The Impact of the Needlestick Safety and Prevention Act on Home Care and Hospice Organizations
|
|
|
- Amos Short
- 6 years ago
- Views:
Transcription
1 The Impact of the Needlestick Safety and Prevention Act on Home Care and Hospice Organizations Mary M. Friedman, MS, RN, CRNI Administrators, managers, and clinicians need to be up-to-date on all aspects of the new Needlestick Safety and Prevention Act recently passed into law. This article gives the background and foundation needed to implement policies, procedures, and actions that will assure compliance with the law and increased worker safety. The Needlestick Safety and Prevention Act (HR 5178), signed into law on November 6, 2000, was designed to protect the 8 million healthcare workers in the United States from injuries caused by needles and other sharp medical devices. This new federal law authorized the Occupational Safety and Health Administration (OSHA) to revise the bloodborne pathogens standard to require employers to identify, evaluate, and make use of effective, safer medical devices, and applies to any facilities where employees may be exposed to blood or other potentially infectious material, including home care and hospice organizations. Since publication of the final rule of the bloodborne pathogens standard in December, 1991 a home care or hospice employee s risk for contracting a bloodborne disease during the course of providing patient care has been significantly reduced. Yet, occupational exposure to bloodborne pathogens from accidental sharps injuries in all healthcare settings has continued to be a serious problem. The exact number of accidental sharps injuries sustained by home care or hospice employees is not known; however, current estimates indicate that 590,000 accidental sharps injuries are Mary M. Friedman, MS, RN, CRNI is a Joint Commission surveyor. Ms. Friedman may be reached at (770) ; fax: (770) ; mmf@mindspring.com sustained annually by healthcare workers in all healthcare settings (OSHA, 1999). This article reviews the historical events behind the legislation and discusses the modifications to the existing bloodborne pathogens standard and their implications for home care and hospice organizations. Out With the Old In December 1991, when the final rule for the bloodborne pathogens standard was published, little data on the efficacy of safer devices were available. In addition, the choice of safer devices was limited. The original bloodborne pathogen standards essentially stated that home care and hospice organizations should be evaluating safety devices, but left a great deal of flexibility in whether their use was adopted. Since then, there has been a substantial increase in the number and assortment of effective safety devices such as needleless systems and sharps with engineered sharps injury protections available to home care, hospice, and other healthcare organizations. Numerous studies have demonstrated that when safety medical devices are part of an overall bloodborne pathogens riskreduction program, there is a reduction in accidental sharps injuries. The Centers for Disease Control and Prevention has estimated that 62% to 88% of the sharps injuries could potentially be 356 Home Healthcare Nurse vol. 19 no. 6 June Lippincott Williams & Wilkins, Inc.
2 prevented by the use of safer medical devices, depending on the type of device used and the procedure involved (OSHA, 1999). OSHA s 1999 Compliance Directive In 1998, OSHA issued a Request for Information (RFI) on engineering and work practice controls used to eliminate or minimize the risk of occupational exposure to bloodborne pathogens due to percutaneous injuries from contaminated sharps. A total of 96 healthcare facilities, groups representing healthcare workers, researchers, educational institutions, professional and industry associations, and manufacturers of medical devices provided comments. In November, 1999, based on feedback from the RFI, OSHA issued a revised Compliance Directive (CPL 22.44D) for the Bloodborne Pathogens Standard, which clarified OSHA s position regarding the use of safety devices as a primary means of preventing bloodborne pathogen exposures. The directive provides guidance to OSHA compliance officers when they are inspecting facilities and enforcing the standard. Since that time, OSHA has cited healthcare facilities for failing to use safety devices. The use of sharps with engineered sharps injury protection is not new and actually is now required. OSHA currently has the authority under the bloodborne pathogens standard to require the use of engineering controls, such as safety devices, to reduce risk to workers. The federal Needlestick Safety and Prevention Act now gives OSHA a legislative mandate to require home care and hospice organizations to provide their employees with safety-engineered sharp devices. State-OSHA Plans Twenty-three states have state-osha plans in effect. The revised bloodborne pathogens standard was published in the Federal Register on January vol. 19 no. 6 June , State-OSHA plans will have to revise their corresponding standards within 6 months of that date. State-OSHA plans are required to have regulations that are at least as effective as those of federal OSHA. Some states with state-osha plans (i.e., California) have already revised their state Bloodborne Pathogens Standard to require the use of safer devices. States With Needle Safety Laws Seventeen states have passed needle safety laws that recognize the risks from occupational needlestick injuries; some mandate that the new technology available to prevent needlesticks be used by healthcare facilities. These states are listed in Table 1. If a state s needle safety law requirements exceed the federal law requirements, the home care or hospice organization must comply with the state s needle safety law requirements as well as the federal requirements. For example, in several states, the needle safety laws require that needlesticks be reported to a state agency. If a state s needle safety law has fewer requirements than the federal law, the federal law s requirements also must be followed. As a guiding principle, the most restrictive law (i.e., either state or federal) must always be followed in practice. In With the New When the legislators drafted the federal Needlestick Safety and Prevention Act, they incorporated much language and content of OSHA s November, 1999 compliance directive (CPL D) regarding requirements for the use of safety devices. As a result, the bill requires healthcare facilities under the federal OSHA to use safety-engineered devices as a primary engineering control to prevent occupational exposures to bloodborne pathogens in the work setting. Home Healthcare Nurse 357
3 Modifications to the Bloodborne Pathogen Standard The Needlestick Safety and Prevention Act required that the bloodborne pathogens standard be revised. The following is a summary of the standard s modifications, along with suggested actions to take to meet the new requirements: Modification to the Definitions The definition of engineering controls within the bloodborne pathogen standard now includes additional examples of controls of safer medical devices, such as sharps with engineered sharps injury protections and needleless systems (Needlestick Safety and Prevention Act of 2000, Pub. L. No , 114 Stat [Nov. 6, 2000]). The term Sharps with Engineered Sharps Injury Protections has been added to the definitions noted within the bloodborne pathogen standard and is defined as: A nonneedle sharp or a needle device used for withdrawing body fluids, accessing a vein or artery, TABLE 1 States With Needlestick Prevention Legislation Passed Alaska California Connecticut Georgia Iowa Maine Maryland Massachusetts Minnesota New Hampshire New Jersey New York Ohio Oklahoma Tennessee Texas West Virginia Pending District of Columbia Michigan Pennsylvania Rhode Island Source: International Health Care Worker Safety Center, EPINet, 11/02/00. Exposure Prevention Information Network. Online: A detailed description of an individual s state s law requirement can be obtained online at: or administering medications or other fluids, with a built-in safety feature or mechanism that effectively reduces the risk of an exposure incident (Needlestick Safety and Prevention Act of 2000, Pub. L. No , 114 Stat [Nov. 6, 2000]). The term Needleless Systems was added to the definitions noted with the bloodborne pathogen standard and defined as: A device that does not use needles for: the collection of bodily fluids or withdrawal of body fluids after initial venous or arterial access is established; the administration of medication or fluids; or any other procedure involving the potential for occupational exposure to bloodborne pathogens due to percutaneous injuries from contaminated sharps (Needlestick Safety and Prevention Act of 2000, Pub. L. No , 114 Stat (Nov. 6, 2000). 1. Review the home care or hospice organization s existing exposure control plan and modify the content to reflect the new/modified definitions. Modification of the Exposure Control Plan Exposure control plans are to be reviewed and updated to: reflect changes in technology that eliminate or reduce exposure to bloodborne pathogens, and document annually consideration and implementation of appropriate commercially available and effective safer medical devices designed to eliminate or minimize occupational exposure (Needlestick Safety and Prevention Act, 2000). 1. Document the revision to the exposure control plan to reflect the plan and timetable for evaluating, selecting, and implementing safety-engineered devices in all device categories with a potential for bloodborne pathogen exposure. 2. Record the procedure for evaluating and documenting the circumstances surrounding an exposure incident in the exposure control plan. 3. Implement the procedures for documenting and evaluating an exposure incident as identified in the exposure control plan. On an annual basis: 1. Document the review of the exposure control plan. 2. As necessary, revise the exposure control plan to reflect changes/updates in commercially 358 Home Healthcare Nurse vol. 19 no. 6 June 2001
