RandomizEd Trial of ENtERal Glutamine to. minimize Thermal Injury. CRS & REDCap Manual. Intended Audience: Research Coordinators & Pharmacists
|
|
|
- Whitney Perkins
- 5 years ago
- Views:
Transcription
1 RandomizEd Trial of ENtERal Glutamine to minimize Thermal Injury CRS & REDCap Manual Intended Audience: Research Coordinators & Pharmacists This study is registered at Clinicaltrials.gov. Identification number NCT Sponsors: Dominique Garrel, MD, University of Montreal Paul Wischmeyer, MD, University of Colorado Funded By: The Canadian Institutes of Health Research The American Burn Association October 20th 2011_NP 1
2 Table of Contents ELECTRONIC DATA CAPTURE SYSTEMS...3 GRANTING CRS & REDCAP ACCESS...3 SCREENING & RANDOMIZATION... 4 SCREENING & ELIGIBILITY... 4 ACCESSING & ENTERING A PATIENT IN THE CRS... 5 INCLUSION CRITERIA...7 EXCLUSION CRITERIA...9 PRE-RANDOMIZATION...10 RANDOMIZATION REDCAP DATA ENTRY NAVIGATING REDCAP My Databases Data Entry Field Event Grid Field Form Links Antibiotics & Microbiology Fields Form Status and Saving DATA CONVENTIONS IN REDCAP EDIT DATA ENTERED ON THE WEB INFECTION ADJUDICATION INFECTION ADJUDICATION DATA ENTRY Infection Adjudication Table...23 STAGES OF DATA ENTRY...28 INVESTIGATOR CONFIRMATION October 20th 2011_NP 2
3 Electronic Data Capture Systems Each site will need to access two different electronic data capture systems for RE-ENERGIZE: 1 The Central Randomization System (CRS) is a web-based system that will be used to screen and randomize eligible patients into the RE-ENERGIZE Study. The CRS may be accessed directly at: or via: 2 REDCap is a web-based system that will be used as the RE-ENERGIZE Study ecrf. REDCap may be accessed directly at: or via: Granting CRS & REDCap Access Access to both CRS and REDCAP will be granted to the Research Coordinator/delegate upon documentation of proper training of study procedures and receipt of Ethics Approval documentation and other essential documents. Research Coordinators that are granted access to the CRS and REDCAP must appear on the Delegation of Authority Log as described in the Implementation Manual. October 20th 2011_NP 3
4 Screening & Randomization Screening & Eligibility The Research Coordinators are expected to screen patients admitted to their ICU daily to see if they meet the inclusion criteria or exclusion criteria as listed below. All screening data should be entered into the Central Randomization System (CRS). For eligible patients, the screening data must be entered onto the CRS in a timely manner in order to randomize the patient and start the study intervention as soon as possible. Since patient eligibility and suitability must be determined by the Site Investigator/delegate, sites are encouraged to use the Inclusion/Exclusion criteria mock ecrfs to document screening and confirmation of eligibility by a physician. Types of Patients to be entered into the CRS The CRS serves as a screening log and is vital in identifying the screening activity at each site. Hence ALL patients that meet the following criteria MUST be entered into the CRS on a regular basis: All patients meeting inclusion criteria, these would be either of the following: o patients that meet an exclusion criteria or o o patients that do not meet any exclusion criteria and consent is obtained (Randomized patients) or patients do not meet any exclusion criteria and consent is not obtained (Eligible but not randomized patients) The table below provides several examples of the types of patients. Inclusion Criteria Present Exclusion Criteria Present Informed Consent Obtained Do not approach for consent as inclusion criteria not met Do not approach for consent as exclusion criteria met Enter into CRS Comments Ineligible patient Randomized patient Eligible but not randomized patient For each patient entered into the CRS, the system will issue a screening number. The screening numbers are assigned sequentially in an 8-digit format: 2 indicates 0 notes the patient patient is being is being screened screened but not but randomized not randomized Site # patient # October 20th 2011_NP 4
5 If the patient is subsequently randomized, they will also be issued an enrollment number. The enrollment numbers are assigned sequentially in an 8-digit format: 3 indicates the patient 0 notes patient is being has been randomized screened but not randomized Site # patient # Accessing & Entering a Patient in the CRS URL: To access the CRS enter your assigned User name and Password. Click LOGIN. Once you have logged in successfully, you will be brought to the Home screen. The Home screen lists all clinical trials for which your site is participating in with CERU that are also using the CRS. Click on RE-ENERGIZE. After selecting the RE-ENERGIZE study from the Home page, you will be brought to the Site Status Page. This page is a listing of all patients screened and randomized to the study. October 20th 2011_NP 5
6 To enter data for a new patient, select the Add patient button on the bottom left of the screen. Each patient entered in the CRS will also have a status associated with it. There are 4 status levels: In progress: inclusion data has been entered. Not Eligible: This patient was excluded. Not Randomized: This patient was eligible but consent was not obtained. Randomized: The patient was eligible, consent was obtained and the patient was enrolled into the study. You will be brought to the Inclusion Criteria form. Complete the fields in the form as appropriate. Dates are to be entered in the DD-MMM-YYYY format. All times should be recorded using the 24- hour (military) clock. All times must include a : to be saved. For example 1200 must be entered as 12:00. Click SAVE. Note: Enter all patients into the Central Randomization System who meet the Inclusion Criteria. See Types of Patients to be Entered section for further detail. October 20th 2011_NP 6
7 Inclusion Criteria Patients must meet all three of the criteria to be included in the study. # Inclusion Criteria 1 Total Burn Surface Area (TBSA) 20% in patients age years Total Burn Surface Area (TBSA) 10% in patients age years This is an assessment that is to be made by the attending surgeon/physician based on his/her clinical judgment. The Lund Browder chart (Appendix 1 mock ecrfs may be used to help make this assessment, if needed). This information must be obtained from the attending surgeon/delegate before randomization can occur. 2 Deep 2 nd and/or deep 3 rd degree burns requiring grafting The presence of deep 2nd degree or deep 3rd degree burns requiring grafting is an assessment that must also be made by the attending surgeon/physician. This information must be obtained from the attending surgeon/delegate before randomization can occur. 3 Age + TBSA = The total of the patient s age and TBSA must be between the range of Consent must be obtained within 48 hrs of admission to the ICU (burn unit) To be eligible, admission to the unit must occur within 48 hrs of burn injury. In the event that the patient received standardized burn care and resuscitation prior to admission to ICU, this may be extended to 96 hrs. In this case, consent must be obtained within 24 hrs of admission to burn ICU October 20th 2011_NP 7
8 Complete the exclusion criteria fields as appropriate. Click on the drop-down boxes to select a Yes or No response for each criterion. If you click Yes to any one criteria, this patient is not eligible for the study. Click SAVE. No futher data entry required. If you click No to all criteria, this patient is eligible. Click SAVE, then proceed to the Pre- Randomization Form. Remember to: Only enter patients who meet ALL the inclusion criteria. Enter the date & time of screening You may want to use the mock ecrfs/worksheets to document screening & eligibility To save time, instead of clicking the drop down box, you may press TAB Y for Yes and TAB N for No October 20th 2011_NP 8
9 Exclusion Criteria Choose all exclusion criteria that apply. If a patient meets any of the exclusion criteria, they are not eligible to participate in the study. # Exclusion Criteria 1 > 72 hrs from admission to ICU to time of consent 2 Patients > 80 years or < 18 years of age (age of maturity for an eligible patient to obtain consent is 18 years in Canada and the US) 3 Liver cirrhosis -Child s class C liver disease 4 Pregnancy (urine/blood tests for pregnancy will be done on all women of childbearing age by each site as part of standard of ICU practice) 5 Absolute contra-indication for EN: intestinal occlusion or perforation, abdominal injury. (Intestinal occlusion or perforation, abdominal injury. Being NPO is not considered a contraindication for Enteral Nutrition). 6 Patients admitted > 48h post burn (for patients that receive standardized burn care and resuscitation prior to admission to ICU, this exclusion criteria may be extended to patients admitted more than 96 hrs post burn: in this case consent must be obtained within 24 hours of admission to burn ICU) 7 Patients with injuries from high voltage electrical shock Patients who are moribund 8 (this may be defined as not expected to be in the ICU for more than 48 hours due to imminent death) PATIENTS WITH EXTREME BODY SIZES: BMI < 18 OR > 50 KG/M 2 10 Enrollment in another industry sponsored ICU intervention study (co-enrollment in academic studies will be considered on a case by case basis) 11 Received glutamine supplement for > 24 hours prior to randomization. 12 Known allergy to maltodextrin, corn starch, corn, or corn products. To minimize any potential contamination, patients that have received glutamine for >24 hrs before randomization, should NOT be included. For such patients, please enter them on to the Central Randomization System and add the reason for no consent as received glutamine for > 24 hrs Patient eligibility must be confirmed by the Site Investigator/MD delegate If a patient is found to meet an exclusion criterion after the patient is randomized into the study, please contact the Project Leader as soon as you become aware for direction on how to proceed. October 20th 2011_NP 9
10 Pre-Randomization Pre-Randomization refers to the period of time between the determination of an eligible patient and randomization of a patient. The patient/next of kin must be approached for consent before you complete this form. If consent is NOT obtained, complete the following form as shown below. Choose one of the following reasons for NOT obtaining consent: Reason No next of kin or substitute decision maker Refused Consent Missed the patient MD refusal Language Barriers Pharmacy not available Not approached for consent Family dynamics Workload Issues Other, please specify Description The SDM or legally acceptable representative (LAR) was not available for consent discussion within the required time frame. The SDM or LAR refused participation. It is important to document the reason for the refusal to consent. The patient was not identified by the site coordinator in time to approach for consent. E.g. the patient came in over the weekend. The MD feels that the patient is not suitable for the study The SDM was not approached because of language barriers. A certified translator was not present. The SDM was not approached for consent because pharmacy is not available to prepare the investigational product. The SDM was not approached due to emotional stress or complicated family dynamics. There was inadequate Research staff present to follow the patient Any other reasons that are not captured above If consent IS obtained, complete the following form as shown below. October 20th 2011_NP 10
