PDF of Trial CTRI Website URL -
|
|
|
- Cecilia Lester
- 5 years ago
- Views:
Transcription
1 CTRI Website URL Clinical Trial Details (PDF Generation Date : Wed, 14 Nov :48:48 GMT) CTRI Number Last Modified On 23/08/2013 Post Graduate Thesis Type of Trial Type of Study Study Design Public Title of Study Scientific Title of Study CTRI/2012/01/ [Registered on: 25/01/2012] Trial Registered Prospectively No Interventional Drug Randomized, Parallel Group, Active Controlled Trial Study to determine the efficacy and safety of Dexlansoprazole in Gastroesophageal Reflux patients A RANDOMIZED, DOUBLE BLIND, ACTIVE CONTROLLED, COMPARATIVE, PARALLEL GROUP, MULTICENTER STUDY TO ASSESS THE EFFICACY AND SAFETY OF DEXLANSOPRAZOLE VERSUS LANSOPRAZOLE FOR GASTROESOPHAGEAL REFLUX DISEASE (GERD) Secondary IDs if Any Secondary ID Identifier Details of Principal Investigator or overall Trial Coordinator (multicenter study) CP/21/10, Version 2.1, Dated Designation Affiliation Protocol Number Details of Principal Investigator Details Contact Person (Scientific Query) Details Contact Person (Public Query) Phone Fax Designation Affiliation Details Contact Person (Scientific Query) Dr Jaideep Gogtay Medical Director Cipla Ltd. Phone Fax Designation Affiliation Bellasis Road, Mumbai Central Mumbai India jgogtay@cipla.com Details Contact Person (Public Query) Dr Sunil Gupta Phone Fax Principal Investigator Gastroenterologist Global Gastro & Liver Centre 6/2 Malviya Nagar, Near Amit Bharadwaj Petrol Pump, India sunilgastro@rediffmail.com page 1 / 6
2 CTRI Website URL Source of Monetary or Material Support Primary Sponsor Details of Secondary Sponsor Countries of Recruitment Sites of Study Source of Monetary or Material Support > Cipla Ltd, Bellasis Road, Mumbai Central, Mumbai, Phone (022) / Fax Cipla Ltd Primary Sponsor Details Bellasis Road, Mumbai Central, Mumbai, Phone: (022) / Fax: (022) Type of Sponsor NIL List of Countries India of Principal Investigator Dr Vijay Sharma Dr Sunil Gupta Dr Mahendra Parmar Dr Sandeep Kansal Dr S D Wagle Dr Unmesh Takalkar Pharmaceutical industryindian NIL of Site Site Phone/Fax/ Dr. Vijay Sharma Gastroenterology Clinic Global Gastro & Liver Centre Dr. Vijay Sharma Gastroenterology Clinic, 170, Indira Colony, Jhotwara Road, Bani Park, Global Gastro & Liver Centre, 6/2 Malviya Nagar, Near Amit Bharadwaj Petrol Pump,17 College & SSG Hospital College & SSG Hospital, Department of Medicine, Vadodara GUJARAT College and New Civil Hospital King Edward Memorial Hospital Kodlikeri Memorial Hospital Department of Surgery, College and New Civil Hospital, Majura Gate, Surat, Gujarat Surat GUJARAT King Edward Memorial Hospital, Department of Gastroenterology, TDH Bldg. 1st Floor,Sardar Moodliar Road, Rasta Peth, Pune Kodlikeri Memorial Hospital, 8, 9, Manjeet Nagar, Opp. Akashwani, Jalna Road, Aurangabad vijaysadashiv@gmail.co m sunilgastro@rediffmail.c om mcparmar1961@gmail. com drkansals@rediffmail.co m sdwagle@gmail.com unmesh_3@sancharnet.in page 2 / 6
3 CTRI Website URL Details of Ethics Committee Dr Punit Mehrotra Gastroenterology & Gynecology Centre Gastroenterology & Gynecology Centre, B/127, Nirala Nagar, UTTAR PRADESH Dr Sanjay Salunkhe Noble Hospital Noble Hospital, 153, Magarpatta City Road, Pune Dr Shrish Bhatnagar Dr Ramesh Satarkar Dr Shobna Bhatia Rajajipuram Hospital & Maternity Centre Satarkar Gastroenterology Centre Rajajipuram Hospital & Maternity Centre, E1075, Rajajipuram, UTTAR PRADESH Satarkar Gastroenterology Centre, Antarang,20,Tilak Nagar, Near Savarkar Chowk, Aurangabad Seth G.S. Medical Seth G.S. Medical College & KEM Hospital College & KEM Hospital, Department of Gastroenterology, MS Bldg, 9th Floor, Parel, Mumbai Dr Sandeep Nijhawan SMS Hospital SMS Hospital, JNL Marg, Dr B Ravi Shankar Yashoda Hospital Yashoda Hospital, Behind Hari Hara Kala Bhavan, S.P. Road, Secunderabad Hyderabad ANDHRA PRADESH punmel@yahoo.com drsanjaysalunkhe@gm ail.com drshrishbhatnagar@gm ail.com rpsatarkar@rediffmail.c om sjb@kem.edu drnijhawansandeep@g mail.com b_ravishankar@yahoo. com of Committee Approval Status Date of Approval Is Independent Ethics Committee? Bio Ethics Forum for Gastroenterology and Gynecology Centre, for Dr. Punit Mehrotra Bio Ethics Forum of, An Independent Ethics Committee (BEFL), Approved 19/02/2012 Yes Approved 16/09/2012 Yes page 3 / 6
4 CTRI Website URL Ethics Committee Kodlikeri Memorial Hospital and CIIGMA Hospital, Aurangabad for Dr. Unmesh Takalkar Approved 28/01/2012 No Ethics Committee of KEM Hospital Research Centre, Pune. for Dr. S D Wagale Approved 23/02/2012 No Human Research Ethics Committee College Surat for Dr Sandeep V. Kansal Committee for Human Research (IECHR) for College & SSG Hospital,Vadodara, Dr. Mahendra Parmar Committee of SMS Hospital, for Dr. Sandeep Nijhawan Committee, Govt. Medical College for Satarkar Gastroenterology Centre, Dr. Ramesh Satarkar, Aurangabad Noble Hospital Committee, Pune for Dr. Sanjay Salunkhe Approved 31/01/2013 No Approved 06/03/2012 No Approved 16/03/2012 No Approved 24/02/2012 No Approved 11/02/2012 No Seth G S Medical College and KEM Hospital, Diamond Jubilee Society Trust Committee, Mumbai Approved 25/01/2012 No Swasthya Kalyan Independant Ethics Committee for Dr. Vijay Sharma Gastroenterology Clinic, Approved 09/02/2012 Yes Swasthya Kalyan Independant Ethics Committee for Global Gastro and Liver Centre, for Dr. Sunil Gupta Yashoda Academy of Medical Education and Research, Hyderabad Approved 16/03/2012 Yes Approved 28/10/2011 No page 4 / 6
5 CTRI Website URL Regulatory Clearance Status from DCGI Health Condition / Problems Studied Intervention / Comparator Agent Inclusion Criteria Status Date Approved/Obtained 16/12/2011 Health Type Patients Condition GASTROESOPHAGEAL REFLUX DISEASE (GERD) Type Details Intervention Comparator Agent Age From Age To Gender Details Dexlansoprazole 30mg(NERD) and 60mg(Erosive Oesophagitis) Lansoprazole 15mg(NERD) and 30mg(Erosive Oesophagitis) Year(s) Year(s) Both Inclusion Criteria Note: There is no upper age limit Single Capsule to be taken once daily before breakfast for 8 weeks. Single Capsule to be taken once daily before breakfast for 8 weeks. Screening Visit 1 1. Written, signed, dated and ethics committee approved informed consent obtained from patients before performing any study related procedures. 2. Patients of both the sexes of age 18 years and above, who can read and write. 