4 available and effective engineering controls designed to eliminate or minimize exposure. Even if the home care or hospice organization is providing safety devices, be prepared to defend their effectiveness against the latest devices being introduced; an OSHA compliance officer may want to review that analysis. New Sharps Injury Log Requirement A home care or hospice organization is now mandated to establish and maintain a sharps injury log to record percutaneous injuries from contaminated sharps, as the previous OSHA 2000 log did not sufficiently reflect injuries that may involve exposure to bloodborne pathogens. The documentation maintained in the sharps injury log must be recorded and maintained in a way that protects the confidentiality of the injured employee. Minimally, the sharps injury log must contain: 1. the type and brand of device involved in the incident, 2. the department or work area where the exposure incident occurred, and 3. an explanation of how the incident occurred (Needlestick Safety and Prevention Act, 2000) 1. Continue to document on the OSHA 200 log. 2. Collect exposure data on sharp-object injuries and blood and body fluid exposures on separate forms that protect the confidentiality of the injured employee. 3. Document the type and brand of device involved in the incident, the department or work area (location) where the exposure incident occurred, and how the incident occurred, documentation should include the: route(s) of the exposure, engineering controls in use at the time, work practices followed, protective equipment or clothing that was used at the time of the exposure incident, procedure being performed when the incident occurred, employee s training, and comparison of similar occurrences and recommendations to avoid future exposures. 4. Aggregate the ongoing exposure incident data collected and analyze the data to determine if any adverse patterns or trends were noted. If an OSHA inspection occurs, copies of the home care and hospice organization s documentation on exposure incidents will be requested to determine if compliance has been met. Solicitation of Input From Nonmanagerial Employees A new section added to the bloodborne pathogen standard requires a home care or hospice organization to solicit input from nonmanagerial employees responsible for direct patient care who are potentially exposed to injuries from contaminated sharps in the identification, evaluation, and selection of effective engineering and work practice controls. The exposure control plan must document their participation (Needlestick Safety and Prevention Act, 2000). Establish a multidisciplinary team that includes clinical management and direct patient care providers potentially exposed to injuries from contaminated sharps to: 1. coordinate the activities of sharps injury prevention program to include the development or revision of a plan for selection, evaluation, and implementation of safety-engineer safety devices and needless systems; 2. gather information on the current use and availability of safety devices; 3. collect data and identify sharps devices in use with the greatest risk of exposure to bloodborne pathogens to be replaced; 4. identify, evaluate, and select safety-engineered sharps devices with protective features that retract, blunt, or otherwise shield the sharp point or edge after use and needless systems; 5. meet with product vendors to identify and choose safety devices to pilot test; 6. pilot-test safety devices for their effectiveness in patient care, reducing injuries, and ease of use; 7. select safety devices to replace targeted devices; 8. educate and train staff on the use of safety devices, needless systems, and safer work practices; 9. replace existing devices with safety devices as soon as possible after training; 10. develop strategies to ensure compliance with the use of safety devices and needless systems; 11. collect data and periodically evaluate the effectiveness of safety devices and needless systems in reducing the risk of injury from vol. 19 no. 6 June Home Healthcare Nurse 359
5 contaminated sharps and their impact on worker injury rates and patient safety; and 12. document the involvement of direct patient care providers in all activities including: evaluation, selection, and implementation of safety-engineered devices in all device categories. Exemptions The modifications to the bloodborne pathogens standard do not mandate that a home care or hospice organization implement the use of any engineering control, including a safer medical device, in any situation where it may jeopardize a patient s or employee s safety, or where it may be medically contraindicated. If a home health agency or hospice chooses not to use an available safety device or a device with a safety component, documentation must be maintained that explains why an available safety device(s) was not used and describes how it would have endangered the patient or employee. Many of the new safety devices are considerably more expensive; however, OSHA does not consider cost an acceptable reason for exemption. OSHA expects safety devices to be implemented if they reduce or eliminate occupational exposures to blood. Following the passage of the bloodborne pathogen standard in 1991, both the dental and home health industries sought exemption from the rule. The American Dental Association v. Martin decision granted a limited exemption to the home care industry (Rhinehart & Friedman, 1999). The court held that OSHA had not adequately considered feasibility problems for home care providers, where employees work at sites that the employer does not control. As a result, OSHA may not cite a home care and hospice organization for site-specific violations such as: housekeeping requirements (e.g., maintenance of a clean and sanitary work site), handling and disposal of regulated waste, ensuring the use of personal protective equipment, ensuring that specific work practices are followed (e.g., hands are washed with running water), and ensuring the use of engineering controls (e.g., safety devices). OSHA may cite a home care or hospice organization for failure to comply with all nonsite-specific requirements of the standard such as: supplying personal protective equipment for the employee s use in the home; providing safety devices to employees (e.g., sharps with engineered sharps injury protection, needleless devices, blunt needles, plastic capillary tubes), and training employees to use the devices properly; maintaining and updating the exposure control plan; offering hepatitis B vaccinations; providing postexposure evaluation and follow-up; maintaining a sharps injury log; and providing initial and ongoing employee education. To avoid an OSHA citation related to the use of engineering controls or safety devices, safety devices must be available for the employee s use in the home and the employee must have received prior trained in how to use the devices. The safety devices may be provided to the staff member on an individual basis and stored in the staff s supply bag taken into the home or the devices may be left in the home for the staff s use. Effective Date The Needlestick Safety and Prevention Act (HR 5178) was signed into law on November 6, The revised Bloodborne Pathogen standard was published in the Federal Register on January 18, The standard takes effect 90 days after publication. Thus, the effective date was April 18, REFERENCES International Health Care Worker Safety Center. (2000). EPINet, exposure prevention information network. Health Sciences Center, University of Virginia, Charlottesville, VA. Retrieved December 16, 2000 from the World Wide Web: Needlestick Safety and Prevention Act of (November 6, 2000). Pub. L. No , 114 Stat Occupational Safety and Health Administration (OSHA). (December 6, 1991). Occupational exposure to bloodborne pathogens; final rule, 29 CFR Part Federal Register, 56(235). Occupational Safety and Health Administration (OSHA). (November 5, 1999). Compliance directive. Enforcement procedures for the occupational exposure to bloodborne pathogens (CPL D). Retrieved December 16, 2000 from the World Wide Web: _2-2_44D.html Rhinehart, E., & Friedman, M. (1999). Effect of court decision on OSHA s Bloodborne Pathogen Regulations (Table 6-1, pp ). Infection Control in Home Care. Gaithersburg, MD: Aspen. 360 Home Healthcare Nurse vol. 19 no. 6 June 2001
