What Require Prior Approval. Debbie Pettitt 2018 SRA Western/Northeast Section Meeting March 2018
|
|
|
- Ferdinand Clark
- 5 years ago
- Views:
Transcription
1 What Require Prior Approval Debbie Pettitt 2018 SRA Western/Northeast Section Meeting March
2 Discussion Topics Definitions to know NIH Standard Terms of Award Expanded Authorities Prior Approval Requirements o Things to Remember o What Constitutes a Change in Scope o Carryover Requirements o Change in PI Resources 2
3 DEFINITIONS NIH STANDARD TERMS OF AWARD EXPANDED AUTHORITY NIH waives the need for prior approval, providing recipient with operating authority for specified issues. PRIOR APPROVAL Specified post award changes that require a written approval from the designated Grants Management Office (GMO) CARRYOVER Reallocate unspent funds from one budget period to the next CHANGE IN SCOPE A change in the methodology, approach or other aspects of the project objectives constitutes a change in scope. ALL changes in scope require prior approval 3
4 NIH Standard Terms of Award Expanded Authorities Expanded Authority provides recipients the flexibility to make some changes in their approved project or budget without prior approval Foreign Grants ARE included under Expanded Authorities unless specifically excluded on the Notice of Award (NoA) NIH Standard Terms of Award 5/section_8/8.1_changes_in_project_and_budg et.htm#nih 4
5 NIH Standard Terms of Award Expanded Authorities Some Award-Expanded Authorities Include: Extend the final budget period of a project for up to 12 months beyond the original expiration date, if: Additional funds are not required Original approved scope will not change Transfer work to a third party through consortium agreement, if: It does not constitute a change in scope Not a foreign subcontract 5
6 NIH Standard Terms of Award Expanded Authorities Make cost-related changes, including significant rebudgeting of funds UNLESS these changes constitute a change in scope An unobligated balance may be carried over into the next budget period without Grants Management Officer approval if the award has automatic carryover authority. Exceptions: Centers (P50, P60, P30 and others), cooperative agreements (U), Kirschsteing-NRSA institutional research training grants (T), non-fast Track Phase I SBIR and STTR awards (R43), clinical trials and awards to individuals, or if the NoA indicates otherwise 6
7 Prior Approval: Things to Remember ALL requests for Prior Approval must be: Submitted in writing via letter or Signed or sent by the Authorizing Official Submitted no later than 30 days before the proposed change 7
8 Prior Approval: Things to Remember Official approval/disapproval of a prior approval request can only be given by the Grants Management Officer Notification of Prior Approval will be issued either by a: Revised NoA noted in the terms and conditions of award Written/ approval or disapproval 8
9 Prior Approval Items Grantees are required to request prior approval for: 2 nd no-cost extension or late notification of initial no-cost extension Change of Grantee Institution (transfer of award) Change of Grantee Organizational Status Change of PI Change from single PI to multi-pi and multi-pi to single PI Reduction of PI effort by 25% or more 9
10 Prior Approval Items Recipients are required to request prior approval for: Deviations from the terms and conditions or restrictions of the Notice of Award (NoA) Actions needing additional NIH funds Addition of a foreign component Alterations & Renovations only minor A&R <$500,000 allowable costs Pre-Award costs more than 90 days (competing award) 10
11 Change in Scope What Constitutes a Change in Scope Change in specific aims Shifting the research emphasis from one disease to another Changing any aspect of research involving animals or human subjects Changing the principal investigator or other key personnel PI on leave for more than 90 days 11
12 Change in Scope What Constitutes a Change in Scope Using new technology Purchasing equipment for $25,000 or more Rebudgeting funds in or out of a budget category by more than 25% of the total costs of the award Rebudgeting funds so that an alterations and renovations project exceeds $500,000 Not sure? Contact your Grants Management Specialist 12
13 Prior Approval Requirements for. Carryover request - If the recipient does not have automatic carryover authority Submit Final FFR Sign and submit carryover request with: Grant number and PI name Reason for unobligated balance Amounts being carried over (DC, F&A and TC) F&A costs using the rate for the year in which the funds are being carried over Budgetary/Scientific justifications Contact information for PI and BO IF new personnel added, appropriate information, HS education certificate, bio sketch and other support Reasons why work cannot be accomplished through rebudgeting from current period funds Must be signed or submitted by Authorizing Official 13
14 Change of PI Institutional Training Grants 2010 Request a change of PI by sending a letter or to the Grants Management Specialist through the Authorizing Official and include: Reasons for change Date original PI will return (if applicable) Bio Sketch Certificate of HS education (if applicable) The request should be signed by the current PI, proposed replacement PI and authorized organizational official 14
15 Overall Prior Approval Requirements Submit the prior approval request in writing by the authorized business official 30 days before the proposed change All request should include: The number in the subject line Your name, title, organization, telephone number and address Justification for the proposed change The Grants Management Officer will work with the Program Official to review the request and required documentation 15
16 Prior Approval Required???? In the competing application the PI s level of effort was listed as 3.6 calendar months per year. In year 2 of the award the PI reduced his level of effort to 3.0 calendar months. In year 3 of the award the PI reduced his level to 2.4 calendar months. Does the recipient need to request NIH prior approval for the reduction in level of effort to 2.4 calendar months? 16
17 Answer Yes, per the Grants Policy Statement, Section Change in Status, Including Absence of PD/PI and Other Senior/Key Personnel Named in the NoA, reductions are cumulative, i.e., the 25% threshold may be reached by two or more successive reductions that total 25% or more. Once agency approval has been given for a significant change in the level of effort, then all subsequent reductions are measured against the approved adjusted level. 17
18 What If The PI comes to you and wants to request a first no cost extension but the project period end date has already passed. Since this is his first no cost extension, is prior approval required. 18
19 Yes, even though this is a first no cost extension, the project period has ended and a formal request must be made to NIH. The request will need to include a justification as to why the extension was not requested through era Commons under expanded authorities. 19
20 The PI wants to use funds from a previous grant year to purchase a piece of equipment costing $38,000. Does this require NIH prior approval since the grant has automatic carryover? 20
21 Yes, NIH prior approval is required since the piece of equipment is over $25,000. A formal request will need to be submitted and must include 3 estimates for the equipment. 21
22 The PI comes to you and wants to take a sabbatical for 6 months. They plan to continue working on the grant and do not plan to list an interim PI. Does the PI need NIH prior approval for the sabbatical even though they are planning to continue their level of effort on the award? Do they have to list an interim PI? 22
23 Yes, once the PI has approval from the institution to take the sabbatical, the institution must submit a request to NIH outlining how the aims of the grant will be carried out during the PIs absence. NIH will not approve a sabbatical.they will approve the plan the institution has in place for the work to be completed during the PIs absence. With a sabbatical of 6 months, NIH most likely will require an interim PI be named for the duration of the sabbatical. 23
24 Resources NIH Grants Policy Statement: TML5/introduction.htm 24
Prior Approval Request What is taking so long?
