Maureen Nolan Director of Nursing National Lead for the Implementation and Audit of Nurse Prescribing of Ionising Radiation and Medicinal Prescribing
|
|
|
- Ariel Garrison
- 6 years ago
- Views:
Transcription
1 Maureen Nolan Director of Nursing National Lead for the Implementation and Audit of Nurse Prescribing of Ionising Radiation and Medicinal Prescribing Dublin Mid Leinster
2 Prescribing Team Clare MacGabhann, National Lead & HSE South Annette Cuddy, HSE West Maureen Nolan, HSE DML Rose Lorenz, HSE DNE Website:
3 2006 Irish Medicines Board (Miscellaneous Provision) Act 2006 July 2006 DoHC consultation process 2006 Drafting of Regulations May 2007 Misuse of Drugs (Amendment) Regulations 2007 Medicinal Products (Prescription and Control of Supply) (Amendment) Regulations 2007 Irish Medicines Board (Miscellaneous Provision) Act 2006 (Commencement Order 2007) New Nurses Rules 2007 (An Bord Altranais) Accessible at and
4 Dual Framework Legislation Irish Medicines Board (Miscellaneous Provisions) Act 2006 Misuse of Drugs (Amendment) Regulations 2007 Medicinal Products (Prescription and Control of Supply) (Amendment) Regulations 2007 Professional Regulation -- Nurses Rules 2007 Misuse of Drugs Regulations 2017 All nurses and midwives employed by a Health Service Provider All prescription drugs Schedule 4 & 5 unrestricted Schedule 2 & 3 (Controlled Drugs) restricted (Schedule 8) Drugs must normally be used in the setting Employer can set criteria RNP must write PIN on every prescription
5 Drugs for Pain Relief in Hospital Morphine Sulphate - Oral, Intramuscular, Intravenous Subcutaneous Morphine Tartrate -Intramuscular, Intravenous, Subcutaneous Buprenorphine - Transdermal Codeine Phosphate - Oral Dihydrocodeine - Oral Fentanyl - Intranasal, Intravenous, Transdermal, Transmucosal, Subcutaneous, Sublingual/Buccal Oxycodone - Oral, Subcutaneous, Intravenous Pethidine - Intramuscular, Intravenous, Subcutaneous Drugs for Palliative Care Morphine Sulphate - Intramuscular, Oral, Subcutaneous Morphine Tartrate - Intramuscular, Subcutaneous Fentanyl - Intranasal, Intravenous, Transdermal, Transmucosal, Subcutaneous, Sublingual/Buccal Hydromorphone - Oral, Subcutaneous Methylphedidiate - Oral Buprenorphine - Transdermal Codeine Phosphate - Oral Oxycodone - Oral, Subcutaneous Drugs for Purposes of Midwifery Pethidine - Intramuscular Drugs for Neonatal Care Fentanyl - Intravenous, Transdermal, Transmucosal Morphine Sulphate - Intramuscular, Intranasal, Intravenous, Oral, Subcutaneous Morphine Tartrate - Intramuscular, Intravenous, Subcutaneous Drugs for use in Mental Health or Intellectual Disability Methylphenidate - Oral Currently under review by Department of Health
6 Nursing and Midwifery Board of Ireland: Education Clinical Competence Clinical Governance Registration Documents: Requirements and Standards for Education Programmes for Nurses and Midwives with Prescriptive Authority (2007) Decision Making Framework for Nurse and Midwife Prescribing Practice Standards and Guidelines for Nurses and Midwives with Prescriptive Authority (2018) Collaborative Practice Agreement (CPA) for Nurses and Midwives with Prescriptive Authority 4th Edn (2016) Website:
7 Requirements to register as RNP Division of the Register for Nurse Prescribers First opened January 2008 Candidate Registration is required Registration linked to other factors Completion of education programme Collaborative Practice Agreement completed Application Form for Registration with NMBI Notification of Collaborative Practice Agreement Registration fee (once off) Accessible at:
8 1 Clinical Decision-making 2 Communication and History Taking 3 Documentation 4 Prescription Writing 5 Prescribing for Self, Family and Significant Others 6 Repeat Prescribing 7 Prescribing of Off-label and Exempt Medicinal Products 8 Prescribing by Means of Other than an Original Prescription 9 Separation of Responsibilities in the Medication Management Cycle 10 Influence of Outside Interests 11 Continuing Professional Development and Continued Competency Accessible at:
9 Revised in 2016 Principles of: professional accountability, responsibility, competence and clinical governance underpin the CPA The Collaborative Practice Agreement (CPA): details the lines of communication established between RNP and the Medical Practitioner regarding the care of their patients and agreed by the employer defines the parameters of the RNPs scope of practice outlines the parameters of the RNPs prescribing authority Accessible at:
10 Employment Voluntary and statutory services of the HSE Name on Division of the Register General, Psychiatric, Children s, Intellectual Disability, Midwife or Public Health Nurse Site Requirements Support from employer to undertake the programme Nomination and confirmation of a designated medical practitioner mentor Experience 3 years recent post-registration clinical experience (within the past 5) With equivalent of 1 year full-time experience in specific area of practice Technical Requirements Competencies recognised at Level 8 (NQAI framework) Evidence of further education Possess a competent level of information technology literacy. Website:
11 Delivered over a Six month period Theoretical instruction no less than 28 days -includes self directed learning) Clinical instruction no less than 12 days (96 hours) Three core modules 1. Professional Accountability in Nurse/Midwife Prescribing 2. Pharmacology and Prescribing 3. Systematic Assessment and Evaluation in Patient care 250 hours in each module Contact time >30 hrs lectures Each module 10 ECTS Level 8 National Qualifications Authority of Ireland
12 Firm commitment (employer/organisation) National Policy Access to a Drugs & Therapeutics Committee/Review Group Prescribing Site Coordinator to oversee the introduction (named individual) A named medical mentor for each candidate Collaborative Practice Arrangement Commitment to continuing education and National Nurse and Midwife Prescribing Minimum Data Set Commitment to submit Attachment B of CPA to D&T committee within 3 months of successful completion of education programme Once CPA approved, commitment to submit documents for registration to NMBI within two weeks Website:
13 Contact between education provider, NMBI and HSE Central point of contact for candidate, RNP, medical mentor, collaborating medical practitioners Approval and implementation of national policy Oversee safe management & quality assurance Liaise with D&T Coordinate CPA process Ensure compliance with audit and monitoring (per policy)
14 Explore with student learning needs and agree a programme/contract for learning Provide support, teaching and learning opportunities equivalent of 12 days history taking and assessment Supplement pharmacology teaching specific to clinical area Meet formally at 3 and 6 months for review Clinical assessment ensure achievement of competence in practice (ABA competency framework) Review and signs off the Competency Booklet
15 Requirement for services introducing nurse/midwife prescribing Multidisciplinary Group Review and advise on the proposed list of drugs of CPA Authorised product (or off-label use) Normally used in setting Appropriate to clinical setting and scope of practice of RNP In keeping with the organisations formulary /guidelines and legislative requirements Generic form of medicinal product
16 Toolkit for the Implementation of Nurse and Midwife Prescribing Other publications Audit, Monitoring, Application Guidelines etc Prescribing Site Coordinators Groups Course Fees funded directly by HSE National Policy Toolkit for Drugs and Therapeutics Committees Clinical Indemnity State Claims Agency Access to GMS and other schemes On going communication Irish RNP enetwork Irish PSC enetwork Newsletters (Health Matters Features), Professional Journals Patient Information Leaflet Website: Nurse and Midwife Prescribing Data Collection System
17 Policy purpose/scope/aims Roles and responsibilities Eligibility to prescribe CPA ensure health service provider is part of the process, final approval issuing commencement date Good prescribing practice Prescription forms/writing/security/loss/theft Role Drugs & Therapeutics Reporting near misses Record keeping Monitoring and audit Verification of status
18 National Nurse and Midwife Prescribing Minimum Data Set Purpose: To develop a system for each individual nurse/midwife prescriber to monitor and report on the number of prescriptions written by them, for what conditions
19 Finding: model chosen for Ireland appropriate and safely implemented Conclusion: national roll out be continued and further strengthened Recommendations: legislation, education, RNP registration, CPA, practice standards, CPD, further developments Recommendations outstanding: Schedule 8; Prescribing of unauthorised (unlicensed) medicines
20 Currently there are 1104 Nurses and Midwives registered to Prescribe
21 Health Service Provider = 198 Candidates funded = 1462 Registered Nurse Prescriber = 1104 (including 63 private) Health Service Provider by HSE Area Dublin MidLeinster Dublin North East South West Variety of Clinical Areas Acute hospitals Older Persons Services Intellectual Disability Mental Health Services Public Health Nursing Services Prison Nursing Specialist Services Private Organisations
22 Number of applicants for education programmes decreased since 2011 Factors contributing to decrease: Public service moratorium in place since 2008 Regulatory requirements relating to Collaborative Practice Agreement arrangements including A) Requirement for annual/biannual review B) Access to Drugs and Therapeutics Committee Requirements for inputting prescriptions to Nurse Midwife Prescribing Data Collection System (HSE)
23 A collaborative approach was established between NMBI and ONMSD (HSE) to review regulatory and implementation systems and processes. Work commenced March 2015 and concluded with Final Report and Recommendations - October 2015
24
25 1. The Collaborative Practice Agreement is retained as a governance tool which must be completed at the point of application for registration with NMBI as a Registered Nurse Prescriber (RNP). 2. The governance for the ongoing review of RNP prescribing practices should be managed through local health service provider policy as directed by the NMBI and HSE. NMBI will continue to require the RNP to attest to having a valid CPA in the short term through the Annual Retention Notification, and subsequently through the NMBI continued competency scheme for nurses and midwives. The NMBI requirement and notification for annual and biannual review will cease.
