Steps when Subjects are Scheduled at CTRC
|
|
|
- Buck Jenkins
- 6 years ago
- Views:
Transcription
1 CTRC Statement of Understanding Scheduling Subjects in the CTRC (5/6/14) As part of our move to the new location on Hatfield 10D, our scheduling processes have changed. The CTRC has begun scheduling all participant visits in Epic as of April 28, This change impacts CTRC visit scheduling in two ways: 1. We now require an OHSU medical record number (MRN) on all subjects 2. We now require that all subjects be linked in Epic to the study Ideally, each subject will have both an MRN, as well as be linked to the study prior to the time of scheduling. To help facilitate understanding of our new processes we have created several informational documents: Additional Steps when Subjects are Scheduled at CTRC Instructions for Obtaining an OHSU MRN VA Study Patient Process Anonymous Patient Information Steps when Subjects are Scheduled at CTRC 1. Study Staff -complete all steps below: a. Submit the study to the IRB b. Submit an OCTRI Resource Request form requesting CTRC services to the OCTRI Research Navigator i. institutes/octri/resources/upload/octri-resource-request-form docx c. If the study also involves medications, upload the CRTC medication order form in the ecris pharmacy smart form d. Access the CTRC Medication Template form on the OCTRI website. Send the request form to Research Pharmacy at
2 i. institutes/octri/resources/policies-forms/upload/ctrc-research- Medication-Template-Form.pdf Note: We are utilizing Epic Therapy plan functionality for all medications that are given at CTRC. We need to start this process 6 weeks from when the subject will first be seen. 2. CTRC receives and reviews service request; provides cost estimate through OCTRI Navigator to study staff; sets up study initiation meeting (see CTRC Guide: Setting up a Study). a Study Calendar in Epic a. CTRC i. Completes the Excel template for the study calendar. 1. Note - the IRB and industrial account number must be included in order to build the study calendar in Epic. ii. the completed form to the Epic research team at EpicResearchTeam@ohsu.edu iii. Please allow up to 10 business days for completion. b. Epic Research Team i. Creates the Study Calendar in Epic and links it to the RSH Record ii. Replies to CTRC to notify when the protocol is complete and available for use If study involves medications: 4. Research Pharmacy a. Completes Epic Catalog Request form within 10 business days of receipt of completed CTRC Medication Template.
3 b. Submits Epic Catalog Request form and order form documents to Pharmacy Informatics via Epic Help Desk 5. Informatics Pharmacist a. Configures build in Epic test environment b. Brings to CTRC Orders Committee for review (1 st and 3 rd Friday each month) a. The Study Coordinator will be asked to attend and will receive an invitation to this meeting b. Build is modified if applicable c. Release CTRC Therapy Plan within Epic Production and complete study specific order forms d. Invdrug@ohsu.edu, as well as the Study Coordinator, that the Therapy Plan is built and in Epic with a link to the final study specific orders. a. Note: Study Coordinator still needs to follow through on the rest of the steps prior to the patient being seen. 6. Study Staff obtains IRB approval and receives Alias from SPA (Sponsored Project Administration) electronically through ecris, if applicable. Provides alias and FAID to OCTRI Navigator. 7. Study Staff receives a Study Account electronically through ecris. Study account is created so we can link the patient to a study. There are two account types: a. Industrial account if charges are filed through Epic b. Zero charge account (or dummy account) if there no billable services through Epic.
4 NOTE: As of July 2013, all studies start in ecris which will automatically process the correct account. b. If study was not started in ecris and there is no research account assigned, Study Staff must request an Industrial Account from Tiffany Thomson in PBS or a Zero Charge Account from CRBO. If you are unsure which account to request, you can contact the CRBO. 8. HB & CRBO creates the RSH record in Epic 9. CTRC & Study Staff After OCTRI approval is received by CTRC and study staff (including study calendar build and Epic therapy plan build, if applicable), the study setup process for visits to CTRC may begin. Go to this link for details: Study Staff starts identifying patients to participate on the study 11. Study Staff - before calling CTRC to schedule the patient for their first visit in CTRC: a. Warm transfer the patient to OHSU Registration to get an MRN and current registration. Note: This is a new process for those subjects being seen at CTRC; you will no longer be faxing an MRN Request form to HIM for these subjects. b. If the study is not in ecris, request the CRBO to associate the patient to the Study in Epic by sending a study status change form to the CRBO. Epic will be updated with a nightly batch file with the research association. a. DY_STATUS_CHANGE_FORM.docx
5 c. If patient hasn t yet signed an informed consent form, the Study Staff should request that a Pre-Consent status is applied to the patient in Epic by the CRBO (note: this links the patient to the study in a pre-active status) by completing a study status change form (see link above) and sending it to CRBO. a. NOTE: The study status change form must be completed and sent to CRBO in this instance regardless of if the study is in ecris. 12. CTRC a. Scheduling is done thru the OCTRI Scheduling address: octrisch@ohsu.edu b. Adds the timeline to the patient the first time before scheduling c. Schedules the patient and links the patient to the Study Timeline If medications are part of the visit: 13. Study Staff a. Ideally, if possible per the design protocol, before the first medication visit, but at a minimum prior to ordering medication (see below), Study Staff will obtain signed Informed Consent from the subject. Have signed Informed Consent form scanned into Epic i. b. Make sure the diagnosis v70.7 (Examination of participant in clinical trial) is listed on the patient s Problem List. If not, have someone associated with the protocol (Principal/co- Investigator, or other LIP listed on the study add the diagnosis to the patient record prior to faxing the medication orders. They should also add any study medications to the patient s medication list.
6 Note: Important - Pharmacy cannot apply the Therapy Plan or release the medications until this diagnosis is on the patient s Problem List. 14. Principle Investigator or LIP listed on the study will then order study medications using the final study specific CTRC medication orders, including medications approved to be continued from home on that patient and then fax the patient med orders to Research Pharmacy, if possible per the design of the protocol at least 2 business days prior to EACH visit at CTRC. a. If you end up having to fax the orders on the day of the patient medication visit, you ll also need to immediately call the CTRC at (Nurse Station) and ask them to tell the Infusion RN to release the orders for that patient, so they are ready when the patient arrives. b. Note: If the protocol design precludes advance ordering, please fax the medication orders as far in advance as possible. i. Note: some medications, due to preparation, may require longer than two days advanced notice. 15. Note: All investigational or EEG patients are to arrive on the HRC10D unit between the hours of 8am and 3pm to ensure medication order identification and verification can be completed. 16. Study Staff may pick up medications from the Research Pharmacy prior to the patient s visit at CTRC or make arrangements to have medication delivered to CTRC prior to treatment.
