Surgical site infections
|
|
|
- Mae Griffin
- 6 years ago
- Views:
Transcription
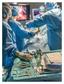
1 Surgical site infections The impact of SSIs on quality of care and the healthcare facility bottom line SSIs caused by airborne contaminants offer an under-addressed area for prevention Hospital operating rooms are among the most infection-sensitive environments in healthcare facilities. Surgical procedures increase patient vulnerability to pathogens transmitted from surgical personnel, surgical equipment, the air and a patient s own skin flora. Despite advancements in surgical techniques and infection-prevention methods, two out of every 100 surgeries in the U.S. result in surgical site infections (SSIs) 1, resulting in an estimated 290,485 surgical site infections per year. 2 Not all SSIs are entirely preventable. However, a better understanding of the impact SSIs have on quality of care and healthcare costs illustrates why additional preventive actions are needed. Likewise, an estimated 40 to 60 percent of SSIs are considered preventable based on current medical practice and technology. 3 Identifying the causes of surgical site infections helps focus on potential areas of improvement. While hygienerelated prevention is the most practiced and proven method, airborne-related contamination control offers one area that could play a much larger role in preventing SSIs. Sources of SSIs The majority of surgical site infections are a result of hygiene-related factors associated with surgical personnel, patient skin and surgical equipment. According to the Center for Healthcare Design (CHD), it is estimated that more than 30 percent of SSIs are caused by airborne pathogens. With respect to bacteria transmitted to the surgical site through the air, squames, or skin scales, are the primary source of transmission. 3 Approximately to 0.9 to 10 8 squames are generated in a typical two to four hour surgical procedure. 6 In the absence of controlled airflow in the operating room and, more importantly, over the sterile field, patients are at risk of squames circulating over and landing in or near the surgical site. Bacteria present in these skin scales are one cause of SSIs. When SSIs do occur, the impact on quality of care and healthcare costs can be significant. Quality of care impacts: Morbidity, mortality and public perception Surgical site infections represent 20 percent of all healthcare acquired infections 1. Impacts on quality of care include morbidity, extended hospital stays and, in extreme cases, mortality. Of the 290,485 SSIs reported in 2009, 13,088 resulted in death, or 4.5 percent of SSIs reported that year. 2 In all instances, the adverse impact of SSIs also has a cumulative effect on the public s perception of a hospital s quality of care. The number of states that mandate reporting of healthcare associated infections (including SSIs) has risen significantly in the past decade, giving healthcare recipients increased access to HAI incidence data at hospitals. Three states had mandated reporting in As of the beginning of 2011, that number has grown to 28 states. 7 Increasing transparency and public access to hospital performance can also be attributed to additional initiatives and incentives by the Centers for Medicare and Medicaid Services (CMS), the U.S. Department of Health and Human Services (HHS) and the Centers for Disease Control and Prevention (CDC). In addition to providing oversight, these organizations, CMS in particular, also play a role in the economic costs of surgical site infections.
2 Economic impacts and where the burdens of added costs fall Beginning October 1, 2008, Medicare can no longer assign an inpatient hospital discharge to a higher paying Medicareseverity diagnosis-related group (MS-DRG) if a selected condition is listed on the claim and was not present on admission. That is, the case will be paid as though the condition were not present. 2 This recent policy change by the CMS puts the financial burden of healthcare acquired conditions (HACs) squarely on the shoulders of hospitals that participate in the Medicare program. Surgical site infections are one of 10 categories of HACs that are no longer eligible for reimbursement. The above statement is part of the CMS s Preventable Hospital- Acquired Conditions (HAC) Provision, one of several value-based purchasing initiatives intended to increase quality and efficiency of care, which also include Present on Admission Indicator Reporting and Hospital Pay-For-Reporting. The added costs of SSIs are broadly attributed to extended hospital stays. On average, the cost to hospitals per SSI is $25,546. 1,2 In aggregate, this amounts to $7.4 billion in additional healthcare costs that Medicare will no longer reimburse every year. 2 The median U.S. hospital performs 3,441 surgeries every year. 8 Assuming two in 100 surgical procedures result in a surgical site infection, the median U.S. hospital would report 69 incidences of SSIs per year. This amounts to a total annual cost to the hospital of $1.76 million when relying on the above average cost per SSI of $25,546. A call for improved prevention: Rethinking the design of the surgical setting to benefit from cleanroom standards offers one solution Despite the significant impact surgical site infections have on healthcare costs and quality of care, a near-zero incidence rate is likely an unrealistic goal. However, improving the estimated two percent national incidence rate is realistic, as methods and systems that support prevention already exist. Table 1. Summary of ASHRAE Standard : Ventilation of Healthcare Facilities Requirements for Surgical Suites Positive pressure differential of +0.01" wc Individual temperature control Airflow shall be unidirectional, downwards Array shall extend a minimum of 12" beyond the footprint of the surgical table on each side < 30% of the array area shall be used for non-diffuser uses At least two low side wall return or exhaust grilles spaced at opposite corners The bottom of these grilles installed approximately 8" above the floor Current requirements for airflow conditions in hospital operating rooms are regulated by ASHRAE Standard (Table 1 lists the basic elements applying to operating rooms). While this standard contains airflow requirements such as the number of exchanges and the temperature and humidity of the air, it does not address airborne contaminant levels. The standard essentially reinforces the old adage, the solution to pollution is dilution. As a result, implementing an airflow system that provides the added benefit of complete contamination control over the sterile field in a surgical setting is often not considered because it is not required by standards or codes. In the absence of standards, it would be difficult, if not impossible, to gauge the effect of this phenomenon on the overall rates of SSIs. After all, hospital operating rooms are designed to a standard that has been deemed sufficient for a surgical Table 2. ISO Standard Cleanroom Class Limits setting. Findings to the contrary could be self-incriminatory from a public relations, if not legal, perspective. Conversely, the standards for airborne contaminant control are extremely well-defined in a cleanroom of a typical semiconductor manufacturer or for other critical manufacturing processes (Table 2). Ranging from Class 1 to 9, cleanrooms are designed to allow no more than a specific number of particles of a specific size to be present in a space at any given time. In meeting these specifications, contamination control systems must account for ancillary equipment such as lighting, machinery, people and other elements in the space that could otherwise work against creating the desired environment. Aseptic procedures for personnel entering the space can be equal to or greater than those required for doctors and nurses entering an operating room for a surgical procedure. Maximum number particles per cubic meter of specified size ISO Particle size Class 0.1 μm 0.2 μm 0.3 μm 0.5 μm 1 μm 5 μm , ,000 2,370 1, ,000 23,700 10,020 3, ,000, , ,000 35,200 8, ,000 83,200 2, ,520, ,000 29, ,520,000 8,320, ,300 Page 2
3 Manufacturers have adopted ISO requirements for cleanrooms with a single purpose: preventing what could be billions of dollars in lost revenue, warranty costs, back charges and liability due to a catastrophic product failure resulting from contaminants present in a sensitive manufacturing environment. It would be foolish to equate the value and well-being of a human life with that of a semiconductor chip or pharmaceutical drug, but a tremendous imbalance exists in the requirements for each critical environment. Benefitting fully from laminar airflow requires proper design So what could be done for the operating room environment to bridge the gap? The obvious answer is to adopt more stringent requirements, similar to those used in cleanroom environments that are inclusive of contamination control. However, the debate over laminar airflow systems illustrates how difficult this process can be without the benefit of similar design guidelines and standards. Laminar airflow systems provide uniform, directional airflow that essentially moves particles floating in the airstream away from areas that are intended to be contaminant-free (i.e. the sterile field) to where they can be disposed of and contained (through the return ducts and filtration system). Conversely, in a turbulent environment, particles are allowed to float undirected, which eliminates any ability to predict where a particle may settle. Laminar airflow is prevalent in most manufacturing cleanrooms and hospital operating rooms, but the difference between designs can be radical. In ISO Class 1 to Class 4 cleanroom environments, nearly every square inch of ceiling space is utilized to optimize airflow and contamination control so that particle containment is not left to chance. Maximizing the amount of surface area in