11 Dates are to be entered in the DD-MMM-YYYY format. All times should be recorded using the 24- hour (military) clock. All times must include a colon (:) to be saved. For example 1200 must be entered as 12:00. Once you click on the Randomize button, the patient will be randomized to the RE-ENERGIZE Study. Randomization Randomization must occur soon after consent so that the intervention can start as soon as possible (within 2 hrs from randomization) You may print a copy of the Randomization Form and file in the Patient Folder/Study files. October 20th 2011_NP 11
12 The Patient Status Page indicates that data entry for this patient is complete. Note: All patient data collected following randomization can be entered on to the ecrf (REDCAP) Click on the Site Status button to view all the patients screened and entered on the CRS. You will note each patient entered into the CRS is issued a screening number. Those patients that are eligible and randomized are issued an enrollment number. To view a patient, click their enrolment number or their screening number, October 20th 2011_NP 12
13 You will then be brought to the Patient Status screen. It shows you each data entry form for the patient as well as the status of the form. You can open a specific form by clicking on it. Each form has a status assigned: Status Symbol Description Completed All data has been completed and saved. Not Completed Data has not yet been entered on the form. Locked The patient has been randomized and the data is no longer able to be edited by the site user. Note: All subsequent data collection must be entered on to the ecrf (REDCAP) as described in the following pages. October 20th 2011_NP 13
14 REDCap Data Entry The REDCap (Research Electronic Data Capture) is a web-based system used for the RE-ENERGIZE Study. REDCap can be accessed at the REDCap login link All authorized study personnel must log onto the web site using their own username and password prior to data entry. Your user password can be changed at any time by clicking My Profile after logging into REDCap. October 20th 2011_NP 14
15 Navigating REDCap My Databases After you log into REDCap, you will be brought to the Home screen. Select the My Databases tab to see a list of the CERU studies you have access to. Select RE-ENERGIZE Data Entry Field The left side of the screen is the main navigation panel where you will see Data Entry. Select Data Entry to choose from a list of patients that are randomized and ready for data entry. REENERGIZE October 20th 2011_NP 15
16 REENERGIZE Click on the drop-down menu to select a randomized patient. Event Grid Field After you have selected a patient, you will be brought to the Event Grid. The Event Grid gives the user a snap shot of the data entry forms for the patient. The type of data entry form is listed in the far left column of the table. The study day is listed on the top row of the table. Each dot on the table represents an individual data entry form. Each individual form can be accessed by clicking on the dot. As you can see below, the circled dot is the Daily Monitoring form for study day 3. October 20th 2011_NP 16
17 Slide the navigation scroll bar at the bottom of the table to reveal the right side of the Event Grid. Form Links You can navigate between forms on the same study day using the form links on the left side navigation menu. REENERGIZE October 20th 2011_NP 17
18 Antibiotics & Microbiology Fields You will note that the antibiotic and microbiology data entry forms have drop down boxes for each day and the events are listed as 1, 2, 3, 4, 5 & 6 (maximum of 6 entries per day). See arrow below. To help identify the status of the antibiotic and microbiology entries, you will find a Form Status Legend at the bottom of the grid (circled). You MUST enter the antibiotic and micro data on the same study day as the antibiotic was started or the day the sample was taken. Form Status and Saving At the end of each form, you will be asked to specify the form status. This legend is to be used to assist you in remembering what data is either incomplete, unverified or complete. The status is indicated on the Event Grid Field using the following convention: Incomplete Unverified Complete Locked No data has been entered on a form. Blanks forms will automatically be set to incomplete. Data entry is partially completed on a form. The RC wants to double check data already entered on a form. Partially completed forms will automatically be set to unverified. Data entry is complete on a form. Further changes to the data are not anticipated. Only forms manually set to complete will have this status. Locked status will appear on all forms after all finalization checks are completed. Data on locked forms can not be changed. October 20th 2011_NP 18
19 There may be up to 4 options at the end of each form to save your progress. The following example is for: Daily Monitoring - Study Day 1 This option will save your progress and bring you to the next form on the same study day. Note: In the case of the Antibiotic and Microbiology forms, this option will bring you to the next corresponding Antibiotic/Microbiology form. For example, if you are working on the third Antibiotic form on Day four, this option will save and bring you to the fourth Antibiotic form on Day four. This option will save your progress and bring you to the same form on the next study day. This option will save your progress and return you to the Event Grid. This option will save your progress and allow you to continue working on that form. This option will allow you to clear the entire form in case the entire form was completed in error. This option will take you to the Event Grid screen. All newly entered data will be lost. Only the last saved version will remain NOTE: Always remember to Save before you navigate away from a form. Navigating from a form without saving will result is loss of data. October 20th 2011_NP 19
20 Data Conventions in REDCap Dates should be entered using the YYYY - MM - DD format i.e A date picker calendar is available to enter dates. Single click on the icon and choose the appropriate month and year from the drop down boxes. Then click the appropriate day. Enter all times using the HH:MM 24-hour period format i.e. 22:37. The semicolon must be entered. Use leading zeros where applicable i.e. 01:28. Midnight should be entered as 00:00 To access individual forms single click the corresponding dot on the event grid. To enter data directly into each field, single click on the left side of the mouse pointer and type information. Do NOT press enter after entering data into a field. This will cause the form to automatically save and bring you to a new screen that will allow you to return to the Event Grid. There should be NO blanks. If data is NOT available use the Not Done/Not Available checkbox options. This includes: Data that is unavailable because the measure wasn t taken or the test was not done. Example: T-Bilirubin was not done on a particular study day. Data that is not known. This assumes every effort has been made to find the data but it is missing from source documents. Example: A particular data point was NOT entered in the medical chart. Or an ICU flow sheet has gone missing. REDCap has an option for user to see the data entry history for each data field. By clicking on the, a window will pop up listing the data entry history for the data field. Edit data entered on the web To edit previously saved information, access the appropriate REDCap form, change the appropriate field(s) and save the form. To ensure Good Clinical Practice is maintained, all changes will be tracked and logged by the computer program. Once a patient has been randomized they cannot be deleted. Please contact the Project Leader for more details. Please keep ALL worksheets/documents that you use as these will be referred to at the time of source verification. October 20th 2011_NP 20
21 Infection Adjudication In order to determine the incidence of newly acquired infections in patients enrolled to the RE-ENERGIZE study, an assessment needs to be made by the Site investigator/md delegate as to whether a newly acquired infection exists and this requires adjudication. A suspicion of infection is determined by the antibiotics received and the data on positive cultures. All antibiotics and cultures that lead to a suspicion of infection will be recorded on the appropriate electronic case report form. Once a clinical suspicion of an infection has been identified, the Site Investigator/MD delegate MUST adjudicate the data to determine the following: Is there an infection or not Degree of certainty of the infection Category of Infection Refer to the algorithm on next page for adjudicating a clinical suspicion of infection. Although the site investigator is responsible for the adjudication, the research coordinator is responsible for facilitating this process. Refer to the following documents for more details 1. Antibiotic, antifungal & antiviral mock ecrf 2. Microbiology mock ecrf 3. Mock ecrf Appendix 7 Categories of Infection 4. Mock ecrf Appendix 8 Definition of No Newly Acquired Infection Infection adjudication MUST be performed by the Site Investigator, or MD delegate. th October _NP 21
22 Was a new antibiotic started > 72 hours from ICU admission? OR Was there a positive culture taken > 72 hours from ICU admission? YES NO NO adjudication required NO adjudication required YES To either question Antibiotic Form Was the antibiotic given for prophylaxis? OR Was this antibiotic a substitute for an antibiotic previously ordered for an infection that occurred within the first 72 hours of ICU admission? Microbiology Form Is this organism a manifestation of an infection that occurred within the first 72 hours of ICU admission? YES This culture represents a relapse/recurrent infection or a persistent infection. NO adjudication required NO This defines a Clinical Suspicion of an infection. This clinical suspicion requires adjudication by the Site Investigator/MD delegate. ADJUDICATION DETERMINATION BY SITE INVESTIGATOR/MD DELEGATE NO Infection YES New infection YES Suspicion of infection that has been previously adjudicated Indicate the Degree of Certainty probable NO or possible NO Refer to mock ecrf Appendix 8 Choose the appropriate Category of Infection AND Degree of Certainty definite yes probable yes or possible yes Refer to Mock ecrf Appendix 7 Indicate which previously adjudicated episode of infection this current suspicion is related to (E.g. suspicion #1, #2, #3, etc). Refers to antibiotic, antifungal or antiviral th October _NP 22