7 Attachment 1 CTRC will be using some new Epic functionality to track study appointments that take place in the CTRC. ONLY CTRC has access to this functionality at this time, but all users will see the changes. Any study which includes patient visits in the CTRC will now have an associated CTRC Protocol. All CTRC outpatient and inpatient visits will be linked not just to the research study but ALSO to a specific point in the CTRC Protocol. This CTRC Protocol and all encounters linked to it will now appear in the Research Studies activity alongside any other non-ctrc encounters that are linked to the study. BEFORE:
8 AFTER:
9
CLINICAL RESEARCH BILLING 101
 CLINICAL RESEARCH BILLING 101 HCCA Research Compliance Conference October 31, 2007 Ann G. Mathias, JD, MHSA Ann E. Mitch-Resignalo, RN, MNEd Prepared September 2007 Updated October 2007 UPMC l University
CLINICAL RESEARCH BILLING 101 HCCA Research Compliance Conference October 31, 2007 Ann G. Mathias, JD, MHSA Ann E. Mitch-Resignalo, RN, MNEd Prepared September 2007 Updated October 2007 UPMC l University
What makes a good research coordinator? 1/16/2015. The Role of the Clinical Research Coordinator. Clinical Research Coordinator
 The Role of the Clinical Research Coordinator Darlene Kitterman, MBA Director, Investigator Support & Integration Services January 22, 2015 Point person that keeps study together Local protocol expert
The Role of the Clinical Research Coordinator Darlene Kitterman, MBA Director, Investigator Support & Integration Services January 22, 2015 Point person that keeps study together Local protocol expert
Research Pharmacy Services
 Introduction and Overview Jennifer Petrolati, PharmD, BCPS Pharmacist January 15, 2014 Research Pharmacy Staff Jennifer Petrolati, PharmD, BCPS Pharmacist Jeanne Liming, PharmD, BCOP Stephanie Kory, PharmD,
Introduction and Overview Jennifer Petrolati, PharmD, BCPS Pharmacist January 15, 2014 Research Pharmacy Staff Jennifer Petrolati, PharmD, BCPS Pharmacist Jeanne Liming, PharmD, BCOP Stephanie Kory, PharmD,
Pamela Richtmyer, MGH Research Compliance
 Pamela Richtmyer, MGH Research Compliance September 27 - Session 1 Basic overview October 4 - Session 2 Tracking patient care charges Invoices to sponsor Insight Patient Care Corrections October 11 - Session
Pamela Richtmyer, MGH Research Compliance September 27 - Session 1 Basic overview October 4 - Session 2 Tracking patient care charges Invoices to sponsor Insight Patient Care Corrections October 11 - Session
Research Pharmacy Services
 Research Pharmacy Services Introduction and Overview Erin Corella, PharmD, BCPS, BCPS, Pharmacist May 25, 2016 Research Pharmacy Staff Pharmacists Jeanne Liming, PharmD, BCOP Amee Kelley, PharmD Sara Blefgen,
Research Pharmacy Services Introduction and Overview Erin Corella, PharmD, BCPS, BCPS, Pharmacist May 25, 2016 Research Pharmacy Staff Pharmacists Jeanne Liming, PharmD, BCOP Amee Kelley, PharmD Sara Blefgen,
Centralized Office of Research
 Centralized Office of Research The driving force for creating this model or type of clinical trials office (CTO) at JHS was noncompliance issues in billing. What we discovered was a general lack of education
Centralized Office of Research The driving force for creating this model or type of clinical trials office (CTO) at JHS was noncompliance issues in billing. What we discovered was a general lack of education
Maestro Care (Epic) and Clinical Research Updates
 Maestro Care (Epic) and Clinical Research Updates Denise Snyder Terry Ainsworth Cory Ennis Duke Office of Clinical Research May 08, 2013 Duke s Complex but Productive Research Environment Duke s 10 schools,
Maestro Care (Epic) and Clinical Research Updates Denise Snyder Terry Ainsworth Cory Ennis Duke Office of Clinical Research May 08, 2013 Duke s Complex but Productive Research Environment Duke s 10 schools,
Go! Guide: Medication Administration
 Go! Guide: Medication Administration Introduction Medication administration is one of the most important aspects of safe patient care. The EHR assists health care professionals with safety by providing
Go! Guide: Medication Administration Introduction Medication administration is one of the most important aspects of safe patient care. The EHR assists health care professionals with safety by providing
Smart Pump Interoperability: A Multi-System Safety Journey. February 23, 2018
 Smart Pump Interoperability: A Multi-System Safety Journey February 23, 2018 Jennifer Biltoft, PharmD, BCPS System Director, Clinical Pharmacy Services, SCL Health Deborah Bonnes, RN, MS Nursing Informatics
Smart Pump Interoperability: A Multi-System Safety Journey February 23, 2018 Jennifer Biltoft, PharmD, BCPS System Director, Clinical Pharmacy Services, SCL Health Deborah Bonnes, RN, MS Nursing Informatics
Yes. Phone encounter? Part of screening process. Consent and enroll patient. Apply Treatment Plan. Verify link to Timeline. Conduct patient assessment
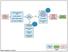 Referral - introduction to study Review consent and study Answer questions Review medical history Phone encounter? IRB approval Request required permission to schedule test (could change at screening)
Referral - introduction to study Review consent and study Answer questions Review medical history Phone encounter? IRB approval Request required permission to schedule test (could change at screening)
3 rd Annual Symposium for Research Administrators
 3 rd Annual Symposium for Research Administrators The UNC-CH Research IT Landscape An overview of the systems that support the research enterprise John Stephenson john@unc.edu July 29, 2016 Research Administration
3 rd Annual Symposium for Research Administrators The UNC-CH Research IT Landscape An overview of the systems that support the research enterprise John Stephenson john@unc.edu July 29, 2016 Research Administration
Effective Date: 11/09 Policy Chronicle:
 Title: Investigational Drug Service Functions Policy Type: Clinical Operations Replaces (supersedes): Title: N/A Policy Chronicle: Date Original Version of Policy was Effective: 09/06 Reviewer Signature:
Title: Investigational Drug Service Functions Policy Type: Clinical Operations Replaces (supersedes): Title: N/A Policy Chronicle: Date Original Version of Policy was Effective: 09/06 Reviewer Signature:
MMC-Einstein Research IT Forum
 MMC-Einstein Research IT Forum March 1, 2017 Harry Shamoon, Associate Dean for Clinical & Translational Research Barrett Katz, Executive Director, Office of Clinical Trials Vic Hatcher, Associate Dean
MMC-Einstein Research IT Forum March 1, 2017 Harry Shamoon, Associate Dean for Clinical & Translational Research Barrett Katz, Executive Director, Office of Clinical Trials Vic Hatcher, Associate Dean
a. It is very important to link a visit before or during the visit. This will drive billing functionality
 Epic FAQs 1. Adding patients to a list a. Only recommended for studies not in Epic i. Use the My Patients on Research Studies or Patients Associated with (Specify Study) Reports to find your patients on
Epic FAQs 1. Adding patients to a list a. Only recommended for studies not in Epic i. Use the My Patients on Research Studies or Patients Associated with (Specify Study) Reports to find your patients on
Introduction to the Parking Lot
 Introduction to the Parking Lot In ARK Epic training sessions, The Parking Lot" is used to capture all questions for which your trainer may not have an immediate answer during session. Your ARK Epic Training
Introduction to the Parking Lot In ARK Epic training sessions, The Parking Lot" is used to capture all questions for which your trainer may not have an immediate answer during session. Your ARK Epic Training
CTRC Lab 1. R (old EC area) Rose Zone Elevator A. 16 beds and 2 chair rooms. Vivian Dorsey RN,BSN, MBA,OCN Nurse Manager
 Clinical & Translational Research Center CTRC Overview Vivian Dorsey RN,BSN, MBA,OCN Nurse Manager 1 7/30/2009 New Location R1.1000 (old EC area) Rose Zone Elevator A 16 beds and 2 chair rooms 2 7/30/2009
Clinical & Translational Research Center CTRC Overview Vivian Dorsey RN,BSN, MBA,OCN Nurse Manager 1 7/30/2009 New Location R1.1000 (old EC area) Rose Zone Elevator A 16 beds and 2 chair rooms 2 7/30/2009
Bethesda Hospital PGY1 Residency Program Learning Experiences
 Bethesda Hospital PGY1 Residency Program Learning Experiences Required rotations Orientation This rotation will orient the resident to hospital pharmacy and the responsibilities of a staff pharmacist.