26 3. The CPA form should be maintained. However, the NMBI should provide more clarity and guidance for the development of Attachments A, B and C.
27 4. The D&T committee is to review and advise on the list of Medications on the CPA and provide support to the DON/M who authorises the CPA on behalf of the health service provider. This reflects its advisory and supportive role with regard to nurse midwife prescribing. The framework in place for use of authorised medicines prescribed for unauthorised indications (off label) as currently established should be retained.
28 5. The D&T committee or the relevant review group is provided with clear directions regarding its role and function specific to nurse and midwife prescribing. This will provide for a national consistent standard involving a tripartheid approach from the DoH, healthcare regulation and HSE.
29 6. The NMBI should revise its professional guidance documents on prescriptive authority. The ONMSD should revise its National Policy for Nurse and Midwife Medicinal Product Prescribing (2012) and accompanying guidelines.
30 7. Each health service provider should have an agreed schedule for routine audit of nurse/midwife prescribing as part of its overall organisational audit programme for prescribing and medication management. The NMPDCS should continue to be available for local use as a support for monitoring and clinical audit of RNP prescribing practice. The HSE national mandatory requirement for RNPs to input their prescriptions into this system should be removed. 8. The HSE ONMSD to engage with HSE ICT regarding the potential to further develop the NMPDCS to generate electronic prescriptions. This would be in collaboration with relevant stakeholders involved in ehealth strategy, eg DoH/HSE.
31 9. The Department of Health to amend the legislative authority for RNPs to prescribe exempt (unauthorised) medicines and draft regulations to enable this. The health service provider should utilise the existing HSE and NMBI guidance frameworks for the use of medicines for unauthorised indication for managing the implementation of exempt medicine prescribing by RNPs.
32 10. Based upon findings of consultation activities regarding exempt and off label products, communication from Advisory Group should be circulated to stakeholders regarding: a. The provision of information updates about exempt (unauthorised) medicines and unauthorised indication medicine usage (off label). b. RNP use of Irish medicine references such as HPRA and the Irish Medicines Formulary vs reliance on UK sources (British National Formulary BNF).
33 11. a) NMBI to examine a process for competency assurance for addressing the current issue of long term candidate nurse prescribers (i.e. candidates who have successfully completed the education programmes but not yet registered as RNP). 11. b) NMBI to examine a process for competency assurance for addressing the current issue of RNPs who have not utilised their prescriptive authority, i.e. those returning from long term leave /maternity leave/career break etc.
34 12. The Director of Nursing/Midwifery/Public Health Nursing/Services must have overall responsibility and authority for the governance of nurse and midwife prescribing to ensure due diligence in their health service provider. This should be in collaboration with Chief Executive Officers (group hospitals), Chief Officer (Community Health Organisations), Superintendent Pharmacist and Clinical Directors as appropriate.
35 Review of CPA guidance document and forms Communication from NMBI regarding CPA annual/biannual review Information provision regarding off label and exempt medicines - reflecting Review findings Development of guidance for D&T committees Communication regarding Nurse Midwife Prescribing Data Collection System Revision of existing guidance for audit practices
36 National Medicinal Product Prescribing Policy revision Ongoing communication with HSE ICT Liaising with DoH regarding exempt (unauthorised) medicines Competency assurance for RNPs Linking with developments of NMBI CC schemes Explore supports for inactive RNPs who are not prescribing Long term candidates not yet registered Guidance to Directors of N/M/PHN services for ongoing and future governance for nurse midwife prescribing. RNP Capacity Building
37 Clare MacGabhann Director of Nursing and Midwifery (Prescribing) Office of the Nursing and Midwifery Services Director HSE South Office: Phone: Maureen Nolan Director of Nursing and National Lead (Prescribing of Ionising Radiation) Office of the Nursing and Midwifery Services Director HSE Dublin MidLeinster Phone: Rose Lorenz Assistant Director of Nursing and Midwifery (Prescribing and Continuing Education) Office of the Nursing and Midwifery Services Director HSE Dublin North East Office: Phone: Annette Cuddy Assistant Director of Nursing and Midwifery (Prescribing) Office of the Nursing and Midwifery Services Director HSE West Office: Phone:
38
Maureen Nolan Director of Nursing National Lead for the Implementation and Audit of Nurse Prescribing of Ionising Radiation and Medicinal Prescribing
 Maureen Nolan Director of Nursing National Lead for the Implementation and Audit of Nurse Prescribing of Ionising Radiation and Medicinal Prescribing Dublin Mid Leinster Prescribing Team Clare MacGabhann,
Maureen Nolan Director of Nursing National Lead for the Implementation and Audit of Nurse Prescribing of Ionising Radiation and Medicinal Prescribing Dublin Mid Leinster Prescribing Team Clare MacGabhann,
(4th Edition, 2016) COLLABORATIVE PRACTICE AGREEMENT GUIDANCE FOR NURSES AND MIDWIVES WITH PRESCRIPTIVE AUTHORITY
 COLLABORATIVE PRACTICE AGREEMENT GUIDANCE FOR NURSES AND MIDWIVES WITH PRESCRIPTIVE AUTHORITY (4th Edition, 2016) 1 Prescriptive Authority for Nurses and Midwives 1. Overview of the purpose and extent
COLLABORATIVE PRACTICE AGREEMENT GUIDANCE FOR NURSES AND MIDWIVES WITH PRESCRIPTIVE AUTHORITY (4th Edition, 2016) 1 Prescriptive Authority for Nurses and Midwives 1. Overview of the purpose and extent
Collaborative Practice Agreement (CPA) for Nurses and Midwives with Prescriptive Authority, 3rd ed
 Collaborative Practice Agreement (CPA) for Nurses and Midwives with Prescriptive Authority, 3rd ed Item type Authors Publisher Book An Bord Altranais (ABA) An Bord Altranais Downloaded 30-Jun-2018 05:13:41
Collaborative Practice Agreement (CPA) for Nurses and Midwives with Prescriptive Authority, 3rd ed Item type Authors Publisher Book An Bord Altranais (ABA) An Bord Altranais Downloaded 30-Jun-2018 05:13:41
Practice standards for nurses and midwives with prescriptive authority
 Practice standards for nurses and midwives with prescriptive authority Item type Authors Publisher Book An Bord Altranais (ABA) An Bord Altranais Downloaded 30-Jun-2018 05:13:36 Link to item http://hdl.handle.net/10147/91385
