNERSHIP JANUARY 12, 2015
 EPA s Implementation of OMB Uniform Grants Guidance FEDERAL DEMONSTRATION PARTNERSHIP JANUARY 12, 2015 Uniform Administrative Requirements Previously located at 40 CFR Parts 30 and 31 and are applicable
EPA s Implementation of OMB Uniform Grants Guidance FEDERAL DEMONSTRATION PARTNERSHIP JANUARY 12, 2015 Uniform Administrative Requirements Previously located at 40 CFR Parts 30 and 31 and are applicable
Federal Rules for Sponsored Programs. Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards 2 CFR 200
 Federal Rules for Sponsored Programs Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards 2 CFR 200 Uniform Guidance (UG) The Basics Presented by Dan Evon Director
Federal Rules for Sponsored Programs Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards 2 CFR 200 Uniform Guidance (UG) The Basics Presented by Dan Evon Director
Uniform Guidance. Overview and Implementation Plan. November 21, 2014
 Uniform Guidance Overview and Implementation Plan November 21, 2014 Uniform Guidance. Change is coming! College or Department name here 1 Overarching goal of the reform is to: streamline the rules and
Uniform Guidance Overview and Implementation Plan November 21, 2014 Uniform Guidance. Change is coming! College or Department name here 1 Overarching goal of the reform is to: streamline the rules and
Subrecipient Profile Questionnaire
 Subrecipient Profile Questionnaire How to use: The questionnaire is used to help determine a subrecipient organization s financial and management strength, which helps assess risk and dictates the monitoring
Subrecipient Profile Questionnaire How to use: The questionnaire is used to help determine a subrecipient organization s financial and management strength, which helps assess risk and dictates the monitoring
PMS Project Closeout. Dan Evon, Michigan State University Nate Martinez Wayman, Duke University
 PMS Project Closeout Dan Evon, Michigan State University Nate Martinez Wayman, Duke University 1 Session Outline GAO Audit Reports Closeout Parameters The FCTR Closeout History Sample Numbers Details Late
PMS Project Closeout Dan Evon, Michigan State University Nate Martinez Wayman, Duke University 1 Session Outline GAO Audit Reports Closeout Parameters The FCTR Closeout History Sample Numbers Details Late
OMB Uniform Guidance 2 CFR Part 200
 OMB Uniform Guidance 2 CFR Part 200 Costing and Administrative Requirements WebEx January 14, 2015 January 22, 2015 OMB Uniform Guidance - 2 CFR Part 200 Uniform Administrative Requirements, Cost Principles,
OMB Uniform Guidance 2 CFR Part 200 Costing and Administrative Requirements WebEx January 14, 2015 January 22, 2015 OMB Uniform Guidance - 2 CFR Part 200 Uniform Administrative Requirements, Cost Principles,
Please fill out the survey forms 2/9/2012. Upcoming ORA Training Opportunities. Agenda
 Agenda Welcome and Announcements Marcia Smith Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Data Management Tool Sharon Farb, Associate University Librarian,
Agenda Welcome and Announcements Marcia Smith Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Data Management Tool Sharon Farb, Associate University Librarian,
Review of Uniform Guidance (2CFR 200)
 Uniform Guidance Workgroup Review of Uniform Guidance (2CFR 200) June, 2014 Submitted to: Caroline Garcia, Associate Vice President, Research Duc Ma, Assistant Vice President, Financial Services Copy to:
Uniform Guidance Workgroup Review of Uniform Guidance (2CFR 200) June, 2014 Submitted to: Caroline Garcia, Associate Vice President, Research Duc Ma, Assistant Vice President, Financial Services Copy to:
Johns Hopkins University Research Administration (JHURA) Fall 2015
 Johns Hopkins University Research Administration (JHURA) Fall 2015 What s New Our Name Our Location Our Organization Our Responsibilities Uniform Guidance Current Initiatives What s New: Our Name Johns
Johns Hopkins University Research Administration (JHURA) Fall 2015 What s New Our Name Our Location Our Organization Our Responsibilities Uniform Guidance Current Initiatives What s New: Our Name Johns
PIRATE. Principal Investigator Research Administration Training and Education: TESTS & ANSWERS. Office of the Vice President for Research