 Prior Approval Request What is taking so long? Mark Langer National Institute of Arthritis Musculoskeletal and Skin Diseases SOCIETY OF RESEARCH ADMINISTRATORS INTERNATIONAL ANNUAL MEETING VANCOUVER, CANADA
Prior Approval Request What is taking so long? Mark Langer National Institute of Arthritis Musculoskeletal and Skin Diseases SOCIETY OF RESEARCH ADMINISTRATORS INTERNATIONAL ANNUAL MEETING VANCOUVER, CANADA
After the Award is Made THEN WHAT?
 After the Award is Made THEN WHAT? George Tucker Chief Grants Management Officer National Institute of Environmental Health Sciences October 2016 TOPICS OF DISCUSSION The ins & outs of the NoA Accessing
After the Award is Made THEN WHAT? George Tucker Chief Grants Management Officer National Institute of Environmental Health Sciences October 2016 TOPICS OF DISCUSSION The ins & outs of the NoA Accessing
IDeA Post Award Management
 IDeA Post Award Management Lisa Hildred Grants Management Specialist Grants Administration Branch National Institute of General Medical Sciences, NIH Meet the Grants Management Team Christy Leake Sam Farrell
IDeA Post Award Management Lisa Hildred Grants Management Specialist Grants Administration Branch National Institute of General Medical Sciences, NIH Meet the Grants Management Team Christy Leake Sam Farrell
Notice of Grant Award (NGA): STANDARD Terms and Conditions
 After the Award is Made Then What? Cheryl Chick Chief Grants Management Officer National Human Genome Research Institute Roger G. Sorensen, Ph.D., MPA Division of Neuroscience and Behavior National Institute
After the Award is Made Then What? Cheryl Chick Chief Grants Management Officer National Human Genome Research Institute Roger G. Sorensen, Ph.D., MPA Division of Neuroscience and Behavior National Institute
THINKING OF MOVING: A GUIDE FOR TRANSFERRING AN NIH GRANT TO A NEW INSTITUTION
 THINKING OF MOVING: A GUIDE FOR TRANSFERRING AN NIH GRANT TO A NEW INSTITUTION It is not unusual for NIH funded Principal Investigators (PIs) to move from one institution to another. We fully expect that
THINKING OF MOVING: A GUIDE FOR TRANSFERRING AN NIH GRANT TO A NEW INSTITUTION It is not unusual for NIH funded Principal Investigators (PIs) to move from one institution to another. We fully expect that
A Walk Through The SF424 (R&R) Marcia Hahn Office of Policy for Extramural Research Administration, OER, NIH January 11, 2006
 A Walk Through The SF424 (R&R) Marcia Hahn Office of Policy for Extramural Research Administration, OER, NIH January 11, 2006 1 Handouts for This Session A Walk Through The SF424 (R&R) (Powerpoint Presentation
A Walk Through The SF424 (R&R) Marcia Hahn Office of Policy for Extramural Research Administration, OER, NIH January 11, 2006 1 Handouts for This Session A Walk Through The SF424 (R&R) (Powerpoint Presentation
Office of Financial Resources Division of Grants Management
 Office of Financial Resources Division of Grants Management MAI New Grantee Meeting December 8-10, 2015 Rockville, MD Andrew Payne Grants Management Specialist SAMHSA Grants/DGM Topics Note: Make sure
Office of Financial Resources Division of Grants Management MAI New Grantee Meeting December 8-10, 2015 Rockville, MD Andrew Payne Grants Management Specialist SAMHSA Grants/DGM Topics Note: Make sure
Research Administration Services Roles & Responsibilities For Grants and Contracts (Excludes Clinical Trials) Version 3.1
 Research Administration Services Roles & Responsibilities For Grants and Contracts (Excludes Clinical Trials) Version 3.1 Category PI/PI Staff Department* RAS Unit School/Unit ORA Offices Relevant SOP
Research Administration Services Roles & Responsibilities For Grants and Contracts (Excludes Clinical Trials) Version 3.1 Category PI/PI Staff Department* RAS Unit School/Unit ORA Offices Relevant SOP
Kathy Hancock, Assistant Grants Compliance Officer, DGCO, OPERA, Office of Extramural Research, NIH, HHS
 Division of Grants Compliance and Oversight (DGCO) Office of Policy for Extramural Research Administration (OPERA), OER National Institutes of Health (NIH), DHHS 19 th Annual SBIR/STTR Conference Milwaukee,
Division of Grants Compliance and Oversight (DGCO) Office of Policy for Extramural Research Administration (OPERA), OER National Institutes of Health (NIH), DHHS 19 th Annual SBIR/STTR Conference Milwaukee,
Welcome and Introductions
 Fiscal Matters: An Overview of Annual Head Start and Early Head Start Grant Requirements U.S. Department of Health and Human Services, Welcome and Introductions Staff Grantee Representatives 1 Goals for
Fiscal Matters: An Overview of Annual Head Start and Early Head Start Grant Requirements U.S. Department of Health and Human Services, Welcome and Introductions Staff Grantee Representatives 1 Goals for
UTHSCSA LAB TRANSFER INFORMATION
 UTHSCSA LAB TRANSFER INFORMATION The information provided below is a list of offices and contacts that are needed for various approvals to move a lab to UTHSCSA. Animal Transfer to UTHSCSA http://research.uthscsa.edu/lar/
UTHSCSA LAB TRANSFER INFORMATION The information provided below is a list of offices and contacts that are needed for various approvals to move a lab to UTHSCSA. Animal Transfer to UTHSCSA http://research.uthscsa.edu/lar/
National Institutes of Health
 National Institutes of Health NIH NIH s mission is to seek fundamental knowledge about the nature and behavior of living systems and the application of that knowledge to enhance health, lengthen life,
National Institutes of Health NIH NIH s mission is to seek fundamental knowledge about the nature and behavior of living systems and the application of that knowledge to enhance health, lengthen life,
Agenda. NIH Update. Other Updates. Proposal & Progress Report Statistics Research Administration Training Topic:
 Agenda NIH Update Salary Cap Guidance NOT OD 17 037: NIH Implementation of the Interim RPPR While a Renewal Application is Under Consideration Prior Approval for No Cost Extensions via the era Commons
Agenda NIH Update Salary Cap Guidance NOT OD 17 037: NIH Implementation of the Interim RPPR While a Renewal Application is Under Consideration Prior Approval for No Cost Extensions via the era Commons
Diane Dean, Director Kathy Hancock, Assistant Grants Compliance Officer Joel Snyderman, Assistant Grants Compliance Officer
 Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar San Diego, CA October 2015 Diane Dean,
Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar San Diego, CA October 2015 Diane Dean,
Preparing for a RPPR Submission
 Preparing for a RPPR Submission Review current NoA Review most current budget and expenditure information. Determine unobligated balance in order to respond to 2590 questions. Review last esnap if applicable
Preparing for a RPPR Submission Review current NoA Review most current budget and expenditure information. Determine unobligated balance in order to respond to 2590 questions. Review last esnap if applicable
Grants Management Issues in Brief National Child Traumatic Stress Initiative Cooperative Agreements. Table of Contents
 Grants Management Issues in Brief National Child Traumatic Stress Initiative Cooperative Agreements Updated October, 2005 This document discusses some issues in the management of your Federal grant that
Grants Management Issues in Brief National Child Traumatic Stress Initiative Cooperative Agreements Updated October, 2005 This document discusses some issues in the management of your Federal grant that
Form Approved Through 03/31/2020 OMB No
 Form Approved Through 03/31/2020 OMB No. 0925-0001 Department of Health and Human Services Public Health Services Grant Application Do not exceed character length restrictions indicated. 1. TITLE OF PROJECT
Form Approved Through 03/31/2020 OMB No. 0925-0001 Department of Health and Human Services Public Health Services Grant Application Do not exceed character length restrictions indicated. 1. TITLE OF PROJECT
Guidance on Grant Award Transfers
 University of the Pacific Office of Sponsored Programs Guidance on Grant Award Transfers It is not unusual for funded Principal Investigators (PIs) to move from one institution to another. However, The
University of the Pacific Office of Sponsored Programs Guidance on Grant Award Transfers It is not unusual for funded Principal Investigators (PIs) to move from one institution to another. However, The
BUDGET PREPARATION: NIH PROPOSALS. Beginning Questions
 BUDGET PREPARATION: NIH PROPOSALS Debbie M. Meltzer, SMPH Christy Schulz, SMPH December 6, 2016 Beginning Questions Institute or Center Cost Principles Direct Cost vs. Indirect Cost Budgets: Modular/Detailed
BUDGET PREPARATION: NIH PROPOSALS Debbie M. Meltzer, SMPH Christy Schulz, SMPH December 6, 2016 Beginning Questions Institute or Center Cost Principles Direct Cost vs. Indirect Cost Budgets: Modular/Detailed
Prior Approval Request
 Prior Approval Request What is taking so long? S O C I ETY O F R ES EA R C H A D MI N I S T R A T O R S I N T ERN A T I O N A L A N N UA L MEETI N G V A N C O UV ER, C A N A D A O C TO B ER 14-18 the only
Prior Approval Request What is taking so long? S O C I ETY O F R ES EA R C H A D MI N I S T R A T O R S I N T ERN A T I O N A L A N N UA L MEETI N G V A N C O UV ER, C A N A D A O C TO B ER 14-18 the only
Grant Application Package
 Generates errors for all applications Grant Application Package Opportunity Title: Research Project Grant (Parent R01) Offering Agency: National Institutes of Health Generates errors in certain situations
Generates errors for all applications Grant Application Package Opportunity Title: Research Project Grant (Parent R01) Offering Agency: National Institutes of Health Generates errors in certain situations
Presentation. Title. Presented Name. Office of Policy for Extramural Research Administration, OER, NIH. Title. Office
 Presentation Title Michelle Presented G. Bulls, By: Director Name Office of Policy for Extramural Research Administration, OER, NIH Title Office Federal Demonstration Partnership, May 2015 NIH is currently
Presentation Title Michelle Presented G. Bulls, By: Director Name Office of Policy for Extramural Research Administration, OER, NIH Title Office Federal Demonstration Partnership, May 2015 NIH is currently
Award Transfer Guidelines
 Award Transfer Guidelines The process of transferring awards from one institution to another is not a simple task and requires negotiation between the sponsoring agency, the old institution and the new
Award Transfer Guidelines The process of transferring awards from one institution to another is not a simple task and requires negotiation between the sponsoring agency, the old institution and the new
Carryover Process Diabetes in Indian Country Conference
 2017 Diabetes in Indian Country Conference Carryover Process John Hoffman, Senior Grants Management Specialist IHS Division of Grant Management September 19, 2017 Objectives 1. Carryover Policy and Procedure
2017 Diabetes in Indian Country Conference Carryover Process John Hoffman, Senior Grants Management Specialist IHS Division of Grant Management September 19, 2017 Objectives 1. Carryover Policy and Procedure
GRANTS MANAGEMENT 411
 GRANTS MANAGEMENT 411 GLS/NSSP Grantee Meeting May 3, 2016 Maria Martinez Grants Management Specialist OFR, Division of Grants Management 240 276 1417; maria.martinez@samhsa.hhs.gov TOPICS Partnership
GRANTS MANAGEMENT 411 GLS/NSSP Grantee Meeting May 3, 2016 Maria Martinez Grants Management Specialist OFR, Division of Grants Management 240 276 1417; maria.martinez@samhsa.hhs.gov TOPICS Partnership