3. The patients with the symptoms of GERD having a history of episodes of heartburn for more than or equal to 2 months prior to screening. Exclusion Criteria Screening Visit 2 and Baseline Visit 1. The patients who experience heartburn on at least 4 of the 7 days of the screening period. Exclusion Criteria Details Screening Visit 1: 1. Patient chronically using non steroidal anti inflammatory drugs including COX2 inhibitors (greater than 12 doses per month) other than aspirin (less than or equal to 325 mg is allowed) within last 14 days. 2. Need for continuous anticoagulant therapy 3. History of hypersensitivity to study drugs or any of its components 4. Severe unstable or uncontrolled disease 5. History of alcohol or drug abuse or dependence 6. Participation in an investigational study within 30 days prior to screening 7. Female who is pregnant or lactating or planning to become pregnant. 8. Woman of child bearing potential who is unwilling to use adequate methods of contraception. Screening Visit 2 and Baseline Visit 1. Patient with GERD complications like Endoscopic Barretts oesophagus and/or definite dysplastic changes in the oesophagus 2. Patient with ZollingerEllision syndrome or other hypersecretory condition 3. Patient with pyloric stenosis, oesophageal stricture, Schatzkis ring, oesophageal or Gastroesophageal surgery and planned surgery during the study duration. page 5 / 6
6 Powered by TCPDF ( PDF of Trial CTRI Website URL Method of Generating Random Sequence Method of Concealment Blinding/Masking Computer generated randomization Prenumbered or coded identical Containers Participant, Investigator and Outcome Assessor Blinded 4. A history of or active gastric or duodenal ulcers within 4 weeks prior or had significant acute upper gastrointestinal hemorrhage within 4 weeks of the baseline endoscopy. 5. Clinically significant laboratory values, as judged by the investigator (as per the assessment on screening visit 2 / Baseline visit) Primary Outcome Outcome Timepoints Mean Change in GSAS questionnaire sum scores (frequency, severity and distress scale) From baseline to 2, 4 and 8 weeks of treatment period. Secondary Outcome Outcome Timepoints Target Sample Size Phase of Trial Phase 3 Date of First Enrollment (India) Date of First Enrollment (Global) Estimated Duration of Trial Recruitment Status of Trial (Global) Recruitment Status of Trial (India) Publication Details Brief Summary Percentage of 24 hours heartburn free days From baseline to week 2, 4 and 8 Percentage of days without daytime heartburn From baseline to week 2, 4 and 8 Percentage of nights without night time heartburn From baseline to week 2, 4 and 8 Reduction in mean severity of heartburn From baseline to week 2, 4 and 8 Percentage of Patients who achieved sustained resolution of heartburn (7 consecutive heartburnfree days) Percentage of days without rescue medication use Percentage of treatment emergent adverse events At week 2, 4 and 8 Clinically significant changes in laboratory values Incidence and nature of adverse events Clinically significant changes in vital signs & systemic examinations Total Sample Size=242 Sample Size from India=242 02/04/2012 No Date Specified Years=1 Months=0 Days=0 Not Applicable Closed to Recruitment of Participants NIL This study is designed to assess the efficacy and safety of dexlansoprazole versus lansoprazole in patients with Gastroesophageal Reflux Disease (GERD). Total 242 patients will be participating in the study from 13 centers throughout India. The study will consist of 714 days of screening period and 8 weeks of treatment period. The primary endpoint of the study is to determine mean change in GSAS questionnaire sum scores from baseline to week 2, week 4 and week 8. Adverse events will be recorded during entire study period of the study. page 6 / 6
WARNING LETTER CERTIFIED MAIL RETURN RECEIPT REQUESTED. Ref: 06-HFD
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville, MD 20857 WARNING LETTER CERTIFIED MAIL RETURN RECEIPT REQUESTED Evangeline G. Gonzalez, M.D. Gonzalez
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville, MD 20857 WARNING LETTER CERTIFIED MAIL RETURN RECEIPT REQUESTED Evangeline G. Gonzalez, M.D. Gonzalez
DEPARTMENT OF HEALTH & HUMAN SERVICES WARNING LETTER. (b) (4) clinical investigation (Protocol entitled A Phase II, Multicenter,
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville, MD 20857 WARNING LETTER CERTIFIED MAIL RETURN RECEIPT REQUESTED Ronald Bukowski, M.D. 28099 Gates Mills
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville, MD 20857 WARNING LETTER CERTIFIED MAIL RETURN RECEIPT REQUESTED Ronald Bukowski, M.D. 28099 Gates Mills
Pablo Tebas, M.D. Joseph Quinn, RN, BSN Yan Jiang, RN, BSN, MSN
 Gilead Sciences, Inc. / Protocol Number GS-US-380-1489 Page 1 of 9 PARTNER PREGNANCY FOLLOW UP CONSENT FORM Sponsor / Study Title: Protocol Number: Principal Investigator: (Study Doctor) Gilead Sciences,
Gilead Sciences, Inc. / Protocol Number GS-US-380-1489 Page 1 of 9 PARTNER PREGNANCY FOLLOW UP CONSENT FORM Sponsor / Study Title: Protocol Number: Principal Investigator: (Study Doctor) Gilead Sciences,
Internal Medicine Curriculum Gastroenterology/Hepatology Rotation
 Internal Medicine Curriculum Gastroenterology/Hepatology Rotation Contact Person: Educational Purpose Gastrointestinal and hepatic disorders frequently cause patients to seek medical attention. Abdominal
Internal Medicine Curriculum Gastroenterology/Hepatology Rotation Contact Person: Educational Purpose Gastrointestinal and hepatic disorders frequently cause patients to seek medical attention. Abdominal
Croydon Health Services NHS Trust (Working in Partnership) Shared Care Guideline: Prescribing Agreement
 Shared Care Guideline: Prescribing Agreement Section A: To be completed by the hospital consultant initiating the treatment GP Practice Details: Name: Address: Tel no: Fax no: NHS.net e-mail: Consultant
Shared Care Guideline: Prescribing Agreement Section A: To be completed by the hospital consultant initiating the treatment GP Practice Details: Name: Address: Tel no: Fax no: NHS.net e-mail: Consultant
PDF of Trial CTRI Website URL -
 Clinical Trial Details (PDF Generation Date :- Sun, 02 Sep 2018 14:54:31 GMT) CTRI Nuber CTRI/2010/091/002909 [Registered on: 08/11/2010] - Last Modified On 26/02/2013 Post Graduate Thesis Type of Trial
Clinical Trial Details (PDF Generation Date :- Sun, 02 Sep 2018 14:54:31 GMT) CTRI Nuber CTRI/2010/091/002909 [Registered on: 08/11/2010] - Last Modified On 26/02/2013 Post Graduate Thesis Type of Trial
Medical research in the UK is a success story!