Practice standards for nurses and midwives with prescriptive authority Item type Authors Publisher Book An Bord Altranais (ABA) An Bord Altranais Downloaded 30-Jun-2018 05:13:36 Link to item http://hdl.handle.net/10147/91385
Collaborative practice agreement for nurses and midwives with prescriptive authority (CPA) second edition
 Collaborative practice agreement for nurses and midwives with prescriptive authority (CPA) second edition Item Type Other Authors An Bord Altranais (ABA) Publisher An Bord Altranais Download date 25/08/2018
Collaborative practice agreement for nurses and midwives with prescriptive authority (CPA) second edition Item Type Other Authors An Bord Altranais (ABA) Publisher An Bord Altranais Download date 25/08/2018
National Nurse and Midwife Medicinal Product Prescribing Policy
 National Nurse and Midwife Medicinal Product Prescribing Policy Office of the Nursing and Midwifery Services Director, Health Service Executive Changing practice to support service delivery Clinical Strategy
National Nurse and Midwife Medicinal Product Prescribing Policy Office of the Nursing and Midwifery Services Director, Health Service Executive Changing practice to support service delivery Clinical Strategy
POLICY ON NURSE PRESCRIBING IN OLCHC EDITION 6
 POLICY ON NURSE PRESCRIBING IN OLCHC EDITION 6 Version Number 6 Date of Issue 23 rd December 2015 Reference Number Review Interval Approved By Name: Rachel Kenna Title: Director of Nursing Title: Drugs
POLICY ON NURSE PRESCRIBING IN OLCHC EDITION 6 Version Number 6 Date of Issue 23 rd December 2015 Reference Number Review Interval Approved By Name: Rachel Kenna Title: Director of Nursing Title: Drugs
Post Graduate Certificate in Nursing and Midwifery Information, Registration and Frequently Asked Questions
 Post Graduate Certificate in Nursing and Midwifery Information, Registration and Frequently Asked Questions For Graduate Staff Nurses and Graduate Staff Midwives December 2013 Introduction The Office of
Post Graduate Certificate in Nursing and Midwifery Information, Registration and Frequently Asked Questions For Graduate Staff Nurses and Graduate Staff Midwives December 2013 Introduction The Office of
National policy for nurse and midwife medicinal product prescribing in primary, community and continuing care
 National policy for nurse and midwife medicinal product prescribing in primary, community and continuing care Item type Authors Publisher Report Health Service Executive (HSE) Office of the Nursing Services
National policy for nurse and midwife medicinal product prescribing in primary, community and continuing care Item type Authors Publisher Report Health Service Executive (HSE) Office of the Nursing Services
Niltional Policy for Nurn and Midwifa Medicinal Produ!:t PrescrIbing
 1 (Idlb,aBNdt.lllSdrta&e!Mllie """"... Niltional Policy for Nurn and Midwifa Medicinal Produ!:t PrescrIbing Document ~SEJNMPf011O Document d... ltlld by: 0Ifk.e 01 hi NU...I~ and MkIwile
1 (Idlb,aBNdt.lllSdrta&e!Mllie """"... Niltional Policy for Nurn and Midwifa Medicinal Produ!:t PrescrIbing Document ~SEJNMPf011O Document d... ltlld by: 0Ifk.e 01 hi NU...I~ and MkIwile
2006 No. 915 MEDICINES. The Medicines for Human Use (Prescribing) (Miscellaneous Amendments) Order 2006
 STATUTORY INSTRUMENTS 2006 No. 915 MEDICINES The Medicines for Human Use (Prescribing) (Miscellaneous Amendments) Order 2006 Made - - - - 23rd March 2006 Laid before Parliament 3rd April 2006 Coming into
STATUTORY INSTRUMENTS 2006 No. 915 MEDICINES The Medicines for Human Use (Prescribing) (Miscellaneous Amendments) Order 2006 Made - - - - 23rd March 2006 Laid before Parliament 3rd April 2006 Coming into
An Bord Altranais. Download date 19/09/ :12:48. Find this and similar works at -
 Information for nurses and midwives educated and trained overseas in non-eu countries and applicants within EU member states who do not qualify under EU directives: regarding eligibility to register with
Information for nurses and midwives educated and trained overseas in non-eu countries and applicants within EU member states who do not qualify under EU directives: regarding eligibility to register with
The introduction of nurse and midwife prescribing inireland: an overview
 The introduction of nurse and midwife prescribing inireland: an overview Item type Authors Other An Bord Altranais (ABA), Health Service Executive, Department of Health & Children, National Council for
The introduction of nurse and midwife prescribing inireland: an overview Item type Authors Other An Bord Altranais (ABA), Health Service Executive, Department of Health & Children, National Council for
Royal College of Surgeons in Ireland
 Royal College of Surgeons in Ireland School of Nursing Certificate in Nursing (Nurse / Midwife Prescribing) (Minor Award, level 8) Application Form October 2010 Please attach 1 passport photograph to this
Royal College of Surgeons in Ireland School of Nursing Certificate in Nursing (Nurse / Midwife Prescribing) (Minor Award, level 8) Application Form October 2010 Please attach 1 passport photograph to this
MSc Nursing Advanced Nursing Practice: Overview
 MSc Nursing Advanced Nursing Practice: Overview Dr Patrick Cotter DN, MSc, HDipN(A&E), BSc, Dip Mgt&ER, RGN, RM, RNP, RANP Advanced Nurse Practitioner, Emergency Dept, CUH Lecturer Practitioner Advanced
MSc Nursing Advanced Nursing Practice: Overview Dr Patrick Cotter DN, MSc, HDipN(A&E), BSc, Dip Mgt&ER, RGN, RM, RNP, RANP Advanced Nurse Practitioner, Emergency Dept, CUH Lecturer Practitioner Advanced
NON-MEDICAL PRESCRIBING POLICY
 NON-MEDICAL PRESCRIBING POLICY To be read in conjunction with the Medicines Policy, Controlled Drug Policy and the FP10 Prescribing Forms Policy Version: 5 Date of issue: August 2017 Review date: August
NON-MEDICAL PRESCRIBING POLICY To be read in conjunction with the Medicines Policy, Controlled Drug Policy and the FP10 Prescribing Forms Policy Version: 5 Date of issue: August 2017 Review date: August
IONISING RADIATION (NURSE PRESCRIPTIVE AUTHORITY)
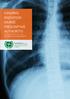 IONISING RADIATION (NURSE PRESCRIPTIVE AUTHORITY) Standards and requirements 1 These standards and requirements were originally published in February 2008 as Requirements and Standards for Nurse Education
IONISING RADIATION (NURSE PRESCRIPTIVE AUTHORITY) Standards and requirements 1 These standards and requirements were originally published in February 2008 as Requirements and Standards for Nurse Education
Practice Standards and Guidelines for Nurses and Midwives with Prescriptive Authority (3rd Edition)
 Practice Standards and Guidelines for Nurses and Midwives with Prescriptive Authority (3rd Edition) Contents INTRODUCTION 2 Medicines Legislation for Nurse/Midwife Prescribing 2 Professional Regulation