Grant Application Package
 Grant Application Package Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Research
Grant Application Package Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Research
NIH Agency Specifics August 11, 2015
 NIH Agency Specifics August 11, 2015 Maria Skinner Office of Sponsored Programs Stacy Salisbury Contract & Grant Administration NIH Pre-Award Presented by: Maria Skinner Office of Sponsored Programs (OSP)
NIH Agency Specifics August 11, 2015 Maria Skinner Office of Sponsored Programs Stacy Salisbury Contract & Grant Administration NIH Pre-Award Presented by: Maria Skinner Office of Sponsored Programs (OSP)
Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS
 Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar Chicago, IL October 2016 Diane Dean, Director
Division of Grants Compliance and Oversight Office of Policy for Extramural Research Administration, OER National Institutes of Health, DHHS NIH Regional Seminar Chicago, IL October 2016 Diane Dean, Director
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP)
 Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Complex Award Management Program Projects/Multi-Fund Awards- Post Award NUMBER: RAS SOP 2001 VERSION: 4.0
Emory University Research Administration Services (RAS) Standard Operating Procedure (SOP) TITLE: Complex Award Management Program Projects/Multi-Fund Awards- Post Award NUMBER: RAS SOP 2001 VERSION: 4.0
Utilizing the era Commons to Manage NIH Grants. Office of Sponsored Research UNC-CH
 Utilizing the era Commons to Manage NIH Grants Office of Sponsored Research UNC-CH Objectives of the workshop Introduce you to the the look and feel of the era Commons web site Familiarize you with the
Utilizing the era Commons to Manage NIH Grants Office of Sponsored Research UNC-CH Objectives of the workshop Introduce you to the the look and feel of the era Commons web site Familiarize you with the
Grant Application Package
 Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Research Project Grant (Parent R01)
Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Research Project Grant (Parent R01)
Pre Award Table of Contents
 Pre Award Table of Contents ORA Internal Procedures for Account Establishment 2 Conflict of Interest: Thomas Jefferson University Conflict of Interest (COI) Policy 4 Cost Sharing and Matching Funds 6 Developing
Pre Award Table of Contents ORA Internal Procedures for Account Establishment 2 Conflict of Interest: Thomas Jefferson University Conflict of Interest (COI) Policy 4 Cost Sharing and Matching Funds 6 Developing
UF Commitment Entry Guidelines
 UF Commitment Entry Guidelines Table of Contents Commitment Entry Guidelines... 2 General Entry... 2 UFIRST General Guidance... 2 myufl General Guidance... 3 Personnel... 3 Minimum Effort... 3 Exceeding
UF Commitment Entry Guidelines Table of Contents Commitment Entry Guidelines... 2 General Entry... 2 UFIRST General Guidance... 2 myufl General Guidance... 3 Personnel... 3 Minimum Effort... 3 Exceeding
NIH Proposal Outline Twelve Page Limit For Activity Codes R01, R10, R15, R18, R21/R33, R24, R33, R34, DP3, G08, G11, G13, SC1, X01
 NIH Proposal Outline Twelve Page Limit For Activity Codes R01, R10, R15, R18, R21/R33, R24, R33, R34, DP3, G08, G11, G13, SC1, X01 1. Introduction to Application (for Resubmission or Revision applications
NIH Proposal Outline Twelve Page Limit For Activity Codes R01, R10, R15, R18, R21/R33, R24, R33, R34, DP3, G08, G11, G13, SC1, X01 1. Introduction to Application (for Resubmission or Revision applications
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018
 CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
Grant Application Package
 Grant Application Package Generates errors for all applications Generates errors in certain situations Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition
Grant Application Package Generates errors for all applications Generates errors in certain situations Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition
Fundamentals of the NIH. Erica Brown, PhD Director, NIH AREA Program Extramural Policy Coordination Officer National Institutes of Health
 Fundamentals of the NIH Erica Brown, PhD Director, NIH AREA Program Extramural Policy Coordination Officer National Institutes of Health 1 Overview The big picture Finding funding opportunities Interacting
Fundamentals of the NIH Erica Brown, PhD Director, NIH AREA Program Extramural Policy Coordination Officer National Institutes of Health 1 Overview The big picture Finding funding opportunities Interacting
Grant Application Package
 Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Grant Application Package Reducing
Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Grant Application Package Reducing
Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed:
 COEUS NIH-GRANTS.GOV PROPOSAL REVIEW CHECKLIST REVIEWER: Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed: Has the Program Announcement been reviewed? What is
COEUS NIH-GRANTS.GOV PROPOSAL REVIEW CHECKLIST REVIEWER: Proposal Development No: Date Due to Sponsor: Target Review by date: Date Review Completed: Has the Program Announcement been reviewed? What is
Children s Discovery Institute Grants Policies
 Table of Contents Section Page #(s) Purpose of Grants 2 Funding Mechanisms 2-3 Grant Identifiers 3 Authority for Making Grants 3 Amendment of Policies 3 Direct Costs 3-4 Facilities & Administrative Costs
Table of Contents Section Page #(s) Purpose of Grants 2 Funding Mechanisms 2-3 Grant Identifiers 3 Authority for Making Grants 3 Amendment of Policies 3 Direct Costs 3-4 Facilities & Administrative Costs
2. DATE SUBMITTED 3. DATE RECEIVED BY STATE. 4. Federal Identifier 7. * TYPE OF APPLICANT: Other (Specify): 9. * NAME OF FEDERAL AGENCY: 10.