6 Continuing Education Test To earn continuing education (CE) credit, follow these instructions: Home Healthcare Nurse CE also available online at 2.5 hours 1. Read the article on page 356. Complete sections A, B, and C* on the enrollment coupon. Each question has only one correct answer. 2. Send the coupon with your $18.75 registration fee to: Continuing Education Department, Lippincott Williams & Wilkins, 345 Hudson Street, New York, NY Within 6 weeks you ll be notified of your test results. A passing score for this test is 12 correct answers. If you pass, you will receive a certificate of completion. If you fail, you have the option of taking the test again at no additional cost. Lippincott Williams & Wilkins is accredited as a provider of continuing education in nursing by the American Nurses Credentialing Center s Commission on Accreditation and by the American Association of Critical Care Nurses (AACN 9722 Category A). Lippincott Williams & Wilkins is also an approved provider of nursing continuing education in Alabama, California, Florida, and Iowa and holds the following provider numbers: AL #ABNP0114; CA #CEP11749; FL #FBN2454; IA #75. All of its home study activities are classified for Texas nursing continuing education requirements as Type I. *In accordance with the Iowa Board of Nursing Administrative rules governing grievances, a copy of your evaluation of the CE offering (section C) may be submitted directly to the Iowa Board of Nursing. CE Test: Occupational Injuries and Needlestick Legislation General Purpose These articles describe measures to prevent occupational hazards. Specific safety devices and recommended procedures for phlebotomy and needle disposal are discussed. Objectives Read the following articles in this issue: The Impact of the Needlestick Safety and Prevention Act on Home Care and Hospice Organizations by Mary M. Friedman, MS, RN, CRNI, and Guide to Needlestick Prevention Devices by Dennis J. Ernst, MT(ASCP). After reading these articles and taking this test the learner will be able to: 1. Identify practices that increase one s risk for occupational injuries. 2. Discuss measures to reduce the incidence of needlesticks. 3. Describe the Occupational Safety and Health Administration s (OSHA) requirements and the Federal Needlestick Safety and Prevention Act. Questions 1. Which of these questions should be considered most important when evaluating safety devices for effectiveness? a. How long does it take to discard or permanently sheath the needle? b. Is the product user friendly? c. How much do these safety devices cost? d. Will the product enhance patient satisfaction? 2. When evaluating a safety device for phlebotomy you should give high priority to devices a. with two-hand activation. b. that will change usual procedures. c. with end-to-end protection. d. that activate with maximum manipulation. 3. The most effective safety disposal device for used needles a. allows the healthcare worker to remove the needle quickly. b. retracts the needle from the patient s arm into a protective holder. c. positions the needle upward after withdrawal from the patient. d. provides a puncture-proof cap to be placed over the used needle. 4. In the event that a sharps container is unavailable at your client s home, you should be prepared to a. immediately wash the used needle. b. conceal the tip with a thimble-size cover. c. twist the needle off with gloved hands. d. place the used needle in an envelope. 5. The contaminated sharps container that meets OSHA standards is one that a. can be easily opened and is reusable. b. is stored outside the patient s room. c. is puncture resistant and leak proof. d. has a needle clip and can be locked. 6. The window of vulnerability refers to the a. time between exposure and developing symptoms. b. time between needle removal from the patient and its discard. c. immune status of the exposed individual. d. virulence of the contaminated organism. 7. Which of these types of venipuncture collection tubes is safest? a. glass b. mercury based c. plastic d. laser 8. If a home care agency s state needle-safety requirements exceed the federal law requirements, it a. can choose which directives to follow. b. should develop its own standards. c. must comply with both the state and federal mandates. d. should adhere to the federal rules. 9. Which of these requirements was included in the Federal Needlestick Safety and Prevention Act? a. requirements for the use of safety devices b. funding for agency changes c. creation of a federal needlestick data base d. agency penalties for excessive injuries 10. Which of these changes meets OSHA s recommended engineering controls standards? a. use of needleless systems b. mandatory training programs c. increased staffing levels d. automatic vacuum systems 11. Which of these actions is required annually for a home care agency? a. evaluate all sharps-related staff injuries b. develop mandatory remedial programs c. recreate agency standards and policies d. document the review of the exposure control plan 12. Required information to be included in the sharps injury log includes a. the name of the injured employee. b. the brand of the device involved in the incident. c. the names of two witnesses to the incident. d. educational training provided after the incident. 13. A home care organization may be cited by OSHA for failure to comply with a. ensuring the use of personal protective equipment. b. ensuring that employees wash their hands correctly. c. proper disposal of contaminated waste in the home. d. offering hepatitis B vaccinations to employees. 14. Which of these practices by a healthcare worker increases the risk for an accidental needlestick? a. recapping used needles with two hands b. drawing blood before wearing gloves c. tearing off the tip of your glove to help find a vein d. removing the tourniquet with one hand after penetrating the vein 15. Which of these procedures is associated with the highest rate of accidental needlesticks? a. drawing blood without gloves b. using a syringe to draw blood c. applying a tourniquet to locate veins d. drawing arterial blood gases 16. Which of these instructions should a nurse educator include when teaching staff safety tips to prevent needlestick injuries? a. The needle should never leave your hand until it is discarded. b. Always completely fill up the sharps containers. c. Place contaminated needles on the bedside table, out of the patient s reach. d. Remove the needle from the syringe before discarding it. 17. What is the recommended time interval between removal of the needle from the patient s arm to permanent discard? a. 0 seconds to 5 seconds b. 5 seconds to 10 seconds c. 10 seconds to 20 seconds d. 20 seconds to 30 seconds A Lippincott Williams & Wilkins CE Home Study Enrollment Course Occupational Injuries and Needlestick Legislation CE Credit: 2.5 Contact Hours Fee: $18.75 Registration Deadline: June 30, 2003 Please check all that apply: LPN RN CNS NP CRNA CNM Other SS# Are you certified? Yes No Certified by Telephone # Last Name First Name MI Address City State Zip State of Licensure #1 License No. #1 State of Licensure #2 License No. #2 Position Title Area of Specialty C 1. Did this CE activity s learning objectives relate to its general purpose? Y N 2. Was the journal home study format an effective way to present the material? Y N 3. Was the content current to nursing practice? Y N 4. How long did it take you to complete this CE activity? hours 5. Suggestions for future topics B Test Responses: Darken one for your answer to each question. 1. a b c d 2. a b c d 3. a b c d 4. a b c d 5. a b c d 6. a b c d 7. a b c d 8. a b c d 9. a b c d 10. a b c d 11. a b c d 12. a b c d 13. a b c d 14. a b c d 15. a b c d 16. a b c d 17. a b c d vol. 19 no. 6 June Home Healthcare Nurse 361
OSHA s Revised Bloodborne Pathogens Standard. Outreach and Education Effort 2001
 OSHA s Revised Bloodborne Pathogens Standard Outreach and Education Effort 2001 Bloodborne Pathogens Standard 29 CFR 1910.1030, Occupational Exposure to Bloodborne Pathogens Published December 1991 Effective
OSHA s Revised Bloodborne Pathogens Standard Outreach and Education Effort 2001 Bloodborne Pathogens Standard 29 CFR 1910.1030, Occupational Exposure to Bloodborne Pathogens Published December 1991 Effective
Creating An Effective OSHA Compliance Program
 Presents Creating An Effective OSHA Compliance Program Bloodborne Pathogens and Your Course Faculty R. Thomas (Tom) Loughrey, MBA, CCS-P Chairman, CEO & Co-Founder of Economedix Certified Coding Specialist