23 Infection Adjudication Data Entry Based on the microbiology and antibiotic data entered, REDCAP will automatically trigger suspicions of newly acquired infections. This is done by generating a series of tables that will guide and assist the Site Investigator/Research Coordinator through the various steps of Infection Adjudication. These steps are described below. You will see the Infection Adjudication tab appear in the left hand side under Resources. Click on Infection Adjudication and you will see a new window as described below. The Infection Adjudication link will open a new window listing all your patients. Patients needing an adjudication are in Stage 2 and 4b. Select Infection Adjudication 4b red and 4b blue indicate that the initial central adjudication queries (4b red) and subsequent queries (4b blue) need to be addressed by site adjudicator. You must complete the infection adjudication for these patients to proceed further. Select the appropriate patient from the list. You will note this table lists the patient ID, # suspicions of newly acquired infection, and the patient s data entry status. th October _NP 23
24 An Infection Adjudication Table is automatically generated that lists all the relevant data that has been entered for the patient. The top of the table identifies the patient, number of infections that need to be adjudicated based on the number of clinical suspicions and baseline and outcome data. The next section is the entire table with the clinical data the Site Investigator/MD delegate will use to adjudicate the infection. This data includes the following: Date, Temperature, Worst PF ratio, WBC highest and lowest, Pressors, Ventilation Status, Microbiology data and Antibiotic data. Refer to the column on the right called Newly Acquired Infection for all the infections that need to be adjudicated th October _NP 24
25 The Site Investigator, or MD delegate, is to pick the most appropriate response by referring to the variables in the table in addition to reviewing the patient s medical chart and condition at the time of infection. Three response options available for each instance of a clinical suspicion of infection are: This is a newly acquired infection This is NOT a newly acquired infection This is a previously adjudicated suspicion of infection As seen in REDCap This is a newly acquired infection Pick this option if the clinical suspicion of infection is considered to be a newly acquired infection. The Site Investigator, or MD delegate, will assign a Category of Infection (Appendix 7 of the Mock ecrf), then the degree of certainty of the infection using the definition from within the assigned Category of Infection. Example: On study day 9 the patient is febrile, has an elevated WBC, CXR reveals a new infiltrate. An endotracheal aspirate specimen was sent for culture, S. aureus is identified. The infection should be adjudicated as follows: This is a newly acquired infection Category of Infection = 11- ICU Pneumonia Probable YES th October _NP 25
26 This is NOT a newly acquired infection Pick this option if the clinical suspicion of infection is not considered to be an infection. Refer to (Appendix 10 of the Mock ecrf), for associated definitions. Example: On study day 17 a blood culture indicates the presence of Staph Epidermis. There are no other clinical indicators of infection (i.e. SIRS). A repeat culture is negative. The initial positive culture is thought to be a contaminant. The infection should be adjudicated as follows: This is NOT a newly acquired infection Probable-NO This is a previously adjudicated infection Pick this option if this clinical suspicion of infection is associated with an infection already adjudicated.you must also indicate the day and the suspicion # of the associated previously adjudicated infection. Although infections that occur within the first 72 hrs of ICU admission are not to be considered newly acquired infections and hence are not be adjudicated per se, there is an option to capture this information for the Central Adjudication Process. If the suspicion of infection was due to an infection that occurred in the first 72 hours after ICU admission, select Baseline Infection. Example: On study day the patient is febrile, has an elevated WBC and CXR reveals a new infiltrate. An endotracheal aspirate specimen was sent for culture, S. aureus is identified. On study day 10 an antibiotic was initiated to treat the S. aureus. The clinical suspicion triggered on study day 10 with the initiation of an antibiotic is related to a previously adjudicated infection. th October _NP 26
27 The adjudication response for study day 19 is: This is a newly acquired infection Category of Infection = 11 - ICU Pneumonia Probable-Yes The adjudication response for study day 20 is: This is a previously adjudicated infection Infection # 1 (same response as study day 19). Before the locking checks are completed (Stage 1), the site may click on the SAVE button to save their adjudication data. After the locking checks are completed (Stage 2) and all incidents of clinical suspicion of infection have been adjudicated for a patient, click on the COMPLETED button. This will save the data you have entered No clinical suspicions of infection After the locking checks are completed (Stage 2), complete the adjudication form. If there are no clinical suspicions of infection in the right hand column, the adjudication form must still be completed by clicking on the COMPLETED button Following the completion of the Infection Adjudication by the Site Investigator, a Central adjudication process will occur in which an external reviewer(s) will review the adjudications done by the Site Investigator. Any queries/discrepancies will need to be addressed by the Site Investigator and the details of this process will be communicated by the Project Leader at CERU when site infection adjudications are completed. th October _NP 27
28 Stages of Data Entry To help you determine the status of the patient data, we have designated different stages of data completion. Each stage marks the completion of a specific set of data. The diagram below summarizes the site responsibilities at these various stages. Stage 1 Enter data on REDCap Address locking checks Lock patient Stage 2 Complete site adjudication If no adjudication, press complete If no adjudications needed Stage 3 Complete 3 & 6 month follow-up If adjudications needed Stage 3 Complete 3 & 6 month follow-up Stage 4a Central infection adjudication Stage 4b Site response to central infection adjudication Stage 5 Patient chart closed Investigator Confirmation Stage 5 Patient chart closed Investigator Confirmation th October _NP 28
29 Once all data has been completed up to and including hosptial overview (6 month follow-up excepted), the user can proceed to Stage 2. The Complete stage 1 button is found at the bottom of the Grid. Once the Complete Stage 1 button has been selected, REDCap will run front-end logic and edit checks. If any data discrepancies are identified the user will see them listed on a new screen. REENERGIZE Once all errors have been addressed by the site and patient is locked, the patient will be in Stage 2 Each error identified must be addressed before you can Lock the patient. There is an individual link to the relevant form to address each error noted. REENERGIZE Once a patient is locked the site will NOT be able to modify the data. Contact the Project Leader if modifications to the data are required. th October _NP 29
30 Investigator Confirmation After the completion of all data entry (i.e. Status of Stage 5 ), the Investigators Confirmation form must be completed and forwarded to the Project Leader. To access the Investigator Confirmation form, select the link from the Resources section on the left side menu. The form will automatically be populated with the site name and patient enrollment number. Print this form and have the site Investigator sign and date. By signing, the site Investigator is attesting to the following: The data collection and entry was conducted under his/her supervision and in accordance with study procedures. The data and statement, including newly acquired hospital infection adjudication are complete and accurate to the best of their knowledge. Forward a scan or fax ( ) of the signed Investigator Conformation form. File the original in your study files. October 20 th 2011_NP 30
A Randomized Trial of Supplemental Parenteral Nutrition in. Under and Over Weight Critically Ill Patients: The TOP UP Trial. CRS & REDCap Manual
 A Randomized Trial of Supplemental Parenteral Nutrition in Under and Over Weight Critically Ill Patients: The TOP UP Trial CRS & REDCap Manual Intended Audience: Research Coordinators This study is registered
A Randomized Trial of Supplemental Parenteral Nutrition in Under and Over Weight Critically Ill Patients: The TOP UP Trial CRS & REDCap Manual Intended Audience: Research Coordinators This study is registered
Study Procedures Manual
 A RandomizEd Trial of ENtERal Glutamine to minimize Thermal Injury Study Procedures Manual Intended Audience: Research Coordinators This study is registered at Clinicaltrials.gov. Identification number
A RandomizEd Trial of ENtERal Glutamine to minimize Thermal Injury Study Procedures Manual Intended Audience: Research Coordinators This study is registered at Clinicaltrials.gov. Identification number
electronic Case Report Form (ecrf) Worksheets and Instructions
 A Randomized Trial of Enteral Glutamine to Minimize Thermal Injury Clinical trials.gov ID #NCT00985205 electronic Case Report Form (ecrf) Worksheets and Instructions 01 September 2016 Please direct questions
A Randomized Trial of Enteral Glutamine to Minimize Thermal Injury Clinical trials.gov ID #NCT00985205 electronic Case Report Form (ecrf) Worksheets and Instructions 01 September 2016 Please direct questions
Downtime Viewer User Guide for All Users
 Downtime Viewer User Guide for All Users Overview... 1 Logging into Downtime Viewer... 1 Opening a Patient Chart in Downtime Viewer... 2 Patient Lists... 2 Clinics... 4 Navigating in the Patient s Chart...
Downtime Viewer User Guide for All Users Overview... 1 Logging into Downtime Viewer... 1 Opening a Patient Chart in Downtime Viewer... 2 Patient Lists... 2 Clinics... 4 Navigating in the Patient s Chart...
User Guide on Jobs Bank Portal (Employers)
 User Guide on Jobs Bank Portal (Employers) Table of Contents 1 INTRODUCTION... 4 2 Employer Dashboard... 5 2.1 Logging In... 5 2.2 First Time Registration... 7 2.2.1 Organisation Information Registration...