 PIRATE Principal Investigator Research Administration Training and Education: TESTS & ANSWERS Office of the Vice President for Research Severna Park MD 21146 443 517 7074 WWW.FOSTERKNOWLEDGE.COM 1. Introduction
PIRATE Principal Investigator Research Administration Training and Education: TESTS & ANSWERS Office of the Vice President for Research Severna Park MD 21146 443 517 7074 WWW.FOSTERKNOWLEDGE.COM 1. Introduction
Cost Sharing. Policy Statement and Purpose
 Cost Sharing Policy Type: Administrative Responsible Office: Office of Sponsored Programs (proposal and award), Office of Research and Innovation, and Grants and Contracts Accounting (fiscal and accounting),
Cost Sharing Policy Type: Administrative Responsible Office: Office of Sponsored Programs (proposal and award), Office of Research and Innovation, and Grants and Contracts Accounting (fiscal and accounting),
Open DFARS Cases as of 12/22/2017 3:45:53PM
 Open DFARS Cases as of 3:45:53PM 2018-D004 252.225-7049, 52.225-7050 State Sponsor of Terrorism-- North Korea 2018-D003 252.222-7007 (R) Repeal of DFARS Provision "Representation Regarding Combating Trafficking
Open DFARS Cases as of 3:45:53PM 2018-D004 252.225-7049, 52.225-7050 State Sponsor of Terrorism-- North Korea 2018-D003 252.222-7007 (R) Repeal of DFARS Provision "Representation Regarding Combating Trafficking
Sponsored Programs and Research Compliance SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a subaward proposal to University of North Carolina Wilmington (UNCW). Please complete this form and send all required
SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a subaward proposal to University of North Carolina Wilmington (UNCW). Please complete this form and send all required
Outgoing Subaward Guide and F-A-Qs
 1. What is a subaward? Outgoing Subaward Guide and F-A-Qs 2. After an award is made to Rutgers, how is a subaward established? 3. What is the outgoing subaward process? 4. How can a subaward be changed/amended?
1. What is a subaward? Outgoing Subaward Guide and F-A-Qs 2. After an award is made to Rutgers, how is a subaward established? 3. What is the outgoing subaward process? 4. How can a subaward be changed/amended?
AAU Association of American Universities APLU Association of Public and Land-grant Universities
 AAU Association of American Universities APLU Association of Public and Land-grant Universities June 2, 2013 Mr. Daniel I. Werfel Controller, Office of Federal Financial Management Office of Management
AAU Association of American Universities APLU Association of Public and Land-grant Universities June 2, 2013 Mr. Daniel I. Werfel Controller, Office of Federal Financial Management Office of Management
Diane Dean, Director Kathy Hancock, Assistant Grants Compliance Officer Joel Snyderman, Assistant Grants Compliance Officer
 Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar San Diego, CA October 2015 Diane Dean,
Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar San Diego, CA October 2015 Diane Dean,
Subaward Policies and Procedures Manual
 JOHNS HOPKINS UNIVERISTY Krieger School of Arts and Sciences Business and Research Administration Subaward Policies and Procedures Manual 2016 Contents Introduction...3 Differentiating types of relationships...3
JOHNS HOPKINS UNIVERISTY Krieger School of Arts and Sciences Business and Research Administration Subaward Policies and Procedures Manual 2016 Contents Introduction...3 Differentiating types of relationships...3
The Lifecycle of a Sponsored Project
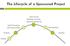 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where does the
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where does the
AURA Meeting. July 23, 2014
 AURA Meeting July 23, 2014 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored
AURA Meeting July 23, 2014 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored
Uniform Guidance Sponsored Projects Services
 Arizona s First University. Uniform Guidance Sponsored Projects Services 520-626-6000 sponsor@email.arizona.edu Agenda What is Uniform Guidance (UG)? Effective dates Structure of the Uniform Guidance Significant
Arizona s First University. Uniform Guidance Sponsored Projects Services 520-626-6000 sponsor@email.arizona.edu Agenda What is Uniform Guidance (UG)? Effective dates Structure of the Uniform Guidance Significant