Practice Standards and Guidelines for Nurses and Midwives with Prescriptive Authority (3rd Edition) Contents INTRODUCTION 2 Medicines Legislation for Nurse/Midwife Prescribing 2 Professional Regulation
Clinical Nurse Specialist and Advanced Nurse Practitioner roles in Emergency Departments
 POSITION PAPER APRIL 2005 Clinical Nurse Specialist and Advanced Nurse Practitioner roles in Emergency Departments The development of specialist and advanced practice roles in Ireland is part of the strategic
POSITION PAPER APRIL 2005 Clinical Nurse Specialist and Advanced Nurse Practitioner roles in Emergency Departments The development of specialist and advanced practice roles in Ireland is part of the strategic
Nurse and Midwife Prescribing Data Collection System. Changing practice to support service delivery. Office of the Nursing Services Director
 Changing practice to support service delivery Office of the Nursing Services Director Introduction Irish law was amended in May 2007 to give prescriptive authority to nurses and midwives under specific
Changing practice to support service delivery Office of the Nursing Services Director Introduction Irish law was amended in May 2007 to give prescriptive authority to nurses and midwives under specific
Post Graduate Certificate in Nursing and Midwifery
 Post Graduate Certificate in Nursing and Midwifery for Graduate Staff Nurses/Midwives Presented by Ms Mary Wynne & Ms Ina Crowley in partnership with RCSI Overview of the Office of Nursing and Midwifery
Post Graduate Certificate in Nursing and Midwifery for Graduate Staff Nurses/Midwives Presented by Ms Mary Wynne & Ms Ina Crowley in partnership with RCSI Overview of the Office of Nursing and Midwifery
Medicine Protocol for the Administration of HPV vaccine (Gardasil) by registered nurses and registered midwives
 Medicine Protocol for the Administration of HPV vaccine (Gardasil) by registered nurses and registered midwives This medicine protocol is a specific written instruction for the administration of Gardasil,
Medicine Protocol for the Administration of HPV vaccine (Gardasil) by registered nurses and registered midwives This medicine protocol is a specific written instruction for the administration of Gardasil,
Requirements and standards for education programmes for nurses and midwives with prescriptive authority
 Requirements and standards for education programmes for nurses and midwives with prescriptive authority Item type Authors Publisher Book An Bord Altranais (ABA) An Bord Altranais Downloaded 6-Jun-2018
Requirements and standards for education programmes for nurses and midwives with prescriptive authority Item type Authors Publisher Book An Bord Altranais (ABA) An Bord Altranais Downloaded 6-Jun-2018
Medicine Protocol for the Administration of Inactivated Influenza Vaccine (Split Virion) BP Version 1, June 2017
 Medicine Protocol for the Administration of Inactivated Influenza Vaccine (Split Virion) BP to nurses, midwives, healthcare workers, agency staff, contract workers and volunteers by registered nurses and
Medicine Protocol for the Administration of Inactivated Influenza Vaccine (Split Virion) BP to nurses, midwives, healthcare workers, agency staff, contract workers and volunteers by registered nurses and
Non- medical Prescribing Policy
 Non- medical Prescribing Policy February 2010 Policy Title: Non-medical Prescribing Policy Policy Reference Number: Acute10/005 Implementation Date: February 2010 Review Date: February 2012 Responsible
Non- medical Prescribing Policy February 2010 Policy Title: Non-medical Prescribing Policy Policy Reference Number: Acute10/005 Implementation Date: February 2010 Review Date: February 2012 Responsible
Dublin: Nursing and Midwifery Board of Ireland available at:
 Medicine Protocol for the Administration of IPV Boostrix (Diphtheria, Tetanus, Pertussis and Poliomyelitis) vaccine (adsorbed, reduced antigen(s) content) by registered nurses and registered midwives This
Medicine Protocol for the Administration of IPV Boostrix (Diphtheria, Tetanus, Pertussis and Poliomyelitis) vaccine (adsorbed, reduced antigen(s) content) by registered nurses and registered midwives This
Prescribing in Specialist Palliative Care Our Journey
 Prescribing in Specialist Palliative Care Our Journey M A I M U R P H Y C N S R N P M A R Y C O R C O R A N C N S R N P L A O I S / O F F A L Y S P E C I A L I S T P A L L I A T I V E C A R E S E R V I
Prescribing in Specialist Palliative Care Our Journey M A I M U R P H Y C N S R N P M A R Y C O R C O R A N C N S R N P L A O I S / O F F A L Y S P E C I A L I S T P A L L I A T I V E C A R E S E R V I
Framework for the establishment of clinical nurse / midwife specialist posts: intermediate pathway - 3rd ed. (778 KB)
 Framework for the establishment of clinical nurse / midwife specialist posts: intermediate pathway - 3rd ed. (778 KB) Item type Authors Rights Report National Council for the Professional Development of
Framework for the establishment of clinical nurse / midwife specialist posts: intermediate pathway - 3rd ed. (778 KB) Item type Authors Rights Report National Council for the Professional Development of
Application form for Stand Alone Modules School of Nursing & Midwifery National University of Ireland, Galway
 School of Nursing & Midwifery National University of Ireland, Galway Postgraduate Stand Alone Module Application Form Important! Please read the supporting information sheet attached to this application
School of Nursing & Midwifery National University of Ireland, Galway Postgraduate Stand Alone Module Application Form Important! Please read the supporting information sheet attached to this application
SPONSORSHIP APPLICATION 2018 PUBLIC HEALTH SERVICE EMPLOYEES WISHING TO TRAIN AS NURSES/MIDWIVES HSE HR Circular 009/2010
 SPONSORSHIP APPLICATION 2018 PUBLIC HEALTH SERVICE EMPLOYEES WISHING TO TRAIN AS NURSES/MIDWIVES HSE HR Circular 009/2010 Closing date for completed applications is: 5pm Friday 15 th June 2018 Before you
SPONSORSHIP APPLICATION 2018 PUBLIC HEALTH SERVICE EMPLOYEES WISHING TO TRAIN AS NURSES/MIDWIVES HSE HR Circular 009/2010 Closing date for completed applications is: 5pm Friday 15 th June 2018 Before you
Office of Nursing and Midwifery Services Priorities Patrick Glackin Area Director of NMPD, West
 Office of Nursing and Midwifery Services Priorities 2017 Patrick Glackin Area Director of NMPD, West 1 ONMSD Role and Function provides professional guidance and expertise at a corporate level and to health
Office of Nursing and Midwifery Services Priorities 2017 Patrick Glackin Area Director of NMPD, West 1 ONMSD Role and Function provides professional guidance and expertise at a corporate level and to health
A start date will be indicated at job offer stage.