Presents Creating An Effective OSHA Compliance Program Bloodborne Pathogens and Your Course Faculty R. Thomas (Tom) Loughrey, MBA, CCS-P Chairman, CEO & Co-Founder of Economedix Certified Coding Specialist
SALEM TOWNSHIP FIRE DEPARTMENT BLOODBORNE EXPOSURE CONTROL PLAN
 PURPOSE SALEM TOWNSHIP FIRE DEPARTMENT BLOODBORNE EXPOSURE CONTROL PLAN The Salem Township Fire Department (STFD) is committed to providing a safe and healthful work environment for our entire staff. The
PURPOSE SALEM TOWNSHIP FIRE DEPARTMENT BLOODBORNE EXPOSURE CONTROL PLAN The Salem Township Fire Department (STFD) is committed to providing a safe and healthful work environment for our entire staff. The
EXPOSURE CONTROL PLAN
 BLOODBORNE PATHOGEN EXPOSURE CONTROL PLAN SALT LAKE COMMUNITY COLLEGE October 2011 ~ 1 ~ POLICY Salt Lake Community College is committed to providing a safe and healthful work environment for our entire
BLOODBORNE PATHOGEN EXPOSURE CONTROL PLAN SALT LAKE COMMUNITY COLLEGE October 2011 ~ 1 ~ POLICY Salt Lake Community College is committed to providing a safe and healthful work environment for our entire
Department: Legal Department. Issued by: Quality Council. Approved by:
 HAWAII HEALTH SYSTEMS C O R P O R A T I O N Touching Lives Everyday" Policies and Procedures Department: Legal Department Issued by: Quality Council Policy No.: PAT 0009 Revision No.: 1 Effective Date:
HAWAII HEALTH SYSTEMS C O R P O R A T I O N Touching Lives Everyday" Policies and Procedures Department: Legal Department Issued by: Quality Council Policy No.: PAT 0009 Revision No.: 1 Effective Date:
Bloodborne Pathogens Cumru Township Fire Department 02/10/2011 Policy 10.5 Page: 1 of 7
 Policy 10.5 Page: 1 of 7 Purpose: The Cumru Township Fire Department is committed to providing a safe and healthful work environment for our entire staff, both career and volunteers. In pursuit of this
Policy 10.5 Page: 1 of 7 Purpose: The Cumru Township Fire Department is committed to providing a safe and healthful work environment for our entire staff, both career and volunteers. In pursuit of this
BLOODBORNE PATHOGENS
 BLOODBORNE PATHOGENS Supplement to Standard Training Module TRAINING REQUIREMENTS OVERVIEW This standard Vivid training module provides a general overview of Bloodborne Pathogens (BBP). It is important
BLOODBORNE PATHOGENS Supplement to Standard Training Module TRAINING REQUIREMENTS OVERVIEW This standard Vivid training module provides a general overview of Bloodborne Pathogens (BBP). It is important
BLOODBORNE PATHOGENS EXPOSURE CONTROL PLAN
 BLOODBORNE PATHOGENS EXPOSURE CONTROL PLAN School Name: Eastern Local School District Date of Preparation: August 2, 2000 (Revised August 22, 2002) In accordance with the PERRP Bloodborne Pathogens standard,
BLOODBORNE PATHOGENS EXPOSURE CONTROL PLAN School Name: Eastern Local School District Date of Preparation: August 2, 2000 (Revised August 22, 2002) In accordance with the PERRP Bloodborne Pathogens standard,
Bloodborne Pathogens. Goal. Objectives. Background
 Texas Department of Insurance Division of Workers Compensation Safety Education and Training Programs Bloodborne Pathogens Goal HS99-152C(2-05) Definitions This program provides information about the requirements
Texas Department of Insurance Division of Workers Compensation Safety Education and Training Programs Bloodborne Pathogens Goal HS99-152C(2-05) Definitions This program provides information about the requirements
Bloodborne Pathogens. Goal. Objectives. Definitions. Background
 Bloodborne Pathogens HS99-152D (03/09) Goal This program provides information about the requirements of the Occupational Health and Safety Administration (OSHA) Bloodborne Pathogens Standard, 29 Code of
Bloodborne Pathogens HS99-152D (03/09) Goal This program provides information about the requirements of the Occupational Health and Safety Administration (OSHA) Bloodborne Pathogens Standard, 29 Code of
COMPLYING WITH OSHA S BLOODBORNE PATHOGEN FINAL RULE OBJECTIVES
 Module B COMPLYING WITH OSHA S BLOODBORNE PATHOGEN FINAL RULE Almost there! OBJECTIVES Provide an overview of the Bloodborne Pathogen (BBP) Standard Highlight OSHA s requirements regarding bloodborne pathogens,
Module B COMPLYING WITH OSHA S BLOODBORNE PATHOGEN FINAL RULE Almost there! OBJECTIVES Provide an overview of the Bloodborne Pathogen (BBP) Standard Highlight OSHA s requirements regarding bloodborne pathogens,
Houston Controls, Inc Safety Management System
 Preparation: Safety Mgr Authority: Dennis Johnston Issuing Dept: Safety Page: Page 1 of 8 Purpose This Bloodborne Pathogen Exposure Control Plan has been established to ensure a safe and healthful working
Preparation: Safety Mgr Authority: Dennis Johnston Issuing Dept: Safety Page: Page 1 of 8 Purpose This Bloodborne Pathogen Exposure Control Plan has been established to ensure a safe and healthful working
ATTACHMENT B: TCSG Exposure Control Plan Model INTRODUCTION
 ATTACHMENT B: TCSG Exposure Control Plan Model 2016-2017 INTRODUCTION Oconee Fall Line Technical College Exposure Control Plan for Occupational Exposure to Bloodborne Pathogens and Airborne Pathogens/Tuberculosis
ATTACHMENT B: TCSG Exposure Control Plan Model 2016-2017 INTRODUCTION Oconee Fall Line Technical College Exposure Control Plan for Occupational Exposure to Bloodborne Pathogens and Airborne Pathogens/Tuberculosis
CAPE ELIZABETH SCHOOL DEPARTMENT Cape Elizabeth, Maine
 In accordance with OSHA Bloodborne Pathogens standards, 29 CFR 1910.1030, the following exposure control plan has been developed. 1. EXPOSURE DETERMINATION The purpose of this plan is to limit occupational
In accordance with OSHA Bloodborne Pathogens standards, 29 CFR 1910.1030, the following exposure control plan has been developed. 1. EXPOSURE DETERMINATION The purpose of this plan is to limit occupational
9/11/2013. Complying with OSHA s Bloodborne Pathogen Final Rule. OSHA and OSHA-NC. OSHA s Mandate. Module B Objectives
 Module B Objectives Complying with OSHA s Bloodborne Pathogen Final Rule Provide an overview of the Bloodborne Pathogen (BBP) Standard Highlight OHSA s requirements regarding bloodborne pathogens, including
Module B Objectives Complying with OSHA s Bloodborne Pathogen Final Rule Provide an overview of the Bloodborne Pathogen (BBP) Standard Highlight OHSA s requirements regarding bloodborne pathogens, including
Student Guide Preview. Bloodborne Pathogens. in the Workplace
 Student Guide Preview Bloodborne Pathogens in the Workplace Bloodborne Pathogens in the Workplace Student Guide Version 7.0 Purpose of this Guide This MEDIC First Aid Bloodborne Pathogens Version 7.0 Student
Student Guide Preview Bloodborne Pathogens in the Workplace Bloodborne Pathogens in the Workplace Student Guide Version 7.0 Purpose of this Guide This MEDIC First Aid Bloodborne Pathogens Version 7.0 Student
CORPORATE SAFETY MANUAL
 CORPORATE SAFETY MANUAL Procedure No. 27-0 Revision: Date: May 2005 Total Pages: 9 PURPOSE To make certain that our employees are duly aware of the hazards of blood exposure or other potentially infectious
CORPORATE SAFETY MANUAL Procedure No. 27-0 Revision: Date: May 2005 Total Pages: 9 PURPOSE To make certain that our employees are duly aware of the hazards of blood exposure or other potentially infectious
Shawnee State University
 Shawnee State University AREA: ACADEMIC AFFAIRS POLICY NO.: 5.21 ADMIN. CODE: 3362-5-22 PAGE NO.: 1 OF 13 EFFECTIVE DATE: 6 / 1 8 / 9 3 RECOMMENDED BY: A.L. Addington SUBJECT: BLOODBORNE PATHOGENS APPROVED
Shawnee State University AREA: ACADEMIC AFFAIRS POLICY NO.: 5.21 ADMIN. CODE: 3362-5-22 PAGE NO.: 1 OF 13 EFFECTIVE DATE: 6 / 1 8 / 9 3 RECOMMENDED BY: A.L. Addington SUBJECT: BLOODBORNE PATHOGENS APPROVED
BLOODBORNE PATHOGENS EXPOSURE PREVENTION POLICY AND PROCEDURE BLOODBORNE PATHOGENS EXPOSURE CONTROL PLAN
 BLOODBORNE PATHOGENS EXPOSURE PREVENTION POLICY AND PROCEDURE This sample plan is provided only as a guide to assist in complying with the OSHA Bloodborne Pathogens standard 29 CFR 1910.1030, as adopted
BLOODBORNE PATHOGENS EXPOSURE PREVENTION POLICY AND PROCEDURE This sample plan is provided only as a guide to assist in complying with the OSHA Bloodborne Pathogens standard 29 CFR 1910.1030, as adopted
MSAD 55. Blood Borne Pathogens Control Plan. 137 South Hiram Road Hiram, Maine (207)
 MSAD 55 Blood Borne Pathogens Control Plan 137 South Hiram Road Hiram, Maine 04041 www.sad55.org (207) 625-2490 MSAD 55 BLOOD BORNE PATHOGENS EXPOSURE CONTROL PLAN 1 PURPOSE In accordance with the OSHA
MSAD 55 Blood Borne Pathogens Control Plan 137 South Hiram Road Hiram, Maine 04041 www.sad55.org (207) 625-2490 MSAD 55 BLOOD BORNE PATHOGENS EXPOSURE CONTROL PLAN 1 PURPOSE In accordance with the OSHA
POLICY & PROCEDURES MEMORANDUM
 Policy No. *SF-1373.6 POLICY & PROCEDURES MEMORANDUM TITLE: BLOODBORNE PATHOGENS: EXPOSURE CONTROL PLAN (ECP) EFFECTIVE DATE: November 25, 2002* (*ORM Regulations Update 9/24/12; Title Updates 5/7/05)
Policy No. *SF-1373.6 POLICY & PROCEDURES MEMORANDUM TITLE: BLOODBORNE PATHOGENS: EXPOSURE CONTROL PLAN (ECP) EFFECTIVE DATE: November 25, 2002* (*ORM Regulations Update 9/24/12; Title Updates 5/7/05)
Infection Prevention Checklist Section I: Policies and Practices I.1 Administrative Measures