Office of Commercialization and Innovation: Contracts & Industry Agreements (CIA)
 Office of Commercialization and Innovation: Contracts & Industry Agreements (CIA) Introduction to CIA and Contract Processing College of Engineering November 13, 2015 1 Overview of Today s Session 1. Introduction
Office of Commercialization and Innovation: Contracts & Industry Agreements (CIA) Introduction to CIA and Contract Processing College of Engineering November 13, 2015 1 Overview of Today s Session 1. Introduction
The Rollout of OMB A-81 and its Effect on UH
 The Rollout of OMB A-81 and its Effect on UH Cris Milligan, Interim Assistant Vice President for Research Administration Beverly Rymer, Executive Director, Office of Contracts and GrantsOffice of Contracts
The Rollout of OMB A-81 and its Effect on UH Cris Milligan, Interim Assistant Vice President for Research Administration Beverly Rymer, Executive Director, Office of Contracts and GrantsOffice of Contracts
Dept/ College. Dept/ College. Dept/ College
 Office of Research Administration Matrix Revision date -2.10.17 Submitting proposals, executing awards, conducting research, and administrating sponsored projects involves many different people and units
Office of Research Administration Matrix Revision date -2.10.17 Submitting proposals, executing awards, conducting research, and administrating sponsored projects involves many different people and units
(a) Article 1. Awards Covered by Research General T&Cs. REFERENCE: ARTICLE 2(a)
 U. S. ARMY RESEARCH OFFICE RESEARCH GENERAL TERMS AND CONDITIONS AGENCY SPECIFIC REQUIREMENTS 1 June 2009 Army Research Office Home Page http://www.aro.army.mil DoDGARS Part 32 http://www.dtic.mil/whs/directives/corres/html/321006r.htm
U. S. ARMY RESEARCH OFFICE RESEARCH GENERAL TERMS AND CONDITIONS AGENCY SPECIFIC REQUIREMENTS 1 June 2009 Army Research Office Home Page http://www.aro.army.mil DoDGARS Part 32 http://www.dtic.mil/whs/directives/corres/html/321006r.htm
Courtney Silverthorn, Host Agency Representative, Executive Board
 DATE: December 9, 2016 TO: Courtney Silverthorn, Host Agency Representative, Executive Board Courtney.silverthorn@nist.gov FROM: Anthony DeCrappeo, President, COGR Mary Sue Coleman, President, AAU Peter
DATE: December 9, 2016 TO: Courtney Silverthorn, Host Agency Representative, Executive Board Courtney.silverthorn@nist.gov FROM: Anthony DeCrappeo, President, COGR Mary Sue Coleman, President, AAU Peter
University of Pittsburgh SPONSORED PROJECT FINANCIAL GUIDELINE Subject: SUBRECIPIENT MONITORING
 I. Scope Subrecipient monitoring is the process by which the University selects, monitors, controls, and accounts for University subcontractors or subrecipients utilized on sponsored projects in accordance
I. Scope Subrecipient monitoring is the process by which the University selects, monitors, controls, and accounts for University subcontractors or subrecipients utilized on sponsored projects in accordance
Emory Research A to Z ERAZ
 Emory Research A to Z ERAZ July 17, 2014 Whitehead Auditorium G01 Whitehead Building Agenda Controlled Substances Sub Award Processing RAS Updates SAM Kiosk Federal Updates OSP/OGCA Training Updates Christine
Emory Research A to Z ERAZ July 17, 2014 Whitehead Auditorium G01 Whitehead Building Agenda Controlled Substances Sub Award Processing RAS Updates SAM Kiosk Federal Updates OSP/OGCA Training Updates Christine
NIH Update. Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH. FDP January 11, 2016
 NIH Update Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH FDP January 11, 2016 1 2 NIH FY 2016 Budget News NIH is currently funded under the Consolidated
NIH Update Michelle G. Bulls, Director Office of Policy for Extramural Research Administration, OER, NIH FDP January 11, 2016 1 2 NIH FY 2016 Budget News NIH is currently funded under the Consolidated
Expense Review Process and Internal Controls for Award Closeout. Semi-annual Networking Event February 2017
 ACCOUNTING SERVICES FOR RESEARCH AND SPONSORED PROGRAMS (ASRSP) Expense Review Process and Internal Controls for Award Closeout Semi-annual Networking Event February 2017 Kathy Mustea, Assistant Director