Bethesda Hospital PGY1 Residency Program Learning Experiences Required rotations Orientation This rotation will orient the resident to hospital pharmacy and the responsibilities of a staff pharmacist.
Transitioning OPAT (Outpatient Antibiotic Therapy) patients from the Acute Care Setting to the Ambulatory Setting
 Transitioning OPAT (Outpatient Antibiotic Therapy) patients from the Acute Care Setting to the Ambulatory Setting American College of Medical Practice Executives Case Study Submitted by Chantay Lucas,
Transitioning OPAT (Outpatient Antibiotic Therapy) patients from the Acute Care Setting to the Ambulatory Setting American College of Medical Practice Executives Case Study Submitted by Chantay Lucas,
Subject Screening, Recruitment, and Retention. Tiffany Morrison, MS, CCRP Director, Clinical Trials Rothman Institute
 Subject Screening, Recruitment, and Retention Tiffany Morrison, MS, CCRP Director, Clinical Trials Rothman Institute Learning objectives List two sources of potential subjects who can be screening for
Subject Screening, Recruitment, and Retention Tiffany Morrison, MS, CCRP Director, Clinical Trials Rothman Institute Learning objectives List two sources of potential subjects who can be screening for
Clinical Data acquisition and management. David A. Dorr, MD, MS Data after Dark 1/2016 OHSU
 Clinical Data acquisition and management David A. Dorr, MD, MS Data after Dark 1/2016 OHSU Clinical data: Four major points (revisited) Point Clinical data has potential to transform research across the
Clinical Data acquisition and management David A. Dorr, MD, MS Data after Dark 1/2016 OHSU Clinical data: Four major points (revisited) Point Clinical data has potential to transform research across the
Full application deadline Noon on April 4, Presentations to Scientific Review Committee (if invited) May 11, 2016
 Request for Applications: Pilot Project Funding for Catalyzing Translational Research Opportunities KEY DATES Full application deadline Noon on April 4, 2016 Presentations to Scientific Review Committee
Request for Applications: Pilot Project Funding for Catalyzing Translational Research Opportunities KEY DATES Full application deadline Noon on April 4, 2016 Presentations to Scientific Review Committee
Quanum Electronic Health Record Frequently Asked Questions
 Quanum Electronic Health Record Frequently Asked Questions Table of Contents... 4 What is Quanum EHR?... 4 What are the current capabilities of Quanum EHR?... 4 Is Quanum EHR an EMR?... 5 Can I have Quanum
Quanum Electronic Health Record Frequently Asked Questions Table of Contents... 4 What is Quanum EHR?... 4 What are the current capabilities of Quanum EHR?... 4 Is Quanum EHR an EMR?... 5 Can I have Quanum
HOME HEALTH AUTHORIZATION PROCESS FREQUENTLY ASKED QUESTIONS
 HOME HEALTH AUTHORIZATION PROCESS FREQUENTLY ASKED QUESTIONS HOME HEALTH AUTHORIZATION PROCESS PROVIDER QUESTIONS Recent updates are in blue italics: The most recent changes and additions to the frequently
HOME HEALTH AUTHORIZATION PROCESS FREQUENTLY ASKED QUESTIONS HOME HEALTH AUTHORIZATION PROCESS PROVIDER QUESTIONS Recent updates are in blue italics: The most recent changes and additions to the frequently
RESEARCH 101: THE KEYS TO GETTING APPROVAL & CONDUCTING RESEARCH AT GRADY. MARCH 22, 2018 Presented By: The Offices of Research & Grant Administration
 RESEARCH 101: THE KEYS TO GETTING APPROVAL & CONDUCTING RESEARCH AT GRADY MARCH 22, 2018 Presented By: The Offices of Research & Grant Administration LEARNING OBJECTIVES By the end of the session, you
RESEARCH 101: THE KEYS TO GETTING APPROVAL & CONDUCTING RESEARCH AT GRADY MARCH 22, 2018 Presented By: The Offices of Research & Grant Administration LEARNING OBJECTIVES By the end of the session, you
AIS Tip Sheet. Velos Patient Registration
 1 Refer to the Velos Epic Integration Velos Requirements for Epic Patient Interface document for more information. You must enter your Username in all lowercase letters, but your password as you would
1 Refer to the Velos Epic Integration Velos Requirements for Epic Patient Interface document for more information. You must enter your Username in all lowercase letters, but your password as you would
Mastering Clinical Research April 19, :30 am
 Mastering Clinical Research April 19, 2017 7:30 am New Question and Answer Response System Log In Directions Use the following link to access pre and post test questions: http://www.socrative.com/ Click
Mastering Clinical Research April 19, 2017 7:30 am New Question and Answer Response System Log In Directions Use the following link to access pre and post test questions: http://www.socrative.com/ Click
EPIC RESEARCH FAQs FOR MGH REVENUE CYCLE IMPLEMENTATION
 EPIC RESEARCH FAQs FOR MGH REVENUE CYCLE IMPLEMENTATION GENERAL 1. What does One Patient/One Record mean for researchers, study staff and patients who participate in research studies? 2. What records feed
EPIC RESEARCH FAQs FOR MGH REVENUE CYCLE IMPLEMENTATION GENERAL 1. What does One Patient/One Record mean for researchers, study staff and patients who participate in research studies? 2. What records feed
Feature Medication Adherence
 Version No: 3.0 Feature Medication Adherence CONTENTS CONTENTS... 2 AUDIENCE... 3 1. INTRODUCTION... 3 2. WORKFLOW... 3 3. MEDICATION MANAGEMENT FOR ADHERENCE (TYPES OF MESSAGES)... 4 3.1 Medication Adherence...