the space from which supply air is flowing is critical to achieve the ultimate goal of laminar airflow minimize turbulence to produce predictable movement of particles away from the sterile field. In comparison (Figure 1), hospital operating room laminar flow systems typically consist of multiple laminar flow diffusers arranged in a variety of arrays, which are intended to optimize airflow to the space. In most cases, the arrangement of these diffusers with large gaps for light troffers and other components would be unacceptable by ISO Class 1 to Class 4 cleanroom standards because the ultimate result of the design would actually create, versus minimize, turbulence in the space. Utilization of a laminar flow diffuser does not necessarily guarantee the desired results of a laminar flow system in a critical environment. Proper laminar flow design requires that a strict set of rules be followed to optimize its effectiveness. Table 3 provides a comparison of a minimum diffuser array, as required per ASHRAE Standard 170 and AIA guidelines, to a single diffuser array designed to achieve laminar flow in the sterile field. Using Computational Fluid Dynamic (CFD) modeling, this comparison illustrates the difference in how the two systems perform with respect to providing laminar airflow. Figure 1. Conventional operating room air delivery system (left) and a system designed around optimizing laminar airflow over the patient (right). Note the large gap in airflow delivery over the patient in the conventional system. Page 3
4 Table 3. Comparison of Laminar Flow Designs Minimum Array Design 35 fpm 62º entering air Single Diffuser Design 35 fpm 62º entering air Thermal Properties: In the minimum array design (left), the temperature varies as much as 10 F due to the thermal effects of lights and people (lighter blue shaded areas). In the single diffuser (right), the temperature remains fairly constant from diffuser to the operating table. Turbulence: While the minimum array design contains laminar diffusers, it does not create a laminar flow environment in the space (left). The single diffuser design does achieve the desired effect of minimizing turbulence in the sterile field (right). Better Performance As Airflow Is Increased: Increasing airflow to the space by as little as 15 fpm to 50 fpm in both the minimum diffuser array (left) and the single diffuser design (right) provides improved performance in both systems. This phenomenon is consistent with cleanroom applications where as much as 100 fpm may be required for a Class 1 cleanroom. Page 4
5 Conclusion Given the quality of care, economic and public relations consequences of SSIs, consideration should be given to rethinking the requirements for the operating room to include some measure of contamination control and aerobiological quality standards. The technology and design practices have already been successfully implemented by semiconductor, pharmaceutical and other critical-process manufacturers facing similar consequences in a different context. International standards currently exist that identify the size and quantity of particulates allowed in a given space over a given amount of time. Granted, many obstacles exist within the operating environment that challenge the feasibility of achieving 100 percent laminar airflow and the contamination control benefits it can provide. Booms, lights, monitors and other equipment in the operating room can interfere with ideal airflow conditions. But facing these challenges head-on to advance the typical operating room beyond current design standards can prove to be a worthy venture toward improving the quality of patient care and benefitting the financial bottom line of healthcare facilities. References 1 The Direct Medical Costs of Healthcare-Associated Infections in U.S. Hospitals and the Benefits of Prevention; R. Douglas Scott II; March, Action Plan to Prevent Healthcare Associated Infections; Department of Health and Human Services; June, Never Events : Not Every Hospital-Acquired Infection Is Preventable; Jack Brown, Fred Doloresco III, and Joseph M. Mylotte; April, The importance of airborne bacterial contamination of wounds; J Hosp Infect; Whyte W, Hodgson R, Tinkler J; 1982; 3: Prevention of airborne infection during surgery; Lancet 1985; 325: Comparisons of Operating Ventilation Systems in Protection of the Surgical Site; Farhad Memarzadeh, P.E., Ph.D. and Andrew P. Manning, Ph.D.; July, Eliminating Healthcare Associated Infections; CDC; March American Hospital Association Hospital Database 9 Effect of Operation Room Geometry and Ventilation System Parameter Variations on the Protection of the Surgical Site; Farhad Memarzadeh, P.E., Ph.D. and Zheng Jiang, Ph.D.; Revisions to the Hospital Interpretive Guidelines for Infection Control; Centers for Medicare and Medicaid Services; November, Update to Hospital Value-Based Purchasing Program Final Rule; Centers for Medicare and Medicaid Services; April, Reducing surgical site infections; Contemporary surgery; November Guidelines for Environmental Infection Control in Health-Care Facilities; U.S. Department of Health and Human Services; Guideline for Isolation Precautions: Preventing Transmission of Infectious Agents in Healthcare Settings; Healthcare Infection Control Practices Advisory Committee; Page 5
6 CLEANSUITE Systems: Bringing cleanroom technology to the operating room CLEANSUITE System (top) and cleanroom system (below): Taking advantage of the similarities in both design and function provides a unique opportunity to improve the prevention of SSIs and HAIs in operating rooms and other sensitive healthcare applications. The HUNTAIR CLEANSUITE is an all-inclusive, modular ceiling diffuser system that is hung directly above the operating table in surgical suites and other applications requiring low turbulence, microbial-free airflow. CLEANSUITE systems help prevent SSIs and other healthcare-acquired infections (HAIs) caused by airborne contaminants in hospital operating rooms, outpatient surgery centers and other sensitive healthcare applications. CLEANSUITE systems have evolved from the manufacturing environments for semiconductors, pharmaceuticals and other sensitive products, where contamination is strictly controlled by Federal and industry standards. CLEANSUITE systems can be designed to Class 1 manufacturing cleanroom standards to meet specific airflow requirements, while providing the added benefit of contamination control. A Class 1 cleanroom is defined as allowing no more than 10 particles at a size of up to 0.1 micrometers per cubic meter and no more than two particles at a size of up to 0.2 micrometers per cubic meter. Currently, hospital operating rooms are required to meet ASHRAE Standard , which includes airflow requirements but does not address contaminant levels. Therefore, while many hospitals refer to an operating room suite as ultra-clean if it contains no more than 10 CFU/m 3, a typical Class 1 cleanroom is required to maintain contaminant levels that are 1,000 times lower. CLEANSUITE systems can help you bridge this gap using proven technology and design practices from thousands of installed and operating cleanroom applications worldwide. Huntair s reputation for providing the highest quality systems for airflow and contamination control has been earned over 20 years of experience in serving mission-critical applications. We are experts in providing aseptic design details with HEPA filtration, antimicrobial coatings and laminar flow for cleanrooms and other sensitive environments. The CLEANSUITE system is a natural extension of these capabilities and can be customdesigned for the unique requirements of your operating room setting. Page 6
7 CLEANSUITE Systems: A different approach offering sole source responsibility CLEANSUITE systems are pre-designed and fabricated at Huntair, arriving at your facility in fully-integrated modules that can be navigated through standard doorways and service elevators, lifted into place and connected to the building structure. Each all-inclusive module is custom-designed and can include integral boom mounts, lights, filtration, air balancing, sprinklers, medical gas connections and more. Single point connections can be provided for all services including controls, communications, electricity, water and/or medical gas. Traditional ceiling diffuser systems for operating room environment control are field-built and require significant coordination between trades representing structural, mechanical and utility services (electrical, plumbing, sheet metal and specialty services such as medical gas). The modular design and sole source responsibility offered by CLEANSUITE systems provides several benefits over the traditional approach: Dramatically reduces the design phase and jobsite coordination of trades while providing a customdesigned system for your operating room environment control. Provides tremendous installation cost savings by eliminating most jobsite labor and costly field workarounds associated with field-built systems. Installs in 1 /6 the time typically required for a field-built system, providing the opportunity to accelerate the timeline from construction to a fully functional, revenue-generating facility. CLEANSUITE Systems: Customdesigned for your application The anatomy of a CLEANSUITE system begins with Advanced Space Frame technology that provides a high strengthto-weight ratio capable of supporting the most demanding loads including radially extending surgical booms, imaging equipment, robotics, etc., supplied by others while performing to the strictest seismic requirements. Each module is pre-plumbed and wired with quick connects for electrical, water and other specialty services such as medical gas. Facing down at the operating room, each module includes a grid that incorporates flush-mounted lighting, sprinkler heads and roomside air balancing equipment. The grid includes accommodations to bottom-load gel-sealed HEPA filters (optional) and patented Clean-Screen laminar diffusers. All metal surfaces are powder coated or stainless steel, with or without antimicrobial treatments. The CLEANSUITE design results in smooth surfaces facing the operating room and allows the maximum possible surface area to be devoted to delivering laminar airflow over the operating table both of which are critical to promoting SSI and HAI prevention. The degree to which your CLEANSUITE module is customized is ultimately determined by your requirements. Highlighted custom features include: Variable height (18 to 72 ) and width (24 to 120 ) allow you to match space and move-in path requirements. Virtually limitless configurations and options for boom mounts and other ceiling-hung equipment within each CLEANSUITE module provides the flexibility to optimize their function while minimizing their impact on laminar airflow. Flexible placement of wiring, medical gas, audio/video feeds and other specialty items that are required to run through each CLEANSUITE module. Items are encased outside the airstream and provided with quick-connects to simplify field connections. Optional air curtain module with HEPA filtration to meet application requirements. Flexible side or top placement of supply air connection(s) to match application requirements. Page 7