ACCOUNTING SERVICES FOR RESEARCH AND SPONSORED PROGRAMS (ASRSP) Expense Review Process and Internal Controls for Award Closeout Semi-annual Networking Event February 2017 Kathy Mustea, Assistant Director
45 CFR 75 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Health and Human Services Awards
 45 CFR 75 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Health and Human Services Awards Frances Hodge Public Health Analyst, Southern Services Branch Division of Metropolitan
45 CFR 75 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Health and Human Services Awards Frances Hodge Public Health Analyst, Southern Services Branch Division of Metropolitan
The OmniCircular - 2 CFR 200
 The OmniCircular - 2 CFR 200 Mary Karen Wills 202-480-2773 mkwills@brg-expert.com Tina Reynolds 703.760.7701 Treynolds@mofo.com September 16, 2014 OMB Final Rule and Applicability Office of Management
The OmniCircular - 2 CFR 200 Mary Karen Wills 202-480-2773 mkwills@brg-expert.com Tina Reynolds 703.760.7701 Treynolds@mofo.com September 16, 2014 OMB Final Rule and Applicability Office of Management
2 CFR Chapter II, Part 200 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards For Auditees
 2 CFR Chapter II, Part 200 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards For Auditees SHERYL L. STEPHENS BURKE, CPA, MST Nashua Office 102 Perimeter Road
2 CFR Chapter II, Part 200 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards For Auditees SHERYL L. STEPHENS BURKE, CPA, MST Nashua Office 102 Perimeter Road
Subrecipient Monitoring Under 2 CFR 200. Kris Rhodes, MS Director, Higher Education Practice
 Subrecipient Monitoring Under 2 CFR 200 Kris Rhodes, MS Director, Higher Education Practice krisrhodes@maximus.com 1 MAXIMUS Higher Education Practice Headquartered in Northbrook, IL Satellite Offices
Subrecipient Monitoring Under 2 CFR 200 Kris Rhodes, MS Director, Higher Education Practice krisrhodes@maximus.com 1 MAXIMUS Higher Education Practice Headquartered in Northbrook, IL Satellite Offices
Indirect Cost Rates Frequently Asked Questions
 Indirect Cost Rates Frequently Asked Questions FAQs ON PROPOSALS 1. What indirect cost rate should be used on a proposal? It is university policy to recover the full indirect cost rate allowed on sponsored
Indirect Cost Rates Frequently Asked Questions FAQs ON PROPOSALS 1. What indirect cost rate should be used on a proposal? It is university policy to recover the full indirect cost rate allowed on sponsored
Open FAR Cases as of 2/9/ :56:25AM
 Open FAR Cases as of 11:56:25AM 2018-010 (S) Use of Products and Services of Kaspersky Lab Implements section 1634 of the NDAA for FY 2018. Section 1634 prohibits the use of products and services developed
Open FAR Cases as of 11:56:25AM 2018-010 (S) Use of Products and Services of Kaspersky Lab Implements section 1634 of the NDAA for FY 2018. Section 1634 prohibits the use of products and services developed
Administrative Burden of Research Compliance
 Administrative Burden of Research Compliance Measuring and Minimizing David L. Wynes, Ph.D. Vice President for Research Administration Emory University 1 FDP Faculty Burden Survey (X2) PIs estimated that
Administrative Burden of Research Compliance Measuring and Minimizing David L. Wynes, Ph.D. Vice President for Research Administration Emory University 1 FDP Faculty Burden Survey (X2) PIs estimated that
Uniform Guidance - Lessons Learned And To Be Learned
 DAY MAY 23, 2017 3:35-4:50PM Uniform Guidance - Lessons Learned And To Be Learned MODERATOR Jerry E. Durham Assistant Director for Research and Compliance, Tennessee Comptroller of the Treasury SPEAKERS
DAY MAY 23, 2017 3:35-4:50PM Uniform Guidance - Lessons Learned And To Be Learned MODERATOR Jerry E. Durham Assistant Director for Research and Compliance, Tennessee Comptroller of the Treasury SPEAKERS
Presentation 2/26/2013. Director, TitleOPERA
 Presentation Title Michelle Presented G. Bulls By: Name Director, TitleOPERA Office OER AASCU Washington Conference February 21, 2013 2 NIH is operating under a continuing resolution. See NOT-OD- 13-018
Presentation Title Michelle Presented G. Bulls By: Name Director, TitleOPERA Office OER AASCU Washington Conference February 21, 2013 2 NIH is operating under a continuing resolution. See NOT-OD- 13-018
DARPA. Doing Business with