Version No: 3.0 Feature Medication Adherence CONTENTS CONTENTS... 2 AUDIENCE... 3 1. INTRODUCTION... 3 2. WORKFLOW... 3 3. MEDICATION MANAGEMENT FOR ADHERENCE (TYPES OF MESSAGES)... 4 3.1 Medication Adherence...
Care Everywhere Integrated Workgroup
 Care Everywhere Integrated Workgroup PAC 5/9/2017 Scope Care Everywhere Description of Group Care Everywhere is Epic s tool to allow you to exchange a patient s record electronically to other healthcare
Care Everywhere Integrated Workgroup PAC 5/9/2017 Scope Care Everywhere Description of Group Care Everywhere is Epic s tool to allow you to exchange a patient s record electronically to other healthcare
Improve Patient Care Utilizing Electronic Whiteboard
 Improve Patient Care Utilizing Electronic Whiteboard Presented By: Improve Patient Care Utilizing Electronic Whiteboard Christa Weghorn Performance Improvement Specialist IDEXX Laboratories Steve Safris,
Improve Patient Care Utilizing Electronic Whiteboard Presented By: Improve Patient Care Utilizing Electronic Whiteboard Christa Weghorn Performance Improvement Specialist IDEXX Laboratories Steve Safris,
Figure 1: Heat map showing zip codes and countries of residence for patients in STARR
 1 / 5 STARR Data Synopsis We operate STARR, a research data repository with 20 years of fully identified clinical data. STARR includes, but is not limited to, nightly clinical data, Epic Clarity, from
1 / 5 STARR Data Synopsis We operate STARR, a research data repository with 20 years of fully identified clinical data. STARR includes, but is not limited to, nightly clinical data, Epic Clarity, from
OBSERVATION CARE EVALUATION AND MANAGEMENT CODES POLICY
 OBSERVATION CARE EVALUATION AND MANAGEMENT CODES POLICY UnitedHealthcare Oxford Reimbursement Policy Policy Number: ADMINISTRATIVE 232.10 T0 Effective Date: March 1, 2017 Table of Contents Page INSTRUCTIONS
OBSERVATION CARE EVALUATION AND MANAGEMENT CODES POLICY UnitedHealthcare Oxford Reimbursement Policy Policy Number: ADMINISTRATIVE 232.10 T0 Effective Date: March 1, 2017 Table of Contents Page INSTRUCTIONS
I. Scope This policy defines unanticipated problems and adverse events and establishes the reporting process and timeline.
 Human Research Protection Program Policies & Procedures Unanticipated Problems and Adverse Events Version 3.0 Date Effective: 11.9.2012 Research Integrity Office Mail code L106-RI Portland, Oregon 97239-3098
Human Research Protection Program Policies & Procedures Unanticipated Problems and Adverse Events Version 3.0 Date Effective: 11.9.2012 Research Integrity Office Mail code L106-RI Portland, Oregon 97239-3098
CMS Oncology Care Model s Standards for Patient Navigation
 CMS Oncology Care Model s Standards for Patient Navigation Nikolas Buescher Executive Director of Cancer Services Penn Medicine, Lancaster November 13, 2017 Ann B Barshinger Health Cancer Institute scale
CMS Oncology Care Model s Standards for Patient Navigation Nikolas Buescher Executive Director of Cancer Services Penn Medicine, Lancaster November 13, 2017 Ann B Barshinger Health Cancer Institute scale
IRES Proposal Tracking (PT) Presented by: Kathi Goodfriend Office of Sponsored Projects Revised 03/15/2018 PRN: 5/14/ :19 PM
 IRES Proposal Tracking (PT) Presented by: Kathi Goodfriend Office of Sponsored Projects Revised 03/15/2018 PRN: 5/14/2018 12:19 PM Topics Covered Class Objective:. To become more familiar with the IRES
IRES Proposal Tracking (PT) Presented by: Kathi Goodfriend Office of Sponsored Projects Revised 03/15/2018 PRN: 5/14/2018 12:19 PM Topics Covered Class Objective:. To become more familiar with the IRES
MEDICARE CCLF ANALYTICS: MEDICARE ANALYTICS DATA ENGINE (MADE)
 MEDICARE CCLF ANALYTICS: MEDICARE ANALYTICS DATA ENGINE (MADE) Frequently Asked Questions 1.0 October 10, 2017 hmetrix hmetrix This document contains frequently asked questions regarding the utility, functionality,
MEDICARE CCLF ANALYTICS: MEDICARE ANALYTICS DATA ENGINE (MADE) Frequently Asked Questions 1.0 October 10, 2017 hmetrix hmetrix This document contains frequently asked questions regarding the utility, functionality,
Inpatient Anticoagulation Management Services to Improve Transitions of Care
 Inpatient Anticoagulation Management Services to Improve Transitions of Care Andrea Resseguie PharmD, RPh, CACP Advanced Practice Clinical Specialist Anticoagulation Management Service Learning Objectives
Inpatient Anticoagulation Management Services to Improve Transitions of Care Andrea Resseguie PharmD, RPh, CACP Advanced Practice Clinical Specialist Anticoagulation Management Service Learning Objectives
Instructions for Completing a Human Research Billing Analysis Form
 Instructions for Completing a Human Research Billing Analysis Form Principal Investigators are required to submit one Human Research Billing Analysis Form per research protocol at the time of the IRB submission
Instructions for Completing a Human Research Billing Analysis Form Principal Investigators are required to submit one Human Research Billing Analysis Form per research protocol at the time of the IRB submission
Frequently Asked Questions about the Physician Quality Reporting System (PQRS)
 Q. What is the reporting period for the 2016 PQRS Diabetes Module? A. The reporting period is January 1 December 31, 2016. Physicians who successfully collect data on 20 unique, separate and distinct patients
Q. What is the reporting period for the 2016 PQRS Diabetes Module? A. The reporting period is January 1 December 31, 2016. Physicians who successfully collect data on 20 unique, separate and distinct patients
A Guide to Working with the Investigational Drug Service for: Coordinators, Investigators, Data Managers and Business Administrators
 A Guide to Working with the Investigational Drug Service for: Coordinators, Investigators, Data Managers and Business Administrators Table of Contents Introduction... 2 Submitting a New Trial to IDS...
A Guide to Working with the Investigational Drug Service for: Coordinators, Investigators, Data Managers and Business Administrators Table of Contents Introduction... 2 Submitting a New Trial to IDS...