8 HUNTAIR Air Handlers: Benefit from sole source responsibility for your entire air delivery system For seamless integration of your entire air delivery system and its controls, consider HUNTAIR air handlers with FANWALL TECHNOLOGY to compliment your CLEANSUITE system. Huntair is a leading supplier of custom air handling solutions for the most sensitive mission critical applications in the healthcare, semiconductor and pharmaceutical industries. Since 1993, Huntair has reengineered how we move air with innovative solutions, including FANWALL TECHNOLOGY, arguably the first true innovation in the air handling equipment industry in years. Based on replacing large fans with a modular array of smaller fans, FANWALL TECHNOLOGY allows for a significantly smaller footprint, redundancy for improved reliability, high energy efficiency, low operating costs, very low sound and significantly reduced maintenance requirements. All of these benefits are ideal for new construction and retrofit applications for healthcare facilities particularly the critical application of serving operating rooms. The experience, attention to detail, quality and innovation of Huntair provide you with the assurance that you are getting the most advanced, capable and reliable air handler custom-made for your requirements. For more information on how HUNTAIR CLEANSUITE systems and air handlers with FANWALL TECHNOLOGY can benefit your next application, contact your local HUNTAIR representative. To locate your representative, visit info@huntair.com FANWALL TECHNOLOGY and FANWALL are trademarks of Huntair, Inc. This product is covered by one or more of the following U.S. patents (7,137,775; 7,179,046; 7,527,468; 7,597,534) and other pending U.S. or Canadian patent applications and/or foreign patents Huntair, Inc. HUNT-CS-WP-1A May 2012
and fungal contamination can also be present in these skin scales. Quality of care impacts: Public perception
 White Paper Advancing Aerobiological Quality Standards For Hospital Operating Rooms (ORs) Hospital ORs are among the most infection-sensitive environments in healthcare facilities. Undergoing a surgical
White Paper Advancing Aerobiological Quality Standards For Hospital Operating Rooms (ORs) Hospital ORs are among the most infection-sensitive environments in healthcare facilities. Undergoing a surgical
Formal Interpretations Guidelines for Design and Construction of Hospitals and Outpatient Facilities, 2014 edition
 Formal Interpretations Guidelines for Design and Construction of Hospitals and Outpatient Facilities, 2014 edition Decisions published here were rendered after a multi-person panel of Health Guidelines
Formal Interpretations Guidelines for Design and Construction of Hospitals and Outpatient Facilities, 2014 edition Decisions published here were rendered after a multi-person panel of Health Guidelines
Isolation Precaution (Part 2) Protective Environment (PE) Room. Combined AII/PE Rooms. Contact Isolation 5/22/2017
 Isolation Precaution (Part 2) Prof (Col) Dr RN Basu Adviser Quality & Academics Medica Superspecialty Hospital And Executive Director Academy of Hospital Administration, Kolkata Chapter Airborne Infection
Isolation Precaution (Part 2) Prof (Col) Dr RN Basu Adviser Quality & Academics Medica Superspecialty Hospital And Executive Director Academy of Hospital Administration, Kolkata Chapter Airborne Infection
Providing Quality Equipment and Services for Specialized Mechanical Air Systems. Serving Kentucky and Southern Indiana for over 70 years
 Providing Quality Equipment and Services for Specialized Mechanical Air Systems Serving Kentucky and Southern Indiana for over 70 years 1 ANSI/ASHRAE/ASHE Standard 170 ANSI/ASHRAE/ASHE Standard 170-2017:
Providing Quality Equipment and Services for Specialized Mechanical Air Systems Serving Kentucky and Southern Indiana for over 70 years 1 ANSI/ASHRAE/ASHE Standard 170 ANSI/ASHRAE/ASHE Standard 170-2017:
QUESTIONS PERTINENT TO PRODUCT SELECTION:
 QUESTIONS PERTINENT TO PRODUCT SELECTION: Impact on patient outcomes Impact on patient/staff safety Economic considerations Use the following pages to help facilitate discussion with vendors, write your
QUESTIONS PERTINENT TO PRODUCT SELECTION: Impact on patient outcomes Impact on patient/staff safety Economic considerations Use the following pages to help facilitate discussion with vendors, write your
PRESENTORS Cyndi McCullough Andrew Yosten
 PRESENTORS Cyndi McCullough Andrew Yosten Applying Principles and Lessons Learned in Biosafety and Biocontainment Facility Design to the Challenges of Handling Patients with Highly Pathogenic Infectious
PRESENTORS Cyndi McCullough Andrew Yosten Applying Principles and Lessons Learned in Biosafety and Biocontainment Facility Design to the Challenges of Handling Patients with Highly Pathogenic Infectious
HEALTHCARE GUIDELINES AND STANDARDS
 HEALTHCARE GUIDELINES AND STANDARDS APPLICATION NOTE LC-126 INTRODUCTION This publication provides excerpts from some of the many guidelines and standards that pertain to the construction and operation
HEALTHCARE GUIDELINES AND STANDARDS APPLICATION NOTE LC-126 INTRODUCTION This publication provides excerpts from some of the many guidelines and standards that pertain to the construction and operation
Jeanne Moldenhauer (c) Jeanne Moldenhauer
 Jeanne Moldenhauer (c) Jeanne Moldenhauer 2013 1 Presentation Overview Conflicts between regulatory and compendial guidance Understanding the requirements for non sterile and terminally sterilized products
Jeanne Moldenhauer (c) Jeanne Moldenhauer 2013 1 Presentation Overview Conflicts between regulatory and compendial guidance Understanding the requirements for non sterile and terminally sterilized products
Environmental Monitoring in NHS Facilities
 NHS QA Symposium 25 th /26 th September 2012 Environmental Monitoring in NHS Facilities Tim Triggs Director of ACT Ltd & Slide 1 Lighthouse Worldwide Solutions R3 Nordic Speaker introduction - Tim Triggs
NHS QA Symposium 25 th /26 th September 2012 Environmental Monitoring in NHS Facilities Tim Triggs Director of ACT Ltd & Slide 1 Lighthouse Worldwide Solutions R3 Nordic Speaker introduction - Tim Triggs
Compounded Sterile Preparations Pharmacy Content Outline May 2018
 Compounded Sterile Preparations Pharmacy Content Outline May 2018 The following domains, tasks, and knowledge statements were identified and validated through a role delineation study. The proportion of
Compounded Sterile Preparations Pharmacy Content Outline May 2018 The following domains, tasks, and knowledge statements were identified and validated through a role delineation study. The proportion of
Evidence-Based Approaches to Hand Hygiene: Best Practices for Collaboration
 Evidence-Based Approaches to Hand Hygiene: Best Practices for Collaboration Written by J. Hudson Garrett Jr., PhD, Senior Director, Clinical Affairs, PDI January 09, 2013 Historical perspective Hand hygiene
Evidence-Based Approaches to Hand Hygiene: Best Practices for Collaboration Written by J. Hudson Garrett Jr., PhD, Senior Director, Clinical Affairs, PDI January 09, 2013 Historical perspective Hand hygiene
Healthcare-Associated Infections
 Healthcare-Associated Infections A healthcare crisis requiring European leadership Healthcare-associated infections (HAIs - also referred to as nosocomial infections) are defined as an infection occurring
Healthcare-Associated Infections A healthcare crisis requiring European leadership Healthcare-associated infections (HAIs - also referred to as nosocomial infections) are defined as an infection occurring
Re: Rewarding Provider Performance: Aligning Incentives in Medicare
 September 25, 2006 Institute of Medicine 500 Fifth Street NW Washington DC 20001 Re: Rewarding Provider Performance: Aligning Incentives in Medicare The American College of Physicians (ACP), representing
September 25, 2006 Institute of Medicine 500 Fifth Street NW Washington DC 20001 Re: Rewarding Provider Performance: Aligning Incentives in Medicare The American College of Physicians (ACP), representing
6/23/2011. Compounded Sterile Products Update and Review. Learning Goals for the Pharmacist. Learning Goals for the Pharmacy Technician
 Compounded Sterile Products- 2011 Update and Review Jo Ann Gibbs, PharmD Director of Pharmacy Byrd Regional Hospital Leesville, LA Learning Goals for the Pharmacist The pharmacist will be able to: 1. Identify
Compounded Sterile Products- 2011 Update and Review Jo Ann Gibbs, PharmD Director of Pharmacy Byrd Regional Hospital Leesville, LA Learning Goals for the Pharmacist The pharmacist will be able to: 1. Identify
BUGS BE GONE: Reducing HAIs and Streamlining Care!