 Early Years Inspector (Assistant Director of Public Health Nursing) Job Specification & Terms and Conditions Job Title and Early Years Inspector (Assistant Director of Public Health Nursing) Grade (Grade
Early Years Inspector (Assistant Director of Public Health Nursing) Job Specification & Terms and Conditions Job Title and Early Years Inspector (Assistant Director of Public Health Nursing) Grade (Grade
CLOSING DATE: 13 th December 2013
 Royal College of Surgeons in Ireland Coláiste Ríoga na Máinleá in Éirinn POSITION: EXECUTIVE DIRECTOR, FACULTY OF NURSING & MIDWIFERY CLOSING DATE: 13 th December 2013 EDUCATIONAL EXCELLENCE IN SURGERY
Royal College of Surgeons in Ireland Coláiste Ríoga na Máinleá in Éirinn POSITION: EXECUTIVE DIRECTOR, FACULTY OF NURSING & MIDWIFERY CLOSING DATE: 13 th December 2013 EDUCATIONAL EXCELLENCE IN SURGERY
Changes to midwives exemptions
 NMC circular 07/2011 Issue date 17 June 2011 Review date 17 June 2013 Replaces circular 06/2010 Category Midwifery Status Action Changes to midwives exemptions Summary On 1 July 2011, amending legislation
NMC circular 07/2011 Issue date 17 June 2011 Review date 17 June 2013 Replaces circular 06/2010 Category Midwifery Status Action Changes to midwives exemptions Summary On 1 July 2011, amending legislation
Guideline for Funding Support for Continuing Professional Development for Nurses & Midwives
 Guideline for Funding Support for Continuing Professional Development for Nurses & Midwives NURSING & MIDWIFERY PLANNING & DEVELOPMENT UNIT (CORK/KERRY) HEALTH SERVICE EXECUTIVE SOUTH ADMINISTRATION BUILDING,
Guideline for Funding Support for Continuing Professional Development for Nurses & Midwives NURSING & MIDWIFERY PLANNING & DEVELOPMENT UNIT (CORK/KERRY) HEALTH SERVICE EXECUTIVE SOUTH ADMINISTRATION BUILDING,
Non Medical Prescribing Policy
 Non Medical Prescribing Policy Author: Sponsor/Executive: Responsible committee: Ratified by: Consultation & Approval: (Committee/Groups which signed off the policy, including date) This document replaces:
Non Medical Prescribing Policy Author: Sponsor/Executive: Responsible committee: Ratified by: Consultation & Approval: (Committee/Groups which signed off the policy, including date) This document replaces:
Framework for the establishment of advanced nurse practitioner posts - 3rd ed. (605 KB)
 Framework for the establishment of advanced nurse practitioner posts - 3rd ed. (605 KB) Item Type Report Authors National Council for the Professional Development of Nursing and Midwifery Rights National
Framework for the establishment of advanced nurse practitioner posts - 3rd ed. (605 KB) Item Type Report Authors National Council for the Professional Development of Nursing and Midwifery Rights National
Standards of proficiency for nurse and midwife prescribers
 Standards of proficiency for nurse and midwife prescribers Protecting the public through professional standards Contents Introduction... 4 Legislation and terminology... 4 Standards of proficiency for
Standards of proficiency for nurse and midwife prescribers Protecting the public through professional standards Contents Introduction... 4 Legislation and terminology... 4 Standards of proficiency for
Changes to midwives exemptions
 NMC circular 07/2011 Issue date 17 June 2011 Review date 17 June 2013 Replaces circular 06/2010 Category Midwifery Status Action Changes to midwives exemptions Summary On 1 July 2011, amending legislation
NMC circular 07/2011 Issue date 17 June 2011 Review date 17 June 2013 Replaces circular 06/2010 Category Midwifery Status Action Changes to midwives exemptions Summary On 1 July 2011, amending legislation
Changing practice to support service delivery
 A Guiding Framework for the Implementation of Nurse Prescribing of Medical Ionising Radiation (X-Ray) in Ireland Changing practice to support service delivery Office of the Nursing Services Director ISBN
A Guiding Framework for the Implementation of Nurse Prescribing of Medical Ionising Radiation (X-Ray) in Ireland Changing practice to support service delivery Office of the Nursing Services Director ISBN
All areas of the Trust All Trust staff All Patients Deputy Chief Nurse & Chief Pharmacist Final
 Trust Policy and Procedure Document Ref. No: PP(15)233 Non-Medical Prescribing Policy For use in: For use by: For use for: Document owner: Status: All areas of the Trust All Trust staff All Patients Deputy
Trust Policy and Procedure Document Ref. No: PP(15)233 Non-Medical Prescribing Policy For use in: For use by: For use for: Document owner: Status: All areas of the Trust All Trust staff All Patients Deputy
St. John s Hospital Limerick. Job Description
 St. John s Hospital Limerick Job Description JOB TITLE: REPORTS TO: Director of Nursing Chief Executive Role Summary The Director of Nursing (DON) is part of the Hospital Senior Management Team that manages
St. John s Hospital Limerick Job Description JOB TITLE: REPORTS TO: Director of Nursing Chief Executive Role Summary The Director of Nursing (DON) is part of the Hospital Senior Management Team that manages
Methods available for the delivery of medicines to patients
 Chapter 1 Methods available for the delivery of medicines to patients A number of different methods are available to healthcare professionals for the delivery of medicines to patients. These methods include
Chapter 1 Methods available for the delivery of medicines to patients A number of different methods are available to healthcare professionals for the delivery of medicines to patients. These methods include
Nursing and Midwifery Services Director - Job Specification, Terms and Conditions
 Nursing and Midwifery Services Director - Job Specification, Terms and Conditions Job Title and Grade Nursing and Midwifery Services Director (Assistant National Director Level) Closing Date 12.00pm Friday,
Nursing and Midwifery Services Director - Job Specification, Terms and Conditions Job Title and Grade Nursing and Midwifery Services Director (Assistant National Director Level) Closing Date 12.00pm Friday,
COMPETENCE ASSESSMENT TOOL FOR MIDWIVES
 Nursing and Midwifery Board of Ireland (NMBI) COMPETENCE ASSESSMENT TOOL FOR MIDWIVES 1 The has been developed for midwives educated and trained outside Ireland who do not qualify for registration under
Nursing and Midwifery Board of Ireland (NMBI) COMPETENCE ASSESSMENT TOOL FOR MIDWIVES 1 The has been developed for midwives educated and trained outside Ireland who do not qualify for registration under
Non Medical Prescribing Policy Register No: Status: Public
 Non Medical Prescribing Policy Policy Register No: 07049 Status: Public Developed in response to: Department of Health Policies, Prescribing Guidance & Legislation Contributes to CQC Outcome: 9 Consulted
Non Medical Prescribing Policy Policy Register No: 07049 Status: Public Developed in response to: Department of Health Policies, Prescribing Guidance & Legislation Contributes to CQC Outcome: 9 Consulted
Recruitment & Retention of Health Professionals Return to Nursing Practice Education Programme 17 th March 2016
 Recruitment & Retention of Health Professionals Return to Nursing Practice Education Programme 17 th March 2016 SANDRA MCCARTHY, HEAD OF LEARNING & DEVELOPMENT, TALLAGHT HOSPITAL, DUBLIN Happy St. Patricks
Recruitment & Retention of Health Professionals Return to Nursing Practice Education Programme 17 th March 2016 SANDRA MCCARTHY, HEAD OF LEARNING & DEVELOPMENT, TALLAGHT HOSPITAL, DUBLIN Happy St. Patricks
Clinical Placement Coordinator (Mental Health) Job Specification, Terms and Conditions
 Clinical Placement Coordinator (Mental Health) Job Specification, Terms and Conditions Job Title and Grade Campaign Reference Clinical Placement Coordinator (Mental Health) (Grade Code: 241Y) NRS04110
Clinical Placement Coordinator (Mental Health) Job Specification, Terms and Conditions Job Title and Grade Campaign Reference Clinical Placement Coordinator (Mental Health) (Grade Code: 241Y) NRS04110
Nurse prescribing in substance misuse February 2005, updated May 2005
 Nurse prescribing in substance misuse February 2005, updated May 2005 1. Introduction This briefing aims to clarify the current situation in relation to nurse prescribing in the substance misuse sector.
Nurse prescribing in substance misuse February 2005, updated May 2005 1. Introduction This briefing aims to clarify the current situation in relation to nurse prescribing in the substance misuse sector.