User Guide on Jobs Bank Portal (Employers) Table of Contents 1 INTRODUCTION... 4 2 Employer Dashboard... 5 2.1 Logging In... 5 2.2 First Time Registration... 7 2.2.1 Organisation Information Registration...
Certification of Employee Time and Effort
 Procedure: Policy: Number: Completing a Personnel Activity Report (PAR) Certification of Employee Time and Effort GP1200.3 ( ) Complete Revision Supersedes: Page: ( ) Partial Revision Page 1 of 21 ( X
Procedure: Policy: Number: Completing a Personnel Activity Report (PAR) Certification of Employee Time and Effort GP1200.3 ( ) Complete Revision Supersedes: Page: ( ) Partial Revision Page 1 of 21 ( X
Using PowerChart: Organizer View
 Slide Agenda Caption 3 1. Finding and logging into PowerChart 2. The Millennium Message Box 3. Toolbar Basics 4. The Organizer Toolbar 5. The Actions Toolbar 4 6. The Links toolbar 7. Patient Search Options
Slide Agenda Caption 3 1. Finding and logging into PowerChart 2. The Millennium Message Box 3. Toolbar Basics 4. The Organizer Toolbar 5. The Actions Toolbar 4 6. The Links toolbar 7. Patient Search Options
N.C.P.M emar-12 Page 1 of 10 BRIGHAM AND WOMEN S HOSPITAL DEPARTMENT OF NURSING ELECTRONIC MEDICATION ADMINISTRATION RECORD (EMAR) DOWNTIME POLICY
 Page 1 of 10 BRIGHAM AND WOMEN S HOSPITAL DEPARTMENT OF NURSING ELECTRONIC MEDICATION ADMINISTRATION RECORD (EMAR) DOWNTIME POLICY APPROVED FOR: RN LPN PCA GENERAL ICU OTHER PURPOSE: To insure a process
Page 1 of 10 BRIGHAM AND WOMEN S HOSPITAL DEPARTMENT OF NURSING ELECTRONIC MEDICATION ADMINISTRATION RECORD (EMAR) DOWNTIME POLICY APPROVED FOR: RN LPN PCA GENERAL ICU OTHER PURPOSE: To insure a process
Privacy (PPI) Training
 Privacy (PPI) Training This training is mandatory. In other words, anyone working in Home and Community Care must take the Privacy Training. This is a 2-3 hour course taken online. You only have to take
Privacy (PPI) Training This training is mandatory. In other words, anyone working in Home and Community Care must take the Privacy Training. This is a 2-3 hour course taken online. You only have to take
Implementation Manual
 RandomizEd Trial of ENtERal Glutamine to minimize Thermal Injury Implementation Manual Intended Audience: Research Coordinators & Pharmacists This study is registered at Clinicaltrials.gov. Identification
RandomizEd Trial of ENtERal Glutamine to minimize Thermal Injury Implementation Manual Intended Audience: Research Coordinators & Pharmacists This study is registered at Clinicaltrials.gov. Identification
Chapter 4. Disbursements
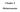 Chapter 4 Disbursements This Page Left Blank Intentionally CTAS User Manual 4-1 Disbursements: Introduction The Claims Module in CTAS allows you to post approved claims into disbursements. If you use a
Chapter 4 Disbursements This Page Left Blank Intentionally CTAS User Manual 4-1 Disbursements: Introduction The Claims Module in CTAS allows you to post approved claims into disbursements. If you use a
LOGIN TO INFINITE CAMPUS: A. In Chrome or Firefox, copy and paste the following URL and hit return and the IC login window will open
 A staff member that has been given IC rights to review and approve the application. Usually Office Support or staff member certified in Census/Enrollment. The staff member can only approve applications
A staff member that has been given IC rights to review and approve the application. Usually Office Support or staff member certified in Census/Enrollment. The staff member can only approve applications
User Guide on Jobs Bank Portal (Employers)
 User Guide on Jobs Bank Portal (Employers) Table of Contents 4 Manage Job Postings... 3 4.1 Create Job Posting... 3 4.1.1 Publish Job Posting... 10 4.2 Create Job Posting As Third Party Employer... 11
User Guide on Jobs Bank Portal (Employers) Table of Contents 4 Manage Job Postings... 3 4.1 Create Job Posting... 3 4.1.1 Publish Job Posting... 10 4.2 Create Job Posting As Third Party Employer... 11
Psychiatric Consultant Guide CMTS. Care Management Tracking System. University of Washington aims.uw.edu
 Psychiatric Consultant Guide CMTS Care Management Tracking System University of Washington aims.uw.edu rev. 8/13/2018 Table of Contents TOP TIPS & TRICKS... 1 INTRODUCTION... 2 PSYCHIATRIC CONSULTANT ACCOUNT
Psychiatric Consultant Guide CMTS Care Management Tracking System University of Washington aims.uw.edu rev. 8/13/2018 Table of Contents TOP TIPS & TRICKS... 1 INTRODUCTION... 2 PSYCHIATRIC CONSULTANT ACCOUNT
User Guide on Jobs Bank Portal (Employers)
 User Guide on Jobs Bank Portal (Employers) Table of Contents 7 Manage Interviews... 3 7.1 Send Interview Invite... 3 7.2 Sort Interview Invites... 6 7.3 Filter Interview Invites... 8 7.4 Confirm Applicant
User Guide on Jobs Bank Portal (Employers) Table of Contents 7 Manage Interviews... 3 7.1 Send Interview Invite... 3 7.2 Sort Interview Invites... 6 7.3 Filter Interview Invites... 8 7.4 Confirm Applicant
Effort Coordinator Training. University of Kansas Summer 2016
 Effort Coordinator Training University of Kansas Summer 2016 Agenda 1. Effort Reporting Overview 2. Effort Workflow and Basic Information 3. Effort Coordinator: Pre-Review 4. PI/Self-Certifier: Certification
Effort Coordinator Training University of Kansas Summer 2016 Agenda 1. Effort Reporting Overview 2. Effort Workflow and Basic Information 3. Effort Coordinator: Pre-Review 4. PI/Self-Certifier: Certification
Module Proposal Creation
 Proposal Creation 1 Module 5 1 2 Objectives In this module you will learn how to: Create a new proposal Navigate the proposal Assign proposal permissions Validate and track errors Lock/Break Lock Copy/Transform
Proposal Creation 1 Module 5 1 2 Objectives In this module you will learn how to: Create a new proposal Navigate the proposal Assign proposal permissions Validate and track errors Lock/Break Lock Copy/Transform
Medical Assistance Provider Incentive Repository. User Guide. For Eligible Hospitals
 Medical Assistance Provider Incentive Repository User Guide For Eligible Hospitals February 25, 2013 Contents Introduction...1 Before You Begin...2 Complete your R&A registration... 2 Identify one individual
Medical Assistance Provider Incentive Repository User Guide For Eligible Hospitals February 25, 2013 Contents Introduction...1 Before You Begin...2 Complete your R&A registration... 2 Identify one individual
Medical Assistance Provider Incentive Repository. User Guide. For Eligible Hospitals
 Medical Assistance Provider Incentive Repository User Guide For Eligible Hospitals February 25, 2013 Contents Introduction... 3 Before You Begin... 3 Complete your R&A registration.... 3 Identify one individual
Medical Assistance Provider Incentive Repository User Guide For Eligible Hospitals February 25, 2013 Contents Introduction... 3 Before You Begin... 3 Complete your R&A registration.... 3 Identify one individual
C. The Assessment Wizard
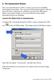 C. The Assessment Wizard The Assessment Wizard in CAPS 2 is used to record service eligibility determination information. The Assessment Wizard information is only one part of the three components of a
C. The Assessment Wizard The Assessment Wizard in CAPS 2 is used to record service eligibility determination information. The Assessment Wizard information is only one part of the three components of a
Peoplesoft Effort Certification. Participant s Manual
 Peoplesoft Effort Certification Participant s Manual Version 1.3.7 Revised April, 2007 TABLE OF CONTENTS COURSE OVERVIEW... 3 INTRODUCTION... 4 LEARNING OBJECTIVES... 4 MODULE 1: WHY COMPLETE EFFORT REPORTS...
Peoplesoft Effort Certification Participant s Manual Version 1.3.7 Revised April, 2007 TABLE OF CONTENTS COURSE OVERVIEW... 3 INTRODUCTION... 4 LEARNING OBJECTIVES... 4 MODULE 1: WHY COMPLETE EFFORT REPORTS...
Office of Clinical Research. CTMS Reference Guide Patient Entry & Visit Tracking
 Se Office of Clinical Research CTMS Reference Guide Patient Entry & Visit Tracking Table of Contents Logging into CTMS... 3 Search and Recruitment / Quick Search... 4 How to Configure Quick Search Fields...
Se Office of Clinical Research CTMS Reference Guide Patient Entry & Visit Tracking Table of Contents Logging into CTMS... 3 Search and Recruitment / Quick Search... 4 How to Configure Quick Search Fields...