 BUGS BE GONE: Reducing HAIs and Streamlining Care! SUSAN WHITNEY, RN, PCCN, MM, BME FLORIDA HOSPITAL ORLANDO, FL SUWHIT@AOL.COM LEARNING OUTCOMES 1. Describe HAI s and the impact disposable ECG leads have
BUGS BE GONE: Reducing HAIs and Streamlining Care! SUSAN WHITNEY, RN, PCCN, MM, BME FLORIDA HOSPITAL ORLANDO, FL SUWHIT@AOL.COM LEARNING OUTCOMES 1. Describe HAI s and the impact disposable ECG leads have
July 10, reduce the risk of staff or patient airborne exposure to communicable diseases during surgical procedures (See Appendix A) and
 TITLE GUIDELINE APPROVING AUTHORITY Infection Prevention and Control Operations PRACTICE SUPPORT DOCUMENT SPONSOR Infection Prevention and Control If you have any questions or comments regarding the information
TITLE GUIDELINE APPROVING AUTHORITY Infection Prevention and Control Operations PRACTICE SUPPORT DOCUMENT SPONSOR Infection Prevention and Control If you have any questions or comments regarding the information
WARD ISOLATION SUITE BASIC CONCEPTS and RESEARCH PROGRAMME. Malcolm Thomas Consulting Engineer March 2006
 WARD ISOLATION SUIT BASIC CONCPTS and RSARCH PROGRAMM Malcolm Thomas Consulting ngineer 01132 582788 March 2006 Isolation Fact and Fiction Isolation is required! When admitted, patients condition unknown
WARD ISOLATION SUIT BASIC CONCPTS and RSARCH PROGRAMM Malcolm Thomas Consulting ngineer 01132 582788 March 2006 Isolation Fact and Fiction Isolation is required! When admitted, patients condition unknown
3.03 Functions of support services personnel Name
 3.03 Functions of support services personnel Name Date Directions: Record notes and classroom discussion about the function and responsibilities of support services personnel. Create a therapeutic environment
3.03 Functions of support services personnel Name Date Directions: Record notes and classroom discussion about the function and responsibilities of support services personnel. Create a therapeutic environment
Microbial contamination risks of the surface of surgical clothing systems an observational study
 European Journal of Parenteral & Pharmaceutical Sciences 2017; 22(1): 6-12 2017 Pharmaceutical and Healthcare Sciences Society Microbial contamination risks of the surface of surgical clothing systems
European Journal of Parenteral & Pharmaceutical Sciences 2017; 22(1): 6-12 2017 Pharmaceutical and Healthcare Sciences Society Microbial contamination risks of the surface of surgical clothing systems
75,000 Approxiamte amount of deaths ,000 Number of patients who contract HAIs each year 1. HAIs: Costing Everyone Too Much
 HAIs: Costing Everyone Too Much July 2015 Healthcare-associated infections (HAIs) are serious, sometimes fatal conditions that have challenged healthcare institutions for decades. They are also largely
HAIs: Costing Everyone Too Much July 2015 Healthcare-associated infections (HAIs) are serious, sometimes fatal conditions that have challenged healthcare institutions for decades. They are also largely
EQUIPMENT MANAGEMENT MEDICAL EQUIPMENT: EC , EC UTILITY SYSTEMS: EC , EC
 EQUIPMENT MANAGEMENT MEDICAL EQUIPMENT: EC.02.04.01, EC.02.04.03 UTILITY SYSTEMS: EC.02.05.01, EC.02.05.05 ONLY APPLIES TO HOSPITAL & CAH PROGRAMS George Mills, Director Engineering Department The Joint
EQUIPMENT MANAGEMENT MEDICAL EQUIPMENT: EC.02.04.01, EC.02.04.03 UTILITY SYSTEMS: EC.02.05.01, EC.02.05.05 ONLY APPLIES TO HOSPITAL & CAH PROGRAMS George Mills, Director Engineering Department The Joint
Hospital Construction, Renovation, and Demolition
 01.38 - Hospital Construction, Renovation, and Demolition Purpose Infection Control Risk Assessment (ICRA) To provide infection control guidelines for hospital construction, renovation, and demolition.
01.38 - Hospital Construction, Renovation, and Demolition Purpose Infection Control Risk Assessment (ICRA) To provide infection control guidelines for hospital construction, renovation, and demolition.
9/14/2017. Best Practices in Instrument Cleaning. Objectives. Healthcare-associated Infections
 in Instrument Cleaning Crit Fisher, CST, FAST Director, Field Operations Protection1 Services Karl Storz Endoscopy-America, Inc. Objectives Discuss regulations, standards and guidelines of equipment management
in Instrument Cleaning Crit Fisher, CST, FAST Director, Field Operations Protection1 Services Karl Storz Endoscopy-America, Inc. Objectives Discuss regulations, standards and guidelines of equipment management
Commonwealth Nurses Federation. A Safe Patient. Jill ILIFFE Executive Secretary. Commonwealth Nurses Federation
 A Safe Patient Jill ILIFFE Executive Secretary Commonwealth Nurses Federation INFECTION CONTROL Every patient encounter should be viewed as potentially infectious Standard Precautions 1. Hand hygiene 2.!
A Safe Patient Jill ILIFFE Executive Secretary Commonwealth Nurses Federation INFECTION CONTROL Every patient encounter should be viewed as potentially infectious Standard Precautions 1. Hand hygiene 2.!
CMS-0044-P; Proposed Rule: Medicare and Medicaid Programs; Electronic Health Record Incentive Program Stage 2
 May 7, 2012 Submitted Electronically Ms. Marilyn Tavenner Acting Administrator Centers for Medicare and Medicaid Services Department of Health and Human Services Room 445-G, Hubert H. Humphrey Building
May 7, 2012 Submitted Electronically Ms. Marilyn Tavenner Acting Administrator Centers for Medicare and Medicaid Services Department of Health and Human Services Room 445-G, Hubert H. Humphrey Building
RESEARCH LABORATORIES CONDUCTING HIV/HBV RESEARCH AND PRODUCTION
 RESEARCH LABORATORIES CONDUCTING HIV/HBV RESEARCH AND PRODUCTION A. Definition of HIV/HBV Research and Production Laboratories Research laboratory means a laboratory which produces or uses research laboratory
RESEARCH LABORATORIES CONDUCTING HIV/HBV RESEARCH AND PRODUCTION A. Definition of HIV/HBV Research and Production Laboratories Research laboratory means a laboratory which produces or uses research laboratory
Facility Tuberculosis (TB) Risk Assessment for Correctional Facilities
 Facility Tuberculosis (TB) Risk Assessment for Correctional Facilities The various areas within correctional facilities have different levels of risk for TB transmission. Apply this worksheet to assess
Facility Tuberculosis (TB) Risk Assessment for Correctional Facilities The various areas within correctional facilities have different levels of risk for TB transmission. Apply this worksheet to assess
Multi-Link. X2 ECG System. Choose flexibility and quality for your reusable and disposable single-patient-use leadwires
 Multi-Link X2 ECG System Choose flexibility and quality for your reusable and disposable single-patient-use leadwires For more than a decade, our engineers have designed the only official, validated ECG
Multi-Link X2 ECG System Choose flexibility and quality for your reusable and disposable single-patient-use leadwires For more than a decade, our engineers have designed the only official, validated ECG
Isolation Categories of Transmission-Based Precautions
 Isolation Categories of Transmission-Based Highlights Policy Statement Standard shall be used when caring for residents at all times regardless of their suspected or confirmed infection status. Transmission-Based
Isolation Categories of Transmission-Based Highlights Policy Statement Standard shall be used when caring for residents at all times regardless of their suspected or confirmed infection status. Transmission-Based
Linens Laundry Logistics. Fight Infection Track Inventory Enhance Image Reduce Costs. Everything Fits.
 Linens Laundry Logistics Fight Infection Track Inventory Enhance Image Reduce Costs Everything Fits. Hidden linen charges? Clean smarter. Look smarter. What s your point of pain? Inventory shortages? A
Linens Laundry Logistics Fight Infection Track Inventory Enhance Image Reduce Costs Everything Fits. Hidden linen charges? Clean smarter. Look smarter. What s your point of pain? Inventory shortages? A
HealthStream Ambulatory Regulatory Course Descriptions
 This course covers three related aspects of medical care. All three are critical for the safety of patients. Avoiding Errors: Communication, Identification, and Verification These three critical issues
This course covers three related aspects of medical care. All three are critical for the safety of patients. Avoiding Errors: Communication, Identification, and Verification These three critical issues
Part D Infection Control
 Part D Infection Control HAAD Guidelines for Health Facility Design, Approvals Construction and Consultant Prequalification Version 3.3 April 2014 Table of Contents 1.0 General Requirements... 3 1.1 General...
Part D Infection Control HAAD Guidelines for Health Facility Design, Approvals Construction and Consultant Prequalification Version 3.3 April 2014 Table of Contents 1.0 General Requirements... 3 1.1 General...