RADIATION ONCOLOGY RESIDENCY SUPERVISION POLICY
 RADIATION ONCOLOGY RESIDENCY SUPERVISION POLICY This policy is intended to guide the activities of radiation oncology residents in insuring that patient care activities in which residents participate are
RADIATION ONCOLOGY RESIDENCY SUPERVISION POLICY This policy is intended to guide the activities of radiation oncology residents in insuring that patient care activities in which residents participate are
Provider Frequently Asked Questions
 Provider Frequently Asked Questions Strengthening Clinical Processes Training CASE MANAGEMENT: Q1: Does Optum allow Case Managers to bill for services provided when the Member is not present? A1: Optum
Provider Frequently Asked Questions Strengthening Clinical Processes Training CASE MANAGEMENT: Q1: Does Optum allow Case Managers to bill for services provided when the Member is not present? A1: Optum
Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network of Clinical Research Professionals
 ORIENTATION FOR NEW CLINICAL RESEARCH COORDINATORS Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network of Clinical Research Professionals Overall Agenda for Orientation Module 1:
ORIENTATION FOR NEW CLINICAL RESEARCH COORDINATORS Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network of Clinical Research Professionals Overall Agenda for Orientation Module 1:
In-Patient Medication Order Entry System - contribution of pharmacy informatics
 In-Patient Medication Order Entry System - contribution of pharmacy informatics Ms S C Chiang BPharm, MRPS, MHA, FACHSE, FHKCHSE, FCPP Senior Pharmacist Chief Pharmacist s Office In-Patient Medication
In-Patient Medication Order Entry System - contribution of pharmacy informatics Ms S C Chiang BPharm, MRPS, MHA, FACHSE, FHKCHSE, FCPP Senior Pharmacist Chief Pharmacist s Office In-Patient Medication
Executive Director s Report
 Executive Director s Report Peter V. Lee Executive Director California Health Benefit Exchange Board Meeting December 18, 2012 Covered California Board Calendar 2013 Month / Date January 17 February 21
Executive Director s Report Peter V. Lee Executive Director California Health Benefit Exchange Board Meeting December 18, 2012 Covered California Board Calendar 2013 Month / Date January 17 February 21
INSPIRing Changes to the IRB Process: New templates and more
 INSPIRing Changes to the IRB Process: New templates and more John F. Ennever, MD, PhD, CIP Director, Human Research Protection Program Office of Human Research Affairs Boston Medical Center and Boston
INSPIRing Changes to the IRB Process: New templates and more John F. Ennever, MD, PhD, CIP Director, Human Research Protection Program Office of Human Research Affairs Boston Medical Center and Boston
Informatics and Technology (elective)
 Informatics Technology (elective) PGY2 - Health-System Pharmacy Administration (87405) Faculty: Link, Nicholas; Moore, Dallas Site: University of Utah Hospitals Clinics Status: Active Not Required Description:
Informatics Technology (elective) PGY2 - Health-System Pharmacy Administration (87405) Faculty: Link, Nicholas; Moore, Dallas Site: University of Utah Hospitals Clinics Status: Active Not Required Description:
Protocols for Migrant Health Promoters
 Protocols for Migrant Health Promoters As a Health Promoter your primary job is to promote health care access. This will be done in a variety of ways including the distribution of information about how
Protocols for Migrant Health Promoters As a Health Promoter your primary job is to promote health care access. This will be done in a variety of ways including the distribution of information about how
Clinical Trial Professional and Technical Fee Billing Procedures
 Clinical Trial Professional and Technical Fee Billing Procedures 1. Responsibilities: The principal investigator and his/her designees are responsible for: a. Accurately billing medical technical and professional
Clinical Trial Professional and Technical Fee Billing Procedures 1. Responsibilities: The principal investigator and his/her designees are responsible for: a. Accurately billing medical technical and professional
Bar Code Medication Administration and MAR Resource Manual
 Bar Code Medication Administration and MAR Resource Manual Administering Medications Administering Meds using CareMobile (PDA)... 2 Viewing Allergies in CareMobile... 8 Determining Which Meds to Give When...
Bar Code Medication Administration and MAR Resource Manual Administering Medications Administering Meds using CareMobile (PDA)... 2 Viewing Allergies in CareMobile... 8 Determining Which Meds to Give When...
Admission Medication History and Reconciliation Documentation. Froedtert Hospital, Milwaukee WI
 Overview of Medication History and Reconciliation Process 2 Overview of Icons Used in the Medication History 2 and Reconciliation Process The Admission Navigator 3 SureScripts Medication Reconciliation
Overview of Medication History and Reconciliation Process 2 Overview of Icons Used in the Medication History 2 and Reconciliation Process The Admission Navigator 3 SureScripts Medication Reconciliation
InfoEd Proposal Development Class
 InfoEd Proposal Development Class Last Updated: 4/17/18 Icahn School of Medicine at Mount Sinai InfoEd Class Goals 1. Broad Overview of Process Including Application Routing 2. Review of the Essential
InfoEd Proposal Development Class Last Updated: 4/17/18 Icahn School of Medicine at Mount Sinai InfoEd Class Goals 1. Broad Overview of Process Including Application Routing 2. Review of the Essential
UNIVERSITY MEDICAL CENTER NEW ORLEANS RESEARCH REVIEW APPLICATION
 UNIVERSITY MEDICAL CENTER NEW ORLEANS RESEARCH REVIEW APPLICATION Submission and Approval Process 1. Submit complete RRC application to Office of Research as early as possible in study process. 2. Quality
UNIVERSITY MEDICAL CENTER NEW ORLEANS RESEARCH REVIEW APPLICATION Submission and Approval Process 1. Submit complete RRC application to Office of Research as early as possible in study process. 2. Quality
MEDICARE CCLF ANALYTICS: MEDICARE ANALYTICS DATA ENGINE (MADE)
 MEDICARE CCLF ANALYTICS: MEDICARE ANALYTICS DATA ENGINE (MADE) Frequently Asked Questions 1.2 November 13, 2017 hmetrix hmetrix This document contains frequently asked questions regarding the utility,
MEDICARE CCLF ANALYTICS: MEDICARE ANALYTICS DATA ENGINE (MADE) Frequently Asked Questions 1.2 November 13, 2017 hmetrix hmetrix This document contains frequently asked questions regarding the utility,
Soarian Clinicals Results Viewing Quick User Guide
 Soarian Clinicals Results Viewing Quick User Guide Physicians, Medical Secretaries, Residents and Nurse Practitioners (For clinicians who provide care in one unit/location) December, 2008 Vs. c5 Table
Soarian Clinicals Results Viewing Quick User Guide Physicians, Medical Secretaries, Residents and Nurse Practitioners (For clinicians who provide care in one unit/location) December, 2008 Vs. c5 Table
Hospital EHR Medical Staff Newsletter
 Hospital EHR Medical Staff Newsletter November 2016, Volume 3, Issue 11 For questions, comments, or suggestions regarding the EHR or this newsletter, please contact me directly. Suzanne Wilkerson, MD_
Hospital EHR Medical Staff Newsletter November 2016, Volume 3, Issue 11 For questions, comments, or suggestions regarding the EHR or this newsletter, please contact me directly. Suzanne Wilkerson, MD_
TITLE: Processing Provider Orders: Inpatient and Outpatient
 POLICY and PROCEDURE TITLE: Processing Provider Orders: Inpatient and Outpatient Number: 13211 Version: 13211.10 Type: Patient Care Author: Carol Vanetti; Provider Order Policy Committee Effective Date:
POLICY and PROCEDURE TITLE: Processing Provider Orders: Inpatient and Outpatient Number: 13211 Version: 13211.10 Type: Patient Care Author: Carol Vanetti; Provider Order Policy Committee Effective Date:
Research & Development. Case Report Form SOP. J H Pacynko and J Illingworth. Research, pharmacy and R&D staff
 Department Title of SOP Research & Development Case Report Form SOP SOP reference no: R&D GCP SOP 03 Authors: Current version number and date: J H Pacynko and J Illingworth Version 2, 01.02.18 Next review
Department Title of SOP Research & Development Case Report Form SOP SOP reference no: R&D GCP SOP 03 Authors: Current version number and date: J H Pacynko and J Illingworth Version 2, 01.02.18 Next review
The presenter has owns Kelly Willenberg, LLC in relation to this educational activity.