Non-Medical Prescriber Registration Policy
 Non-Medical Prescriber Registration Policy REFERENCE NUMBER VERSION V1b APPROVING COMMITTEE Clinical Executive Committee & DATE 11.08.2015 REVIEW DUE DATE August 2018 V1a change of CSU email address April
Non-Medical Prescriber Registration Policy REFERENCE NUMBER VERSION V1b APPROVING COMMITTEE Clinical Executive Committee & DATE 11.08.2015 REVIEW DUE DATE August 2018 V1a change of CSU email address April
Practising as a midwife in the UK
 Practising as a midwife in the UK An overview of midwifery regulation CONTENTS Introduction 3 Section 1: Education 4 Section 2: Joining the register and maintaining registration 6 Section 3: Standards
Practising as a midwife in the UK An overview of midwifery regulation CONTENTS Introduction 3 Section 1: Education 4 Section 2: Joining the register and maintaining registration 6 Section 3: Standards
Clinical Governance Group for the HSE Home Birth Service. Document reference number. developed by. Revision number
 Guideline for the Continuing Professional, Clinical Education and Development Needs and Requirements of the Self-Employed Community Midwife providing Home Birth Services on behalf of the Health Service
Guideline for the Continuing Professional, Clinical Education and Development Needs and Requirements of the Self-Employed Community Midwife providing Home Birth Services on behalf of the Health Service
Non-Medical Prescribing
 Non-Medical Prescribing Registration Policy Dr Lisa Rogan 9/11/2016 Review Date: November 2019 Version 1 This policy outlines the CCG authorisation process required to add and maintain a nonmedical prescriber
Non-Medical Prescribing Registration Policy Dr Lisa Rogan 9/11/2016 Review Date: November 2019 Version 1 This policy outlines the CCG authorisation process required to add and maintain a nonmedical prescriber
PSYCHIATRIC NURSE POST REGISTRATION PROGRAMME
 Nursing and Midwifery Board of Ireland (NMBI) PSYCHIATRIC NURSE POST REGISTRATION PROGRAMME Standards and requirements 1 These standards and requirements were originally published in 2007 as Requirements
Nursing and Midwifery Board of Ireland (NMBI) PSYCHIATRIC NURSE POST REGISTRATION PROGRAMME Standards and requirements 1 These standards and requirements were originally published in 2007 as Requirements
IRISH ASSOCIATION OF DIRECTORS OF NURSING AND MIDWIFERY. ARTICLES OF ASSOCIATION
 IRISH ASSOCIATION OF DIRECTORS OF NURSING AND MIDWIFERY. ARTICLES OF ASSOCIATION September 2011 Articles of Association Irish Association of Directors of Nursing & Midwifery September 2011 Definitions
IRISH ASSOCIATION OF DIRECTORS OF NURSING AND MIDWIFERY. ARTICLES OF ASSOCIATION September 2011 Articles of Association Irish Association of Directors of Nursing & Midwifery September 2011 Definitions
JOB DESCRIPTION. Director of Midwifery / Nursing. Department of Midwifery / Nursing. Director of Midwifery / Nursing
 JOB DESCRIPTION Director of Midwifery / Nursing Department: Title of Post: Accountable to: Department of Midwifery / Nursing Director of Midwifery / Nursing The Master (Chief Executive Officer) The Director
JOB DESCRIPTION Director of Midwifery / Nursing Department: Title of Post: Accountable to: Department of Midwifery / Nursing Director of Midwifery / Nursing The Master (Chief Executive Officer) The Director
Liaison / Coordinator Nurse for Spinal Injury Pathway, Spinal Cord System of Care Programme at NRH. Permanent, Full time
 Liaison / Coordinator Nurse for Spinal Injury Pathway, Spinal Cord System of Care Programme at NRH. Permanent, Full time Particulars of Office (Generic) 1. The appointment permanent, whole time and pensionable.
Liaison / Coordinator Nurse for Spinal Injury Pathway, Spinal Cord System of Care Programme at NRH. Permanent, Full time Particulars of Office (Generic) 1. The appointment permanent, whole time and pensionable.
(b) Is administered via a transdermal route; or
 ACTION: To Be Refiled DATE: 10/10/2018 2:31 PM 4723-9-10 Formulary; standards of prescribing for advanced practice registered nurses designated as clinical nurse specialists, certified nurse-midwives,
ACTION: To Be Refiled DATE: 10/10/2018 2:31 PM 4723-9-10 Formulary; standards of prescribing for advanced practice registered nurses designated as clinical nurse specialists, certified nurse-midwives,
Non-Medical Prescribing
 Non-Medical Prescribing Registration Policy Dr Lisa Rogan 9/11/2016 Review Date: November 2019 Version 7 updated January 18 v7.1 This policy outlines the CCG authorisation process required to add and maintain
Non-Medical Prescribing Registration Policy Dr Lisa Rogan 9/11/2016 Review Date: November 2019 Version 7 updated January 18 v7.1 This policy outlines the CCG authorisation process required to add and maintain
Fact sheet: New obligations for Nurses and Midwives
 Fact sheet: New obligations for Nurses and Midwives Registration standards The Nursing and Midwifery Board of Australia (the Board) has developed registration standards, which have been approved by the
Fact sheet: New obligations for Nurses and Midwives Registration standards The Nursing and Midwifery Board of Australia (the Board) has developed registration standards, which have been approved by the
This is a repository copy of Non-medical prescribing in palliative care: a regional survey.
 This is a repository copy of Non-medical prescribing in palliative care: a regional survey. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/879/ Version: Accepted Version
This is a repository copy of Non-medical prescribing in palliative care: a regional survey. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/879/ Version: Accepted Version
HIQA s Medication Safety Monitoring Programme in Public Acute Hospitals. One Year Later
 HIQA s Medication Safety Monitoring Programme in Public Acute Hospitals One Year Later Sean Egan Head of Healthcare Regulation Health Information and Quality Authority Presentation outline Recap on the
HIQA s Medication Safety Monitoring Programme in Public Acute Hospitals One Year Later Sean Egan Head of Healthcare Regulation Health Information and Quality Authority Presentation outline Recap on the
Requirements and standards for the midwife registration education programme (replaced by 2005 edition)
 Requirements and standards for the midwife registration education programme (replaced by 2005 edition) Item Type Report Authors An Bord Altranais (ABA) Rights An Bord Altranais Download date 08/10/2018
Requirements and standards for the midwife registration education programme (replaced by 2005 edition) Item Type Report Authors An Bord Altranais (ABA) Rights An Bord Altranais Download date 08/10/2018
Transcribing Medicines for Adults Policy. Policy Register No:09076 Status: Public. NHSLA Risk Assessment standards
 ` Transcribing Medicines for Adults Policy Policy Register No:09076 Status: Public Developed in response to: Contributes to CQC Core Standard number: Dept of Health Medicines Regulations, NHSLA Risk Assessment
` Transcribing Medicines for Adults Policy Policy Register No:09076 Status: Public Developed in response to: Contributes to CQC Core Standard number: Dept of Health Medicines Regulations, NHSLA Risk Assessment
Non-Medical Prescribing Policy
 Non-Medical Prescribing Policy This policy describes the context in which qualified non-medical prescribers may prescribe, sets out individual roles and responsibilities in relation to non-medical prescribing
Non-Medical Prescribing Policy This policy describes the context in which qualified non-medical prescribers may prescribe, sets out individual roles and responsibilities in relation to non-medical prescribing
Best Practice Guidance for Supplementary Prescribing by Nurses Within the HPSS in Northern Ireland. patient CMP
 Best Practice Guidance for Supplementary Prescribing by Nurses Within the HPSS in Northern Ireland patient CMP nurse doctor For further information relating to Nurse Prescribing please contact the Nurse
Best Practice Guidance for Supplementary Prescribing by Nurses Within the HPSS in Northern Ireland patient CMP nurse doctor For further information relating to Nurse Prescribing please contact the Nurse
APPLICATION FOR NON-MEDICAL PRESCRIBING