 Research in Scotland Medical research in the UK is a success story! Funding Publications University standing Novel drugs, treatments, service improvements We could do better! Threats: The costs of research
Research in Scotland Medical research in the UK is a success story! Funding Publications University standing Novel drugs, treatments, service improvements We could do better! Threats: The costs of research
Roles of Investigators in the Managements of Clinical Trials
 Roles of Investigators in the Managements of Clinical Trials Chii-Min Hwu, M.D. Section of General Medicine Department of Medicine Taipei Veterans General Hospital Learning Objectives PI Outlines How to
Roles of Investigators in the Managements of Clinical Trials Chii-Min Hwu, M.D. Section of General Medicine Department of Medicine Taipei Veterans General Hospital Learning Objectives PI Outlines How to
FERCI MODEL SOPs. [The IEC members (author/s, reviewer/s) and Chairperson will sign and date the SOP on this first page]
![FERCI MODEL SOPs. [The IEC members (author/s, reviewer/s) and Chairperson will sign and date the SOP on this first page] FERCI MODEL SOPs. [The IEC members (author/s, reviewer/s) and Chairperson will sign and date the SOP on this first page]](/thumbs/87/95993773.jpg) Title: SOP Code: SOP 12/V1 [The IEC members (author/s, reviewer/s) and Chairperson will sign and date the SOP on this first page] Prepared by: Dr. Padmaja Marathe, FERCI Member (Signature with Date) Reviewed
Title: SOP Code: SOP 12/V1 [The IEC members (author/s, reviewer/s) and Chairperson will sign and date the SOP on this first page] Prepared by: Dr. Padmaja Marathe, FERCI Member (Signature with Date) Reviewed
Fellowship Training Program in Digestive Diseases and Hepatology Stony Brook University Medical Center Northport Veterans Affairs Medical Center
 Fellowship Training Program in Digestive Diseases and Hepatology Stony Brook University Medical Center Northport Veterans Affairs Medical Center Inpatient GI Curriculum Goals and Objectives Revised December
Fellowship Training Program in Digestive Diseases and Hepatology Stony Brook University Medical Center Northport Veterans Affairs Medical Center Inpatient GI Curriculum Goals and Objectives Revised December
Upper GI Endoscopy a guide for patients and carers
 Upper GI Endoscopy a guide for patients and carers Welcome to the Endoscopy Unit. This information leaflet is intended to provide you with information about an upper endoscopy. It is not expected to cover
Upper GI Endoscopy a guide for patients and carers Welcome to the Endoscopy Unit. This information leaflet is intended to provide you with information about an upper endoscopy. It is not expected to cover
Internal Medicine Residency Program Rotation Curriculum
 University of California, Irvine Department of Medicine Internal Medicine Residency Program Rotation Curriculum I. Rotation Sites and Supervision Rotation Name: GASTROENTEROLOGY CONSULT Site Faculty Supervisor
University of California, Irvine Department of Medicine Internal Medicine Residency Program Rotation Curriculum I. Rotation Sites and Supervision Rotation Name: GASTROENTEROLOGY CONSULT Site Faculty Supervisor
DUKE INTERNAL MEDICINE RESIDENCY PROGRAM. GASTROENTEROLOGY SUBSPECIALTY CONSULTS (ELECTIVE) ROTATION DESCRIPTION Biliary, General GI and Hepatology
 Department of Medicine Internal Medicine Residency Program DUKE INTERNAL MEDICINE RESIDENCY PROGRAM GASTROENTEROLOGY SUBSPECIALTY CONSULTS (ELECTIVE) ROTATION DESCRIPTION Biliary, General GI and Hepatology
Department of Medicine Internal Medicine Residency Program DUKE INTERNAL MEDICINE RESIDENCY PROGRAM GASTROENTEROLOGY SUBSPECIALTY CONSULTS (ELECTIVE) ROTATION DESCRIPTION Biliary, General GI and Hepatology
E U R O P E A N F O R M A T
 E U R O P E A N F O R M A T F O R C U R R I C U L U M V I T A E DSC00951.JPG PERSONAL INFORMATIONS Name Antonio Iannetti Nationality Italian Date of birth 27/09/1949 WORKING EXPERIENCE From January 1980
E U R O P E A N F O R M A T F O R C U R R I C U L U M V I T A E DSC00951.JPG PERSONAL INFORMATIONS Name Antonio Iannetti Nationality Italian Date of birth 27/09/1949 WORKING EXPERIENCE From January 1980
Inpatient GI Consult Service LBJ General Hospital Curriculum for Year I, II & III Fellows
 Inpatient GI Consult Service LBJ General Hospital Curriculum for Year I, II & III Fellows Educational Purpose: The LBJ rotation comprises several aspects of clinical gastroenterology: consultation on inpatients
Inpatient GI Consult Service LBJ General Hospital Curriculum for Year I, II & III Fellows Educational Purpose: The LBJ rotation comprises several aspects of clinical gastroenterology: consultation on inpatients
Effect of a self-management program on patients with chronic disease Lorig K R, Sobel D S, Ritter P L, Laurent D, Hobbs M
 Effect of a self-management program on patients with chronic disease Lorig K R, Sobel D S, Ritter P L, Laurent D, Hobbs M Record Status This is a critical abstract of an economic evaluation that meets
Effect of a self-management program on patients with chronic disease Lorig K R, Sobel D S, Ritter P L, Laurent D, Hobbs M Record Status This is a critical abstract of an economic evaluation that meets
Division of Gastroenterology, Hepatology and Nutrition
 Jewish Hospital Goals: 1. Consultative and management prevalence in hepatology, pre- and post-liver transplantation. 2. Offer diagnostic and therapeutic procedure experience. Learning Objectives: Patient
Jewish Hospital Goals: 1. Consultative and management prevalence in hepatology, pre- and post-liver transplantation. 2. Offer diagnostic and therapeutic procedure experience. Learning Objectives: Patient
UNIVERSITY OF PENNSYLVANIA HEALTH SYSTEM
 Gilead Sciences, Inc. GS-US-248-0123, Amendment 1, 19-JUN-2012 A Long Term Follow-up Registry Study of Subjects Who Did Not Achieve Sustained Virologic Response in Gilead-Sponsored Trials in Subjects with
Gilead Sciences, Inc. GS-US-248-0123, Amendment 1, 19-JUN-2012 A Long Term Follow-up Registry Study of Subjects Who Did Not Achieve Sustained Virologic Response in Gilead-Sponsored Trials in Subjects with
Priority Program Translational Oncology Applicants' Guidelines Letter of Intent / Project Outlines
 Stiftung Deutsche Krebshilfe Dr. h.c. Fritz Pleitgen Präsident Spendenkonto Kreissparkasse Köln IBAN DE65 3705 0299 0000 9191 91 BIC COKSDE33XXX Priority Program Translational Oncology Applicants' Guidelines
Stiftung Deutsche Krebshilfe Dr. h.c. Fritz Pleitgen Präsident Spendenkonto Kreissparkasse Köln IBAN DE65 3705 0299 0000 9191 91 BIC COKSDE33XXX Priority Program Translational Oncology Applicants' Guidelines
Using Clinical Criteria for Evaluating Short Stays and Beyond. Georgeann Edford, RN, MBA, CCS-P. The Clinical Face of Medical Necessity
 Using Clinical Criteria for Evaluating Short Stays and Beyond Georgeann Edford, RN, MBA, CCS-P The Clinical Face of Medical Necessity 1 The Documentation Faces of Medical Necessity ç3 Setting the Stage
Using Clinical Criteria for Evaluating Short Stays and Beyond Georgeann Edford, RN, MBA, CCS-P The Clinical Face of Medical Necessity 1 The Documentation Faces of Medical Necessity ç3 Setting the Stage
Registry eform Data Entry Guidelines Version Apr 2014 Updated for eform on 20 Jun 2016
 Registry eform Data Entry Guidelines Version 1.0 02 Apr 2014 Updated for eform on 20 Jun 2016 Part 3 General recommendation for data entry in ProMISe and instructions of completion for the Follow up Form
Registry eform Data Entry Guidelines Version 1.0 02 Apr 2014 Updated for eform on 20 Jun 2016 Part 3 General recommendation for data entry in ProMISe and instructions of completion for the Follow up Form
DRAFT TEMPLATE FOR WRITING HOMEOPATHIC CLINICAL TRIAL PROTOCOLS VERSION-I. Study Summary Title. Title
 Title [The Title is short yet powerful description of the study to be undertaken. It may include phase, design, site, drug, and target disease(s)] Example: A phase II, randomized, double-blind, placebo-controlled,
Title [The Title is short yet powerful description of the study to be undertaken. It may include phase, design, site, drug, and target disease(s)] Example: A phase II, randomized, double-blind, placebo-controlled,
San Francisco Health Network Committee on Interdisciplinary Practice STANDARDIZED PROCEDURE NURSE PRACTITIONER / PHYSICIAN ASSISTANT PREAMBLE
 Community Health Network of San Francisco Health Network Committee on Interdisciplinary Practice STANDARDIZED PROCEDURE NURSE PRACTITIONER / PHYSICIAN ASSISTANT Title: CHILDREN S HEALTH CENTER I. Policy
Community Health Network of San Francisco Health Network Committee on Interdisciplinary Practice STANDARDIZED PROCEDURE NURSE PRACTITIONER / PHYSICIAN ASSISTANT Title: CHILDREN S HEALTH CENTER I. Policy
Adverse Drug Events: A Focus on Anticoagulation Steve Meisel, Pharm.D., CPPS Director of Patient Safety Fairview Health Services, Minneapolis, MN
 Adverse Drug Events: A Focus on Anticoagulation Steve Meisel, Pharm.D., CPPS Director of Patient Safety Fairview Health Services, Minneapolis, MN Fairview Health Services 6 hospitals, ranging from rural
Adverse Drug Events: A Focus on Anticoagulation Steve Meisel, Pharm.D., CPPS Director of Patient Safety Fairview Health Services, Minneapolis, MN Fairview Health Services 6 hospitals, ranging from rural
Long-Term Care Medication Management: A Demonstration Project. Training Support Deck July 2016
 Long-Term Care Medication Management: A Demonstration Project Training Support Deck July 2016 Overview Background Purpose and Guiding Principles of Demonstration Project Targeted Drug Classes Sample Scenario
Long-Term Care Medication Management: A Demonstration Project Training Support Deck July 2016 Overview Background Purpose and Guiding Principles of Demonstration Project Targeted Drug Classes Sample Scenario
ENDOSCOPY NURSE LED CONSENT PROCESS
 ENDOSCOPY NURSE LED CONSENT PROCESS Date issued January 2011 Reviewed By Next Review August 2012 Responsible H Chisholm C/N G Greenhill, C/N Halcrow, C/N Chisholm Contents Guideline Content 1.0 Introduction
ENDOSCOPY NURSE LED CONSENT PROCESS Date issued January 2011 Reviewed By Next Review August 2012 Responsible H Chisholm C/N G Greenhill, C/N Halcrow, C/N Chisholm Contents Guideline Content 1.0 Introduction
CLINICAL GUIDELINE FOR Management of NON-VARICEAL Upper GI haemorrhage
 CLINICAL GUIDELINE FOR Management of NON-VARICEAL Upper GI haemorrhage Suspected Non Variceal upper GI haemorrhage If any features suggest liver disease consult the variceal haemorrhage guideline http://www.rcht.nhs.uk/documentslibrary/royalcornw
CLINICAL GUIDELINE FOR Management of NON-VARICEAL Upper GI haemorrhage Suspected Non Variceal upper GI haemorrhage If any features suggest liver disease consult the variceal haemorrhage guideline http://www.rcht.nhs.uk/documentslibrary/royalcornw
Comprehensive Protocol Feasibility Questionnaire
 Protocol Title: Potential Principal Investigator: Regulatory Coordinators: Department Chair: PROJECT FEASIBILITY PI and Study Team: YOUR RESPONSES TO THIS SURVEY CONSTITUTE A BEST ESTIMATE OF RESOURCES
Protocol Title: Potential Principal Investigator: Regulatory Coordinators: Department Chair: PROJECT FEASIBILITY PI and Study Team: YOUR RESPONSES TO THIS SURVEY CONSTITUTE A BEST ESTIMATE OF RESOURCES
ROTATION DESCRIPTION - PGY1 Adult Internal Medicine
 ROTATION DESCRIPTION - PGY1 Adult Internal Medicine PURPOSE The IM rotation provides the opportunity for PGY1 residents to improve their knowledge base and pharmacotherapeutic skills while enhancing care
ROTATION DESCRIPTION - PGY1 Adult Internal Medicine PURPOSE The IM rotation provides the opportunity for PGY1 residents to improve their knowledge base and pharmacotherapeutic skills while enhancing care
Nurse Led Follow Up: Is It The Best Way Forward for Post- Operative Endometriosis Patients?