 Infection Prevention Checklist Section I: Policies and Practices I.1 Administrative Measures Facility name:... Completed by:... Date:... A. Written infection prevention policies and procedures specific
Infection Prevention Checklist Section I: Policies and Practices I.1 Administrative Measures Facility name:... Completed by:... Date:... A. Written infection prevention policies and procedures specific
BLOODBORNE PATHOGENS EXPOSURE CONTROL PLAN
 BLOODBORNE PATHOGENS EXPOSURE CONTROL PLAN School Name: MSAD #33 Date of Preparation: March 1993 In accordance with the OSHA Bloodborne Pathogens standard, 29 CFR 1910.1030, the following exposure control
BLOODBORNE PATHOGENS EXPOSURE CONTROL PLAN School Name: MSAD #33 Date of Preparation: March 1993 In accordance with the OSHA Bloodborne Pathogens standard, 29 CFR 1910.1030, the following exposure control
STUDENT BOOK PREVIEW STUDENT BOOK. Bloodborne Pathogens. in the Workplace
 STUDENT BOOK STUDENT BOOK PREVIEW Bloodborne Pathogens in the Workplace Bloodborne Pathogens In the Workplace Student Book Version 8.0 Purpose of this Guide This MEDIC First Aid Bloodborne Pathogens Version
STUDENT BOOK STUDENT BOOK PREVIEW Bloodborne Pathogens in the Workplace Bloodborne Pathogens In the Workplace Student Book Version 8.0 Purpose of this Guide This MEDIC First Aid Bloodborne Pathogens Version
Rice University Exposure Control Plan
 Rice University Exposure Control Plan Environmental Health and Safety MS 123 P.O. Box 1892 Houston, TX 77251-1892 713 348 4444 February 2015 1 Rice University Exposure Control Plan Rice University is committed
Rice University Exposure Control Plan Environmental Health and Safety MS 123 P.O. Box 1892 Houston, TX 77251-1892 713 348 4444 February 2015 1 Rice University Exposure Control Plan Rice University is committed
NEEDLE STICK SAFETY & BLOODBORNE PATHOGENS (BBP)
 NEEDLE STICK SAFETY & BLOODBORNE PATHOGENS (BBP) THIS MATERIAL WAS PRODUCED UNDER GRANT SH-29634-SH6 FROM OSHA, THE OCCUPATIONAL SAFETY AND HEALTH ADMINISTRATION, U.S. DEPARTMENT OF LABOR. IT DOES NOT
NEEDLE STICK SAFETY & BLOODBORNE PATHOGENS (BBP) THIS MATERIAL WAS PRODUCED UNDER GRANT SH-29634-SH6 FROM OSHA, THE OCCUPATIONAL SAFETY AND HEALTH ADMINISTRATION, U.S. DEPARTMENT OF LABOR. IT DOES NOT
The Safe Use of Sharps in Healthcare Guidance for managers and staff
 The Safe Use of Sharps in Healthcare Guidance for managers and staff This guide has been written to highlight the main requirements of the Health and Safety (Sharps Instruments in Healthcare) Regulations
The Safe Use of Sharps in Healthcare Guidance for managers and staff This guide has been written to highlight the main requirements of the Health and Safety (Sharps Instruments in Healthcare) Regulations
Appendix AX: B Occupational Exposure to Bloodborne Pathogens Exposure Control Plan
 Occupational Exposure to Bloodborne Pathogens Exposure Control Plan Employer: Nevada State Health Division Effective Date: May 5, 1992 Compliance Statement: In accordance with OSHA Bloodborne Pathogens
Occupational Exposure to Bloodborne Pathogens Exposure Control Plan Employer: Nevada State Health Division Effective Date: May 5, 1992 Compliance Statement: In accordance with OSHA Bloodborne Pathogens
Blood-borne Pathogen Exposure Control Plan
 Purpose Blood-borne Pathogen Exposure Control Plan 2010 The purpose of this plan is to minimize exposure of blood-borne pathogens to College Staff and Students, and to meet the requirements of the OSHA
Purpose Blood-borne Pathogen Exposure Control Plan 2010 The purpose of this plan is to minimize exposure of blood-borne pathogens to College Staff and Students, and to meet the requirements of the OSHA
Regulations that Govern the Disposal of Medical Waste
 Regulations that Govern the Disposal of Medical Waste In Louisiana, there are three (3) sources of regulations for medical wastes: OSHA, the Louisiana Department of Health and Hospitals, and the Louisiana
Regulations that Govern the Disposal of Medical Waste In Louisiana, there are three (3) sources of regulations for medical wastes: OSHA, the Louisiana Department of Health and Hospitals, and the Louisiana
Bloodborne Pathogens & Exposure Control Plan
 Bloodborne Pathogens & Exposure Control Plan Rev. 9/8/16 Page 1 of 8 Purpose: To ensure that Wayne County employees are aware and trained in bloodborne pathogens to eliminate and minimize employee exposure
Bloodborne Pathogens & Exposure Control Plan Rev. 9/8/16 Page 1 of 8 Purpose: To ensure that Wayne County employees are aware and trained in bloodborne pathogens to eliminate and minimize employee exposure
Bloodborne Pathogens Exposure Control Plan Dumas Independent School District
 Bloodborne Pathogens Exposure Control Plan Dumas Independent School District Part I: Purpose The purpose of this exposure control plan is to eliminate or minimize work-related exposure to bloodborne pathogens,
Bloodborne Pathogens Exposure Control Plan Dumas Independent School District Part I: Purpose The purpose of this exposure control plan is to eliminate or minimize work-related exposure to bloodborne pathogens,
EXPOSURE CONTROL PLAN
 OVERVIEW Revised, 2/14/12 OSHA EXPOSURE TO BLOODBORNE PATHOGENS 29 CFR 1910.1030 WESTERN NEW ENGLAND UNIVERSITY DEPARTMENT OF ATHLETICS EXPOSURE CONTROL PLAN The purpose of this Exposure Control Plan is
OVERVIEW Revised, 2/14/12 OSHA EXPOSURE TO BLOODBORNE PATHOGENS 29 CFR 1910.1030 WESTERN NEW ENGLAND UNIVERSITY DEPARTMENT OF ATHLETICS EXPOSURE CONTROL PLAN The purpose of this Exposure Control Plan is
We Have Your Back A Worker Safety Collaborative An Initiative of the Florida Hospital Association
 1 We Have Your Back A Worker Safety Collaborative An Initiative of the Florida Hospital Association WORKER SAFETY WEDNESDAY WEBINAR SERIES: SHARPS INJURY AND BLOOD EXPOSURE PREVENTION BUNDLE OVERVIEW THURSDAY,
1 We Have Your Back A Worker Safety Collaborative An Initiative of the Florida Hospital Association WORKER SAFETY WEDNESDAY WEBINAR SERIES: SHARPS INJURY AND BLOOD EXPOSURE PREVENTION BUNDLE OVERVIEW THURSDAY,
HEALTH AND SAFETY COMMITTEES Getting Started
 HEALTH AND SAFETY COMMITTEES Getting Started Many AFT locals have established health and safety committees. The primary goal of these committees is to assure that all members have a hazard-free, safe and
HEALTH AND SAFETY COMMITTEES Getting Started Many AFT locals have established health and safety committees. The primary goal of these committees is to assure that all members have a hazard-free, safe and
SEP Memorandum Report: "Trends in Nursing Home Deficiencies and Complaints," OEI
 DEPARTMENT OF HEALTH &. HUMAN SERVICES Office of Inspector General SEP 18 2008 Washington, D.C. 20201 TO: FROM: Kerry Weems Acting Administrator Centers for Medicare & Medicaid Services Daniel R. Levinson~
DEPARTMENT OF HEALTH &. HUMAN SERVICES Office of Inspector General SEP 18 2008 Washington, D.C. 20201 TO: FROM: Kerry Weems Acting Administrator Centers for Medicare & Medicaid Services Daniel R. Levinson~
SOCCCD. Bloodborne Pathogens Exposure Control Program
 SOCCCD Bloodborne Pathogens Exposure Control Program Office of Risk Management District Business Services Revised: 06/07/2016 Updated: 07/31/2017 SOUTH ORANGE COUNTY COMMUNITY COLLEGE DISTRICT BLOODBORNE
SOCCCD Bloodborne Pathogens Exposure Control Program Office of Risk Management District Business Services Revised: 06/07/2016 Updated: 07/31/2017 SOUTH ORANGE COUNTY COMMUNITY COLLEGE DISTRICT BLOODBORNE
Infection Prevention and Control: How to Meet the Conditions of Participation for Home Health
 Infection Prevention and Control: How to Meet the Conditions of Participation for Home Health Mary McGoldrick, MS, RN, CRNI Home Care and Hospice Consultant Saint Simons Island, GA Nothing to Disclose
Infection Prevention and Control: How to Meet the Conditions of Participation for Home Health Mary McGoldrick, MS, RN, CRNI Home Care and Hospice Consultant Saint Simons Island, GA Nothing to Disclose
Guidance for the Selection and Use of Personal Protective Equipment (PPE) in Healthcare Settings
 Guidance for the Selection and Use of Personal Protective Equipment (PPE) in Healthcare Settings : Program Goal Improve personnel safety in the healthcare environment through appropriate use of PPE. :
Guidance for the Selection and Use of Personal Protective Equipment (PPE) in Healthcare Settings : Program Goal Improve personnel safety in the healthcare environment through appropriate use of PPE. :
EAST CAROLINA UNIVERSITY INFECTION CONTROL POLICY
 EAST CAROLINA UNIVERSITY INFECTION CONTROL POLICY Office of Prospective Health Infection Control Plan Date Originated: August 26, 2003 Date Reviewed: 10/22/03; 9/04/07; 03/09/10; 9/01/15; Date Approved:
EAST CAROLINA UNIVERSITY INFECTION CONTROL POLICY Office of Prospective Health Infection Control Plan Date Originated: August 26, 2003 Date Reviewed: 10/22/03; 9/04/07; 03/09/10; 9/01/15; Date Approved:
EXPOSURE CONTROL PLAN BLOODBORNE PATHOGENS COUNTY OF INYO
 EXPOSURE CONTROL PLAN BLOODBORNE PATHOGENS COUNTY OF INYO Contacts: Supervising Nurse Anita Richardson (760) 873-4312 (760) 937-8567 Health Officer Dr. James Richardson (760) 873-7868 (760) 920-0433 Risk