Reimbursements: Submit a Flat Rate Reimbursement
 Reimbursements: Submit a Flat Rate Reimbursement Overview Tax-Aide volunteers may elect to receive a one-time, flat-rate expense reimbursement for which volunteers receive $35 and volunteer leaders receive
Reimbursements: Submit a Flat Rate Reimbursement Overview Tax-Aide volunteers may elect to receive a one-time, flat-rate expense reimbursement for which volunteers receive $35 and volunteer leaders receive
Site Manager Guide CMTS. Care Management Tracking System. University of Washington aims.uw.edu
 Site Manager Guide CMTS Care Management Tracking System University of Washington aims.uw.edu rev. 8/13/2018 Table of Contents INTRODUCTION... 1 SITE MANAGER ACCOUNT ROLE... 1 ACCESSING CMTS... 2 SITE NAVIGATION
Site Manager Guide CMTS Care Management Tracking System University of Washington aims.uw.edu rev. 8/13/2018 Table of Contents INTRODUCTION... 1 SITE MANAGER ACCOUNT ROLE... 1 ACCESSING CMTS... 2 SITE NAVIGATION
An Introduction to FirstNet for Nurses
 V3 : 17-01-2017 An Introduction to FirstNet for Nurses Nursing Staff Induction Program The Townsville Hospital June 2017 1. Log into FirstNet 1. Double click on iemr icon form desktop screen 2. Enter user
V3 : 17-01-2017 An Introduction to FirstNet for Nurses Nursing Staff Induction Program The Townsville Hospital June 2017 1. Log into FirstNet 1. Double click on iemr icon form desktop screen 2. Enter user
Atlas LabWorks User Guide Table of Contents
 http://lab.parkview.com Atlas LabWorks User Guide Table of Contents Technical Support 2 Online Directory of Services.......3 Log into Connect.Parkview.com Account... 4 Log into Atlas Account....6 Patient
http://lab.parkview.com Atlas LabWorks User Guide Table of Contents Technical Support 2 Online Directory of Services.......3 Log into Connect.Parkview.com Account... 4 Log into Atlas Account....6 Patient
N C MPASS. Clinical Self-Scheduling. Version 6.8
 N C MPASS Clinical Self-Scheduling Version 6.8 Ontario Telemedicine Network (OTN) All rights reserved. Last update: May 24, 2018 This document is the property of OTN. No part of this document may be reproduced
N C MPASS Clinical Self-Scheduling Version 6.8 Ontario Telemedicine Network (OTN) All rights reserved. Last update: May 24, 2018 This document is the property of OTN. No part of this document may be reproduced
Psychiatric Consultant Guide SPIRIT CMTS. Care Management Tracking System. University of Washington aims.uw.edu
 Psychiatric Consultant Guide SPIRIT CMTS Care Management Tracking System University of Washington aims.uw.edu rev. 9/20/2016 Table of Contents TOP TIPS & TRICKS... 1 INTRODUCTION... 2 PSYCHIATRIC CONSULTANT
Psychiatric Consultant Guide SPIRIT CMTS Care Management Tracking System University of Washington aims.uw.edu rev. 9/20/2016 Table of Contents TOP TIPS & TRICKS... 1 INTRODUCTION... 2 PSYCHIATRIC CONSULTANT
Paragon Clinician Hub for Physicians (PCH) Reference
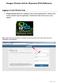 Paragon Clinician Hub for Physicians (PCH) Reference Logging in to the Clinician Hub Paragon Clinician Hub (PCH) is available on any Carroll Hospital Network. VMWare View must be utilized to open the application.
Paragon Clinician Hub for Physicians (PCH) Reference Logging in to the Clinician Hub Paragon Clinician Hub (PCH) is available on any Carroll Hospital Network. VMWare View must be utilized to open the application.
Kansas University Medical Center ecrt Department Administrator Training. June 2008
 Kansas University Medical Center ecrt Department Administrator Training June 2008 KUMC Process Timeline Effort Reporting Period 3 Week Pre-Review Period 3 Week Certification Period Post Certification Period
Kansas University Medical Center ecrt Department Administrator Training June 2008 KUMC Process Timeline Effort Reporting Period 3 Week Pre-Review Period 3 Week Certification Period Post Certification Period
Overview What is effort? What is effort reporting? Why is Effort Reporting necessary?... 2
 Effort Certification Training Guide Contents Overview... 2 What is effort?... 2 What is effort reporting?... 2 Why is Effort Reporting necessary?... 2 Effort Certification Process: More than just Certification...
Effort Certification Training Guide Contents Overview... 2 What is effort?... 2 What is effort reporting?... 2 Why is Effort Reporting necessary?... 2 Effort Certification Process: More than just Certification...
Venous Thromboembolism (VTE) Audit Day
 Venous Thromboembolism (VTE) Audit Day Questions If you have any questions or require clarification, please contact Artemis Diamantouros. Email: artemis.diamantouros@sunnybrook.ca Welcome to the Canadian
Venous Thromboembolism (VTE) Audit Day Questions If you have any questions or require clarification, please contact Artemis Diamantouros. Email: artemis.diamantouros@sunnybrook.ca Welcome to the Canadian
Audit of Communication, CarE Planning, and DocumenTation
 Audit of Communication, CarE Planning, and DocumenTation A multicenter, prospective study Implementation Manual Short Version This study is registered at ClinicalTrials.gov Identification #: NCT01362855
Audit of Communication, CarE Planning, and DocumenTation A multicenter, prospective study Implementation Manual Short Version This study is registered at ClinicalTrials.gov Identification #: NCT01362855
Booking Elective Trauma Surgery for Inpatients
 ADT31 Version 3.1 Trauma Team Operational Areas Included Trauma Co-ordinator Roles Responsible for Carrying out this Process All other areas Operational Areas Excluded GEN01 Logging into Lorenzo GEN02
ADT31 Version 3.1 Trauma Team Operational Areas Included Trauma Co-ordinator Roles Responsible for Carrying out this Process All other areas Operational Areas Excluded GEN01 Logging into Lorenzo GEN02
RETRIEVAL AND CRITICAL HEALTH INFORMATION SYSTEM
 RETRIEVAL AND CRITICAL HEALTH INFORMATION SYSTEM USER GUIDE May 2017 Contents Introduction... 3 Access to REACH... 3 Homepage... 3 Roles within REACH... 4 Hospital Administrator... 4 Hospital User... 4
RETRIEVAL AND CRITICAL HEALTH INFORMATION SYSTEM USER GUIDE May 2017 Contents Introduction... 3 Access to REACH... 3 Homepage... 3 Roles within REACH... 4 Hospital Administrator... 4 Hospital User... 4
PATIENT PORTAL USERS GUIDE
 PATIENT PORTAL USERS GUIDE V 5.0 December 2012 eclinicalworks, 2012. All rights reserved Login and Pre-Registration Patients enter a valid Username and secure Password, then click the Sign In button to
PATIENT PORTAL USERS GUIDE V 5.0 December 2012 eclinicalworks, 2012. All rights reserved Login and Pre-Registration Patients enter a valid Username and secure Password, then click the Sign In button to
Instructions for Navigating Your Awarded Grant
 Instructions for Navigating Your Awarded Grant proposalcentral s Post-Award allows grantees to submit progress reports, project documents, financial/budget information, communicate with the funding organization,
Instructions for Navigating Your Awarded Grant proposalcentral s Post-Award allows grantees to submit progress reports, project documents, financial/budget information, communicate with the funding organization,
Surgical Appliance Walk-in patients
 APS02 Version 3.0 Appliance Services Operational Areas Included HCA Roles Responsible for Carrying out this Process All Other Areas Operational Areas Excluded GEN01 Logging into Lorenzo Associated Procedures
APS02 Version 3.0 Appliance Services Operational Areas Included HCA Roles Responsible for Carrying out this Process All Other Areas Operational Areas Excluded GEN01 Logging into Lorenzo Associated Procedures
MONITORING PATIENTS. Responding to Readings
 CHAPTER 6 MONITORING PATIENTS Responding to Readings This section covers the steps required to respond to patient readings within LifeStream. You can view patient readings on the Current Status or Tabular
CHAPTER 6 MONITORING PATIENTS Responding to Readings This section covers the steps required to respond to patient readings within LifeStream. You can view patient readings on the Current Status or Tabular
Care Planning User Guide June 2011
 User Guide June 2011 2011, ADL Data Systems, Inc. All rights reserved Table of Contents Introduction... 1 About Care Plan... 1 About this Information... 1 Logon... 2 Care Planning Module Basics... 5 Starting
User Guide June 2011 2011, ADL Data Systems, Inc. All rights reserved Table of Contents Introduction... 1 About Care Plan... 1 About this Information... 1 Logon... 2 Care Planning Module Basics... 5 Starting
Find & Apply. User Guide
 Find & Apply User Guide Version 2.0 Prepared April 9, 2008 Grants.gov Find and Apply User Guide Table of Contents Introduction....3 Find Grant Opportunities...4 Search Grant Opportunities...5 Email Subscription...8
Find & Apply User Guide Version 2.0 Prepared April 9, 2008 Grants.gov Find and Apply User Guide Table of Contents Introduction....3 Find Grant Opportunities...4 Search Grant Opportunities...5 Email Subscription...8
Entering Direct Service & Fundraising Hours in the My Service Log System
 Entering Direct Service & Fundraising Hours in the My Service Log System A Step by Step Overview 1 This presentation will show you how to enter hours that you serve in the ADVANCE AmeriCorps program participating
Entering Direct Service & Fundraising Hours in the My Service Log System A Step by Step Overview 1 This presentation will show you how to enter hours that you serve in the ADVANCE AmeriCorps program participating
Bar Code Medication Administration and MAR Resource Manual
 Bar Code Medication Administration and MAR Resource Manual Administering Medications Administering Meds using CareMobile (PDA)... 2 Viewing Allergies in CareMobile... 8 Determining Which Meds to Give When...