 DATE SUBMITTED Applicant Identifier RSP will enter the assigned PALS # here DATE RECEIVED BY STATE State Application Identifier APPLICATION FOR FEDERAL ASSISTANCE SF 424 (R&R) Read agency specific guidelines
DATE SUBMITTED Applicant Identifier RSP will enter the assigned PALS # here DATE RECEIVED BY STATE State Application Identifier APPLICATION FOR FEDERAL ASSISTANCE SF 424 (R&R) Read agency specific guidelines
The Patrick and Catherine Weldon Donaghue Foundation for Medical Research POLICIES FOR GRANTEES
 The Patrick and Catherine Weldon Donaghue Foundation for Medical Research POLICIES FOR GRANTEES A. Change in Grant Award It is the obligation of the investigator and host institution to notify the Donaghue
The Patrick and Catherine Weldon Donaghue Foundation for Medical Research POLICIES FOR GRANTEES A. Change in Grant Award It is the obligation of the investigator and host institution to notify the Donaghue
Outgoing Subagreements: Subawards and Subcontracts
 Effective Date: March 1, 2014 Version: 1 Page: 1 of 6 Overview Sponsored programs may include partnerships with other organizations or institutions whereby a portion of the project is conducted by investigators
Effective Date: March 1, 2014 Version: 1 Page: 1 of 6 Overview Sponsored programs may include partnerships with other organizations or institutions whereby a portion of the project is conducted by investigators
Ins and Outs of SBIR Grants and Contracts. A Grants Manager Perspective
 Ins and Outs of SBIR Grants and Contracts A Grants Manager Perspective Andre D. Walker Outline Differences Between a Grant and Contract Eligibility Budget Just In Time Post Award Activities 1 So What s
Ins and Outs of SBIR Grants and Contracts A Grants Manager Perspective Andre D. Walker Outline Differences Between a Grant and Contract Eligibility Budget Just In Time Post Award Activities 1 So What s
Grant Application Package
 Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Research Project Grant (Parent R01)
Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Research Project Grant (Parent R01)
Grant Application Package
 Grant Application Package Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Methodology
Grant Application Package Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Methodology
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates!
 FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
Page 1 of 10 1/31/2008
 Page 1 of 10 1/31/2008 Step by Step Tips to Avoid Errors and Warnings o Full instructions can be found at: http://grants1.nih.gov/grants/funding/424/index.htm. o This document is intended to assist you
Page 1 of 10 1/31/2008 Step by Step Tips to Avoid Errors and Warnings o Full instructions can be found at: http://grants1.nih.gov/grants/funding/424/index.htm. o This document is intended to assist you
Presentation. Title. Presented Name. Office of Policy for Extramural Research Administration, OER, NIH. Title. Office
 Presentation Title Michelle Presented G. Bulls, By: Director Name Office of Policy for Extramural Research Administration, OER, NIH Title Office Federal Demonstration Partnership, January 2015 NIH is currently
Presentation Title Michelle Presented G. Bulls, By: Director Name Office of Policy for Extramural Research Administration, OER, NIH Title Office Federal Demonstration Partnership, January 2015 NIH is currently
The Lifecycle of a Sponsored Project
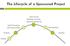 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where does the
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where does the
Grant Application Package
 Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Health Promotion Among Racial and Ethnic
Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Health Promotion Among Racial and Ethnic
NIH R-Mechanism Mini Guide for Completing SF424 (R&R) Applications Using Grants.gov *Adobe Forms Version B2*
 NIH R-Mechanism Mini Guide for Completing SF424 (R&R) Applications Using Grants.gov *Adobe Forms Version B2* Please note that this mini guide is a tool for assisting in correctly preparing a grant application
NIH R-Mechanism Mini Guide for Completing SF424 (R&R) Applications Using Grants.gov *Adobe Forms Version B2* Please note that this mini guide is a tool for assisting in correctly preparing a grant application
October electronic Research Administration (era) OER, OD, National Institutes of Health
 October 2016 electronic Research Administration (era) OER, OD, National Institutes of Health Scarlett Gibb era Customer Relationship Manager, era Commons Joe Schumaker era Communications and Outreach 2
October 2016 electronic Research Administration (era) OER, OD, National Institutes of Health Scarlett Gibb era Customer Relationship Manager, era Commons Joe Schumaker era Communications and Outreach 2
SPONSORED PROGRAM ADMINISTRATION MEETING March 2017 WELCOME
 SPONSORED PROGRAM ADMINISTRATION MEETING March 2017 WELCOME OVPR/SPS News and Information Technology Commercialization Services Andrew Zehner 2017 Individual Financial Disclosure in Research Internal Routing
SPONSORED PROGRAM ADMINISTRATION MEETING March 2017 WELCOME OVPR/SPS News and Information Technology Commercialization Services Andrew Zehner 2017 Individual Financial Disclosure in Research Internal Routing
MOC AACN Research Grant
 MOC AACN Research Grant The MOC AACN Research Grant is funded and supported by MOC AACN. We encourage clinicians and researchers to propose projects that support inquiry and systematic research that generates
MOC AACN Research Grant The MOC AACN Research Grant is funded and supported by MOC AACN. We encourage clinicians and researchers to propose projects that support inquiry and systematic research that generates
The Hope Foundation SEED Fund for SWOG Early Exploration and Development 2016 Announcement
 The Hope Foundation SEED Fund for SWOG Early Exploration and Development 2016 Announcement OVERVIEW SWOG s mission is to improve the practice of cancer medicine in preventing, detecting, and treating cancer,