 Kelly M Willenberg, MBA, BSN, CCRP, CHC, CHRC 1 The presenter has owns Kelly Willenberg, LLC in relation to this educational activity. 2 1 Medical Necessity when you submit claims Coding for qualifying
Kelly M Willenberg, MBA, BSN, CCRP, CHC, CHRC 1 The presenter has owns Kelly Willenberg, LLC in relation to this educational activity. 2 1 Medical Necessity when you submit claims Coding for qualifying
Standard Operating Procedures
 Clinical Monitoring and Site Verification Procedure Overview To define the standard procedures for preparation and documentation of site visits for clinical monitoring and spoke verification for any NETT
Clinical Monitoring and Site Verification Procedure Overview To define the standard procedures for preparation and documentation of site visits for clinical monitoring and spoke verification for any NETT
Congestive Heart Failure (CHF) Improvement
 Congestive Heart Failure (CHF) Improvement December 3, 2015 Beth Averbeck, MD Senior Medical Director, HPMG Primary Care HealthPartners Health Plan 1.5 million members Medical Clinics 1,700 physicians
Congestive Heart Failure (CHF) Improvement December 3, 2015 Beth Averbeck, MD Senior Medical Director, HPMG Primary Care HealthPartners Health Plan 1.5 million members Medical Clinics 1,700 physicians
Long Term Care (LTC) Claims Forwarding Webinar for Nursing Facility Users Frequently Asked Questions (FAQ)
 Long Term Care (LTC) Claims Forwarding Webinar for Nursing Facility Users Frequently Asked Questions (FAQ) 1. What assistance is available if providers have additional questions regarding claims billing
Long Term Care (LTC) Claims Forwarding Webinar for Nursing Facility Users Frequently Asked Questions (FAQ) 1. What assistance is available if providers have additional questions regarding claims billing
PATIENT ADMISSIONS 2.0
 PATIENT ADMISSIONS 2.0 HOSPITAL DIVISION FINANCIAL POLICIES and PROCEDURES MANUAL SECTION 2.0 PATIENT ADMISSIONS 2.4 Admissions General Information Patient Classification Inpatient An inpatient is a person
PATIENT ADMISSIONS 2.0 HOSPITAL DIVISION FINANCIAL POLICIES and PROCEDURES MANUAL SECTION 2.0 PATIENT ADMISSIONS 2.4 Admissions General Information Patient Classification Inpatient An inpatient is a person
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP)
 Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Research Proposal Application Process NUMBER: RAS SOP 1002 VERSION: 4.0 LAST REVISED: PREPARED BY: Office
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Research Proposal Application Process NUMBER: RAS SOP 1002 VERSION: 4.0 LAST REVISED: PREPARED BY: Office
How to Analyze and Implement a Research Protocol
 How to Analyze and Implement a Research Protocol Phyllis Carelllo, CCRC Bridget Adams, MSHS, CCRA What is a protocol? A detailed plan that sets forth the study: Objectives Design Methodology Why do we
How to Analyze and Implement a Research Protocol Phyllis Carelllo, CCRC Bridget Adams, MSHS, CCRA What is a protocol? A detailed plan that sets forth the study: Objectives Design Methodology Why do we
Oregon Clinical and Translational Research KL2 Program
 Request for Applications www.octri.org/octrikl2 2014 OCTRI KL2 RFA (updated 12/19/2013) Page 1 of 8 Oregon Clinical and Translational Research KL2 Program Letter of Intent deadline: January 13, 2014 Extended
Request for Applications www.octri.org/octrikl2 2014 OCTRI KL2 RFA (updated 12/19/2013) Page 1 of 8 Oregon Clinical and Translational Research KL2 Program Letter of Intent deadline: January 13, 2014 Extended
Oncology Quick Start Guide
 Oncology Quick Start Guide Oncology Provider - Beacon Version Date: 5/31/2017 Table of Contents Oncology Reports... 1 Treatment Plan Springboard... 1 Summary Phys Onc... 1 Summary Nurse Onc... 2 Scheduler
Oncology Quick Start Guide Oncology Provider - Beacon Version Date: 5/31/2017 Table of Contents Oncology Reports... 1 Treatment Plan Springboard... 1 Summary Phys Onc... 1 Summary Nurse Onc... 2 Scheduler
MA/Office Staff: Proposing Surgical Procedure Orders and PowerPlans (Order Sets)
 Acute Surgical Procedure Orders and PowerPlans Affiliated MA/Office Staff: Proposing Surgical Procedure Orders and PowerPlans (Order Sets) This document walks you through: 1. Requesting a FIN (Financial
Acute Surgical Procedure Orders and PowerPlans Affiliated MA/Office Staff: Proposing Surgical Procedure Orders and PowerPlans (Order Sets) This document walks you through: 1. Requesting a FIN (Financial
SUBMIT DISCLOSURE TO THE CFB 1. Go to Submit Disclosure. Click Submission and then Submit Disclosure.
 Help Content: CFB Submission This module explains how to view a draft of a disclosure statement, submit a final statement to the CFB, and amend your CFB filings. It also explains how to submit monetary
Help Content: CFB Submission This module explains how to view a draft of a disclosure statement, submit a final statement to the CFB, and amend your CFB filings. It also explains how to submit monetary
CAH SWING BED BILLING, CODING AND DOCUMENTATION. Lisa Pando, Sr. Consultant GPS Healthcare Consultants
 CAH SWING BED BILLING, CODING AND Lisa Pando, Sr. Consultant GPS Healthcare Consultants Learning Objectives: 1. Review Medical Necessity documentation specific to swing bed patients 2. Reasons to use the
CAH SWING BED BILLING, CODING AND Lisa Pando, Sr. Consultant GPS Healthcare Consultants Learning Objectives: 1. Review Medical Necessity documentation specific to swing bed patients 2. Reasons to use the
Side-by-Side Triage Activity Quick Start Guide
 Side-by-Side Triage Activity Quick Start Guide Physicians, RNs, APNs, Paramedics, Medical Students Triage Activity Tab: The Triage Activity Tab allows clinicians to review relevant triage information,
Side-by-Side Triage Activity Quick Start Guide Physicians, RNs, APNs, Paramedics, Medical Students Triage Activity Tab: The Triage Activity Tab allows clinicians to review relevant triage information,
Go! Guide: Patient Orders (Non-Medication)
 Go! Guide: Patient Orders (Non-Medication) Introduction The Orders tab in the EHR is where all members of the healthcare team find orders, or instructions, to care for, diagnose, and treat each patient.