 Research Article Nurse Led Follow Up: Is It The Best Way Forward for Post- Operative Endometriosis Patients? R Mallick *, Z Magama, C Neophytou, R Oliver, F Odejinmi Barts Health NHS Trust, Whipps Cross
Research Article Nurse Led Follow Up: Is It The Best Way Forward for Post- Operative Endometriosis Patients? R Mallick *, Z Magama, C Neophytou, R Oliver, F Odejinmi Barts Health NHS Trust, Whipps Cross
DEPARTMENT OF SURGERY SECTION OF PEDIATRIC SURGERY PEDIATRIC SURGERY ROTATION (DSP)
 DEPARTMENT OF SURGERY SECTION OF PEDIATRIC SURGERY PEDIATRIC SURGERY ROTATION (DSP) C.S. Mott Children s Hospital Von Voigtlander Women s Hospital House Officer I House Officer II House Officer III Curriculum/Rotation
DEPARTMENT OF SURGERY SECTION OF PEDIATRIC SURGERY PEDIATRIC SURGERY ROTATION (DSP) C.S. Mott Children s Hospital Von Voigtlander Women s Hospital House Officer I House Officer II House Officer III Curriculum/Rotation
IBUPROFEN PATIENT GROUP DIRECTION CHILD HEALTH 1. Aim/Purpose of this Guideline
 IBUPROFEN PATIENT GROUP DIRECTION CHILD HEALTH 1. Aim/Purpose of this Guideline 1.1. This Patient Group Direction (PGD) applies to all nursing and clinical staff in the Child Health Department and its
IBUPROFEN PATIENT GROUP DIRECTION CHILD HEALTH 1. Aim/Purpose of this Guideline 1.1. This Patient Group Direction (PGD) applies to all nursing and clinical staff in the Child Health Department and its
Quality in Your Endoscopy Unit. David A. Greenwald, MD Mount Sinai Hospital Nancy S. Schlossberg, BSN, RN, CGRN NYSGE Course 2015 December 17, 2015
 Quality in Your Endoscopy Unit David A. Greenwald, MD Mount Sinai Hospital Nancy S. Schlossberg, BSN, RN, CGRN NYSGE Course 2015 December 17, 2015 Two Case Scenarios Patient with concerns about safety
Quality in Your Endoscopy Unit David A. Greenwald, MD Mount Sinai Hospital Nancy S. Schlossberg, BSN, RN, CGRN NYSGE Course 2015 December 17, 2015 Two Case Scenarios Patient with concerns about safety
Impact of Implementing Designed Nursing Intervention Protocol on Clinical Outcome of Patient with Peptic Ulcer. Amal Mohamed Ahmad
 Impact of Implementing Designed Nursing Intervention Protocol on Clinical Outcome of Patient with Peptic Ulcer By Amal Mohamed Ahmad Assistant Professor, Medical-Surgical Nursing, Faculty of Nursing, Aswan
Impact of Implementing Designed Nursing Intervention Protocol on Clinical Outcome of Patient with Peptic Ulcer By Amal Mohamed Ahmad Assistant Professor, Medical-Surgical Nursing, Faculty of Nursing, Aswan
Department of Surgery Surgical Endoscopy Goals and Objectives
 Department of Surgery Surgical Endoscopy Goals and Objectives Medical Knowledge and Patient Care: Residents must demonstrate understanding of anatomy and physiology of the gastrointestinal tract, with
Department of Surgery Surgical Endoscopy Goals and Objectives Medical Knowledge and Patient Care: Residents must demonstrate understanding of anatomy and physiology of the gastrointestinal tract, with
Deprescribing in a family health team: a study of chronic proton pump inhibitor use
 Deprescribing in a family health team: a study of chronic proton pump inhibitor use Kate Walsh BSc.H, BScPhm; 1 Debbie Kwan BScPhm, MSc; 2 Patricia Marr BScPhm, PharmD; 2 Christine Papoushek PharmD; 2
Deprescribing in a family health team: a study of chronic proton pump inhibitor use Kate Walsh BSc.H, BScPhm; 1 Debbie Kwan BScPhm, MSc; 2 Patricia Marr BScPhm, PharmD; 2 Christine Papoushek PharmD; 2
NEWCASTLE CLINICAL TRIALS UNIT STANDARD OPERATING PROCEDURES
 NEWCASTLE CLINICAL TRIALS UNIT STANDARD OPERATING PROCEDURES SOP details SOP title: Safety Reporting in CTIMPs and ATMPs SOP number: TM 003 SOP category: Trial Management Version number: 03 Version date:
NEWCASTLE CLINICAL TRIALS UNIT STANDARD OPERATING PROCEDURES SOP details SOP title: Safety Reporting in CTIMPs and ATMPs SOP number: TM 003 SOP category: Trial Management Version number: 03 Version date:
ETHICS COMMITTEE: ROLE, RESPONSIBILITIES AND FUNCTIONS K.R.CHANDRAMOHANAN NAIR DEPARTMENT OF ANATOMY, MEDICAL COLLEGE, THIRUVANANTHAPURAM
 ETHICS COMMITTEE: ROLE, RESPONSIBILITIES AND FUNCTIONS K.R.CHANDRAMOHANAN NAIR DEPARTMENT OF ANATOMY, MEDICAL COLLEGE, THIRUVANANTHAPURAM Outline Introduction Composition Responsibilities of IEC Responsibilities
ETHICS COMMITTEE: ROLE, RESPONSIBILITIES AND FUNCTIONS K.R.CHANDRAMOHANAN NAIR DEPARTMENT OF ANATOMY, MEDICAL COLLEGE, THIRUVANANTHAPURAM Outline Introduction Composition Responsibilities of IEC Responsibilities
GROUP PROTOCOL FOR THE MANAGEMENT of HEARTBURN and ACID REFLUX. Version 4 January 2014
 GROUP PROTOCOL FOR THE MANAGEMENT of HEARTBURN and ACID REFLUX Version 4 January 2014 RATIFYING COMMITTEE Drugs and Therapeutics Group DATE RATIFIED MAUP EXPIRES January 2017 EXECUTIVE SPONSOR Executive
GROUP PROTOCOL FOR THE MANAGEMENT of HEARTBURN and ACID REFLUX Version 4 January 2014 RATIFYING COMMITTEE Drugs and Therapeutics Group DATE RATIFIED MAUP EXPIRES January 2017 EXECUTIVE SPONSOR Executive
CLIC Clinical Investigator (And Site Staff) Certification
 CLIC Clinical Investigator (And Site Staff) Certification CLIC Level 1: 23-24 March 2015 CLIC Level 2: 25-27 March 2015 Cape Town (Venue to be confirmed) C PD points L e v e l 1 : 1 4 G e n e r a l, 2
CLIC Clinical Investigator (And Site Staff) Certification CLIC Level 1: 23-24 March 2015 CLIC Level 2: 25-27 March 2015 Cape Town (Venue to be confirmed) C PD points L e v e l 1 : 1 4 G e n e r a l, 2
Curriculum Vitae. Currently part of core team involved in establishing a Phase I and Proof of Concept research unit
 Curriculum Vitae Hari Ballabh Gupta B- 12, 13, Flat No 39, Indra Enclave Neb Sarai, Saket Delhi- 110068 Ph. No. 0091-9810604685, 9716776657 E. mail: hariballabhg@yahoo.co.in, Hariballabh.gupta@medanta.org
Curriculum Vitae Hari Ballabh Gupta B- 12, 13, Flat No 39, Indra Enclave Neb Sarai, Saket Delhi- 110068 Ph. No. 0091-9810604685, 9716776657 E. mail: hariballabhg@yahoo.co.in, Hariballabh.gupta@medanta.org
PROTOCOL FOR THE ADMINISTRATION OF SENNA. Formulary and Prescribing Guidelines
 PROTOCOL FOR THE ADMINISTRATION OF SENNA Formulary and Prescribing Guidelines Introduction This protocol allows for the administration of Senna by a registered nurse without a prescription from a doctor
PROTOCOL FOR THE ADMINISTRATION OF SENNA Formulary and Prescribing Guidelines Introduction This protocol allows for the administration of Senna by a registered nurse without a prescription from a doctor
NEWCASTLE CLINICAL TRIALS UNIT STANDARD OPERATING PROCEDURES
 NEWCASTLE CLINICAL TRIALS UNIT STANDARD OPERATING PROCEDURES SOP details SOP title: Safety Reporting in CTIMPs and ATMPs SOP number: TM-003 SOP category: Trial Management Version number: 04 Version date:
NEWCASTLE CLINICAL TRIALS UNIT STANDARD OPERATING PROCEDURES SOP details SOP title: Safety Reporting in CTIMPs and ATMPs SOP number: TM-003 SOP category: Trial Management Version number: 04 Version date:
managing or activities.