The Hope Foundation SEED Fund for SWOG Early Exploration and Development 2016 Announcement OVERVIEW SWOG s mission is to improve the practice of cancer medicine in preventing, detecting, and treating cancer,
Follow Just-in-Time Procedures to Get Your Award Faster
 Follow Just-in-Time Procedures to Get Your Award Faster Mindy Bixby NICHD November 08 09, 2017 What is Just Time? Just in Time (JIT) refers to the information that NIH requests after an application has
Follow Just-in-Time Procedures to Get Your Award Faster Mindy Bixby NICHD November 08 09, 2017 What is Just Time? Just in Time (JIT) refers to the information that NIH requests after an application has
ACL GRANTS MANAGEMENT AND AMENDMENTS. SMP/SHIP Conference August 2-5, 2016
 1 ACL GRANTS MANAGEMENT AND AMENDMENTS SMP/SHIP Conference August 2-5, 2016 Agenda 2 Introductions Understanding and Reading the Notice of Award (NOA) Requesting Amendments Reporting Requirements Website
1 ACL GRANTS MANAGEMENT AND AMENDMENTS SMP/SHIP Conference August 2-5, 2016 Agenda 2 Introductions Understanding and Reading the Notice of Award (NOA) Requesting Amendments Reporting Requirements Website
Grant Application Package
 Grant Application Package Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: NIH Research
Grant Application Package Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: NIH Research
Grant Application Package
 Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Grant Application Package NIH Research
Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Grant Application Package NIH Research
Letter of Intent to Establish a Consortium Agreement Saint Louis University as Primary Applicant
 Letter of Intent to Establish a Consortium Agreement Saint Louis University as Primary Applicant Saint Louis University PI: Subcontract Organization: SLU PI Department: Subcontract PI: Sponsor Name: Proposal
Letter of Intent to Establish a Consortium Agreement Saint Louis University as Primary Applicant Saint Louis University PI: Subcontract Organization: SLU PI Department: Subcontract PI: Sponsor Name: Proposal
The AOFAS Research Grants Program Description, Policies, and Guidelines for Applicants and Institutional Representatives
 The AOFAS Research Grants Program Description, Policies, and Guidelines for Applicants and Institutional Representatives Updated October 2016 for the 2017 Grants Cycle Table of Contents ABOUT THE AOFAS
The AOFAS Research Grants Program Description, Policies, and Guidelines for Applicants and Institutional Representatives Updated October 2016 for the 2017 Grants Cycle Table of Contents ABOUT THE AOFAS
Form Approved Through 09/30/2007 OMB No Department of Health and Human Services Public Health Services Grant Application
 Form Approved Through 9/3/27 OMB No. 925-1 Department of Health and Human Services Public Health Services Grant Application LEAVE BLANK FOR PHS USE ONLY. Type Activity Number Review Group Formerly Do not
Form Approved Through 9/3/27 OMB No. 925-1 Department of Health and Human Services Public Health Services Grant Application LEAVE BLANK FOR PHS USE ONLY. Type Activity Number Review Group Formerly Do not
Grant Application Package
 Grant Application Package Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Agriculture
Grant Application Package Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Agriculture
 Page 1 of 6 Search: Contact Us Print Version Advanced Search Site Map Funding Opportunities Funding Opportunities (RFAs, PAs) & Notices Unsolicited Applications (Parent Announcements) Research Training
Page 1 of 6 Search: Contact Us Print Version Advanced Search Site Map Funding Opportunities Funding Opportunities (RFAs, PAs) & Notices Unsolicited Applications (Parent Announcements) Research Training
(Insert additional Principal Investigators in the Comments section.) Co-Investigator Data Investigators Employee # School
 University of Southern California Department of Contracts & Grants (DCG) Proposal Approval Record (PAR) For Dept. of Contracts & Grants Use Only USC Proposal # Date Received Deadline Date Principal Investigator
University of Southern California Department of Contracts & Grants (DCG) Proposal Approval Record (PAR) For Dept. of Contracts & Grants Use Only USC Proposal # Date Received Deadline Date Principal Investigator
Cancer Research Education Grants Program to Promote Diversity (R25) Courses for Skills Development (PAR ) Research Experiences (PAR )
 Cancer Research Education Grants Program to Promote Diversity (R25) Courses for Skills Development (PAR-16-139) Research Experiences (PAR-16-138) Davyd Chung, Ph.D. Diversity Training Branch Center to
Cancer Research Education Grants Program to Promote Diversity (R25) Courses for Skills Development (PAR-16-139) Research Experiences (PAR-16-138) Davyd Chung, Ph.D. Diversity Training Branch Center to
The Lifecycle of a Sponsored Project
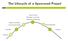 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
FAER RESEARCH GRANTS OVERVIEW & REQUIREMENTS
 FAER RESEARCH GRANTS OVERVIEW & REQUIREMENTS APPLICATION INSTRUCTIONS updated 08/02/2018 UPCOMING APPLICATION CYCLES Fall 2018 Application Deadline: August 15, 2018 Earliest possible funding date: January
FAER RESEARCH GRANTS OVERVIEW & REQUIREMENTS APPLICATION INSTRUCTIONS updated 08/02/2018 UPCOMING APPLICATION CYCLES Fall 2018 Application Deadline: August 15, 2018 Earliest possible funding date: January
Ball State University
 Ball State University CONTRACTS AND GRANTS MANUAL 1 - Introduction... 3 1.1 Purpose of the CGO Manual... 3 1.2 Roles and Responsibilities... 3 A. Responsibilities of the Contracts and Grants Office...
Ball State University CONTRACTS AND GRANTS MANUAL 1 - Introduction... 3 1.1 Purpose of the CGO Manual... 3 1.2 Roles and Responsibilities... 3 A. Responsibilities of the Contracts and Grants Office...