 Doing Business with DARPA The Defense Advanced Research Projects Agency (DARPA) is the central research and development agency within the Department of Defense. DARPA s mission is to maintain the technological
Doing Business with DARPA The Defense Advanced Research Projects Agency (DARPA) is the central research and development agency within the Department of Defense. DARPA s mission is to maintain the technological
Policy on Cost Allocation, Cost Recovery, and Cost Sharing
 President Page 1 of 11 PURPOSE: Provide guidance and structure when allocating and documenting costs (direct and indirect) for extramurally funded awards. Serves to provide direction for budgeting, allocating
President Page 1 of 11 PURPOSE: Provide guidance and structure when allocating and documenting costs (direct and indirect) for extramurally funded awards. Serves to provide direction for budgeting, allocating
SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients must complete this form when submitting a proposal to KSU, along with documents and certifications required by sponsors. This form must be endorsed by the
SUBRECIPIENT COMMITMENT FORM All subrecipients must complete this form when submitting a proposal to KSU, along with documents and certifications required by sponsors. This form must be endorsed by the
University of North Carolina Finance Improvement & Transformation Contracts and Grants Standards. January 2015 Version 8
 University of North Carolina Finance Improvement & Transformation Contracts and Grants Standards January 2015 Version 8 Table of Contents Introduction.......3 Account Setup.....10 Time and Effort Reporting......14
University of North Carolina Finance Improvement & Transformation Contracts and Grants Standards January 2015 Version 8 Table of Contents Introduction.......3 Account Setup.....10 Time and Effort Reporting......14
Ryan White HIV/AIDS Program Fiscal Health Series. Systems to Sustainability TM. Federal Grants
 Ryan White HIV/AIDS Program Fiscal Health Series Systems to Sustainability TM Federal Grants INTRODUCTION Sound fiscal management is critical for organizations to improve access to health care, advance
Ryan White HIV/AIDS Program Fiscal Health Series Systems to Sustainability TM Federal Grants INTRODUCTION Sound fiscal management is critical for organizations to improve access to health care, advance
UC San Diego Policy & Procedure Manual
 UC San Diego Policy & Procedure Manual Search A Z Index Numerical Index Classification Guide What s New CONTRACTS AND GRANTS (RESEARCH) Section: 150-45 Effective: 08/01/2001 Supersedes: 05/26/1999 Review
UC San Diego Policy & Procedure Manual Search A Z Index Numerical Index Classification Guide What s New CONTRACTS AND GRANTS (RESEARCH) Section: 150-45 Effective: 08/01/2001 Supersedes: 05/26/1999 Review
Award Transfer Guidelines
 Award Transfer Guidelines The process of transferring awards from one institution to another is not a simple task and requires negotiation between the sponsoring agency, the old institution and the new
Award Transfer Guidelines The process of transferring awards from one institution to another is not a simple task and requires negotiation between the sponsoring agency, the old institution and the new
UNIFORM ADMINISTRATIVE REQUIREMENTS, COST PRINCIPLES, AND AUDIT REQUIREMENTS FOR FEDERAL AWARDS. AOA Conference Sacramento, CA January 12, 2014
 UNIFORM ADMINISTRATIVE REQUIREMENTS, COST PRINCIPLES, AND AUDIT REQUIREMENTS FOR FEDERAL AWARDS AOA Conference Sacramento, CA January 12, 2014 Agenda 1. Introduction 2. History 3. Learning Objectives 4.
UNIFORM ADMINISTRATIVE REQUIREMENTS, COST PRINCIPLES, AND AUDIT REQUIREMENTS FOR FEDERAL AWARDS AOA Conference Sacramento, CA January 12, 2014 Agenda 1. Introduction 2. History 3. Learning Objectives 4.
The Other Transaction Authority Basic Legal Principles*
 GENERAL DYNAMICS PROPRIETARY This document in printed form may not be the latest issue. Verify latest issue online. The Other Transaction Authority Basic Legal Principles* Presented by: Benjamin McMartin,
GENERAL DYNAMICS PROPRIETARY This document in printed form may not be the latest issue. Verify latest issue online. The Other Transaction Authority Basic Legal Principles* Presented by: Benjamin McMartin,