 STANDARD OPERATING PROCEDURE Clinical Research Monitoring TITLE: Site Initiation Visit TITLE: Site Initiation Visit 1. PURPOSE SOP Number: Version: 1.0 MICHR CRM MON 002 Effective Date: 19Dec2013 1.1 This
STANDARD OPERATING PROCEDURE Clinical Research Monitoring TITLE: Site Initiation Visit TITLE: Site Initiation Visit 1. PURPOSE SOP Number: Version: 1.0 MICHR CRM MON 002 Effective Date: 19Dec2013 1.1 This
20, avenue Appia CH-1211 Geneva 27 Switzerland Tel central Fax central
 SOP 408.4 Annex C 20, avenue Appia CH-1211 Geneva 27 Switzerland Tel central +41 22 791 2111 Fax central +41 22 791 3111 www.who.int WHO PUBLIC INSPECTION REPORT (WHOPIR) Contract Research Organization
SOP 408.4 Annex C 20, avenue Appia CH-1211 Geneva 27 Switzerland Tel central +41 22 791 2111 Fax central +41 22 791 3111 www.who.int WHO PUBLIC INSPECTION REPORT (WHOPIR) Contract Research Organization
Paper for the Health Board Quality and Safety Committee. Out of Hours Upper GI Haemorrhage
 Paper for the Health Board Quality and Safety Committee Out of Hours Upper GI Haemorrhage This short paper describes the current pathways within the Health Board for the management of out of hours emergency
Paper for the Health Board Quality and Safety Committee Out of Hours Upper GI Haemorrhage This short paper describes the current pathways within the Health Board for the management of out of hours emergency
GROUP PROTOCOL FOR THE MANAGEMENT OF SIMPLE INDIGESTION. Version 5 December 2017
 GROUP PROTOCOL FOR THE MANAGEMENT OF SIMPLE INDIGESTION Version 5 December 2017 RATIFYING COMMITTEE Drugs and Therapeutics Group DATE RATIFIED MAUP EXPIRES December 2020 EXECUTIVE SPONSOR Chief Nurse MAUP
GROUP PROTOCOL FOR THE MANAGEMENT OF SIMPLE INDIGESTION Version 5 December 2017 RATIFYING COMMITTEE Drugs and Therapeutics Group DATE RATIFIED MAUP EXPIRES December 2020 EXECUTIVE SPONSOR Chief Nurse MAUP
TITLE: Reporting Adverse Events SOP #: RCO-204 Page: 1 of 5 Effective Date: 01/31/18
 SOP #: RCO-204 Page: 1 of 5 1. POLICY STATEMENT: The research team is responsible for recognizing changes in subject health that may qualify as adverse events, classifying those results as defined in the
SOP #: RCO-204 Page: 1 of 5 1. POLICY STATEMENT: The research team is responsible for recognizing changes in subject health that may qualify as adverse events, classifying those results as defined in the
Pfizer Independent Grants for Learning & Change Request for Proposals (RFP) Antimicrobial Stewardship in the Asia-Pacific Region
 Pfizer Independent Grants for Learning & Change Request for Proposals (RFP) Antimicrobial Stewardship in the Asia-Pacific Region I. Background The Joint Commission, in collaboration with Pfizer Independent
Pfizer Independent Grants for Learning & Change Request for Proposals (RFP) Antimicrobial Stewardship in the Asia-Pacific Region I. Background The Joint Commission, in collaboration with Pfizer Independent
WARNING LETTER. Dear Dr. Wright : DEPARTMENT OF HEALTH & HUMAN SERVICES CERTIFIED MAIL RETURN RECEIPT REQUESTED
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville, MD 20857 WARNING LETTER CERTIFIED MAIL RETURN RECEIPT REQUESTED Richard A. Wright. M.D. Ref: 09-HFD-45-02-0
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville, MD 20857 WARNING LETTER CERTIFIED MAIL RETURN RECEIPT REQUESTED Richard A. Wright. M.D. Ref: 09-HFD-45-02-0
Submission of a clinical trial for access to ECRIN services Notice to the Applicant
 Submission of a clinical trial for access to ECRIN services Notice to the Applicant BEFORE SUBMITTING YOUR PROTOCOL Please, contact the European Correspondent (EuCo) in your country. The list of EuCos
Submission of a clinical trial for access to ECRIN services Notice to the Applicant BEFORE SUBMITTING YOUR PROTOCOL Please, contact the European Correspondent (EuCo) in your country. The list of EuCos
New Patient Registration Form NJR_NP_F100
 New Patient Registration Form NJR_NP_F100 Patient Last Name First Name Middle Name Maiden Name Address (Street or Box) City State Zip Code Home Phone Number Cell Phone Number Work Phone Number E-Mail Patient
New Patient Registration Form NJR_NP_F100 Patient Last Name First Name Middle Name Maiden Name Address (Street or Box) City State Zip Code Home Phone Number Cell Phone Number Work Phone Number E-Mail Patient
Study Management SM STANDARD OPERATING PROCEDURE FOR Adverse Event Reporting
 Study Management SM 306.00 STANDARD OPERATING PROCEDURE FOR Adverse Event Reporting Approval: Nancy Paris, MS, FACHE President and CEO 24 May 2017 (Signature and Date) Approval: Frederick M. Schnell, MD,
Study Management SM 306.00 STANDARD OPERATING PROCEDURE FOR Adverse Event Reporting Approval: Nancy Paris, MS, FACHE President and CEO 24 May 2017 (Signature and Date) Approval: Frederick M. Schnell, MD,
PATIENT GROUP DIRECTION (PGD) FOR Metronidazole 400mg Tablets
 Antibiotic Oral (tablet/capsule/suspension) PATIENT GROUP DIRECTION (PGD) FOR YOU MUST BE AUTHORISED BY NAME, UNDER THE CURRENT VERSION OF THIS PGD BEFORE YOU ATTEMPT TO WORK ACCORDING TO IT Caution: This
Antibiotic Oral (tablet/capsule/suspension) PATIENT GROUP DIRECTION (PGD) FOR YOU MUST BE AUTHORISED BY NAME, UNDER THE CURRENT VERSION OF THIS PGD BEFORE YOU ATTEMPT TO WORK ACCORDING TO IT Caution: This
CLIENT REFERRAL PACKAGE
 p HEARTWOOD CENTRE FOR WOMEN CLIENT REFERRAL PACKAGE REFERRAL INFORMATION PACKAGE Heartwood, a residential treatment program, is a provincial tertiary 30 bed resource for women with substance dependence,
p HEARTWOOD CENTRE FOR WOMEN CLIENT REFERRAL PACKAGE REFERRAL INFORMATION PACKAGE Heartwood, a residential treatment program, is a provincial tertiary 30 bed resource for women with substance dependence,
Patient: Gender: Male Female. Mailing Address: Ethnicity: Not Hispanic or Latin Hispanic/Latin Home Phone #:
 5002 Highway 39 N Bldg. A Meridian, MS 39301 Phone: 601-512-0500 Fax: 601-512-0505 Patient Information Patient: Gender: Male Female First Middle Last Primary Language: English Spanish Other Mailing Address:
5002 Highway 39 N Bldg. A Meridian, MS 39301 Phone: 601-512-0500 Fax: 601-512-0505 Patient Information Patient: Gender: Male Female First Middle Last Primary Language: English Spanish Other Mailing Address:
Patient survey report Survey of people who use community mental health services 2011 Pennine Care NHS Foundation Trust
 Patient survey report 2011 Survey of people who use community mental health services 2011 The national Survey of people who use community mental health services 2011 was designed, developed and co-ordinated