Bar Code Medication Administration and MAR Resource Manual Administering Medications Administering Meds using CareMobile (PDA)... 2 Viewing Allergies in CareMobile... 8 Determining Which Meds to Give When...
Educational Grant and Outcomes Database User Guide
 Educational Grant and Outcomes Database User Guide June 06 Table of Contents Getting Started System Tips and Useful Hints p.3 Where to Find Us p.4 Logging in as a Registered User p.5 Registering as a First-Time
Educational Grant and Outcomes Database User Guide June 06 Table of Contents Getting Started System Tips and Useful Hints p.3 Where to Find Us p.4 Logging in as a Registered User p.5 Registering as a First-Time
Soarian Clinicals View Only
 Soarian Clinicals View Only Participant Guide Table of Contents 1. Welcome!... 5 Course Description... 5 Learning Objectives... 5 What to Expect... 5 Evaluation... 5 Agenda... 5 2. Getting Started... 6
Soarian Clinicals View Only Participant Guide Table of Contents 1. Welcome!... 5 Course Description... 5 Learning Objectives... 5 What to Expect... 5 Evaluation... 5 Agenda... 5 2. Getting Started... 6
Serious Adverse Events
 The REDOXS Study REducing Deaths due to OXidative Stress A randomized trial of glutamine and antioxidant supplementation in critically ill patients Serious Adverse Events This study is registered at Clinicaltrials.gov.
The REDOXS Study REducing Deaths due to OXidative Stress A randomized trial of glutamine and antioxidant supplementation in critically ill patients Serious Adverse Events This study is registered at Clinicaltrials.gov.
e-sdrt User Guide, Update April 2014 First Nations and Inuit Home and Community Care Program: e-sdrt User Guide
 e-sdrt User Guide, Update April 2014 First Nations and Inuit Home and Community Care Program: e-sdrt User Guide 1 e-sdrt User Guide, Update April 2014 2 e-sdrt User Guide, Update April 2014 TABLE OF CONTENTS
e-sdrt User Guide, Update April 2014 First Nations and Inuit Home and Community Care Program: e-sdrt User Guide 1 e-sdrt User Guide, Update April 2014 2 e-sdrt User Guide, Update April 2014 TABLE OF CONTENTS
RETRIEVAL AND CRITICAL HEALTH INFORMATION SYSTEM
 RETRIEVAL AND CRITICAL HEALTH INFORMATION SYSTEM USER GUIDE November 2014 Contents Introduction... 4 Access to REACH... 4 Homepage... 4 Roles within REACH... 5 Hospital Administrator... 5 Hospital User...
RETRIEVAL AND CRITICAL HEALTH INFORMATION SYSTEM USER GUIDE November 2014 Contents Introduction... 4 Access to REACH... 4 Homepage... 4 Roles within REACH... 5 Hospital Administrator... 5 Hospital User...
Appendix 3 Record Review Workbook Instructions
 Appendix 3 Record Review Workbook Instructions NCQA PCMH Standards and Guidelines (2017 Edition, Version 2) September 30, 2017 Appendix 3 PCMH Record Review Workbook General Instructions 3-1 APPENDIX 3
Appendix 3 Record Review Workbook Instructions NCQA PCMH Standards and Guidelines (2017 Edition, Version 2) September 30, 2017 Appendix 3 PCMH Record Review Workbook General Instructions 3-1 APPENDIX 3
Post-Production, Visual Effects and Digital Animation Grant GUIDE TO APPLICATION SUBMISSION
 Post-Production, Visual Effects and Digital Animation Grant GUIDE TO APPLICATION SUBMISSION Introduction This guide will assist Post-Production, Visual Effects and Digital Animation Grant (PPG) applicants
Post-Production, Visual Effects and Digital Animation Grant GUIDE TO APPLICATION SUBMISSION Introduction This guide will assist Post-Production, Visual Effects and Digital Animation Grant (PPG) applicants
RD Apply Automated Intake Application System for Water and Environmental Programs December 2015
 RD Apply Automated Intake Application System for Water and Environmental Programs December 2015 Michele J. Cardwell, Acting State Director and Community Programs Director Jane Patchin, Rural Development
RD Apply Automated Intake Application System for Water and Environmental Programs December 2015 Michele J. Cardwell, Acting State Director and Community Programs Director Jane Patchin, Rural Development
HELLO HEALTH TRAINING MANUAL
 HELLO HEALTH TRAINING MANUAL Please note: As with all training materials, the names and data used in this training manual are purely fictitious and for information and training purposes only Login/What
HELLO HEALTH TRAINING MANUAL Please note: As with all training materials, the names and data used in this training manual are purely fictitious and for information and training purposes only Login/What
Medication Module Tutorial
 Medication Module Tutorial An Introduction to the Medication module Whether completing a clinic patient evaluation, a hospital admission history and physical, a discharge summary, a hospital order set,
Medication Module Tutorial An Introduction to the Medication module Whether completing a clinic patient evaluation, a hospital admission history and physical, a discharge summary, a hospital order set,
Optima POC PARTICIPANT GUIDE
 Optima POC Point of Care PARTICIPANT GUIDE 2017 Optima Healthcare Solutions Page 1 CONTENTS CONTENTS... 2 ABOUT THIS GUIDE... 3 LEARNING OUTCOMES... 4 1. ACCESSING POINT OF CARE... 5 2. CLOCKING IN...
Optima POC Point of Care PARTICIPANT GUIDE 2017 Optima Healthcare Solutions Page 1 CONTENTS CONTENTS... 2 ABOUT THIS GUIDE... 3 LEARNING OUTCOMES... 4 1. ACCESSING POINT OF CARE... 5 2. CLOCKING IN...
MAR Training Guide for Nurses
 MAR Training Guide for Nurses Medication Ordering Fields Verbal Orders Workflow And Navigating the MAR Contents HOW DO I BEGIN?... 3 Update Adverse Drug Reactions... 3 Enter Verbal Orders from Nursing
MAR Training Guide for Nurses Medication Ordering Fields Verbal Orders Workflow And Navigating the MAR Contents HOW DO I BEGIN?... 3 Update Adverse Drug Reactions... 3 Enter Verbal Orders from Nursing
KN-CLAIM. Kansas Nutrition - CLaims And Information Management Quick Reference for Verification Reporting
 KN-CLAIM Kansas Nutrition - CLaims And Information Management 2016-2017 Quick Reference for Verification Reporting Child Nutrition & Wellness Kansas State Department of Education Child Nutrition & Wellness
KN-CLAIM Kansas Nutrition - CLaims And Information Management 2016-2017 Quick Reference for Verification Reporting Child Nutrition & Wellness Kansas State Department of Education Child Nutrition & Wellness
User Guide on Jobs Bank (Individuals)
 User Guide on Jobs Bank (Individuals) Table of Contents 1 Individual Dashboard... 3 1.1 Logging In... 3 1.2 Logging Out... 5 2 Profile... 6 2.1 Make Selected Profile Information Not Viewable To All Employers...
User Guide on Jobs Bank (Individuals) Table of Contents 1 Individual Dashboard... 3 1.1 Logging In... 3 1.2 Logging Out... 5 2 Profile... 6 2.1 Make Selected Profile Information Not Viewable To All Employers...
EFFORT CERTIFICATION GUIDE
 SOUTH DAKOTA SCHOOL OF MINES AND TECHNOLOGY EFFORT CERTIFICATION GUIDE 1/1/2011 WEB-BASED EFFORT CERTIFICATION Version 2 What is Effort Certification? Effort Certification is the institution s process
SOUTH DAKOTA SCHOOL OF MINES AND TECHNOLOGY EFFORT CERTIFICATION GUIDE 1/1/2011 WEB-BASED EFFORT CERTIFICATION Version 2 What is Effort Certification? Effort Certification is the institution s process
QOF queries in SystmOne
 QOF queries in SystmOne For further help with QOF: 1. See the Primary Care Contracting (www.primarycarecontracting.nhs.uk) website for more information 2. Contact your PCT Information or Data Quality team
QOF queries in SystmOne For further help with QOF: 1. See the Primary Care Contracting (www.primarycarecontracting.nhs.uk) website for more information 2. Contact your PCT Information or Data Quality team
RCPsych CPD Submission User Guide March 2015
 RCPsych CPD Submission User Guide March 2015 Submitting your CPD returns online CPD submission, the electronic system for recording your CPD activities and submitting returns and generating Certificates
RCPsych CPD Submission User Guide March 2015 Submitting your CPD returns online CPD submission, the electronic system for recording your CPD activities and submitting returns and generating Certificates
EMAR Medication Pass with Pre-Pour
 EMAR Medication Pass with Pre-Pour This manual includes the setup of medications with Pre-Pour and the recording of resident medication passes. The Pre- Pour options must be turned on in File Setup Community.