 APPLICATION FOR NON-MEDICAL PRESCRIBING Sections 1, 2 & 3 All Applicants to complete Section 4 Only Independent/Supplementary Prescribing Applicants & their DMP to complete Section 5 Only Community Practitioner
APPLICATION FOR NON-MEDICAL PRESCRIBING Sections 1, 2 & 3 All Applicants to complete Section 4 Only Independent/Supplementary Prescribing Applicants & their DMP to complete Section 5 Only Community Practitioner
INFORMATION FOR ANP/AMP APPLICANTS
 1. INTRODUCTION Please read this booklet carefully before completing any part of the application form. http://www.nursingboard.ie/en/apply_registration.aspx Failure to do so may delay your application
1. INTRODUCTION Please read this booklet carefully before completing any part of the application form. http://www.nursingboard.ie/en/apply_registration.aspx Failure to do so may delay your application
Protocol for Completion of the Midwifery Practice Record Book
 Protocol for Completion of the Midwifery Practice Record Book Document reference number Revision number PPPGC- MND-5 Document approved by: Mary Doyle Document developed by Mary Doyle Midwifery Practice
Protocol for Completion of the Midwifery Practice Record Book Document reference number Revision number PPPGC- MND-5 Document approved by: Mary Doyle Document developed by Mary Doyle Midwifery Practice
Appendix 2 to NMP policy Prescribing Governance Framework Standards for Supplementary and Independent Non-Medical Prescribers at SCH
 Appendix 2 to NMP policy Prescribing Governance Framework Standards for Supplementary and Independent Non-Medical Prescribers at SCH All prescribers and their managers/professional leads should ensure
Appendix 2 to NMP policy Prescribing Governance Framework Standards for Supplementary and Independent Non-Medical Prescribers at SCH All prescribers and their managers/professional leads should ensure
Non-Medical Prescriber Registration Policy
 Non-Medical Prescriber Registration Policy REFERENCE NUMBER Non medical prescribing policy VERSION V1 APPROVING COMMITTEE & DATE Clinical Executive Committee 4.8.15 REVIEW DUE DATE August 2018 1 1. Introduction
Non-Medical Prescriber Registration Policy REFERENCE NUMBER Non medical prescribing policy VERSION V1 APPROVING COMMITTEE & DATE Clinical Executive Committee 4.8.15 REVIEW DUE DATE August 2018 1 1. Introduction
Community Nurse Prescribing (V100) Portfolio of Evidence
 ` School of Health and Human Sciences Community Nurse Prescribing (V100) Portfolio of Evidence Start date: September 2016 Student Name: Student Number:. Practice Mentor:.. Personal Tutor:... Submission
` School of Health and Human Sciences Community Nurse Prescribing (V100) Portfolio of Evidence Start date: September 2016 Student Name: Student Number:. Practice Mentor:.. Personal Tutor:... Submission
Psychiatric Nurse. Competency Assessment Document (CAD) for the Undergraduate Nursing Student. Year One. (Pilot Document, 2017)
 Psychiatric Nurse Competency Assessment Document (CAD) for the Undergraduate Nursing Student Year One (Pilot Document, 2017) WELCOME TO YOUR COMPETENCY ASSESSMENT DOCUMENT This guide has been developed
Psychiatric Nurse Competency Assessment Document (CAD) for the Undergraduate Nursing Student Year One (Pilot Document, 2017) WELCOME TO YOUR COMPETENCY ASSESSMENT DOCUMENT This guide has been developed
Advisory Group of the Irish Association for Palliative Care. Department of Health (DoH) Download date 18/09/ :34:10
 The report of the advisory group of The Irish Association for Palliative Care: guidelines on the possession, supply and administration of controlled drugs by registered nurses working in the community
The report of the advisory group of The Irish Association for Palliative Care: guidelines on the possession, supply and administration of controlled drugs by registered nurses working in the community
SHAPING THE FUTURE OF INTELLECTUAL DISABILITY NURSING IN IRELAND
 Supporting people with an intellectual disability to live ordinary lives in ordinary places SHAPING THE FUTURE OF INTELLECTUAL DISABILITY NURSING IN IRELAND Commenced in 2013 Draft report 2016 Published
Supporting people with an intellectual disability to live ordinary lives in ordinary places SHAPING THE FUTURE OF INTELLECTUAL DISABILITY NURSING IN IRELAND Commenced in 2013 Draft report 2016 Published
Programme specification: MSc Advanced Practice (Health)
 Programme specification: MSc Advanced Practice (Health) 1. Awarding Institution/Body University of Gloucestershire 2. Teaching Institution University of Gloucestershire 3. Recognition by Professional Body.
Programme specification: MSc Advanced Practice (Health) 1. Awarding Institution/Body University of Gloucestershire 2. Teaching Institution University of Gloucestershire 3. Recognition by Professional Body.
Non-Medical Prescribing Policy December 2016
 1 Policy Title Policy Reference Number Acute17/002 Implementation Date Review Date 30 September 2018 or earlier subject to changes in legislation or review Responsible Officer Head of Pharmacy & Medicines
1 Policy Title Policy Reference Number Acute17/002 Implementation Date Review Date 30 September 2018 or earlier subject to changes in legislation or review Responsible Officer Head of Pharmacy & Medicines
Review of the Implementation of the Nurse Prescribing Role
 Review of the Implementation of the Nurse Prescribing Role On behalf of the Trust Nurses Association in Northern Ireland L.M. Barrowman TABLE OF CONTENTS Acknowledgements 4 Page No Executive Summary 5
Review of the Implementation of the Nurse Prescribing Role On behalf of the Trust Nurses Association in Northern Ireland L.M. Barrowman TABLE OF CONTENTS Acknowledgements 4 Page No Executive Summary 5
Module Name: Historical, Legal and Theoretical Foundations of Nursing Module Code: NU1S03 ECTS: 5 No. of Hours: 20
 Module Name: Historical, Legal and Theoretical Foundations of Nursing Module Code: NU1S03 ECTS: 5 No. of Hours: 20 Term: Michaelmas Assessment Date: Week 16 Module Leader: Dr Fiona Timmins Lecturers: Jacqueline
Module Name: Historical, Legal and Theoretical Foundations of Nursing Module Code: NU1S03 ECTS: 5 No. of Hours: 20 Term: Michaelmas Assessment Date: Week 16 Module Leader: Dr Fiona Timmins Lecturers: Jacqueline
March The Nursing and Midwifery Board of Ireland A Guide to Fitness to Practise
 The Nursing and Midwifery Board of Ireland A Guide to Fitness to Practise March 2017 The Nursing and Midwifery Board of Ireland A Guide to Fitness to Practise 1 The Nursing and Midwifery Board of Ireland
The Nursing and Midwifery Board of Ireland A Guide to Fitness to Practise March 2017 The Nursing and Midwifery Board of Ireland A Guide to Fitness to Practise 1 The Nursing and Midwifery Board of Ireland
NON-MEDICAL PRESCRIBING POLICY
 NON-MEDICAL PRESCRIBING POLICY PROCEDURE NUMBER Clinical.186 PROCEDURE VERSION 2 (Review). RATIFYING COMMITTEE Policy and Professional Practice Forum DATE RATIFIED 20 October 2015 DATE OF EQUALITY & September
NON-MEDICAL PRESCRIBING POLICY PROCEDURE NUMBER Clinical.186 PROCEDURE VERSION 2 (Review). RATIFYING COMMITTEE Policy and Professional Practice Forum DATE RATIFIED 20 October 2015 DATE OF EQUALITY & September
Registration prescribed information handbook
 Registration prescribed information handbook Guidance for registered providers submitting prescribed information as part of a registration pack or a registration notification form. October 2016 Page 2
Registration prescribed information handbook Guidance for registered providers submitting prescribed information as part of a registration pack or a registration notification form. October 2016 Page 2
Non-Medical Prescribing Strategy
 Non-Medical Prescribing Strategy 2014-2017 Nursing & Partnerships Directorate Page 1 of 13 Section Contents Page No. 1. STATEMENT OF INTENT 3 2. PURPOSE 3 3. SCOPE 3 4. BACKGROUND 3 5. STRATEGIC GOALS
Non-Medical Prescribing Strategy 2014-2017 Nursing & Partnerships Directorate Page 1 of 13 Section Contents Page No. 1. STATEMENT OF INTENT 3 2. PURPOSE 3 3. SCOPE 3 4. BACKGROUND 3 5. STRATEGIC GOALS
V100 Community Practitioner Nurse Prescriber