SOFT SURFACE BACTERIAL MANAGEMENT:
 SOFT SURFACE BACTERIAL MANAGEMENT: NEBRASKA Methodist Hospital Establishes a New Standard for Soft Surface Fabrics Prepared By: Peggy Prinz Luebbert MS, MT(ASCP), CIC, CHSP Introduction Evidence exposing
SOFT SURFACE BACTERIAL MANAGEMENT: NEBRASKA Methodist Hospital Establishes a New Standard for Soft Surface Fabrics Prepared By: Peggy Prinz Luebbert MS, MT(ASCP), CIC, CHSP Introduction Evidence exposing
Healthcare- Associated Infections in North Carolina
 2018 Healthcare- Associated Infections in North Carolina Reference Document Revised June 2018 NC Surveillance for Healthcare-Associated and Resistant Pathogens Patient Safety Program NC Department of Health
2018 Healthcare- Associated Infections in North Carolina Reference Document Revised June 2018 NC Surveillance for Healthcare-Associated and Resistant Pathogens Patient Safety Program NC Department of Health
Personal Hygiene & Protective Equipment. NEO111 M. Jorgenson, RN BSN
 Personal Hygiene & Protective Equipment NEO111 M. Jorgenson, RN BSN Hand Hygiene the single most effective way to help prevent the spread of infections agents. (CDC, 2002.) Consistency & Compliancy 50%
Personal Hygiene & Protective Equipment NEO111 M. Jorgenson, RN BSN Hand Hygiene the single most effective way to help prevent the spread of infections agents. (CDC, 2002.) Consistency & Compliancy 50%
An economic - quality business case for infection control & Prof. dr. Dominique Vandijck
 An economic - quality business case for infection control & prevention @VandijckD Prof. dr. Dominique Vandijck What you/we all know, (hopefully) but do our healthcare executives, and politicians know this?
An economic - quality business case for infection control & prevention @VandijckD Prof. dr. Dominique Vandijck What you/we all know, (hopefully) but do our healthcare executives, and politicians know this?
Medicare Value Based Purchasing August 14, 2012
 Medicare Value Based Purchasing August 14, 2012 Wes Champion Senior Vice President Premier Performance Partners Copyright 2012 PREMIER INC, ALL RIGHTS RESERVED Premier is the nation s largest healthcare
Medicare Value Based Purchasing August 14, 2012 Wes Champion Senior Vice President Premier Performance Partners Copyright 2012 PREMIER INC, ALL RIGHTS RESERVED Premier is the nation s largest healthcare
April 10, 2018 York Chan, CHFM, CHC, SASHE
 1 The Physical Environment In Healthcare Facilities April 10, 2018 York Chan, CHFM, CHC, SASHE 2 YORK CHAN, CHFM, CHSP, SASHE Vice President, Facility Services Board of Directors 2008-2012 Faculty 2013
1 The Physical Environment In Healthcare Facilities April 10, 2018 York Chan, CHFM, CHC, SASHE 2 YORK CHAN, CHFM, CHSP, SASHE Vice President, Facility Services Board of Directors 2008-2012 Faculty 2013
USP 797: A FOCUS ON ANTIMICROBIAL RISK LEVEL KAREN MILKIEWICZ, PHARMD
 USP 797: A FOCUS ON ANTIMICROBIAL RISK LEVEL KAREN MILKIEWICZ, PHARMD USP 797: A FOCUS ON ANTIMICROBIAL RISK LEVEL ACTIVITY DESCRIPTION On June 1, 2008, The Revision Bulletin to USP Chapter 797, Pharmaceutical
USP 797: A FOCUS ON ANTIMICROBIAL RISK LEVEL KAREN MILKIEWICZ, PHARMD USP 797: A FOCUS ON ANTIMICROBIAL RISK LEVEL ACTIVITY DESCRIPTION On June 1, 2008, The Revision Bulletin to USP Chapter 797, Pharmaceutical
FEATURE. Back to. A Fresh Look at Asepsis BASICS. Alecia Cooper, RN, BS, MBA, CNOR 14 THE OR CONNECTION
 FEATURE Back to A Fresh Look at Asepsis BASICS Alecia Cooper, RN, BS, MBA, CNOR 14 THE OR CONNECTION PATIENT SAFETY A Back to Basics series should start with the principles of asepsis. What does asepsis
FEATURE Back to A Fresh Look at Asepsis BASICS Alecia Cooper, RN, BS, MBA, CNOR 14 THE OR CONNECTION PATIENT SAFETY A Back to Basics series should start with the principles of asepsis. What does asepsis
WHY IMPLEMENT CENTRAL LINE INSERTION BUNDLES
 WHY IMPLEMENT CENTRAL LINE INSERTION BUNDLES WHY IMPLEMENT A CENTRAL LINE BUNDLE? Hospital-acquired infections (HAIs) are the fourth largest killer in America. The death toll from HAIs is estimated at
WHY IMPLEMENT CENTRAL LINE INSERTION BUNDLES WHY IMPLEMENT A CENTRAL LINE BUNDLE? Hospital-acquired infections (HAIs) are the fourth largest killer in America. The death toll from HAIs is estimated at
Tuberculosis (TB) risk assessment worksheet
 128 Tuberculosis (TB) Risk MMWR Assessment Worksheet December 30, 2005 Tuberculosis (TB) risk assessment worksheet This model worksheet should be considered for use in performing TB risk assessments for
128 Tuberculosis (TB) Risk MMWR Assessment Worksheet December 30, 2005 Tuberculosis (TB) risk assessment worksheet This model worksheet should be considered for use in performing TB risk assessments for
Prevention of Orthopaedic Surgical Site Infections in the Perioperative Setting. Disclosures. Objectives
 Prevention of Orthopaedic Surgical Site Infections in the Perioperative Setting Mary Atkinson Smith, DNP, FNP-BC, ONP-C, RNFA, CNOR & W. Todd Smith, MD, FAAOS Disclosures We hereby certify that, to the
Prevention of Orthopaedic Surgical Site Infections in the Perioperative Setting Mary Atkinson Smith, DNP, FNP-BC, ONP-C, RNFA, CNOR & W. Todd Smith, MD, FAAOS Disclosures We hereby certify that, to the
Building a Reliable, Accurate and Efficient Hand Hygiene Measurement System
 Building a Reliable, Accurate and Efficient Hand Hygiene Measurement System Growing concern about the frequency of healthcare-associated infections (HAIs) has made hand hygiene an increasingly important
Building a Reliable, Accurate and Efficient Hand Hygiene Measurement System Growing concern about the frequency of healthcare-associated infections (HAIs) has made hand hygiene an increasingly important
Building a Culture of Patient Safety in Today s Healthcare Environment
 Building a Culture of Patient Safety in Today s Healthcare Environment A Discussion of the Tools and Healthcare Policies Driving Improvements in Patient Safety PANELISTS Paul Hiltz, MHA, FACHE Kelly E.
Building a Culture of Patient Safety in Today s Healthcare Environment A Discussion of the Tools and Healthcare Policies Driving Improvements in Patient Safety PANELISTS Paul Hiltz, MHA, FACHE Kelly E.
Incentives and Penalties
 Incentives and Penalties CAUTI & Value Based Purchasing and Hospital Associated Conditions Penalties: How Your Hospital s CAUTI Rate Affects Payment Linda R. Greene, RN, MPS,CIC UR Highland Hospital Rochester,
Incentives and Penalties CAUTI & Value Based Purchasing and Hospital Associated Conditions Penalties: How Your Hospital s CAUTI Rate Affects Payment Linda R. Greene, RN, MPS,CIC UR Highland Hospital Rochester,
Hospital-Based Ambulatory Care
 C H A P T E R 2 Hospital-Based Ambulatory Care ANSWERS TO KNOWLEDGE-BASED QUESTIONS 1. What has been the trend in the utilization of hospital-based services? What factors help to account for this trend?
C H A P T E R 2 Hospital-Based Ambulatory Care ANSWERS TO KNOWLEDGE-BASED QUESTIONS 1. What has been the trend in the utilization of hospital-based services? What factors help to account for this trend?
LESSON ASSIGNMENT. After completing this lesson, you should be able to: 2-3. Distinguish between medical and surgical aseptic technique.