EMAR Medication Pass with Pre-Pour This manual includes the setup of medications with Pre-Pour and the recording of resident medication passes. The Pre- Pour options must be turned on in File Setup Community.
SigmaCare Care Management
 This document will walk a Nurse through all SBAR tasks in SigmaCare, including: Adding a new SBAR from Resident Summary Page 1 Adding a new SBAR from an open Stop and Watch Page 2 SBAR Entry... Page 3
This document will walk a Nurse through all SBAR tasks in SigmaCare, including: Adding a new SBAR from Resident Summary Page 1 Adding a new SBAR from an open Stop and Watch Page 2 SBAR Entry... Page 3
Reference Guide for Applicants
 Grants Ontario System Reference Guide for Applicants Please inform us if you need a different format or other accommodation to access this information. Version Date: July 2017 Table of Contents Introduction...
Grants Ontario System Reference Guide for Applicants Please inform us if you need a different format or other accommodation to access this information. Version Date: July 2017 Table of Contents Introduction...
University of Miami Clinical Enterprise Technologies
 Provider Manual 1 Our Mission: To design and deliver ongoing support for a network of Business and Clinical Information Management Systems which enhance the academic and research vision while implementing
Provider Manual 1 Our Mission: To design and deliver ongoing support for a network of Business and Clinical Information Management Systems which enhance the academic and research vision while implementing
ED Disposition Diagnosis. Training Manual for. ED Physicians
 ED Disposition Diagnosis Training Manual for ED Physicians Warning: In Post Train do not select the Display Board button as it will freeze your window and you will not be able to close out of the window.
ED Disposition Diagnosis Training Manual for ED Physicians Warning: In Post Train do not select the Display Board button as it will freeze your window and you will not be able to close out of the window.
MEDICAL SPECIALISTS OF THE PALM BEACHES, INC. Chronic Care Management (CCM) Program Training Manual
 MEDICAL SPECIALISTS OF THE PALM BEACHES, INC. Chronic Care Management (CCM) Program Training Manual September 2017 Table of Contents CCM PROGRAM OVERVIEW... 4 3 STEPS TO BEGIN CCM:... 5 Identify the Patient...
MEDICAL SPECIALISTS OF THE PALM BEACHES, INC. Chronic Care Management (CCM) Program Training Manual September 2017 Table of Contents CCM PROGRAM OVERVIEW... 4 3 STEPS TO BEGIN CCM:... 5 Identify the Patient...
Sepsis Screening Tool
 S I E M E N S NET A C C E S S Sepsis Screening Tool C L I N I C A L I N F O R M A T I C S M A R C H 2 0 1 2 Screening Objectives Early identification of patients for SEVERE Sepsis Initiate early resuscitation
S I E M E N S NET A C C E S S Sepsis Screening Tool C L I N I C A L I N F O R M A T I C S M A R C H 2 0 1 2 Screening Objectives Early identification of patients for SEVERE Sepsis Initiate early resuscitation
CPOM TRAINING. Page 1
 CPOM TRAINING Page 1 Physician Training For CPOM Patient list columns, Flag Management, Icons Icons added for CPOM: Columns added: Flags New Orders: GREEN - are general orders. RED means STAT orders included
CPOM TRAINING Page 1 Physician Training For CPOM Patient list columns, Flag Management, Icons Icons added for CPOM: Columns added: Flags New Orders: GREEN - are general orders. RED means STAT orders included
Completing a Medication History Inpatient Nurses
 Completing a Medication History Inpatient Nurses Inpatient nurses may complete a medication history completing the following steps: Open the patient s chart Click the Ad hoc button Double click the Nursing
Completing a Medication History Inpatient Nurses Inpatient nurses may complete a medication history completing the following steps: Open the patient s chart Click the Ad hoc button Double click the Nursing
Wisconsin Department of Justice. Egrants System User Guide
 Wisconsin Department of Justice Egrants System User Guide Last updated 11-17/2017 What is in this guide? This guide describes technical steps describing how to apply for and manage a grant in Egrants.
Wisconsin Department of Justice Egrants System User Guide Last updated 11-17/2017 What is in this guide? This guide describes technical steps describing how to apply for and manage a grant in Egrants.
Amalga FAQs. When I print my patient s Form, there are no printer options. How do I get this fixed? Call the Support Center at
 Amalga FAQs Amalga Access: How do I access Amalga from home or outside of Novant Health facilities? You would require Phone Factor or FOB to access Amalga from outside of Novant. Once you log in using
Amalga FAQs Amalga Access: How do I access Amalga from home or outside of Novant Health facilities? You would require Phone Factor or FOB to access Amalga from outside of Novant. Once you log in using
Mobile Lite Training Key Notes
 Mobile Lite Training Key Notes Home Health Aides can do nothing on the mobile device until their schedule has been created. The Scheduler is responsible for assuring that an Aide care plan has been initiated
Mobile Lite Training Key Notes Home Health Aides can do nothing on the mobile device until their schedule has been created. The Scheduler is responsible for assuring that an Aide care plan has been initiated
EMAR Medication Pass
 EMAR Medication Pass This manual includes recording of resident medication passes on a computer. To begin your Medication Pass, click on the EMAR icon, then select a Med Provider. The listing of Med Providers
EMAR Medication Pass This manual includes recording of resident medication passes on a computer. To begin your Medication Pass, click on the EMAR icon, then select a Med Provider. The listing of Med Providers
User Guide. Shortlisting Advertised Job
 User Guide Shortlisting Advertised Job Brief Document Description Overview This User Guide explains how to review applications and shortlist using the erecruitment system for an advertised job. About this
User Guide Shortlisting Advertised Job Brief Document Description Overview This User Guide explains how to review applications and shortlist using the erecruitment system for an advertised job. About this
Table of Contents INTRODUCTION NEW APPLICATION GETTING STARTED POST SUBMISSION. Return to Table of Contents
 External User Guide Table of Contents INTRODUCTION GETTING STARTED Mission Statement Commitment to Research G Vision Investigator/Sponsor Responsibilities Visiontracker Web Portal Registration New User
External User Guide Table of Contents INTRODUCTION GETTING STARTED Mission Statement Commitment to Research G Vision Investigator/Sponsor Responsibilities Visiontracker Web Portal Registration New User
AHP Online Guide Contact Changes Sponsor or Member
 INTRODUCTION Congratulations on your Affordable Housing Program (AHP) award! An AHP award requires the member and sponsor to provide timely reporting on the status of the project. Compliance reporting
INTRODUCTION Congratulations on your Affordable Housing Program (AHP) award! An AHP award requires the member and sponsor to provide timely reporting on the status of the project. Compliance reporting
Now that we have reviewed the agenda and objectives for today, let s proceed with the EC Grants Overview (PPT SLIDE 1).
 Welcome / Introductions Hello. I m Dr. Khalilah O Farrow-Boulware, Regional Trainer for the Piedmont-Triad and Southwest Districts. I d like to welcome you to this EC Grants training webinar. Please note
Welcome / Introductions Hello. I m Dr. Khalilah O Farrow-Boulware, Regional Trainer for the Piedmont-Triad and Southwest Districts. I d like to welcome you to this EC Grants training webinar. Please note
AIRPORT SPONSOR USER GUIDE
 AIRPORT SPONSOR USER GUIDE Table of Contents Section 1: Introduction... 2 1.1 What is BlackCat Grant Management System?... 2 1.2 This User s Guide... 2 Section 2: Getting Started... 3 2.1 User Access...
AIRPORT SPONSOR USER GUIDE Table of Contents Section 1: Introduction... 2 1.1 What is BlackCat Grant Management System?... 2 1.2 This User s Guide... 2 Section 2: Getting Started... 3 2.1 User Access...
Bar Code Medication Administration and MAR Resource Manual
 Bar Code Medication Administration and MAR Resource Manual Creating Orders Creating an Order in CareMobile (Ad Hoc Order Entry)...2 Creating an Order for med that is already ordered with a different dose/frequency....4
Bar Code Medication Administration and MAR Resource Manual Creating Orders Creating an Order in CareMobile (Ad Hoc Order Entry)...2 Creating an Order for med that is already ordered with a different dose/frequency....4
FY 2014 Amendments Instructional Guide for Recipients
 e-snaps Training Series FY 2014 Amendments Instructional Guide for Recipients Completing e-snaps step C1.9b 2015, Version 1 Table of Contents Introduction... 2 Objectives... 2 Overview of the Amendment
e-snaps Training Series FY 2014 Amendments Instructional Guide for Recipients Completing e-snaps step C1.9b 2015, Version 1 Table of Contents Introduction... 2 Objectives... 2 Overview of the Amendment
ipm Information Sheet
 Research Data Entry into IPM Purposes of entering research participation data: ipm Information Sheet 1. To set up patient alerts to notify IPM users if patients are enrolled in a clinical trial/research
Research Data Entry into IPM Purposes of entering research participation data: ipm Information Sheet 1. To set up patient alerts to notify IPM users if patients are enrolled in a clinical trial/research
Using DonorCentral. With DonorCentral, you can: Keep Fund summary information at your fingertips:
 Using DonorCentral DonorCentral is a secure web portal that gives you online access to information about the Fund you have established at the Calgary Foundation (referred to as the Fund ). Amongst its
Using DonorCentral DonorCentral is a secure web portal that gives you online access to information about the Fund you have established at the Calgary Foundation (referred to as the Fund ). Amongst its
Grants Ontario Application Instructions Step by Step Guide
 Grants Ontario Application Instructions Step by Step Guide These steps are designed to help you to submit your Grants Ontario application, attach documents to your application, view payments, and retrieve
Grants Ontario Application Instructions Step by Step Guide These steps are designed to help you to submit your Grants Ontario application, attach documents to your application, view payments, and retrieve
Back Office-General Quick Reference Guide. Enter a Home Health Referral
 Back Office-General Quick Reference Guide Enter a Home Health Referral Table of Contents Enter a Referral... 3 Common Buttons & Icons... 3 Enter a New Referral... 4 Document Basic Info... 5 Document Demographics...