Go! Guide: Patient Orders (Non-Medication) Introduction The Orders tab in the EHR is where all members of the healthcare team find orders, or instructions, to care for, diagnose, and treat each patient.
Chronic Care Management. Sharon A. Shover, CPC, CEMC 2650 Eastpoint Parkway, Suite 300 Louisville, Kentucky
 Chronic Care Management Sharon A. Shover, CPC, CEMC 2650 Eastpoint Parkway, Suite 300 Louisville, Kentucky 40223 502.992.3511 sshover@blueandco.com Agenda Chronic Care Management (CCM) History Define Requirements
Chronic Care Management Sharon A. Shover, CPC, CEMC 2650 Eastpoint Parkway, Suite 300 Louisville, Kentucky 40223 502.992.3511 sshover@blueandco.com Agenda Chronic Care Management (CCM) History Define Requirements
Who Has Been Doing Clinical Trials in my Hospital? Objectives
 Who Has Been Doing Clinical Trials in my Hospital? Research Compliance for the Community Hospital Kevin McPoyle, CPA April 24, 2007 2007 Compliance Institute Objectives Understand Clinical Trials and how
Who Has Been Doing Clinical Trials in my Hospital? Research Compliance for the Community Hospital Kevin McPoyle, CPA April 24, 2007 2007 Compliance Institute Objectives Understand Clinical Trials and how
Deriving Value from a Health Information Exchange. HIMSS17 DA-CH Community Conference Healthix I New York I February 20, 2017
 Deriving Value from a Health Information Exchange HIMSS17 DA-CH Community Conference Healthix I New York I February 20, 2017 About Healthix About Healthix Hundreds of healthcare organizations at more than
Deriving Value from a Health Information Exchange HIMSS17 DA-CH Community Conference Healthix I New York I February 20, 2017 About Healthix About Healthix Hundreds of healthcare organizations at more than
Office of Clinical Research Informatics
 Office of Clinical Research Informatics With Mary Samuels, Rachel Navarro, Adriel Gorsuch, James Dursch Tim Burdick MD MSc Chief Clinical Research Informatics Officer, OHSU Department of Medical Informatics
Office of Clinical Research Informatics With Mary Samuels, Rachel Navarro, Adriel Gorsuch, James Dursch Tim Burdick MD MSc Chief Clinical Research Informatics Officer, OHSU Department of Medical Informatics
Oregon Clinical and Translational Research KL2 Program
 Request for Applications www.octri.org/octrikl2 Oregon Clinical and Translational Research KL2 Program Letter of Intent deadline: April 3, 2017 The OCTRI Education Program is accepting letters of intent
Request for Applications www.octri.org/octrikl2 Oregon Clinical and Translational Research KL2 Program Letter of Intent deadline: April 3, 2017 The OCTRI Education Program is accepting letters of intent
Health Care Home Risk Stratification Tool User Guide
 Health Care Home Risk Stratification Tool User Guide Contents 1. Identifying Potential Health Care Home Patients. Page 3 2. Eligibility Notification.. Page 4 3. Patient Consent. Page 5 4. Completing the
Health Care Home Risk Stratification Tool User Guide Contents 1. Identifying Potential Health Care Home Patients. Page 3 2. Eligibility Notification.. Page 4 3. Patient Consent. Page 5 4. Completing the
Implementation guidance report Mental Health Inpatient Discharge Standard
 Implementation guidance report Mental Health Inpatient Discharge Standard 1 Introduction 1 2 Purpose 1 3 Guidance applicable to all standards 2 3.1 General guidance 2 3.2 Mandatory and optional 3 3.3 Coding
Implementation guidance report Mental Health Inpatient Discharge Standard 1 Introduction 1 2 Purpose 1 3 Guidance applicable to all standards 2 3.1 General guidance 2 3.2 Mandatory and optional 3 3.3 Coding
CMS OASIS Q&As: CATEGORY 2 - COMPREHENSIVE ASSESSMENT
 CMS OASIS Q&As: CATEGORY 2 - COMPREHENSIVE ASSESSMENT Q1. When are we required to collect OASIS? [Q&A EDITED 06/14] A1. The Condition of Participation (CoP) published in January 1999 requires a comprehensive
CMS OASIS Q&As: CATEGORY 2 - COMPREHENSIVE ASSESSMENT Q1. When are we required to collect OASIS? [Q&A EDITED 06/14] A1. The Condition of Participation (CoP) published in January 1999 requires a comprehensive
Bulletin. DHS Provides Policy for Certified Community Behavioral Health Clinics TOPIC PURPOSE CONTACT SIGNED TERMINOLOGY NOTICE NUMBER DATE
 Bulletin NUMBER 17-51-01 DATE February 27, 2017 OF INTEREST TO County Directors Social Services Supervisors and Staff Case Managers and Care Coordinators Managed Care Organizations Mental Health Providers
Bulletin NUMBER 17-51-01 DATE February 27, 2017 OF INTEREST TO County Directors Social Services Supervisors and Staff Case Managers and Care Coordinators Managed Care Organizations Mental Health Providers
Loyola University Chicago Health Sciences Division Maywood, IL. Human Subject Research Project Start-Up Guide
 Loyola University Chicago Health Sciences Division Maywood, IL Human Subject Research Project Start-Up Guide This Start-Up Guide is intended to guide you through the process of designing a research project
Loyola University Chicago Health Sciences Division Maywood, IL Human Subject Research Project Start-Up Guide This Start-Up Guide is intended to guide you through the process of designing a research project
University of Miami Clinical Enterprise Technologies
 Provider Manual 1 Our Mission: To design and deliver ongoing support for a network of Business and Clinical Information Management Systems which enhance the academic and research vision while implementing
Provider Manual 1 Our Mission: To design and deliver ongoing support for a network of Business and Clinical Information Management Systems which enhance the academic and research vision while implementing
ecw Integration PIX, XACML, CCD with Basic Clinical Event Notifications Project Scope Definition
 ecw Integration PIX, XACML, CCD with Basic Clinical Event otifications Project Scope Definition April 27, 2017 I. Key Contacts: Healthix Project Manager and Contact Information: Healthix Business Development
ecw Integration PIX, XACML, CCD with Basic Clinical Event otifications Project Scope Definition April 27, 2017 I. Key Contacts: Healthix Project Manager and Contact Information: Healthix Business Development
South East London Interface Prescribing Policy including the NHS and Private Interface Prescribing Guide
 South East London Interface Prescribing Policy including the NHS and Private Interface Prescribing Guide 1. Introduction 1.1 This policy has been developed by the South East London Clinical Commissioning
South East London Interface Prescribing Policy including the NHS and Private Interface Prescribing Guide 1. Introduction 1.1 This policy has been developed by the South East London Clinical Commissioning
This document provides information on conducting the Perindopril New To Therapy Program using GuildCare software.