 LESSON ASSIGNMENT LESSON 2 Medical Asepsis. LESSON OBJECTIVES After completing this lesson, you should be able to: 2-1. Identify the meaning of aseptic technique. 2-2. Identify the measures treatment personnel
LESSON ASSIGNMENT LESSON 2 Medical Asepsis. LESSON OBJECTIVES After completing this lesson, you should be able to: 2-1. Identify the meaning of aseptic technique. 2-2. Identify the measures treatment personnel
A strategy for building a value-based care program
 3M Health Information Systems A strategy for building a value-based care program How data can help you shift to value from fee-for-service payment What is value-based care? Value-based care is any structure
3M Health Information Systems A strategy for building a value-based care program How data can help you shift to value from fee-for-service payment What is value-based care? Value-based care is any structure
August 15, Dear Mr. Slavitt:
 Andrew M. Slavitt Acting Administrator Centers for Medicare & Medicaid Services Department of Health and Human Services P.O. Box 8010 Baltimore, MD 21244 Re: CMS 3295-P, Medicare and Medicaid Programs;
Andrew M. Slavitt Acting Administrator Centers for Medicare & Medicaid Services Department of Health and Human Services P.O. Box 8010 Baltimore, MD 21244 Re: CMS 3295-P, Medicare and Medicaid Programs;
Identify patients with Active Surveillance Cultures (ASC)
 MRSA CHANGE STRATEGIES The following tables include change strategies proven to be effective in healthcare settings. Implementing these changes through current or new processes may result in reducing healthcare
MRSA CHANGE STRATEGIES The following tables include change strategies proven to be effective in healthcare settings. Implementing these changes through current or new processes may result in reducing healthcare
Patient Safety Course Descriptions
 Adverse Events Antibiotic Resistance This course will teach you how to deal with adverse events at your facility. You will learn: What incidents are, and how to respond to them. What sentinel events are,
Adverse Events Antibiotic Resistance This course will teach you how to deal with adverse events at your facility. You will learn: What incidents are, and how to respond to them. What sentinel events are,
The Evolving Landscape of Healthcare Payment: Incentive Programs and ACO Model Optimization. Quality Forum August 19, 2015
 The Evolving Landscape of Healthcare Payment: Incentive Programs and ACO Model Optimization Quality Forum August 19, 2015 Ross Manson rmanson@eidebailly.com 701.239.8634 Barb Pritchard bpritchard@eidebailly.com
The Evolving Landscape of Healthcare Payment: Incentive Programs and ACO Model Optimization Quality Forum August 19, 2015 Ross Manson rmanson@eidebailly.com 701.239.8634 Barb Pritchard bpritchard@eidebailly.com
Chapter 10. medical and Surgical Asepsis. safe, effective Care environment. Practices that Promote Medical Asepsis
 chapter 10 Unit 1 Section Chapter 10 safe, effective Care environment safety and Infection Control medical and Surgical Asepsis Overview Asepsis The absence of illness-producing micro-organisms. Asepsis
chapter 10 Unit 1 Section Chapter 10 safe, effective Care environment safety and Infection Control medical and Surgical Asepsis Overview Asepsis The absence of illness-producing micro-organisms. Asepsis
PROGRAM OPPORTUNITY NOTICE EFFICIENCY MAINE TRUST CUSTOM INCENTIVE PROGRAM FOR ELECTRIC EFFICIENCY PROJECTS PON EM
 PROGRAM OPPORTUNITY NOTICE EFFICIENCY MAINE TRUST CUSTOM INCENTIVE PROGRAM FOR ELECTRIC EFFICIENCY PROJECTS PON Opening: July 1, 2017 Closing: June 30, 2018 Revised: February 6, 2018 {P1472575.1} CONTENTS
PROGRAM OPPORTUNITY NOTICE EFFICIENCY MAINE TRUST CUSTOM INCENTIVE PROGRAM FOR ELECTRIC EFFICIENCY PROJECTS PON Opening: July 1, 2017 Closing: June 30, 2018 Revised: February 6, 2018 {P1472575.1} CONTENTS
CENTRAL SERVICE (CS) IS A VITAL DEPARTMENT IN ANY HOSPITAL
 CRCST Self-Study Lesson Plan Lesson No. CRCST 158 (Technical Continuing Education - TCE) by Jon Wood, BAAS, IAHCSMM Clinical Educator Sponsored by: Understanding and Preventing Cross Contamination LEARNING
CRCST Self-Study Lesson Plan Lesson No. CRCST 158 (Technical Continuing Education - TCE) by Jon Wood, BAAS, IAHCSMM Clinical Educator Sponsored by: Understanding and Preventing Cross Contamination LEARNING
REGULATORY COMPLIANCE: HOW READY IS YOUR HEALTHCARE SYSTEM?
 REGULATORY COMPLIANCE: HOW READY IS YOUR HEALTHCARE SYSTEM? POP QUIZ: CAN YOU ANSWER THESE 10 QUESTIONS? 1. Is a bloody tissue considered trash or regulated medical waste? 2. What is the proper mix of
REGULATORY COMPLIANCE: HOW READY IS YOUR HEALTHCARE SYSTEM? POP QUIZ: CAN YOU ANSWER THESE 10 QUESTIONS? 1. Is a bloody tissue considered trash or regulated medical waste? 2. What is the proper mix of
HOSPITAL ACQUIRED COMPLICATIONS. Shruti Scott, DO, MPH Department of Medicine UCI Hospitalist Program
 HOSPITAL ACQUIRED COMPLICATIONS Shruti Scott, DO, MPH Department of Medicine UCI Hospitalist Program HOSPITAL ACQUIRED COMPLICATIONS (HACS) A medical condition or complication that a patient develops during
HOSPITAL ACQUIRED COMPLICATIONS Shruti Scott, DO, MPH Department of Medicine UCI Hospitalist Program HOSPITAL ACQUIRED COMPLICATIONS (HACS) A medical condition or complication that a patient develops during
PRECAUTIONS IN INFECTION CONTROL
 PRECAUTIONS IN INFECTION CONTROL Standard precautions Transmission-based precautions Contact precautions Airborne precautions Droplet precautions 1 2/25/2015 WHO HAVE TO PROTECT IN HOSPITALS? Patients
PRECAUTIONS IN INFECTION CONTROL Standard precautions Transmission-based precautions Contact precautions Airborne precautions Droplet precautions 1 2/25/2015 WHO HAVE TO PROTECT IN HOSPITALS? Patients
Care Redesign: An Essential Feature of Bundled Payment
 Issue Brief No. 11 September 2013 Care Redesign: An Essential Feature of Bundled Payment Jett Stansbury Director, New Payment Strategies, Integrated Healthcare Association Gabrielle White, RN, CASC Executive
Issue Brief No. 11 September 2013 Care Redesign: An Essential Feature of Bundled Payment Jett Stansbury Director, New Payment Strategies, Integrated Healthcare Association Gabrielle White, RN, CASC Executive
Number: Ratio of the airflow to the space volume per unit time, usually expressed as the number of air changes per hour.
 POLICIES & PROCEDURES Number: 40 175 Title: Tuberculosis (TB) Management Program Authorization: [X] SHR Infection Control Committee [ ] Facility Board of Directors Source: Infection Prevention & Control
POLICIES & PROCEDURES Number: 40 175 Title: Tuberculosis (TB) Management Program Authorization: [X] SHR Infection Control Committee [ ] Facility Board of Directors Source: Infection Prevention & Control
LPN 8 Hour Didactic IV Education
 LPN 8 Hour Didactic IV Education Infection Prevention and Control By Pamela Truscott, MSN, Nurse Educator, RN Infection Prevention and Control Background Healthcare-acquired infections are increasing 1
LPN 8 Hour Didactic IV Education Infection Prevention and Control By Pamela Truscott, MSN, Nurse Educator, RN Infection Prevention and Control Background Healthcare-acquired infections are increasing 1
The Design Professional and Infection Prevention for Healthcare Facilities
 The Design Professional and Infection Prevention for Healthcare Facilities David J. Wells, AIA October 7, 2016 Design Professional & Infection Prevention in Healthcare Facilities, David Wells 1 Project
The Design Professional and Infection Prevention for Healthcare Facilities David J. Wells, AIA October 7, 2016 Design Professional & Infection Prevention in Healthcare Facilities, David Wells 1 Project
Reimbursement guide. IODOSORB/IODOFLEX are Cadexomer Iodine-based products, available in two forms gel or pad.
 Reimbursement guide IODOSORB/IODOFLEX are Cadexomer Iodine-based products, available in two forms gel or pad. IODOSORB/IODOFLEX remove barriers to healing by its dual action antimicrobial and desloughing
Reimbursement guide IODOSORB/IODOFLEX are Cadexomer Iodine-based products, available in two forms gel or pad. IODOSORB/IODOFLEX remove barriers to healing by its dual action antimicrobial and desloughing
DEDICATED CLEAN AND SOILED SERVICE ELEVATORS
 DEDICATED CLEAN AND SOILED SERVICE ELEVATORS By Hubert P. Roach, General Manager Lerch, Bates & Associates Material Management and Materials Handling Group There has been on-going debate within the Healthcare
DEDICATED CLEAN AND SOILED SERVICE ELEVATORS By Hubert P. Roach, General Manager Lerch, Bates & Associates Material Management and Materials Handling Group There has been on-going debate within the Healthcare
To provide information about the role of the pharmacy in Infection Prevention and Control.