Patient survey report 2011 Survey of people who use community mental health services 2011 The national Survey of people who use community mental health services 2011 was designed, developed and co-ordinated
MEDICINES CONTROL COUNCIL
 MEDICINES CONTROL COUNCIL SECTION 21 APPLICATION FORM Only to be used for orthodox/allopathic medicines for human use. 1. Fax completed form (i.e. pages 1-10), proof of payment of application fee (if applicable)
MEDICINES CONTROL COUNCIL SECTION 21 APPLICATION FORM Only to be used for orthodox/allopathic medicines for human use. 1. Fax completed form (i.e. pages 1-10), proof of payment of application fee (if applicable)
Patient Group Direction for Aspirin 300mg Version: 02 Start Date: 1 st October 2017 Expiry Date: 30 th September 2019
 THIS PATIENT GROUP DIRECTION HAS BEEN AGREED BY THE FOLLOWING ORGANISATIONS: CLINICAL COMMISSIONING GROUP: Doncaster CCG, Lancashire CCGs including East Lancashire, Fylde and Wyre and North Lancashire
THIS PATIENT GROUP DIRECTION HAS BEEN AGREED BY THE FOLLOWING ORGANISATIONS: CLINICAL COMMISSIONING GROUP: Doncaster CCG, Lancashire CCGs including East Lancashire, Fylde and Wyre and North Lancashire
Setting The economic study was conducted in a large teaching hospital in Amsterdam, the Netherlands.
 Early switch from intravenous to oral antibiotics: guidelines and implementation in a large teaching hospital Sevinc F, Prins J M, Koopmans R P, Langendijk P N, Bossuyt P M, Dankert J, Speelman P Record
Early switch from intravenous to oral antibiotics: guidelines and implementation in a large teaching hospital Sevinc F, Prins J M, Koopmans R P, Langendijk P N, Bossuyt P M, Dankert J, Speelman P Record
Efficacy of Tympanostomy Tubes for Children with Recurrent Acute Otitis Media Randomization Phase
 CONSENT FOR A CHILD TO BE A SUBJECT IN MEDICAL RESEARCH AND AUTHORIZATION TO PERMIT THE USE AND SHARING OF IDENTIFIABLE MEDICAL INFORMATION FOR RESEARCH PURPOSES TITLE Efficacy of Tympanostomy Tubes for
CONSENT FOR A CHILD TO BE A SUBJECT IN MEDICAL RESEARCH AND AUTHORIZATION TO PERMIT THE USE AND SHARING OF IDENTIFIABLE MEDICAL INFORMATION FOR RESEARCH PURPOSES TITLE Efficacy of Tympanostomy Tubes for
Crisis Triage, Walk-ins and Mobile Crisis Services
 Section 10.15 Crisis Triage, Walk-ins and Mobile Crisis Services 10.15.1 Introduction 10.15.2 References 10.15.3 Scope 10.15.4 Did you know? 10.15.5 Definitions 10.15.6 Procedures 10.15.6-A Triage 10.15.6-B
Section 10.15 Crisis Triage, Walk-ins and Mobile Crisis Services 10.15.1 Introduction 10.15.2 References 10.15.3 Scope 10.15.4 Did you know? 10.15.5 Definitions 10.15.6 Procedures 10.15.6-A Triage 10.15.6-B
General Administration GA STANDARD OPERATING PROCEDURE FOR Sponsor Responsibility and Delegation of Responsibility
 General Administration GA 102.01 STANDARD OPERATING PROCEDURE FOR Sponsor Responsibility and Delegation of Responsibility Approval: Nancy Paris, MS, FACHE President and CEO (17 July 2014) (Signature and
General Administration GA 102.01 STANDARD OPERATING PROCEDURE FOR Sponsor Responsibility and Delegation of Responsibility Approval: Nancy Paris, MS, FACHE President and CEO (17 July 2014) (Signature and
Gastroenterology. & Hepatology. Gastroenterology. Saturday, September 12, Best Practices & New Developments In
 8th Annual Course Fall 2015 Best Practices & New Developments In 8th Annual Course Fall 2015 Best Practices & New Developments In Gastroenterology & Hepatology Gastroenterology & Hepatology Saturday, September
8th Annual Course Fall 2015 Best Practices & New Developments In 8th Annual Course Fall 2015 Best Practices & New Developments In Gastroenterology & Hepatology Gastroenterology & Hepatology Saturday, September
Upper gastro-intestinal (GI) endoscopy
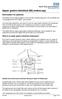 Information for patients This leaflet can be made available in other formats including large print, CD and Braille and in languages other than English, upon request. You have been advised by your doctor
Information for patients This leaflet can be made available in other formats including large print, CD and Braille and in languages other than English, upon request. You have been advised by your doctor
a. General E Code Coding Guidelines
 19. Supplemental Classification of External Causes of Injury and Poisoning (E-codes, E800-E999) Introduction: These guidelines are provided for those who are currently collecting E codes in order that
19. Supplemental Classification of External Causes of Injury and Poisoning (E-codes, E800-E999) Introduction: These guidelines are provided for those who are currently collecting E codes in order that
Job Title Name Signature Date
 Supply of Fluconazole 150mg capsule by Community Pharmacists working in Forth Valley Pharmacies under NHS Minor Ailment Service. Protocol Number 125 Version 5 Date protocol prepared: November 2014 Date
Supply of Fluconazole 150mg capsule by Community Pharmacists working in Forth Valley Pharmacies under NHS Minor Ailment Service. Protocol Number 125 Version 5 Date protocol prepared: November 2014 Date
36 th Annual Meeting Preconference Workshop P4 Handout
 36 th Annual Meeting Preconference Workshop P4 Handout Clinical Trial Management in Multicenter Trials: Collaborating for Success Meet Our Team Dixie Ecklund, RN, MSN, MBA Associate Director, University
36 th Annual Meeting Preconference Workshop P4 Handout Clinical Trial Management in Multicenter Trials: Collaborating for Success Meet Our Team Dixie Ecklund, RN, MSN, MBA Associate Director, University
Checklists for Preventing and Controlling
 Checklists for Preventing and Controlling Clostridium difficile Infection (CDI) This document has been developed to specifically assist senior management and all ward staff to take appropriate actions,
Checklists for Preventing and Controlling Clostridium difficile Infection (CDI) This document has been developed to specifically assist senior management and all ward staff to take appropriate actions,
Preparing GI ASCs for October 2012
 Preparing GI ASCs for October 2012 Anita J. Bhatia, PHD, MPH, Centers for Medicare and Medicaid Services Lawrence B. Cohen, MD, FACG, AGAF, FASGE, New York Gastroenterology Associates Lawrence R. Kosinski,
Preparing GI ASCs for October 2012 Anita J. Bhatia, PHD, MPH, Centers for Medicare and Medicaid Services Lawrence B. Cohen, MD, FACG, AGAF, FASGE, New York Gastroenterology Associates Lawrence R. Kosinski,
https://register.clinicaltrials.gov/prs/app/template/help%2chelpprotocolmo...
