Annual Symposium for Research Administrators
|
|
|
- Andra Moore
- 5 years ago
- Views:
Transcription
1 Annual Symposium for Research Administrators Hosted by The Office of Sponsored Research Wednesday, July 29, :30 am - 9:00 am Registration Check-in & Coffee Chapman Hall 211 9:00 am - 9:10 am Welcome Chapman Hall 211 9:10 am - 10:00 am Keynote Speaker: Tim Quigg Associate Chair Emeritus Administration, Finance and Entrepreneurship Computer Science Department, UNC-Chapel Hill SRA Distinguished Faculty Emeritus 10:00 am - 10:20 am Break Attendees traverse to Computer Science Complex: Sitterson and Brooks (see insert for map)
2 10:20 am - 11:20 am Concurrent Sessions A A1 A2 014 A3 008 A4 Annual Symposium July 29th 2015 Grant Reporting in InfoPorte (also offered during Concurrent Session B) Learn what resources are available to manage grant information through InfoPorte s OSR Ledger Rollup as well as through the Transactions tab for OSR budget and expenses. Learn how you can use the Grant Profile tab to see detailed information on the project and award and how SAS reporting can be utilized for detail account expenditure information. Presenter: Nicole Sebik, Business Systems Analyst, Finance Business Analysis, (former ITS Trainer) Uniform Guidance: Key Points of the UNC Implementation Getting to the Real Impacts of 2 CFR Part 200 New guidance in the management of federal funds went into effect late last year. You ve likely heard about 2 CFR Part 200 (at OSR Quarterly Forums) and perhaps even read about it (in OSR Newsletters). Did you know that we are now classified as an IHE Institution of Higher Education? So much is new, so let s take the dive and get into the most impactful aspects of these regulations and UNC s interpretation of this federal guidance/law. Topics for discussion will include direct costing, prior approval, indirect cost (F&A), subrecipient monitoring and reporting changes. Join us to get into the weeds with these new regulations and understand when they are applied to your federal funding. Presenters: Robin Cyr, AVC for Research & Director Office of Sponsored Research Sharon Brooks, Director, Post Award & Cash Management, Office of Sponsored Research Conflict of Interest As research administrators you play an important role in the Conflict of Interest process both at the individual and institutional level. In this session, you will get an overview of the federal requirements and tips on being proactive in getting COI disclosures and training completed in a timely fashion for your grants. Presenter: Joy Bryde, Conflict of Interest Officer, Office of the Vice Chancellor for Research eipf as a Compliance Tool The top ten things to know for a successful pre-award submission. Presenter: Laurie Henry, Director of Pre-Award Services, Office of Sponsored Research 11:30 am 12:30 pm Concurrent Sessions B B1 B2 011 B3 Grant Reporting in InfoPorte (also offered during Concurrent Sesson A) Learn what resources are available to manage grant information through InfoPorte s OSR Ledger Rollup as well as through the Transactions tab for OSR budget and expenses. Learn how you can use the Grant Profile tab to see detailed information on the project and award and how SAS reporting can be utilized for detail account expenditure information. Presenter: Nicole Sebik, Business Systems Analyst, Finance Business Analysis, (former ITS Trainer) F&A Rate Overview Overview of the Facilities and Administrative (F&A) rate proposal development process including definitions, indirect vs. direct costs and grouping in pools and bases, rate calculations, and the rate negotiation process with the Federal Government. Presentation will also discuss UNC-CH s F&A waiver policy. Presenter: Brian Bertlshofer, Director of Cost Analysis, Office of Sponsored Research NIH Compliance: Public Access Policy & Biosketch This session will cover the NIH Public Access Policy and compliance requirements, including a tour of the bibliography management and compliance monitoring functions in My NCBI. This session will also introduce the new NIH Biographical Sketch format requirements effective for NIH and AHRQ grant applications submitted as of May 25, This class will include a walk-through of the logistical steps in My NCBI for 1) NIH Policy compliance and 2) preparing a bibliography with a public link for addition to the new Biosketch. Presenter: Barrie Hayes, Bioinformatics & Translational Science Librarian, HSL-Library Administration