 MODULE SPECIFICATION KEY FACTS Module name V100 Community Practitioner Nurse Prescriber Module code PHM009 School School of Health Sciences Department or equivalent Department of Health Services Research
MODULE SPECIFICATION KEY FACTS Module name V100 Community Practitioner Nurse Prescriber Module code PHM009 School School of Health Sciences Department or equivalent Department of Health Services Research
Group Assistant Director of Nursing / Group Sepsis
 Job Description and Person Specification Group Assistant Director of Nursing / Group Sepsis Reference: IEHG/15/009 Permanent contract Closing Date: Monday, 7 th September 2015 Mary Day, Chief Executive
Job Description and Person Specification Group Assistant Director of Nursing / Group Sepsis Reference: IEHG/15/009 Permanent contract Closing Date: Monday, 7 th September 2015 Mary Day, Chief Executive
ALABAMA BOARD OF NURSING ADMINISTRATIVE CODE CHAPTER 610-X-5 ADVANCED PRACTICE NURSING COLLABORATIVE PRACTICE TABLE OF CONTENTS
 Nursing Chapter 610-X-5 ALABAMA BOARD OF NURSING ADMINISTRATIVE CODE CHAPTER 610-X-5 ADVANCED PRACTICE NURSING COLLABORATIVE PRACTICE TABLE OF CONTENTS 610-X-5-.01 610-X-5-.02 610-X-5-.03 610-X-5-.04 610-X-5-.05
Nursing Chapter 610-X-5 ALABAMA BOARD OF NURSING ADMINISTRATIVE CODE CHAPTER 610-X-5 ADVANCED PRACTICE NURSING COLLABORATIVE PRACTICE TABLE OF CONTENTS 610-X-5-.01 610-X-5-.02 610-X-5-.03 610-X-5-.04 610-X-5-.05
Return to Practice: A handbook for Nurses and Midwives
 Return to Practice: A handbook for Nurses and Midwives Introduction The Department of Health welcomes you to the Health Service Executive (HSE) as a valuable member of the healthcare team. The health
Return to Practice: A handbook for Nurses and Midwives Introduction The Department of Health welcomes you to the Health Service Executive (HSE) as a valuable member of the healthcare team. The health
LOCAL SUPERVISING AUTHORITY ANNUAL REPORT
 LOCAL SUPERVISING AUTHORITY ANNUAL REPORT 2006 Table of Contents 1.0 PURPOSE OF REPORT...1 2.0 ORGANISATION OF SUPERVISION OF MIDWIVES...1 2.1 Appointment of Supervisor of Midwives...1 2.2 Resignation/De-Selection
LOCAL SUPERVISING AUTHORITY ANNUAL REPORT 2006 Table of Contents 1.0 PURPOSE OF REPORT...1 2.0 ORGANISATION OF SUPERVISION OF MIDWIVES...1 2.1 Appointment of Supervisor of Midwives...1 2.2 Resignation/De-Selection
Reference. No. 02/16 Issue: 1 Page: 1 of 13 Issue Date: 16/05/16 Focal: Aircrew
 Page: 1 of 13 The purpose of this Memorandum is to outline the requirements as well as IAA instructions and procedures for the initial approval and ongoing maintenance of Cabin Crew Training Organisations
Page: 1 of 13 The purpose of this Memorandum is to outline the requirements as well as IAA instructions and procedures for the initial approval and ongoing maintenance of Cabin Crew Training Organisations
Revalidation for Nurses
 Why we have a procedure? Standard Operating Procedure 1 (SOP 1) Revalidation for Nurses An outcome of the Mid Staffordshire NHS Foundation Trust Public Inquiry chaired by Robert Francis QC (2013) was NMC
Why we have a procedure? Standard Operating Procedure 1 (SOP 1) Revalidation for Nurses An outcome of the Mid Staffordshire NHS Foundation Trust Public Inquiry chaired by Robert Francis QC (2013) was NMC
and Midwifery National Council for the Professional Development of Nursing and Midwifery Download date 24/09/ :31:36
 Framework for the establishment of advanced nurse practitioner and advanced midwife practitioner posts / National Council for the Professional Development of Nursing and Midwifery Item Type Report Authors
Framework for the establishment of advanced nurse practitioner and advanced midwife practitioner posts / National Council for the Professional Development of Nursing and Midwifery Item Type Report Authors
Developing roles, skills and competencies of nurses in Irish Acute Medical Units
 Developing roles, skills and competencies of nurses in Irish Acute Medical Units Richard Walsh. D.O.N. Lead, Nat. Acute Medicine Programme Avilene Casey. Exec. Performance Improvement Lead Unscheduled
Developing roles, skills and competencies of nurses in Irish Acute Medical Units Richard Walsh. D.O.N. Lead, Nat. Acute Medicine Programme Avilene Casey. Exec. Performance Improvement Lead Unscheduled
Progression Policy for Nursing Students when undertaking practice component of practice placement module. Document developed by
 APPROVED BY: Seamus Tuohy, Catherine Page 1 of 9 Progression Policy for Nursing Students when undertaking practice component of Document reference number Nurs/BSc/Und/10 /06 Document developed by GMIT/HSE
APPROVED BY: Seamus Tuohy, Catherine Page 1 of 9 Progression Policy for Nursing Students when undertaking practice component of Document reference number Nurs/BSc/Und/10 /06 Document developed by GMIT/HSE
JOB DESCRIPTION. Pharmacy Technician
 JOB DESCRIPTION Pharmacy Technician Issued by AT Medics Primary Care Pharmacy Technician Job Description Job Title: Reporting to: Location: Salary: Job status: Contract: Notice Period: Primary care pharmacy
JOB DESCRIPTION Pharmacy Technician Issued by AT Medics Primary Care Pharmacy Technician Job Description Job Title: Reporting to: Location: Salary: Job status: Contract: Notice Period: Primary care pharmacy
Volunteering in NHSScotland Future of the Volunteering Programme Draft Action Plan
 NG08-09 Volunteering in NHSScotland Future of the Volunteering Programme Draft Action Plan Introduction This paper provides an overview of the suggested activity that an extension of the Programme through
NG08-09 Volunteering in NHSScotland Future of the Volunteering Programme Draft Action Plan Introduction This paper provides an overview of the suggested activity that an extension of the Programme through
Uncontrolled when printed NHS AYRSHIRE & ARRAN CODE OF PRACTICE FOR MEDICINES GOVERNANCE. SECTION 9(a) UNLICENSED MEDICINES
 Uncontrolled when printed NHS AYRSHIRE & ARRAN CODE OF PRACTICE FOR MEDICINES GOVERNANCE SECTION 9(a) UNLICENSED MEDICINES BACKGROUND and PURPOSE Under the Medicines Act 1968 (EEC Directive 65/65), a company
Uncontrolled when printed NHS AYRSHIRE & ARRAN CODE OF PRACTICE FOR MEDICINES GOVERNANCE SECTION 9(a) UNLICENSED MEDICINES BACKGROUND and PURPOSE Under the Medicines Act 1968 (EEC Directive 65/65), a company
PgCert Neonatal Nurse Practitioner MSc/PgDip Advanced Neonatal Nurse Practitioner
 Plymouth University Faculty of Health and Human Sciences School of Nursing and Midwifery Programme Specification PgCert Neonatal Nurse Practitioner MSc/PgDip Advanced Neonatal Nurse Practitioner Special
Plymouth University Faculty of Health and Human Sciences School of Nursing and Midwifery Programme Specification PgCert Neonatal Nurse Practitioner MSc/PgDip Advanced Neonatal Nurse Practitioner Special
Effective utilisation of professional skills of nurses and midwives: report of the working group / chaired by Michael Shannon
 Effective utilisation of professional skills of nurses and midwives: report of the working group / chaired by Michael Shannon Item type Authors Rights Report Department of Health and Children (DOHC) DOHC
Effective utilisation of professional skills of nurses and midwives: report of the working group / chaired by Michael Shannon Item type Authors Rights Report Department of Health and Children (DOHC) DOHC
JOB DESCRIPTION & PERSON SPECIFICATION
 JOB DESCRIPTION & PERSON SPECIFICATION Physiotherapy Manager 1.0 WTE 1 PERSON SPECIFICATION Job Title: Physiotherapy Manager Department: Physiotherapy Report to/accountable Master of Coombe Women & Infants
JOB DESCRIPTION & PERSON SPECIFICATION Physiotherapy Manager 1.0 WTE 1 PERSON SPECIFICATION Job Title: Physiotherapy Manager Department: Physiotherapy Report to/accountable Master of Coombe Women & Infants
NMC programme of change for education Prescribing and standards for medicines management
 NMC programme of change for education Prescribing and standards for medicines management This response form relates to our consultation on nurse and midwifery prescribing competency proposals, programme
NMC programme of change for education Prescribing and standards for medicines management This response form relates to our consultation on nurse and midwifery prescribing competency proposals, programme