EXPOSURE CONTROL PLAN BLOODBORNE PATHOGENS COUNTY OF INYO Contacts: Supervising Nurse Anita Richardson (760) 873-4312 (760) 937-8567 Health Officer Dr. James Richardson (760) 873-7868 (760) 920-0433 Risk
Bloodborne Pathogens Exposure Control Plan. Approved by The College at Brockport, Office of Environmental Health and Safety, February 2018
 Kinesiology, Sport Studies and Physical Education Athletic Training Program Bloodborne Pathogens Exposure Control Plan Approved by The College at Brockport, Office of Environmental Health and Safety, February
Kinesiology, Sport Studies and Physical Education Athletic Training Program Bloodborne Pathogens Exposure Control Plan Approved by The College at Brockport, Office of Environmental Health and Safety, February
Step 1A: Before entering patient room, be sure you have all the material ready and available:
 RECOMMENDATIONS FOR SAFELY COLLECTION AND PROPERLY MANAGEMENT OF POTENTIALLY INFECTED SAMPLES WITH HIGHLY PATHOGENIC AGENTS 1 (Adapted from How to safely collect blood samples from persons suspected to
RECOMMENDATIONS FOR SAFELY COLLECTION AND PROPERLY MANAGEMENT OF POTENTIALLY INFECTED SAMPLES WITH HIGHLY PATHOGENIC AGENTS 1 (Adapted from How to safely collect blood samples from persons suspected to
 3+ 3+ N = 155, 442 3+ R 2 =.32 < < < 3+ N = 149, 685 3+ R 2 =.27 < < < 3+ N = 99, 752 3+ R 2 =.4 < < < 3+ N = 98, 887 3+ R 2 =.6 < < < 3+ N = 52, 624 3+ R 2 =.28 < < < 3+ N = 36, 281 3+ R 2 =.5 < < < 7+
3+ 3+ N = 155, 442 3+ R 2 =.32 < < < 3+ N = 149, 685 3+ R 2 =.27 < < < 3+ N = 99, 752 3+ R 2 =.4 < < < 3+ N = 98, 887 3+ R 2 =.6 < < < 3+ N = 52, 624 3+ R 2 =.28 < < < 3+ N = 36, 281 3+ R 2 =.5 < < < 7+
BLOODBORNE PATHOGEN EXPOSURE CONTROL PLAN POLICY
 POLICY: BLOODBORNE PATHOGEN EXPOSURE CONTROL PLAN POLICY In accordance with the OSHA Bloodborne Pathogens Standard, 29 CFR 1910.1030, UMCHS will adhere to the agency s Bloodborne Pathogen Exposure Control
POLICY: BLOODBORNE PATHOGEN EXPOSURE CONTROL PLAN POLICY In accordance with the OSHA Bloodborne Pathogens Standard, 29 CFR 1910.1030, UMCHS will adhere to the agency s Bloodborne Pathogen Exposure Control
International Trends in Sharpes Injury Prevention Dr. Terry Grimmond, Grimmond and Associates, New Zeland A Webber Training Teleclass
 International Trends in Sharps Injury Prevention Terry Grimmond FASM, GrDpAdEd, BAgrSc tg@gandassoc.com Disclosures Grimmond and Associates are consultants to The Daniels Corporation, a producer of reusable
International Trends in Sharps Injury Prevention Terry Grimmond FASM, GrDpAdEd, BAgrSc tg@gandassoc.com Disclosures Grimmond and Associates are consultants to The Daniels Corporation, a producer of reusable
The University at Albany s Exposure Control Plan for Bloodborne Pathogens
 The University at Albany s Exposure Control Plan for Bloodborne Pathogens Effective Date: 10/92 Office of Environmental Health and Safety Latest Revision: October 2011 Chemistry B 73/ 442 3495 Section
The University at Albany s Exposure Control Plan for Bloodborne Pathogens Effective Date: 10/92 Office of Environmental Health and Safety Latest Revision: October 2011 Chemistry B 73/ 442 3495 Section
Bloodborne Pathogen Exposure Control Plan
 Bloodborne Pathogen Exposure Control Plan September 19, 2017 1 2 Table of Contents Review/Revision Summary... 5 Introduction... 6 Purpose... 6 General Program Structure... 6 Personnel... 6 Accessibility
Bloodborne Pathogen Exposure Control Plan September 19, 2017 1 2 Table of Contents Review/Revision Summary... 5 Introduction... 6 Purpose... 6 General Program Structure... 6 Personnel... 6 Accessibility
The American Legion NATIONAL MEMBERSHIP RECORD
 The American Legion NATIONAL MEMBERSHIP RECORD www.legion.org 2016 The American Legion NATIONAL MEMBERSHIP RECORD 1920-1929 Department 1920 1921 1922 1923 1924 1925 1926 1927 1928 1929 Alabama 4,474 3,246
The American Legion NATIONAL MEMBERSHIP RECORD www.legion.org 2016 The American Legion NATIONAL MEMBERSHIP RECORD 1920-1929 Department 1920 1921 1922 1923 1924 1925 1926 1927 1928 1929 Alabama 4,474 3,246
Sharps Injury Prevention in the Intraoperative Setting
 Sharps Injury Prevention in the Intraoperative Setting Describe recommended safe practices for cleaning instrumentation. Objectives Describe methods to reduce sharps injury to the health care team. Describe
Sharps Injury Prevention in the Intraoperative Setting Describe recommended safe practices for cleaning instrumentation. Objectives Describe methods to reduce sharps injury to the health care team. Describe
Current Medicare Advantage Enrollment Penetration: State and County-Level Tabulations
 Current Advantage Enrollment : State and County-Level Tabulations 5 Slide Series, Volume 40 September 2016 Summary of Tabulations and Findings As of September 2016, 17.9 million of the nation s 56.1 million
Current Advantage Enrollment : State and County-Level Tabulations 5 Slide Series, Volume 40 September 2016 Summary of Tabulations and Findings As of September 2016, 17.9 million of the nation s 56.1 million
TABLE 3c: Congressional Districts with Number and Percent of Hispanics* Living in Hard-to-Count (HTC) Census Tracts**
 living Alaska 00 47,808 21,213 44.4 Alabama 01 20,661 3,288 15.9 Alabama 02 23,949 6,614 27.6 Alabama 03 20,225 3,247 16.1 Alabama 04 41,412 7,933 19.2 Alabama 05 34,388 11,863 34.5 Alabama 06 34,849 4,074
living Alaska 00 47,808 21,213 44.4 Alabama 01 20,661 3,288 15.9 Alabama 02 23,949 6,614 27.6 Alabama 03 20,225 3,247 16.1 Alabama 04 41,412 7,933 19.2 Alabama 05 34,388 11,863 34.5 Alabama 06 34,849 4,074
TABLE 3b: Congressional Districts Ranked by Percent of Hispanics* Living in Hard-to- Count (HTC) Census Tracts**
 Rank State District Count (HTC) 1 New York 05 150,499 141,567 94.1 2 New York 08 133,453 109,629 82.1 3 Massachusetts 07 158,518 120,827 76.2 4 Michigan 13 47,921 36,145 75.4 5 Illinois 04 508,677 379,527
Rank State District Count (HTC) 1 New York 05 150,499 141,567 94.1 2 New York 08 133,453 109,629 82.1 3 Massachusetts 07 158,518 120,827 76.2 4 Michigan 13 47,921 36,145 75.4 5 Illinois 04 508,677 379,527
HOME HEALTH AIDE TRAINING REQUIREMENTS, DECEMBER 2016
 BACKGROUND HOME HEALTH AIDE TRAINING REQUIREMENTS, DECEMBER 2016 Federal legislation (42 CFR 484.36) requires that Medicare-certified home health agencies employ home health aides who are trained and evaluated
BACKGROUND HOME HEALTH AIDE TRAINING REQUIREMENTS, DECEMBER 2016 Federal legislation (42 CFR 484.36) requires that Medicare-certified home health agencies employ home health aides who are trained and evaluated
Section 29 Brieser Construction SH&E Manual
 Brieser Construction SH&E Manual May 30 2008 Company will ensure that all potentially infectious hazards within our facility(s) are evaluated and controlled. This standard practice instruction is intended
Brieser Construction SH&E Manual May 30 2008 Company will ensure that all potentially infectious hazards within our facility(s) are evaluated and controlled. This standard practice instruction is intended
BLOODBORNE PATHOGENS EXPOSURE CONTROL PLAN February 2018
 A. SCOPE AND APPLICATION BLOODBORNE PATHOGENS EXPOSURE CONTROL PLAN February 2018 The Bloodborne Pathogens (BBP) Exposure Control Plan is established in accordance with 29 CFR 1910.1030, Bloodborne Pathogens,
A. SCOPE AND APPLICATION BLOODBORNE PATHOGENS EXPOSURE CONTROL PLAN February 2018 The Bloodborne Pathogens (BBP) Exposure Control Plan is established in accordance with 29 CFR 1910.1030, Bloodborne Pathogens,
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by February 2018 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 Hawaii 2.1 19 Alabama 3.7 33 Ohio 4.5 2 New Hampshire 2.6 19 Missouri 3.7 33 Rhode Island 4.5
States Ranked by February 2018 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 Hawaii 2.1 19 Alabama 3.7 33 Ohio 4.5 2 New Hampshire 2.6 19 Missouri 3.7 33 Rhode Island 4.5
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by November 2015 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.7 19 Indiana 4.4 37 Georgia 5.6 2 Nebraska 2.9 20 Ohio 4.5 37 Tennessee 5.6
States Ranked by November 2015 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.7 19 Indiana 4.4 37 Georgia 5.6 2 Nebraska 2.9 20 Ohio 4.5 37 Tennessee 5.6
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by April 2017 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 Colorado 2.3 17 Virginia 3.8 37 California 4.8 2 Hawaii 2.7 20 Massachusetts 3.9 37 West Virginia
States Ranked by April 2017 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 Colorado 2.3 17 Virginia 3.8 37 California 4.8 2 Hawaii 2.7 20 Massachusetts 3.9 37 West Virginia
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by August 2017 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.3 18 Maryland 3.9 36 New York 4.8 2 Colorado 2.4 18 Michigan 3.9 38 Delaware 4.9
States Ranked by August 2017 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.3 18 Maryland 3.9 36 New York 4.8 2 Colorado 2.4 18 Michigan 3.9 38 Delaware 4.9