Successful Submissions
 Successful Submissions Prepare Complete-Submit-Track-View Created by: Geeta Chhabra Electronic Submission of NIH Grant Applications Find and download announcement and application package Prepares the application
Successful Submissions Prepare Complete-Submit-Track-View Created by: Geeta Chhabra Electronic Submission of NIH Grant Applications Find and download announcement and application package Prepares the application
Key Dates: All correspondence and draft applications are to be submitted by to
 24-NOV-2017 Lysosomal Disease Network (LDN) Request for Proposals (RFP) (for studies aiming to participate in the competitive re-application to the National Institutes of Health Rare Diseases Clinical
24-NOV-2017 Lysosomal Disease Network (LDN) Request for Proposals (RFP) (for studies aiming to participate in the competitive re-application to the National Institutes of Health Rare Diseases Clinical
OSP Forum: The New Federal Research Terms and Conditions Prior Approval Matrix. August 7,
 OSP Forum: The New Federal Research Terms and Conditions Prior Approval Matrix August 7, 2017-1 Agenda Recent changes in Federal terms OSP resources and how to use them Similarities & differences among
OSP Forum: The New Federal Research Terms and Conditions Prior Approval Matrix August 7, 2017-1 Agenda Recent changes in Federal terms OSP resources and how to use them Similarities & differences among
PI Eligibility: Managing Changes in Academic Status - Case Studies
 PI Eligibility: Managing Changes in Academic Status - Case Studies Case Study 1: Professor Smith resigns from McGill effective January 1, 2015, moves to University of Toronto, but keeps an adjunct status
PI Eligibility: Managing Changes in Academic Status - Case Studies Case Study 1: Professor Smith resigns from McGill effective January 1, 2015, moves to University of Toronto, but keeps an adjunct status
2017 Program Announcement Texas Space Grant Consortium New Investigator Program $10,000 Maximum
 2017 Program Announcement Texas Space Grant Consortium $10,000 Maximum 1 I Introduction The (TSGC NIP), established in 2003, is designed to assist in professional development of faculty members or researchers
2017 Program Announcement Texas Space Grant Consortium $10,000 Maximum 1 I Introduction The (TSGC NIP), established in 2003, is designed to assist in professional development of faculty members or researchers
Life Sciences Simons Collaboration on the Origins of Life Fellowship Policies and Procedures
 Life Sciences Simons Collaboration on the Origins of Life Fellowship Policies and Procedures Table of Contents 1. Our mission Page 2 2. Online fellowship management Page 2 3. Fellowship activation Page
Life Sciences Simons Collaboration on the Origins of Life Fellowship Policies and Procedures Table of Contents 1. Our mission Page 2 2. Online fellowship management Page 2 3. Fellowship activation Page
IRB Approval of Research Using Survey Procedures Kerry Agnitsch, Office for Responsible Research October 1, 2009
 IRB Approval of Research Using Survey Procedures Kerry Agnitsch, Office for Responsible Research October 1, 2009 Key Definitions (per 45 CFR 46.102(d) & (f): Research a systematic investigation, including
IRB Approval of Research Using Survey Procedures Kerry Agnitsch, Office for Responsible Research October 1, 2009 Key Definitions (per 45 CFR 46.102(d) & (f): Research a systematic investigation, including
GUIDELINES FOR PREPARING RESEARCH PROPOSALS
 GUIDELINES FOR PREPARING RESEARCH PROPOSALS Each application should have one Principal investigator (PI). A Co-PI can be named by the PI and is someone making a major contribution to a project. The Co-Principal
GUIDELINES FOR PREPARING RESEARCH PROPOSALS Each application should have one Principal investigator (PI). A Co-PI can be named by the PI and is someone making a major contribution to a project. The Co-Principal
Grant Application Package
 Grant Application Package Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Research
Grant Application Package Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Research
NOVA SOUTHEASTERN UNIVERSITY OFFICE OF SPONSORED PROGRAMS POLICIES AND PROCEDURES
 PAGE 1 OF 14 PURPOSE: The increasing involvement of academic researchers and educators with industry and private entrepreneurial ventures has raised the potential for conflict of interest. Such real or
PAGE 1 OF 14 PURPOSE: The increasing involvement of academic researchers and educators with industry and private entrepreneurial ventures has raised the potential for conflict of interest. Such real or
advertising and public relations, other than advertising for help wanted or for the procurement of goods or services necessary for the performance of
 Award management, also referred to as post award administration includes compliance with university and sponsor policies and procedures in accomplishing the statement of work within the awarded budget.
Award management, also referred to as post award administration includes compliance with university and sponsor policies and procedures in accomplishing the statement of work within the awarded budget.
Carnegie Mellon University Sponsored Projects Accounting (PA17) Interim Financial Reporting Procedures Updated: June 30, 2017 Frequency:
 Carnegie Mellon University Sponsored Projects Accounting (PA17) Interim Financial Reporting Procedures Updated: June 30, 2017 Frequency: As Needed Responsible Staff: Post Award Accountant Approval Required:
Carnegie Mellon University Sponsored Projects Accounting (PA17) Interim Financial Reporting Procedures Updated: June 30, 2017 Frequency: As Needed Responsible Staff: Post Award Accountant Approval Required:
Conditions of Award NOAA & California Natural Resources Agency
 The image part with relationship ID rid8 was not found in the file. Conditions of Award NOAA & California Natural Resources Agency 2 C.F.R. Part 200 Uniform Administrative Requirements, Cost Principles,
The image part with relationship ID rid8 was not found in the file. Conditions of Award NOAA & California Natural Resources Agency 2 C.F.R. Part 200 Uniform Administrative Requirements, Cost Principles,
Rosemarie Filart, MD MPH MBA NIH Program Officer National Center of Research Resources, NIH NCRR
 Na ationa al Ins stitut tes of Hea alth The Basics of SBIR/STTR Grants and Other Public & Private Funding Sources for Innovation and Technology Weill Cornell CTSC February 27, 2009 New York, NY Rosemarie
Na ationa al Ins stitut tes of Hea alth The Basics of SBIR/STTR Grants and Other Public & Private Funding Sources for Innovation and Technology Weill Cornell CTSC February 27, 2009 New York, NY Rosemarie
Trigger / Timing / Frequency: When a new award is received by the University and OSP determines that the award can be accepted.