 TITLE/DESCRIPTION: Pharmacy DEPARTMENT: Pharmacy PERSONNEL: Pharmacy Personnel EFFECTIVE DATE: 1/97 REVISED: 4/97, 7/08, 12/11, 1/15 I. PURPOSE To provide information about the role of the pharmacy in
TITLE/DESCRIPTION: Pharmacy DEPARTMENT: Pharmacy PERSONNEL: Pharmacy Personnel EFFECTIVE DATE: 1/97 REVISED: 4/97, 7/08, 12/11, 1/15 I. PURPOSE To provide information about the role of the pharmacy in
Pharmacy General Personnel
 Pharmacy The Pharmacy Department is an important area for infection control because its products are potentially dispensed to all patients. Contamination of medications or other pharmaceuticals whether
Pharmacy The Pharmacy Department is an important area for infection control because its products are potentially dispensed to all patients. Contamination of medications or other pharmaceuticals whether
Prefabricated Materials Help To Innovate Health Care Construction
 Prefabricated Materials Help To Innovate Health Care Construction A Case Study at the University of Kentucky Albert B Chandler Hospital Presented By Prefabricated Materials Help To Innovate Health Care
Prefabricated Materials Help To Innovate Health Care Construction A Case Study at the University of Kentucky Albert B Chandler Hospital Presented By Prefabricated Materials Help To Innovate Health Care
INFECTION CONTROL ORIENTATION TRAINING 2006
 INFECTION CONTROL ORIENTATION TRAINING 2006 INFECTION CONTROL OSHA BLOODBORNE PATHOGEN STANDARD STANDARD PRECAUTIONS RISK OF EXPOSURE TO CONTAMINATED MATERIALS USE OF PROTECTIVE EQUIPMENT FOLLOW-UP OF
INFECTION CONTROL ORIENTATION TRAINING 2006 INFECTION CONTROL OSHA BLOODBORNE PATHOGEN STANDARD STANDARD PRECAUTIONS RISK OF EXPOSURE TO CONTAMINATED MATERIALS USE OF PROTECTIVE EQUIPMENT FOLLOW-UP OF
Fall HOLLY ALEXANDER Academic Coordinator of Clinical Education MS157
 Fall 2010 HOLLY ALEXANDER Academic Coordinator of Clinical Education 609-570-3478 AlexandH@mccc.edu MS157 To reduce infection & prevent disease transmission Nosocomial Infection: an infection acquired
Fall 2010 HOLLY ALEXANDER Academic Coordinator of Clinical Education 609-570-3478 AlexandH@mccc.edu MS157 To reduce infection & prevent disease transmission Nosocomial Infection: an infection acquired
Understanding USP 797
 Baxa Corporation Understanding USP 797 Technical Paper An Overview of USP General Chapter Pharmaceutical Compounding Sterile Preparations Mike Hurst, RPh, MBA 2004 Baxa Corporation Introduction USP
Baxa Corporation Understanding USP 797 Technical Paper An Overview of USP General Chapter Pharmaceutical Compounding Sterile Preparations Mike Hurst, RPh, MBA 2004 Baxa Corporation Introduction USP
Medicare P4P -- Medicare Quality Reporting, Incentive and Penalty Programs
 Medicare P4P -- Medicare Quality Reporting, Incentive and Penalty Programs Presenter: Daniel J. Hettich King & Spalding; Washington, DC dhettich@kslaw.com 1 I. Introduction Evolution of Medicare as a Purchaser
Medicare P4P -- Medicare Quality Reporting, Incentive and Penalty Programs Presenter: Daniel J. Hettich King & Spalding; Washington, DC dhettich@kslaw.com 1 I. Introduction Evolution of Medicare as a Purchaser
TUBERCULOSIS INFECTION CONTROL
 OBJECTIVES TUBERCULOSIS INFECTION CONTROL At the end of this presentation, you will be able to: List infection control approaches to TB prevention and control Describe the type of protective equipment
OBJECTIVES TUBERCULOSIS INFECTION CONTROL At the end of this presentation, you will be able to: List infection control approaches to TB prevention and control Describe the type of protective equipment
2017 Midwest Healthcare Engineering Conference FGI Guidelines Major Changes
 2017 Midwest Healthcare Engineering Conference- 2018 FGI Guidelines Major Changes Todd W. Hite, P.E. Program Manager, Health Care Engineering November 29, 2017 Acknowledgements The information provided
2017 Midwest Healthcare Engineering Conference- 2018 FGI Guidelines Major Changes Todd W. Hite, P.E. Program Manager, Health Care Engineering November 29, 2017 Acknowledgements The information provided
 As healthcare moves toward value-based care and risk-sharing payment models, many hospitals are taking a new look at ambulatory surgery centers (ASCs) as a transformational outpatient strategy with potential
As healthcare moves toward value-based care and risk-sharing payment models, many hospitals are taking a new look at ambulatory surgery centers (ASCs) as a transformational outpatient strategy with potential
ANSWERING TO A HIGHER CALLING
 ANSWERING TO A HIGHER CALLING Verifying Laundry Processes, Quantifying Cleanliness Quality Assurance Best Management Practices Continuous Improvement Process and Outcome Measures ANSWERING TO A HIGHER
ANSWERING TO A HIGHER CALLING Verifying Laundry Processes, Quantifying Cleanliness Quality Assurance Best Management Practices Continuous Improvement Process and Outcome Measures ANSWERING TO A HIGHER
Implementing Technologies for the Reduction of Hospital Acquired Infections. AHCA 2015 October 12, 2015 / 1:00 2:00 pm
 Implementing Technologies for the Reduction of Hospital Acquired Infections AHCA 2015 October 12, 2015 / 1:00 2:00 pm Learning Objectives Identify the human and financial costs impacting healthcare facilities
Implementing Technologies for the Reduction of Hospital Acquired Infections AHCA 2015 October 12, 2015 / 1:00 2:00 pm Learning Objectives Identify the human and financial costs impacting healthcare facilities
Frequently Asked Questions about TB Protocols at Duke Hospital and Clinics ( Revision)
 Frequently Asked Questions about TB Protocols at Duke Hospital and Clinics (7-2018 Revision) A. PAPRs B. Portable HEPAs C. N95 Respirator Masks D. Tuberculin Skin Testing (TST) E. Negative Pressure Isolation
Frequently Asked Questions about TB Protocols at Duke Hospital and Clinics (7-2018 Revision) A. PAPRs B. Portable HEPAs C. N95 Respirator Masks D. Tuberculin Skin Testing (TST) E. Negative Pressure Isolation
SCORING METHODOLOGY APRIL 2014
 SCORING METHODOLOGY APRIL 2014 HOSPITAL SAFETY SCORE Contents What is the Hospital Safety Score?... 4 Who is The Leapfrog Group?... 4 Eligible and Excluded Hospitals... 4 Scoring Methodology... 5 Measures...
SCORING METHODOLOGY APRIL 2014 HOSPITAL SAFETY SCORE Contents What is the Hospital Safety Score?... 4 Who is The Leapfrog Group?... 4 Eligible and Excluded Hospitals... 4 Scoring Methodology... 5 Measures...