 Page 1 of 12 Help: Protocol Modules > All Help: Protocol Modules - All Protocol module help pages contain an introduction, examples, data entry tips and a review checklist for each module in the protocol
Page 1 of 12 Help: Protocol Modules > All Help: Protocol Modules - All Protocol module help pages contain an introduction, examples, data entry tips and a review checklist for each module in the protocol
WARNING LETTER CERTIFIED MAIL RETURN RECEIPT REQUESTED
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville, MD 20857 WARNING LETTER CERTIFIED MAIL RETURN RECEIPT REQUESTED Charles J. Coté, M.D. Ref: 09-HFD-45-02-04
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville, MD 20857 WARNING LETTER CERTIFIED MAIL RETURN RECEIPT REQUESTED Charles J. Coté, M.D. Ref: 09-HFD-45-02-04
DANNOAC-AF synopsis. [Version 7.9v: 5th of April 2017]
![DANNOAC-AF synopsis. [Version 7.9v: 5th of April 2017] DANNOAC-AF synopsis. [Version 7.9v: 5th of April 2017]](/thumbs/77/75232655.jpg) DANNOAC-AF synopsis. [Version 7.9v: 5th of April 2017] A quality of care assessment comparing safety and efficacy of edoxaban, apixaban, rivaroxaban and dabigatran for oral anticoagulation in patients
DANNOAC-AF synopsis. [Version 7.9v: 5th of April 2017] A quality of care assessment comparing safety and efficacy of edoxaban, apixaban, rivaroxaban and dabigatran for oral anticoagulation in patients
QUALITY ASSURANCE PROGRAM
 QUALITY ASSURANCE PROGRAM Elaine Armstrong, MS Quality Assurance Manager PURPOSE Verify accuracy of submitted data Verify compliance with protocol and regulatory requirements Provide educational support
QUALITY ASSURANCE PROGRAM Elaine Armstrong, MS Quality Assurance Manager PURPOSE Verify accuracy of submitted data Verify compliance with protocol and regulatory requirements Provide educational support
Dr. Simi Jesto Joseph, DNP, APN, NP-C
 1 Dr. Simi Jesto Joseph, DNP, APN, NP-C sjoseph@gisolutionsinc.com 7447 W Talcott Ave Suite 209, Chicago IL Ph: 773-631-2728 Fax:773-631-8083 Summary of Qualifications Dedicated doctorally prepared nurse
1 Dr. Simi Jesto Joseph, DNP, APN, NP-C sjoseph@gisolutionsinc.com 7447 W Talcott Ave Suite 209, Chicago IL Ph: 773-631-2728 Fax:773-631-8083 Summary of Qualifications Dedicated doctorally prepared nurse
Policies Approved by the 2017 ASHP House of Delegates
 House of Delegates Policies Approved by the 2017 ASHP House of Delegates 1701 Ensuring Patient Safety and Data Integrity During Cyber-attacks Source: Council on Pharmacy Management To advocate that healthcare
House of Delegates Policies Approved by the 2017 ASHP House of Delegates 1701 Ensuring Patient Safety and Data Integrity During Cyber-attacks Source: Council on Pharmacy Management To advocate that healthcare
Platinum Local Access+ HMO $25 OffEx
 Platinum Local Access+ HMO $25 OffEx Benefit Summary (For groups 1 to 50) (Uniform Health Plan Benefits and Coverage Matrix) Blue Shield of California Effective January 1, 2015 THIS MATRIX IS INTENDED
Platinum Local Access+ HMO $25 OffEx Benefit Summary (For groups 1 to 50) (Uniform Health Plan Benefits and Coverage Matrix) Blue Shield of California Effective January 1, 2015 THIS MATRIX IS INTENDED
Indicator description
 Patients with a primary care visit within 7 days of acute discharge for Quality Improvement Plans - Primary Care Resource for Indicator Standards (RIS) Health Analytics Branch, Ministry of Health and Long-Term
Patients with a primary care visit within 7 days of acute discharge for Quality Improvement Plans - Primary Care Resource for Indicator Standards (RIS) Health Analytics Branch, Ministry of Health and Long-Term
STATE INSURANCE CORPORATION PANCHDEEP BHAVAN: C.I.G. MARG. NEW DELHI
 EMPLOYEES' CONTRACTUAL STATE INSURANCE CORPORATION PANCHDEEP BHAVAN: C.I.G. MARG. NEW DELHI- 110002 ENGAGEMENT OF CONSULTANTS. SPECIALISTS GR. 11 (SR. SCALE) & SPECIALIST GR. 11 (JR. SCALE) The Employees'
EMPLOYEES' CONTRACTUAL STATE INSURANCE CORPORATION PANCHDEEP BHAVAN: C.I.G. MARG. NEW DELHI- 110002 ENGAGEMENT OF CONSULTANTS. SPECIALISTS GR. 11 (SR. SCALE) & SPECIALIST GR. 11 (JR. SCALE) The Employees'
Study of socio-demographic determinants of class IV employees in a tertiary care teaching hospital in Mumbai
 Original article: Study of socio-demographic determinants of class IV employees in a tertiary care teaching hospital in Mumbai Prachi V. Chakor (Palwe) 1, Armaity S. Dehmubed 2, Purushottam A. Giri 3,
Original article: Study of socio-demographic determinants of class IV employees in a tertiary care teaching hospital in Mumbai Prachi V. Chakor (Palwe) 1, Armaity S. Dehmubed 2, Purushottam A. Giri 3,
Study Title: Optimal resuscitation in pediatric trauma an EAST multicenter study
 Study Title: Optimal resuscitation in pediatric trauma an EAST multicenter study PI/senior researcher: Richard Falcone Jr. MD, MPH Co-primary investigator: Stephanie Polites MD, MPH; Juan Gurria MD My
Study Title: Optimal resuscitation in pediatric trauma an EAST multicenter study PI/senior researcher: Richard Falcone Jr. MD, MPH Co-primary investigator: Stephanie Polites MD, MPH; Juan Gurria MD My
PREOPERATIVE PATIENT QUESTIONAIRE
 PREOPERATIVE PATIENT QUESTIONAIRE Name Age Sex Ht Wt PATIENT INFORMATION New Patient Name Change Address Change Insurance Change This questionnaire is designed to assist the anesthesiologist who will be
PREOPERATIVE PATIENT QUESTIONAIRE Name Age Sex Ht Wt PATIENT INFORMATION New Patient Name Change Address Change Insurance Change This questionnaire is designed to assist the anesthesiologist who will be
3HP A WAY TO DO IT INITIATION OF 3HP IN A STATEWIDE TB PROGRAM MISSISSIPPI STATE DEPARTMENT OF HEALTH
 3HP A WAY TO DO IT INITIATION OF 3HP IN A STATEWIDE TB PROGRAM MISSISSIPPI STATE DEPARTMENT OF HEALTH NTCA April 2017, Atlanta GA TB Education and Training Projects: Updates from the Field August 10, 2017
3HP A WAY TO DO IT INITIATION OF 3HP IN A STATEWIDE TB PROGRAM MISSISSIPPI STATE DEPARTMENT OF HEALTH NTCA April 2017, Atlanta GA TB Education and Training Projects: Updates from the Field August 10, 2017
STATUS OF MATERNAL, INFANT, AND YOUNG CHILD NUTRITION (MIYCN) IN MEDICAL COLLEGES & HOSPITALS
 STATUS OF MATERNAL, INFANT, AND YOUNG CHILD NUTRITION (MIYCN) IN MEDICAL COLLEGES & HOSPITALS KEY FINDINGS BASELINE ASSESSMENT 2017 UTTAR PRADESH & BIHAR Image: Velocity Creative Introduction Despite a
STATUS OF MATERNAL, INFANT, AND YOUNG CHILD NUTRITION (MIYCN) IN MEDICAL COLLEGES & HOSPITALS KEY FINDINGS BASELINE ASSESSMENT 2017 UTTAR PRADESH & BIHAR Image: Velocity Creative Introduction Despite a
Version 4 January 18, Principal Investigator: James F. Marion, M.D. The Mount Sinai School of Medicine
 Guidelines for Completing Case Report Forms For A Six-Week Randomized Double-Blind, Controlled Trial of High Dose Asacol (6.0 g/day) Versus Low Dose Asacol (2.4 or 3.6 g/day) for the Treatment of Mild
Guidelines for Completing Case Report Forms For A Six-Week Randomized Double-Blind, Controlled Trial of High Dose Asacol (6.0 g/day) Versus Low Dose Asacol (2.4 or 3.6 g/day) for the Treatment of Mild
Stanford Multiorgan Transplant Surgery: R-1 Tuesday, February 02, 2016