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by March 2016 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 South Dakota 2.5 19 Delaware 4.4 37 Georgia 5.5 2 New Hampshire 2.6 19 Massachusetts 4.4 37 North
States Ranked by March 2016 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 South Dakota 2.5 19 Delaware 4.4 37 Georgia 5.5 2 New Hampshire 2.6 19 Massachusetts 4.4 37 North
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by September 2017 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.4 17 Indiana 3.8 36 New Jersey 4.7 2 Colorado 2.5 17 Kansas 3.8 38 Pennsylvania
States Ranked by September 2017 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.4 17 Indiana 3.8 36 New Jersey 4.7 2 Colorado 2.5 17 Kansas 3.8 38 Pennsylvania
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by December 2017 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 Hawaii 2.0 16 South Dakota 3.5 37 Connecticut 4.6 2 New Hampshire 2.6 20 Arkansas 3.7 37 Delaware
States Ranked by December 2017 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 Hawaii 2.0 16 South Dakota 3.5 37 Connecticut 4.6 2 New Hampshire 2.6 20 Arkansas 3.7 37 Delaware
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by September 2015 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.8 17 Oklahoma 4.4 37 South Carolina 5.7 2 Nebraska 2.9 20 Indiana 4.5 37 Tennessee
States Ranked by September 2015 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.8 17 Oklahoma 4.4 37 South Carolina 5.7 2 Nebraska 2.9 20 Indiana 4.5 37 Tennessee
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by November 2014 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.7 19 Pennsylvania 5.1 35 New Mexico 6.4 2 Nebraska 3.1 20 Wisconsin 5.2 38 Connecticut
States Ranked by November 2014 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 North Dakota 2.7 19 Pennsylvania 5.1 35 New Mexico 6.4 2 Nebraska 3.1 20 Wisconsin 5.2 38 Connecticut
Unemployment Rate (%) Rank State. Unemployment
 States Ranked by July 2018 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 Hawaii 2.1 19 Massachusetts 3.6 37 Kentucky 4.3 2 Iowa 2.6 19 South Carolina 3.6 37 Maryland 4.3
States Ranked by July 2018 Unemployment Rate Seasonally Adjusted Unemployment Unemployment Unemployment 1 Hawaii 2.1 19 Massachusetts 3.6 37 Kentucky 4.3 2 Iowa 2.6 19 South Carolina 3.6 37 Maryland 4.3
SHARPS-RELATED INJURIES IN THE OR
 SHARPS-RELATED INJURIES IN THE OR Rose Moss, MN, RN, CNOR Perioperative Nurse Consultant/Medical Writer C & R Moss LLC Casa Grande, AZ Sharps-related injuries are a significant issue for health care workers
SHARPS-RELATED INJURIES IN THE OR Rose Moss, MN, RN, CNOR Perioperative Nurse Consultant/Medical Writer C & R Moss LLC Casa Grande, AZ Sharps-related injuries are a significant issue for health care workers
STATUTORY/REGULATORY NURSE ANESTHETIST RECOGNITION
 Alabama NPA and SBON R&R CRNAs are a type of advanced practice nurse. Advanced practice nurses are "certified by the Board of Nursing to engage in the practice of advanced practice nursing." [Alabama Nurse
Alabama NPA and SBON R&R CRNAs are a type of advanced practice nurse. Advanced practice nurses are "certified by the Board of Nursing to engage in the practice of advanced practice nursing." [Alabama Nurse
Manhattan Fire Protection District
 SOP #: 102-1 Effective Date: 04/02/11 Revised Date: 06/13/016 Section: Administraton Subject: Infection/Exposure Control PURPOSE: The purpose of this SOP is to establish an Infection Control Policy for
SOP #: 102-1 Effective Date: 04/02/11 Revised Date: 06/13/016 Section: Administraton Subject: Infection/Exposure Control PURPOSE: The purpose of this SOP is to establish an Infection Control Policy for
Bloodborne Pathogens Exposure Control Plan for Elwood C. C. School District #203
 Bloodborne Pathogens Exposure Control Plan for Elwood C. C. School District #203 Adopted by School Board on (date) The following person(s) is responsible for implementation and review of the Exposure Control
Bloodborne Pathogens Exposure Control Plan for Elwood C. C. School District #203 Adopted by School Board on (date) The following person(s) is responsible for implementation and review of the Exposure Control
EAST CAROLINA UNIVERSITY INFECTION CONTROL POLICY
 EAST CAROLINA UNIVERSITY INFECTION CONTROL POLICY Department: Neurology (Hemby Lane) Date Originated: 2/20/14 Date Reviewed: 6.5.18 Date Approved: 6/3/14 Page 1 of 7 Approved by: Department Chairman Administrator/Manager
EAST CAROLINA UNIVERSITY INFECTION CONTROL POLICY Department: Neurology (Hemby Lane) Date Originated: 2/20/14 Date Reviewed: 6.5.18 Date Approved: 6/3/14 Page 1 of 7 Approved by: Department Chairman Administrator/Manager
Bloodborne Pathogens Exposure Control Program Revised 1/3/2013
 Bloodborne Pathogens Exposure Control Program Revised 1/3/2013 REGULATORY AUTHORITY The California Code of Regulations (CCR), Title 8, Section 5193, requires employers to develop and implement an exposure
Bloodborne Pathogens Exposure Control Program Revised 1/3/2013 REGULATORY AUTHORITY The California Code of Regulations (CCR), Title 8, Section 5193, requires employers to develop and implement an exposure
Infection Control Policy and Procedure Manual. Post-Anesthesia Care Unit (Recovery Room) Page 1 of 6
 (Recovery Room) Page 1 of 6 Purpose: The purpose of this policy is to establish infection prevention guidelines to prevent or minimize transmission of infections in the. Policy: All personnel will adhere
(Recovery Room) Page 1 of 6 Purpose: The purpose of this policy is to establish infection prevention guidelines to prevent or minimize transmission of infections in the. Policy: All personnel will adhere
NORTHEAST TEXAS COMMUNITY COLLEGE Professional Education and Allied Health
 Phlebotomy Program APPLICATION INFORMATION The Phlebotomy Program at Northeast Texas Community College is a course series designed to prepare students to take the national certification test with the American
Phlebotomy Program APPLICATION INFORMATION The Phlebotomy Program at Northeast Texas Community College is a course series designed to prepare students to take the national certification test with the American
OSHA Inspections: Real Life Story
 OSHA Inspections: Real Life Story Stephanie Martin, BSN, RN, CNOR, CASC Administrator St. Augustine Surgery Center August 14, 2012, 6:00 AM August 14, 2012, 6:00 AM The day started like any other... Arriving
OSHA Inspections: Real Life Story Stephanie Martin, BSN, RN, CNOR, CASC Administrator St. Augustine Surgery Center August 14, 2012, 6:00 AM August 14, 2012, 6:00 AM The day started like any other... Arriving
Bloodborne Pathogen Exposure Control Plan
 AU Medical Center Policy Library Policy Owner: Epidemiology POLICY STATEMENT Augusta University Medical Center (AUMC) shall establish a written Exposure Control Plan (ECP) designed to eliminate or minimize
AU Medical Center Policy Library Policy Owner: Epidemiology POLICY STATEMENT Augusta University Medical Center (AUMC) shall establish a written Exposure Control Plan (ECP) designed to eliminate or minimize
Fundraising Registration Update 2013
 Fundraising Registration Update 2013 Marc Lee, CFRE, President, Affinity Fundraising Registration February 2013 Handout: www.fundraisingregistration.com/documents/registrationupdate2013.pdf Presenter I
Fundraising Registration Update 2013 Marc Lee, CFRE, President, Affinity Fundraising Registration February 2013 Handout: www.fundraisingregistration.com/documents/registrationupdate2013.pdf Presenter I
Chapter 4 - Employee First Aid, Medical and Emergency Procedures
 Chapter 4 Employee First Aid, Medical and Emergency Procedures Chapter 4 - Employee First Aid, Medical and Emergency Procedures Non-Occupational Illness or Injury Diagnosis and treatment of non-occupational
Chapter 4 Employee First Aid, Medical and Emergency Procedures Chapter 4 - Employee First Aid, Medical and Emergency Procedures Non-Occupational Illness or Injury Diagnosis and treatment of non-occupational
Routine Venipuncture Guidelines
 Department: Administration Procedure Name: Specimen Collection Policy Page: 1 of 5 Procedure Number: Adm. 020 Replaces Policy Dated: Effective Date: October 23, 2006 Retired: Routine Venipuncture Guidelines
Department: Administration Procedure Name: Specimen Collection Policy Page: 1 of 5 Procedure Number: Adm. 020 Replaces Policy Dated: Effective Date: October 23, 2006 Retired: Routine Venipuncture Guidelines
BLOODBORNE PATHOGENS EXPOSURE CONTROL PLAN
 SUNY College at Brockport BLOODBORNE PATHOGENS EXPOSURE CONTROL PLAN Revised: October, 2016 ENVIRONMENTAL HEALTH & SAFETY OFFICE (EHS) TABLE OF CONTENTS TABLE OF CONTENTS 2 PURPOSE OF THE PLAN 3 GENERAL
SUNY College at Brockport BLOODBORNE PATHOGENS EXPOSURE CONTROL PLAN Revised: October, 2016 ENVIRONMENTAL HEALTH & SAFETY OFFICE (EHS) TABLE OF CONTENTS TABLE OF CONTENTS 2 PURPOSE OF THE PLAN 3 GENERAL
EAST CAROLINA UNIVERSITY INFECTION CONTROL POLICY
 EAST CAROLINA UNIVERSITY INFECTION CONTROL POLICY Department: Family Practice Dental Clinic Date Originated: 05-31-2006 Date Reviewed: 06-21-2006 Date Approved: Page 1 of 7 Approved by: Department Chairman