 Perindopril New To Therapy Program PROTOCOL This document provides information on conducting the Perindopril New To Therapy Program using GuildCare software. April 2015 Table of Contents Executive Summary...
Perindopril New To Therapy Program PROTOCOL This document provides information on conducting the Perindopril New To Therapy Program using GuildCare software. April 2015 Table of Contents Executive Summary...
Illinois Medicaid EHR Incentive Program for EPs
 The Chicago HIT Regional Extension Center Bringing Chicago together through health IT < INSERT PICTURE > Illinois Medicaid EHR Incentive Program for EPs A Guide to Attesting for the 2016 Program Year in
The Chicago HIT Regional Extension Center Bringing Chicago together through health IT < INSERT PICTURE > Illinois Medicaid EHR Incentive Program for EPs A Guide to Attesting for the 2016 Program Year in
ChartMaker PatientPortal
 ChartMaker PatientPortal The ChartMaker PatientPortal is an online service that allows a patient to keep track of their personal health information. It will also allow practices to share and receive information
ChartMaker PatientPortal The ChartMaker PatientPortal is an online service that allows a patient to keep track of their personal health information. It will also allow practices to share and receive information
Appendix 1. Immediate Postpartum Long-Acting Reversible Contraception (LARC)
 Appendix 1. Immediate Postpartum Long-Acting Reversible Contraception (LARC) Program Implementation Guide: Exploration Stage Implementation Guide Overview Each stage of the implementation guide is organized
Appendix 1. Immediate Postpartum Long-Acting Reversible Contraception (LARC) Program Implementation Guide: Exploration Stage Implementation Guide Overview Each stage of the implementation guide is organized
Standard Operating Procedure. References Physician Guideline: Chronic Pain, Management of
 Subject Chronic Pain Management Index Number GL-6171 Section Patient Care Subsection Procedures/Treatments Category Corporate Contact Holly Boisen, Ext. 55970 References Physician Guideline: Chronic Pain,
Subject Chronic Pain Management Index Number GL-6171 Section Patient Care Subsection Procedures/Treatments Category Corporate Contact Holly Boisen, Ext. 55970 References Physician Guideline: Chronic Pain,
Frequently Asked Questions MN Prescription Monitoring Program (PMP)
 Frequently Asked Questions MN Prescription Monitoring Program (PMP) Topics: 1. Purpose of the PMP 2. Reporting/Frequency of Reporting 3. Dispensing for Animals 4. Inappropriate Prescribing 5. Accessing
Frequently Asked Questions MN Prescription Monitoring Program (PMP) Topics: 1. Purpose of the PMP 2. Reporting/Frequency of Reporting 3. Dispensing for Animals 4. Inappropriate Prescribing 5. Accessing
Frequently Asked Questions about Lung Cancer Screening and Medicare Coverage
 Frequently Asked Questions about Lung Cancer Screening and Medicare Coverage Eligibility, Coding and Reimbursement Can we start performing screening on Medicare beneficiaries immediately? Yes, but you
Frequently Asked Questions about Lung Cancer Screening and Medicare Coverage Eligibility, Coding and Reimbursement Can we start performing screening on Medicare beneficiaries immediately? Yes, but you
3/27/2017. Historical Perspective. Innovative Model of Healthcare Delivery Using Telemedicine
 Innovative Model of Healthcare Delivery Using Telemedicine Vinita Kamath MS RDN MHA Clinical Director, Nutrition Therapy Cincinnati Children s Hospital Medical Center CNM Conference March 20, 2017 Outline
Innovative Model of Healthcare Delivery Using Telemedicine Vinita Kamath MS RDN MHA Clinical Director, Nutrition Therapy Cincinnati Children s Hospital Medical Center CNM Conference March 20, 2017 Outline
2017 Edition. MIPS Guide. The rule is in and Medicare physician payments are changing. What does that mean for you?
 2017 Edition MIPS Guide The rule is in and Medicare physician payments are changing. What does that mean for you? MERIT-BASED INCENTIVE payment system The Merit-based Incentive Payment System (MIPS) combines
2017 Edition MIPS Guide The rule is in and Medicare physician payments are changing. What does that mean for you? MERIT-BASED INCENTIVE payment system The Merit-based Incentive Payment System (MIPS) combines
2012 CoC Standards: University of Kansas Hospital Cancer Committee Goals. Tim Metcalf, BS, CTR Cancer Registry Manager
 2012 CoC Standards: University of Kansas Hospital Cancer Committee Goals Tim Metcalf, BS, CTR Cancer Registry Manager 1 Standard 4:Outcomes 4.7 Quality Improvement: QI Coordinator develops, analyzes &
2012 CoC Standards: University of Kansas Hospital Cancer Committee Goals Tim Metcalf, BS, CTR Cancer Registry Manager 1 Standard 4:Outcomes 4.7 Quality Improvement: QI Coordinator develops, analyzes &
Administrators. Medical Directors. 61% The negative impact on our hospital-based program s. 44% We will need to consider the most appropriate or most
 2016 This annual survey, which began in 2009, provides key insight into nationwide developments in the business of cancer care. To better capture information from its multidisciplinary membership, this
2016 This annual survey, which began in 2009, provides key insight into nationwide developments in the business of cancer care. To better capture information from its multidisciplinary membership, this
Oregon s Safety Net Incorporating Value-based payment into system reform. Don Ross, Manager Program and Planning October 18, 2016
 Oregon s Safety Net Incorporating Value-based payment into system reform Don Ross, Manager Program and Planning October 18, 2016 Oregon chose a new way Better Health, Better Care and Lower Costs Transform
Oregon s Safety Net Incorporating Value-based payment into system reform Don Ross, Manager Program and Planning October 18, 2016 Oregon chose a new way Better Health, Better Care and Lower Costs Transform
Table 1: Limited Access Summary of Capabilities
 What is the Practice Fusion Limited Access EHR product? The Practice Fusion Limited Access EHR product will be provided to current Practice Fusion customers who have not purchased an EHR subscription plan
What is the Practice Fusion Limited Access EHR product? The Practice Fusion Limited Access EHR product will be provided to current Practice Fusion customers who have not purchased an EHR subscription plan
Brittany Turner, 2015 PharmD Candidate 1 Justin Campbell, PharmD 2 Katie McKinney, PharmD, MS, BCPS 2
 Discharge Medication Concierge Program: A pilot project in heart failure to reduce readmission rates, improve patient satisfaction, and increase pharmacy business metrics Brittany Turner, 2015 PharmD Candidate
Discharge Medication Concierge Program: A pilot project in heart failure to reduce readmission rates, improve patient satisfaction, and increase pharmacy business metrics Brittany Turner, 2015 PharmD Candidate