Back Office-General Quick Reference Guide Enter a Home Health Referral Table of Contents Enter a Referral... 3 Common Buttons & Icons... 3 Enter a New Referral... 4 Document Basic Info... 5 Document Demographics...
Grant E-Management System Help User Guide for Applicants, Reviewers and Co-Signatories
 Grant E-Management System Help User Guide for Applicants, Reviewers and Co-Signatories About this document This document describes how to register as a user of the Grant E-Management System, and how to
Grant E-Management System Help User Guide for Applicants, Reviewers and Co-Signatories About this document This document describes how to register as a user of the Grant E-Management System, and how to
DEPARTMENT OF COUNSELOR EDUCATION AND FAMILY STUDIES. LiveText Field Experience Manual Practicum & Internship
 DEPARTMENT OF COUNSELOR EDUCATION AND FAMILY STUDIES LiveText Field Experience Manual Practicum & Internship 2018-2019 1 Table of Contents Introduction...3 How To Purchase LiveText...3 How To Register
DEPARTMENT OF COUNSELOR EDUCATION AND FAMILY STUDIES LiveText Field Experience Manual Practicum & Internship 2018-2019 1 Table of Contents Introduction...3 How To Purchase LiveText...3 How To Register
A Guide to the National Institute for Health Research Central Commissioning Facility Research Management System (Grant Tracker)
 INTERNAL USER GUIDE A Guide to the National Institute for Health Research Central Commissioning Facility Research Management System (Grant Tracker) A Quick Guide for applicants applying for funding from
INTERNAL USER GUIDE A Guide to the National Institute for Health Research Central Commissioning Facility Research Management System (Grant Tracker) A Quick Guide for applicants applying for funding from
AbbVie Grant Management System (GMS) Requestor Training, Grant Request Training: General Program Support
 AbbVie Grant Management System (GMS) Requestor Training, Grant Request Training: General Program Support After Reviewing this Module, You Will be Able to: Log Onto the GMS Setting up Your Security Password
AbbVie Grant Management System (GMS) Requestor Training, Grant Request Training: General Program Support After Reviewing this Module, You Will be Able to: Log Onto the GMS Setting up Your Security Password
Mobile App Process Guide
 Mobile App Process Guide Agency Setup and Management Copyright 2018 Homecare Software Solutions, LLC One Court Square 44th Floor Long Island City, NY 11101 Phone: (718) 407-4633 Fax: (718) 679-9273 Document
Mobile App Process Guide Agency Setup and Management Copyright 2018 Homecare Software Solutions, LLC One Court Square 44th Floor Long Island City, NY 11101 Phone: (718) 407-4633 Fax: (718) 679-9273 Document
NEW HAMPSHIRE MEDICAID EHR INCENTIVE PROGRAM
 NEW HAMPSHIRE MEDICAID EHR INCENTIVE PROGRAM Eligible Professional Reference Guide for Modified Stage 2 Meaningful Use EP REVISION HISTORY Version Number Date Comments 1.0 September 2013 Posted on NH Medicaid
NEW HAMPSHIRE MEDICAID EHR INCENTIVE PROGRAM Eligible Professional Reference Guide for Modified Stage 2 Meaningful Use EP REVISION HISTORY Version Number Date Comments 1.0 September 2013 Posted on NH Medicaid
Transfer student application guide
 Transfer student application guide Table of Contents Introduction... 3 Application Dates and Deadlines... 3 Create Account... 4-6 Complete Your Profile... 6-9 Changing Your Profile... 10 Select Programs...
Transfer student application guide Table of Contents Introduction... 3 Application Dates and Deadlines... 3 Create Account... 4-6 Complete Your Profile... 6-9 Changing Your Profile... 10 Select Programs...
Care Manager Guide SPIRIT CMTS. Care Management Tracking System. University of Washington aims.uw.edu
 Care Manager Guide SPIRIT CMTS Care Management Tracking System University of Washington aims.uw.edu rev. 12/4/2017 Table of Contents TOP TIPS & TRICKS... 1 INTRODUCTION... 2 CARE MANAGER ACCOUNT ROLE...
Care Manager Guide SPIRIT CMTS Care Management Tracking System University of Washington aims.uw.edu rev. 12/4/2017 Table of Contents TOP TIPS & TRICKS... 1 INTRODUCTION... 2 CARE MANAGER ACCOUNT ROLE...
Personnel Services Delivery Guide 8 March
 Personnel Services Delivery Guide 8 March 2013 1 INTRODUCTION ASSIGNMENT NOTIFICATION The virtual automated Assignment Notification process replaces the manual Assignment Notification Report on Individual
Personnel Services Delivery Guide 8 March 2013 1 INTRODUCTION ASSIGNMENT NOTIFICATION The virtual automated Assignment Notification process replaces the manual Assignment Notification Report on Individual
Patient s Guide to The Waiting Room. Version 1.1 Date: 17-Feb-17
 Patient s Guide to The Waiting Room Version 1.1 Date: 17-Feb-17 Contents Registering for The Waiting Room with your practice... 3 Account Details... 4 Creating an account at TWR... 4 First visit to The
Patient s Guide to The Waiting Room Version 1.1 Date: 17-Feb-17 Contents Registering for The Waiting Room with your practice... 3 Account Details... 4 Creating an account at TWR... 4 First visit to The
Creating A Patient Portal Link From More Patient Button
 Creating A Patient Portal Link From More Patient Button Go to More Patient and click on the Export PHI tab. From this tab, click on Create Patient Portal Link. Note: Allow Internet Based Delivery Of Reminders
Creating A Patient Portal Link From More Patient Button Go to More Patient and click on the Export PHI tab. From this tab, click on Create Patient Portal Link. Note: Allow Internet Based Delivery Of Reminders
MEDITECH 6.14 HOW TO ACCESS THE PATIENT S ELECTRONIC MEDICAL RECORD (EMR)
 MEDITECH 6.14 HOW TO ACCESS THE PATIENT S ELECTRONIC MEDICAL RECORD (EMR) Table of Contents How to Log on to MEDITECH 6.14... 3 How to Access Patient Information... 5 Recently Accessed... 5 Find Patient...
MEDITECH 6.14 HOW TO ACCESS THE PATIENT S ELECTRONIC MEDICAL RECORD (EMR) Table of Contents How to Log on to MEDITECH 6.14... 3 How to Access Patient Information... 5 Recently Accessed... 5 Find Patient...
DonorCentral Handbook
 DonorCentral Handbook www.stlgives.org Click on My Fund to log in The St. Louis Community Foundation s DonorCentral provides you online access to your fund activity and history. Depending on the parameters
DonorCentral Handbook www.stlgives.org Click on My Fund to log in The St. Louis Community Foundation s DonorCentral provides you online access to your fund activity and history. Depending on the parameters
Guide to Enterprise Zone Pre-Certification
 This user guide provides web screen shots to reach the application portal from OEDIT s website, navigate the application portal, and complete an Enterprise Zone Pre-Certification application. The OEDIT
This user guide provides web screen shots to reach the application portal from OEDIT s website, navigate the application portal, and complete an Enterprise Zone Pre-Certification application. The OEDIT
How to Add an Annual Facility Survey
 Add an Annual Facility Survey https://nhsn.cdc.gov/nhsndemo/help/patient_safety_component/how_to/add_an_annual... Page 1 of 1 10/9/2017 Show Patient Safety Component > How To > Facility > Add an Annual
Add an Annual Facility Survey https://nhsn.cdc.gov/nhsndemo/help/patient_safety_component/how_to/add_an_annual... Page 1 of 1 10/9/2017 Show Patient Safety Component > How To > Facility > Add an Annual
CLINICIAN MANUAL. LATITUDE Patient Management System
 CLINICIAN MANUAL LATITUDE Patient Management System Table of Contents LATITUDE PATIENT MANAGEMENT INTRODUCTION........................... 1 Intended Use..........................................................
CLINICIAN MANUAL LATITUDE Patient Management System Table of Contents LATITUDE PATIENT MANAGEMENT INTRODUCTION........................... 1 Intended Use..........................................................