 Stanford University General Surgery Residency Program Abdominal Transplant Surgery Goals and Objectives for Residents: R-1 Rotation Director: Carlos Esquivel, M.D., Ph.D. Description The Abdominal Transplant
Stanford University General Surgery Residency Program Abdominal Transplant Surgery Goals and Objectives for Residents: R-1 Rotation Director: Carlos Esquivel, M.D., Ph.D. Description The Abdominal Transplant
Abstract Rules & Regulations
 Overview 1. Deadline: Abstracts may be submitted until Tuesday, July 5, 2016, 5:00 PM Pacific Time US. 2. Payment: There is no charge for submission. 3. Prior Publication Policy: a. ACP policy allows for
Overview 1. Deadline: Abstracts may be submitted until Tuesday, July 5, 2016, 5:00 PM Pacific Time US. 2. Payment: There is no charge for submission. 3. Prior Publication Policy: a. ACP policy allows for
Study Responsibilities. Choose all that apply. f. Draw/collect laboratory specimens
 Wichita State University Institutional Review Board (IRB) New Study Application Investigator Information Principal Investigator must be a WSU faculty member. Students and anyone outside of WSU are listed
Wichita State University Institutional Review Board (IRB) New Study Application Investigator Information Principal Investigator must be a WSU faculty member. Students and anyone outside of WSU are listed
A descriptive study to assess the burden among family care givers of mentally ill clients
 IOSR Journal of Nursing and Health Science (IOSR-JNHS) e-issn: 2320 1959.p- ISSN: 2320 1940 Volume 3, Issue 3 Ver. IV (May-Jun. 2014), PP 61-67 A descriptive study to assess the burden among family care
IOSR Journal of Nursing and Health Science (IOSR-JNHS) e-issn: 2320 1959.p- ISSN: 2320 1940 Volume 3, Issue 3 Ver. IV (May-Jun. 2014), PP 61-67 A descriptive study to assess the burden among family care
RULE RESPONSIBILITIES OF A PHYSICIAN WHO ENGAGES IN DRUG THERAPY MANAGEMENT WITH A COLORADO LICENSED PHARMACIST
 DEPARTMENT OF REGULATORY AGENCIES Colorado Medical Board RULE 900 - RESPONSIBILITIES OF A PHYSICIAN WHO ENGAGES IN DRUG THERAPY MANAGEMENT WITH A COLORADO LICENSED PHARMACIST 3 CCR 713-32 [Editor s Notes
DEPARTMENT OF REGULATORY AGENCIES Colorado Medical Board RULE 900 - RESPONSIBILITIES OF A PHYSICIAN WHO ENGAGES IN DRUG THERAPY MANAGEMENT WITH A COLORADO LICENSED PHARMACIST 3 CCR 713-32 [Editor s Notes
CLINICAL PRACTICE EVALUATION II: CLINICAL SYSTEMS REVIEW
 Diplomate: CLINICAL PRACTICE EVALUATION II: CLINICAL SYSTEMS REVIEW A. INFORMATION MANAGEMENT 1. Does your practice currently use an electronic medical record system? Yes No 2. If Yes, how long has the
Diplomate: CLINICAL PRACTICE EVALUATION II: CLINICAL SYSTEMS REVIEW A. INFORMATION MANAGEMENT 1. Does your practice currently use an electronic medical record system? Yes No 2. If Yes, how long has the
Questions and Answers on the CMS Comprehensive Care for Joint Replacement Model
 Questions and Answers on the CMS Comprehensive Care for Joint Replacement Model MEGGAN BUSHEE, ESQ. 704.343.2360 mbushee@mcguirewoods.com 201 North Tryon Street, Suite 3000 Charlotte, North Carolina 28202-2146
Questions and Answers on the CMS Comprehensive Care for Joint Replacement Model MEGGAN BUSHEE, ESQ. 704.343.2360 mbushee@mcguirewoods.com 201 North Tryon Street, Suite 3000 Charlotte, North Carolina 28202-2146
Medical Intensive Care Unit Rotation EUHM
 PGY 2 Residency Training Program Medical Intensive Care Unit Rotation EUHM Preceptor: Derek M. Polly, PharmD Office: EUHM, 2 nd Floor, Room 2182 Hours: ~ 7:30 4:00 Desk: 404 686 5674 Pager: 404 686 5500
PGY 2 Residency Training Program Medical Intensive Care Unit Rotation EUHM Preceptor: Derek M. Polly, PharmD Office: EUHM, 2 nd Floor, Room 2182 Hours: ~ 7:30 4:00 Desk: 404 686 5674 Pager: 404 686 5500
PAYMENT IS REQUIRED AT THE TIME SERVICES ARE RENDERED. THANK YOU!
 PATIENT INFORMATION FORM PATIENT DATA: - - PATIENT NAME (LAST, FIRST, MIDDLE) SOCIAL SECURITY # SEX ( ) - ( ) - ADDRESS HOME PHONE NUMBER MOBILE PHONE NUMBER CITY STATE ZIP CODE OCCUPATION / / DATE OF
PATIENT INFORMATION FORM PATIENT DATA: - - PATIENT NAME (LAST, FIRST, MIDDLE) SOCIAL SECURITY # SEX ( ) - ( ) - ADDRESS HOME PHONE NUMBER MOBILE PHONE NUMBER CITY STATE ZIP CODE OCCUPATION / / DATE OF
International Journal of Scientific and Research Publications, Volume 4, Issue 1, January ISSN
 International Journal of Scientific and Research Publications, Volume 4, Issue 1, January 2014 1 A study to assess the effectiveness of planned teaching programme on of staff nurses regarding prevention
International Journal of Scientific and Research Publications, Volume 4, Issue 1, January 2014 1 A study to assess the effectiveness of planned teaching programme on of staff nurses regarding prevention
September 24, Dear Administrator Verma:
 Seema Verma, MD Centers for Medicare and Medicaid Services Department of Health and Human Services Attention: P.O. Box 8013 Baltimore, MD 21244-1850 RE: [] Medicare Program: Proposed Changes to Hospital
Seema Verma, MD Centers for Medicare and Medicaid Services Department of Health and Human Services Attention: P.O. Box 8013 Baltimore, MD 21244-1850 RE: [] Medicare Program: Proposed Changes to Hospital
Hospital at home or acute hospital care: a cost minimisation analysis Coast J, Richards S H, Peters T J, Gunnell D J, Darlow M, Pounsford J
 Hospital at home or acute hospital care: a cost minimisation analysis Coast J, Richards S H, Peters T J, Gunnell D J, Darlow M, Pounsford J Record Status This is a critical abstract of an economic evaluation
Hospital at home or acute hospital care: a cost minimisation analysis Coast J, Richards S H, Peters T J, Gunnell D J, Darlow M, Pounsford J Record Status This is a critical abstract of an economic evaluation
Madhya Pradesh Biotechnology Council
 Madhya Pradesh Biotechnology Council 26, Kisan Bhavan, IIIrd floor, Jail road, Arera Hills, Bhopal - 4620011 e- mail - mpbiotechnologycouncil @ gmail.com Tel.No. 0755-2577185, 186 Fax- 0755-2577187 APPLICATION
Madhya Pradesh Biotechnology Council 26, Kisan Bhavan, IIIrd floor, Jail road, Arera Hills, Bhopal - 4620011 e- mail - mpbiotechnologycouncil @ gmail.com Tel.No. 0755-2577185, 186 Fax- 0755-2577187 APPLICATION
Standard Operating Procedures
 Clinical Monitoring and Site Verification Procedure Overview To define the standard procedures for preparation and documentation of site visits for clinical monitoring and spoke verification for any NETT
Clinical Monitoring and Site Verification Procedure Overview To define the standard procedures for preparation and documentation of site visits for clinical monitoring and spoke verification for any NETT
Registration of Clinical Trials: Background and Implementation
 Registration of Clinical Trials: Background and Implementation Ida Sim, MD, PhD Project Coordinator Department of Research Policy and Cooperation World Health Organization Geneva, Switzerland; and University
Registration of Clinical Trials: Background and Implementation Ida Sim, MD, PhD Project Coordinator Department of Research Policy and Cooperation World Health Organization Geneva, Switzerland; and University