EAST CAROLINA UNIVERSITY INFECTION CONTROL POLICY Department: Family Practice Dental Clinic Date Originated: 05-31-2006 Date Reviewed: 06-21-2006 Date Approved: Page 1 of 7 Approved by: Department Chairman
Administration OCCUPATIONAL HEALTH AND SAFETY
 ACCREDITATION STANDA RDS OCCUPATIONAL HEALTH AND SAFETY The accreditation standards relating to occupational health and safety include those most critical to staff safety in the non-hospital setting; however,
ACCREDITATION STANDA RDS OCCUPATIONAL HEALTH AND SAFETY The accreditation standards relating to occupational health and safety include those most critical to staff safety in the non-hospital setting; however,
NEW JERSEY ESRD REGULATORY UPDATE
 NEW JERSEY ESRD REGULATORY UPDATE New Jersey Department of Health Stefanie Mozgai, BA, RN, CPM, Director Anna Sousa, MS, RD, Supervising Healthcare Evaluator October 2014 REPORTABLE EVENTS New Jersey Department
NEW JERSEY ESRD REGULATORY UPDATE New Jersey Department of Health Stefanie Mozgai, BA, RN, CPM, Director Anna Sousa, MS, RD, Supervising Healthcare Evaluator October 2014 REPORTABLE EVENTS New Jersey Department
Safety Meeting. Meeting Leader Instructions. Safety, Teamwork & Our Customer s 1 st Choice
 Meeting Leader Instructions These meetings are more than safety meetings. While they address safety as a top priority, these meetings are also an opportunity for you to interact with your team; a chance
Meeting Leader Instructions These meetings are more than safety meetings. While they address safety as a top priority, these meetings are also an opportunity for you to interact with your team; a chance
Food Stamp Program State Options Report
 United States Department of Agriculture Food and Nutrition Service Fourth Edition Food Stamp Program State s Report September 2004 vember 2002 Program Development Division Program Design Branch Food Stamp
United States Department of Agriculture Food and Nutrition Service Fourth Edition Food Stamp Program State s Report September 2004 vember 2002 Program Development Division Program Design Branch Food Stamp
Student Guidelines for Preventing Occupational Exposure to Bloodborne Pathogens (BBP)
 University of Michigan-Flint School of Health Professions and Studies (SHPS) Student Guidelines for Preventing Occupational Exposure to Bloodborne Pathogens (BBP) Report all exposures immediately Refer
University of Michigan-Flint School of Health Professions and Studies (SHPS) Student Guidelines for Preventing Occupational Exposure to Bloodborne Pathogens (BBP) Report all exposures immediately Refer
Food Stamp Program State Options Report
 United States Department of Agriculture Food and Nutrition Service Fifth Edition Food Stamp Program State s Report August 2005 vember 2002 Program Development Division Food Stamp Program State s Report
United States Department of Agriculture Food and Nutrition Service Fifth Edition Food Stamp Program State s Report August 2005 vember 2002 Program Development Division Food Stamp Program State s Report
Operating Room Sharp Injuries in a Teaching Hospital. Poonam Kutre MPH 2015
 Operating Room Sharp Injuries in a Teaching Hospital Poonam Kutre MPH 2015 What is sharp injury A sharp injury is a penetrating stab wound from a needle, scalpel, or other sharp object that may result
Operating Room Sharp Injuries in a Teaching Hospital Poonam Kutre MPH 2015 What is sharp injury A sharp injury is a penetrating stab wound from a needle, scalpel, or other sharp object that may result
NORTHEAST TEXAS COMMUNITY COLLEGE Professional Education and Allied Health
 Phlebotomy Program APPLICATION INFORMATION The Phlebotomy Program at Northeast Texas Community College is a course series designed to prepare students to take the national certification test with the American
Phlebotomy Program APPLICATION INFORMATION The Phlebotomy Program at Northeast Texas Community College is a course series designed to prepare students to take the national certification test with the American
SHARPS POLICY & PROCEDURES
 Section: D Policy Number: D-018 Subject: Sharps Policy & Procedure Total Pages: 6 Approval Date: May 20, 2015. Revision Date(s) SHARPS POLICY & PROCEDURES Policy: Community Living-Central Huron is responsible
Section: D Policy Number: D-018 Subject: Sharps Policy & Procedure Total Pages: 6 Approval Date: May 20, 2015. Revision Date(s) SHARPS POLICY & PROCEDURES Policy: Community Living-Central Huron is responsible
MAP 1: Seriously Delinquent Rate by State for Q3, 2008
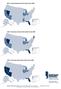 MAP 1: Seriously Delinquent Rate by State for Q3, 2008 Seriously Delinquent Rate Greater than 6.93% 5.18% 6.93% 0 5.17% Source: MBA s National Deliquency Survey MAP 2: Foreclosure Inventory Rate by State
MAP 1: Seriously Delinquent Rate by State for Q3, 2008 Seriously Delinquent Rate Greater than 6.93% 5.18% 6.93% 0 5.17% Source: MBA s National Deliquency Survey MAP 2: Foreclosure Inventory Rate by State
NEW GRADUATE PROVISIONS
 Alabama Yes A nurse anesthetist "may request Provisional Approval for practice as a graduate nurse anesthetist by submitting: (a) Official evidence of authorization to test from the Council on Certification
Alabama Yes A nurse anesthetist "may request Provisional Approval for practice as a graduate nurse anesthetist by submitting: (a) Official evidence of authorization to test from the Council on Certification
Employee First Aid, Medical and Emergency Procedures
 Chapter 4 - Employee First Aid, Medical and Emergency Procedures Chapter 4 Employee First Aid, Medical and Emergency Procedures Non-Occupational Illness or Injury Diagnosis and treatment of non-occupational
Chapter 4 - Employee First Aid, Medical and Emergency Procedures Chapter 4 Employee First Aid, Medical and Emergency Procedures Non-Occupational Illness or Injury Diagnosis and treatment of non-occupational
I MPROVING INFECTION CONTROL in HOME CARE:
 I MPROVING INFECTION CONTROL in HOME CARE: From Ritual to Science-Based Practice While many home care policies and procedures are developed with the best of intentions, they frequently lack a scientific
I MPROVING INFECTION CONTROL in HOME CARE: From Ritual to Science-Based Practice While many home care policies and procedures are developed with the best of intentions, they frequently lack a scientific
ADVANCED EDUCATION REQUIREMENTS
 EDUCATION REQUIREMENTS STATE Alabama Yes The applicant for approval to practice as a certified registered nurse anesthetist shall have... [e]arned at least a master's degree, or post-master's certificate
EDUCATION REQUIREMENTS STATE Alabama Yes The applicant for approval to practice as a certified registered nurse anesthetist shall have... [e]arned at least a master's degree, or post-master's certificate
Introduction. Current Law Distribution of Funds. MEMORANDUM May 8, Subject:
 MEMORANDUM May 8, 2018 Subject: TANF Family Assistance Grant Allocations Under the Ways and Means Committee (Majority) Proposal From: Gene Falk, Specialist in Social Policy, gfalk@crs.loc.gov, 7-7344 Jameson
MEMORANDUM May 8, 2018 Subject: TANF Family Assistance Grant Allocations Under the Ways and Means Committee (Majority) Proposal From: Gene Falk, Specialist in Social Policy, gfalk@crs.loc.gov, 7-7344 Jameson
Bloodborne Pathogens Exposure Control Plan. Northern Illinois University
 Bloodborne Pathogens Exposure Control Plan Northern Illinois University Department of Environmental Health and Safety Updated 7/24/2013 Review and Updates Date Reviewed by Changes Made 2 Contents Introduction...
Bloodborne Pathogens Exposure Control Plan Northern Illinois University Department of Environmental Health and Safety Updated 7/24/2013 Review and Updates Date Reviewed by Changes Made 2 Contents Introduction...
Bloodborne Pathogens
 1.0 REGULATORY AUTHORITY 2.0 BACKGROUND 3.0 POLICY 4.0 PURPOSE 5.0 SCOPE 6.0 DEFINITIONS Bloodborne Pathogens The Code of Federal Regulations, 29CFR 1910.1030, December, 1991 and the California Code of
1.0 REGULATORY AUTHORITY 2.0 BACKGROUND 3.0 POLICY 4.0 PURPOSE 5.0 SCOPE 6.0 DEFINITIONS Bloodborne Pathogens The Code of Federal Regulations, 29CFR 1910.1030, December, 1991 and the California Code of
Interstate Pay Differential
 Interstate Pay Differential APPENDIX IV Adjustments for differences in interstate pay in various locations are computed using the state average weekly pay. This appendix provides a table for the second
Interstate Pay Differential APPENDIX IV Adjustments for differences in interstate pay in various locations are computed using the state average weekly pay. This appendix provides a table for the second
Estimated Economic Impacts of the Small Business Jobs and Tax Relief Act National Report
 Regional Economic Models, Inc. Estimated Economic Impacts of the Small Business Jobs and Tax Relief Act National Report Prepared by Frederick Treyz, CEO June 2012 The following is a summary of the Estimated
Regional Economic Models, Inc. Estimated Economic Impacts of the Small Business Jobs and Tax Relief Act National Report Prepared by Frederick Treyz, CEO June 2012 The following is a summary of the Estimated