 Kuali Research User Guide: Create a New Parent Award Version October 06 Purpose: To create a new parent award record in the system. Trigger / Timing / Frequency: When a new award is received by the University
Kuali Research User Guide: Create a New Parent Award Version October 06 Purpose: To create a new parent award record in the system. Trigger / Timing / Frequency: When a new award is received by the University
RESEARCH ADMINISTRATION STANDARD OPERATING PROCEDURES (SOPS)
 RESEARCH ADMINISTRATION STANDARD OPERATING PROCEDURES (SOPS) Award Obligation Setup (AOS) University Prior Approval Form (UPAF) Agreements Reviewed & Approved by RA Northeastern University Office Of Research
RESEARCH ADMINISTRATION STANDARD OPERATING PROCEDURES (SOPS) Award Obligation Setup (AOS) University Prior Approval Form (UPAF) Agreements Reviewed & Approved by RA Northeastern University Office Of Research
NIH Transition to the SF424 (R&R) Application & Electronic Submission Plus A Walk Through The SF424 (R&R)
 NIH Transition to the SF424 (R&R) Application & Electronic Submission Plus A Walk Through The SF424 (R&R) Nancy Saunders, PhD National Institute of Allergy and Infectious Diseases National Institutes of
NIH Transition to the SF424 (R&R) Application & Electronic Submission Plus A Walk Through The SF424 (R&R) Nancy Saunders, PhD National Institute of Allergy and Infectious Diseases National Institutes of
National Science Foundation Update. SRA Annual Meeting October 20, 2015
 National Science Foundation Update SRA Annual Meeting October 20, 2015 Speakers Jean Feldman Head, Policy Office Division of Institution & Award Support Office of Budget, Finance & Award Management 703.292.8243;
National Science Foundation Update SRA Annual Meeting October 20, 2015 Speakers Jean Feldman Head, Policy Office Division of Institution & Award Support Office of Budget, Finance & Award Management 703.292.8243;
GOVERNMENT SHUTDOWN FREQUENTLY ASKED QUESTIONS
 GOVERNMENT SHUTDOWN FREQUENTLY ASKED QUESTIONS C&G Contact Info: 919-515-8008 or mailto:cnghelpdesk@ncsu.edu SPARCS Contact Info: 919-515-2444 or mailto:sps@ncsu.edu Q: Is my award/account still open for
GOVERNMENT SHUTDOWN FREQUENTLY ASKED QUESTIONS C&G Contact Info: 919-515-8008 or mailto:cnghelpdesk@ncsu.edu SPARCS Contact Info: 919-515-2444 or mailto:sps@ncsu.edu Q: Is my award/account still open for
Guidance on Direct Charging of Administrative and Clerical Salaries
 Guidance on Direct Charging of Administrative and Clerical Salaries Updated: August 10, 2015 For proposals submitted and awards made on or after 12/26/14 that are subject to the Uniform Guidance. Key Notes
Guidance on Direct Charging of Administrative and Clerical Salaries Updated: August 10, 2015 For proposals submitted and awards made on or after 12/26/14 that are subject to the Uniform Guidance. Key Notes
2017 NIH Update. Presented by Stephanie Smith and Stacey Wade
 2017 NIH Update Presented by Stephanie Smith and Stacey Wade PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs NIH INSTITUTES National
2017 NIH Update Presented by Stephanie Smith and Stacey Wade PRESENTERS Stephanie Smith Post-Award Office of Sponsored Programs Stacey Wade Pre-Award Office of Sponsored Programs NIH INSTITUTES National
University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures
 Summary 1. Subaward Definitions A. Subaward B. Subrecipient University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures C. Office of Contracts and Grants (OCG) 2. Distinguishing
Summary 1. Subaward Definitions A. Subaward B. Subrecipient University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures C. Office of Contracts and Grants (OCG) 2. Distinguishing
Financial Oversight of Sponsored Projects Principal Investigator and Department Administrator Responsibilities
 Principal Investigator and Department Administrator Responsibilities Boston College Office for Sponsored Programs Office for Research Compliance and Intellectual Property March 2004 Introduction This guide
Principal Investigator and Department Administrator Responsibilities Boston College Office for Sponsored Programs Office for Research Compliance and Intellectual Property March 2004 Introduction This guide
National Institute of Health (NIH)
 National Institute of Health (NIH) Funding Presented by: Samantha J. Taylor Senior Research Officer Wyss Institute for Biologically Inspired Engineering, Harvard University What is NIH s primary mission?
National Institute of Health (NIH) Funding Presented by: Samantha J. Taylor Senior Research Officer Wyss Institute for Biologically Inspired Engineering, Harvard University What is NIH s primary mission?
New Investigator Research Grant Guidelines
 New Investigator Research Grant Guidelines News and Updates PSI Foundation s new online application system is now in use for New Investigator Grant applications. The PSI Foundation no longer has deadlines.
New Investigator Research Grant Guidelines News and Updates PSI Foundation s new online application system is now in use for New Investigator Grant applications. The PSI Foundation no longer has deadlines.
NIH. Electronic Submissions
 NIH Electronic Submissions 1 HOW DOES IT WORK? PI finds FOA (Funding Opportunity Announcement) through Grants.gov and prints it out. PI double-checks that he/she has the correct Adobe software on his/her
NIH Electronic Submissions 1 HOW DOES IT WORK? PI finds FOA (Funding Opportunity Announcement) through Grants.gov and prints it out. PI double-checks that he/she has the correct Adobe software on his/her
FINANCE-315 7/1/2017 SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a proposal to UACES. It provides a checklist of documents and certifications required by sponsors, as well as an
SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a proposal to UACES. It provides a checklist of documents and certifications required by sponsors, as well as an
The Metis Foundation Office of Grant and Contract Administration SUBRECIPIENT INFORMATION AND COMPLIANCE FORM
 The Metis Foundation Office of Grant and Contract Administration SUBRECIPIENT INFORMATION AND COMPLIANCE FORM All subrecipients must complete this form when submitting a proposal to the Metis Foundation
The Metis Foundation Office of Grant and Contract Administration SUBRECIPIENT INFORMATION AND COMPLIANCE FORM All subrecipients must complete this form when submitting a proposal to the Metis Foundation
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM. December 2017
 Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Life Sciences Simons Collaboration on the Global Brain (SCGB) Fellowships
 Life Sciences Simons Collaboration on the Global Brain (SCGB) Fellowships Fellowship Policies and Procedures Table of Contents 1. Our mission Page 2 2. Online fellowship management Page 2 3. Fellowship
Life Sciences Simons Collaboration on the Global Brain (SCGB) Fellowships Fellowship Policies and Procedures Table of Contents 1. Our mission Page 2 2. Online fellowship management Page 2 3. Fellowship