PARTNERING TO CREATE A CLEANER, HEALTHIER AND SAFER ENVIRONMENT FOR SENIORS
 PARTNERING TO CREATE A CLEANER, HEALTHIER AND SAFER ENVIRONMENT FOR SENIORS A sustainable cleaning program designed with residents, custodians and staff in mind! Proven Strategies for a Cleaner, Healthier
PARTNERING TO CREATE A CLEANER, HEALTHIER AND SAFER ENVIRONMENT FOR SENIORS A sustainable cleaning program designed with residents, custodians and staff in mind! Proven Strategies for a Cleaner, Healthier
Infection Prevention and Control in Ambulatory Care Settings: Minimum Expectations for Safe Care
 Infection Prevention and Control in Ambulatory Care Settings: Minimum Expectations for Safe Care Melissa Schaefer, MD Division of Healthcare Quality Promotion Centers for Disease Control and Prevention
Infection Prevention and Control in Ambulatory Care Settings: Minimum Expectations for Safe Care Melissa Schaefer, MD Division of Healthcare Quality Promotion Centers for Disease Control and Prevention
Submitted electronically:
 Mr. Andy Slavitt Acting Administrator Centers for Medicare and Medicaid Services Department of Health and Human Services Attention: CMS-5517-FC P.O. Box 8013 7500 Security Boulevard Baltimore, MD 21244-8013
Mr. Andy Slavitt Acting Administrator Centers for Medicare and Medicaid Services Department of Health and Human Services Attention: CMS-5517-FC P.O. Box 8013 7500 Security Boulevard Baltimore, MD 21244-8013
June 25, Dear Administrator Verma,
 June 25, 2018 Seema Verma Administrator Centers for Medicare and Medicaid Services U.S. Department of Health and Human Services Room 445 G, Hubert H. Humphrey Building 200 Independence Avenue SW Washington,
June 25, 2018 Seema Verma Administrator Centers for Medicare and Medicaid Services U.S. Department of Health and Human Services Room 445 G, Hubert H. Humphrey Building 200 Independence Avenue SW Washington,
Environmental Services: Delivering on the Patient-Centered Promise
 Environmental Services: Delivering on the Patient-Centered Promise A patient s perception of hospital cleanliness is highly correlated with multiple safety, quality and experience measures. Executive Summary
Environmental Services: Delivering on the Patient-Centered Promise A patient s perception of hospital cleanliness is highly correlated with multiple safety, quality and experience measures. Executive Summary
Reducing Infections and Improving Engagement St. Luke's Nephrology Associates. Contact Information: Robert Gayner, M.D., FASN
 BEST PRACTICES Vascular Access and CLABSI Reduction Reducing Infections and Improving Engagement St. Luke's Nephrology Associates Contact Information: Robert Gayner, M.D., FASN St. Luke's Nephrology Associates
BEST PRACTICES Vascular Access and CLABSI Reduction Reducing Infections and Improving Engagement St. Luke's Nephrology Associates Contact Information: Robert Gayner, M.D., FASN St. Luke's Nephrology Associates
Global Healthcare Accreditation Standards Brief 4.0
 Global Healthcare Accreditation Standards Brief 4.0 for Medical Travel Services Effective June 1, 2017 Copyright 2017, Global Healthcare Accreditation Program All rights Version reserved. 4.0 No Reproduction
Global Healthcare Accreditation Standards Brief 4.0 for Medical Travel Services Effective June 1, 2017 Copyright 2017, Global Healthcare Accreditation Program All rights Version reserved. 4.0 No Reproduction
Replaces: 08/11/16. Formulated: 1/2000 TRANSMISSION-BASED PRECAUTIONS
 CMHC INFECTION CONTROL Effective : 08/10/17 Page 1 of 4 POLICY: TDCJ and any medical contractors will implement Transmission-Based Precautions as needed to interrupt the transmission of potentially contagious
CMHC INFECTION CONTROL Effective : 08/10/17 Page 1 of 4 POLICY: TDCJ and any medical contractors will implement Transmission-Based Precautions as needed to interrupt the transmission of potentially contagious
Joy in Medicine Physician well-being: A discussion on burnout and achieving joy in practice
 Joy in Medicine Physician well-being: A discussion on burnout and achieving joy in practice AMA s SL2 (Share, Listen, Speak, Learn) Series December 2017 Share, Listen, Speak, Learn (SL2) Series Share existing
Joy in Medicine Physician well-being: A discussion on burnout and achieving joy in practice AMA s SL2 (Share, Listen, Speak, Learn) Series December 2017 Share, Listen, Speak, Learn (SL2) Series Share existing
Healthcare Associated Infections Know No Boundaries: A View Across the Continuum of Care
 Healthcare Associated Infections Know No Boundaries: A View Across the Continuum of Care J. Hudson Garrett Jr., PhD, MSN, MPH, FNP, CSRN, VA-BC Vice President Clinical Affairs, PDI Healthcare Healthcare
Healthcare Associated Infections Know No Boundaries: A View Across the Continuum of Care J. Hudson Garrett Jr., PhD, MSN, MPH, FNP, CSRN, VA-BC Vice President Clinical Affairs, PDI Healthcare Healthcare
LESSONS LEARNED IN LENGTH OF STAY (LOS)
 FEBRUARY 2014 LESSONS LEARNED IN LENGTH OF STAY (LOS) USING ANALYTICS & KEY BEST PRACTICES TO DRIVE IMPROVEMENT Overview Healthcare systems will greatly enhance their financial status with a renewed focus
FEBRUARY 2014 LESSONS LEARNED IN LENGTH OF STAY (LOS) USING ANALYTICS & KEY BEST PRACTICES TO DRIVE IMPROVEMENT Overview Healthcare systems will greatly enhance their financial status with a renewed focus
Prime TC. Transport Chair. Patient Transport Redefined.
 Prime TC Transport Chair Patient Transport Redefined. Stryker and the Michael Graves Design Group: A Patient-driven Partnership For over 70 years, Stryker has been a partner in patient transport for hospitals
Prime TC Transport Chair Patient Transport Redefined. Stryker and the Michael Graves Design Group: A Patient-driven Partnership For over 70 years, Stryker has been a partner in patient transport for hospitals
Adopting Accountable Care An Implementation Guide for Physician Practices
 Adopting Accountable Care An Implementation Guide for Physician Practices EXECUTIVE SUMMARY November 2014 A resource developed by the ACO Learning Network www.acolearningnetwork.org Executive Summary Our
Adopting Accountable Care An Implementation Guide for Physician Practices EXECUTIVE SUMMARY November 2014 A resource developed by the ACO Learning Network www.acolearningnetwork.org Executive Summary Our
VALUE BASED ORTHOPEDIC CARE
 VALUE BASED ORTHOPEDIC CARE Becker's 14th Annual Spine, Orthopedic and Pain Management- Driven ASC Conference + The Future of Spine June 9-11, 2016 Swissotel, Chicago, IL LES JEBSON Administrator, Adjunct
VALUE BASED ORTHOPEDIC CARE Becker's 14th Annual Spine, Orthopedic and Pain Management- Driven ASC Conference + The Future of Spine June 9-11, 2016 Swissotel, Chicago, IL LES JEBSON Administrator, Adjunct
42 CFR Infection Control
 42 CFR 482.42 Infection Control Dodjie B. Guioa, MBA Hospital/ASC Program Lead Region VI Dallas dodjie.guioa@cms.hhs.gov Condition of Participation Infection Control The hospital must provide a sanitary
42 CFR 482.42 Infection Control Dodjie B. Guioa, MBA Hospital/ASC Program Lead Region VI Dallas dodjie.guioa@cms.hhs.gov Condition of Participation Infection Control The hospital must provide a sanitary
time to replace adjusted discharges
 REPRINT May 2014 William O. Cleverley healthcare financial management association hfma.org time to replace adjusted discharges A new metric for measuring total hospital volume correlates significantly
REPRINT May 2014 William O. Cleverley healthcare financial management association hfma.org time to replace adjusted discharges A new metric for measuring total hospital volume correlates significantly
Advancing Accountability for Improving HCAHPS at Ingalls
 iround for Patient Experience Advancing Accountability for Improving HCAHPS at Ingalls A Case Study Webconference 2 Managing your audio Use Telephone If you select the use telephone option please dial
iround for Patient Experience Advancing Accountability for Improving HCAHPS at Ingalls A Case Study Webconference 2 Managing your audio Use Telephone If you select the use telephone option please dial
POLICY & PROCEDURE POLICY NO: IPAC 3.2
 POLICY & PROCEDURE POLICY NO: IPAC 3.2 SUBJECT SUPERCEDES August 2007, July 2008 S 1of 5 APPROVAL: Infection Prevention & Control Committee DATE: September, 2010 Professional Advisory Committee DATE: January
POLICY & PROCEDURE POLICY NO: IPAC 3.2 SUBJECT SUPERCEDES August 2007, July 2008 S 1of 5 APPROVAL: Infection Prevention & Control Committee DATE: September, 2010 Professional Advisory Committee DATE: January
PROGRAM OPPORTUNITY NOTICE EFFICIENCY MAINE TRUST CUSTOM INCENTIVE PROGRAM FOR DISTRIBUTED GENERATION PROJECTS PON EM
 PROGRAM OPPORTUNITY NOTICE EFFICIENCY MAINE TRUST CUSTOM INCENTIVE PROGRAM FOR DISTRIBUTED GENERATION PROJECTS PON Opening: July 1, 2017 Closing: June 30, 2018 Updated: July 1, 2017 CONTENTS SECTION 1:
PROGRAM OPPORTUNITY NOTICE EFFICIENCY MAINE TRUST CUSTOM INCENTIVE PROGRAM FOR DISTRIBUTED GENERATION PROJECTS PON Opening: July 1, 2017 Closing: June 30, 2018 Updated: July 1, 2017 CONTENTS SECTION 1:
Infection Prevention & Control Orientation for Housestaff Welcome to Shands at UF!
 Infection Prevention & Control Orientation for Housestaff 2011 Welcome to Shands at UF! Hot Topics: Prevention Initiatives National Patient Safety Goal 07: Prevent Healthcare Associated Infections Prevent
Infection Prevention & Control Orientation for Housestaff 2011 Welcome to Shands at UF! Hot Topics: Prevention Initiatives National Patient Safety Goal 07: Prevent Healthcare Associated Infections Prevent
Infection prevention & control
 Infection control in Australian medical practice: Current practice and future developments John Ferguson Infectious Diseases & Microbiology Director, Infection Prevention & Control, Hunter New England
Infection control in Australian medical practice: Current practice and future developments John Ferguson Infectious Diseases & Microbiology Director, Infection Prevention & Control, Hunter New England