3 B4 141 (on 1st floor above ) Annual Symposium July 29th 2015 Subrecipient Assessment and Monitoring It Takes a Village! Given enhanced language in the Uniform Guidance (2 CFR Part 200) related to the oversight of subrecipient relationships, this session will delve into the: Who (is involved and responsible)? What (needs to be done)? When (at what point in the subrecipient life cycle)? How (do we document our oversight)? Each of us, including the PIs, as we are involved in the life cycle of these funding relationships will have a role in helping to ensure that the external parties are providing the level of stewardship and responsibility that is expected in the management of sponsored funds as a partner to UNC. Presenters: Laurie Henry, Director of Pre-Award Services, Office of Sponsored Research Sharon Brooks, Director, Post Award & Cash Management, Office of Sponsored Research 12:30 pm - 1:30 pm Box Lunch Round Table Discussions 1:30 pm - 2:30 pm Concurrent Sessions C C1 011 C2 008 C3 C4/ D4 141 (on 1st floor above ) Sitterson Hall Lower Lobby See insert for descriptions and room location Grant Transactions: Preparing and Tracking Voucher and Journal Entries in ConnectCarolina Most transactions go through 5 basic steps in the ConnectCarolina. Learn more about these steps and what to watch for from a grants perspective. For example, budget checking/errors; payment posting; document and approval requirements; budget versus actuals. This session will help you better navigate in ConnectCarolina and how to follow the lifecycle of a transaction through the system. Presenter: Susan McDonough, ITS Trainer Cost Transfer Policy In response to the Office of the Inspector General Audit findings, the Cost Transfer policy and procedure have been updated. This overview will provide information about timeliness and documentation requirements as well as the need for proper allocability, allowability and reasonableness when justifying a cost transfer. Presenter: Vanessa Peoples, Deputy Director, Office of Sponsored Research Audits and Reviews Who Is Looking, Lessons Learned and How Can We be Prepared? Oh no! The phone call or the letter comes - our systems and practices are under review - we are being audited. Join us to take a peek at recent audits of sponsored programs activities and what actual findings can tell us. We will share the 'hot topics' that are getting attention and what we need to do as a community to be prepared when our time(s) comes. We will also spend time discussing the various responsibilities in meeting the requests of the auditors and involvement of central OSR in these engagements. Presenters: Robin Cyr, AVC for Research & Director Office of Sponsored Research Sharon Brooks, Director, Post Award & Cash Management, Office of Sponsored Research Grants Budgeting 101 (Concurrent Sessions C and D) Successful grantsmanship is the result of sound scientific principles with an acceptable budget. Some view developing the budget as the most challenging aspect of preparing grant proposals. During this 2-Part interactive workshop, the instructors will review budgeting requirements unique to the world of sponsored projects. We will offer insights into the budget planning process, including determining direct costs, indirect costs and cost sharing. Participants will also learn about budgeting for patient care costs, participant support costs, consultants, and sub-awards. Bring your grant budgeting questions and your calculators and come prepared to participate. NOTE: this is a 2-hour session 1:30 4:00 p.m. that includes a break. Presenters: Kathryn Calderone, Univ. Program Specialist, Finance & Business Operations, School of Medicine Laurie Henry, Director of Pre-Award Services, Office of Sponsored Research 2:30 pm - 2:50 pm Refreshment Break Sitterson Hall Lower Lobby
4 2:50 pm - 3:50 pm Concurrent Sessions D D1 D2 D3 014 C4/ D4 The Final Countdown: Project Closeout The Last Step in Meeting Compliance Requirements Let' talk...when does project closeout really begin? What happens in the closeout process in preparation for the project to be closed? Join us for a discussion on the collaborative steps that need to take place in order to provide timely and accurate financial invoices and reports to the sponsors. We will take a look at the NOTR and provide tips and tools that should be used in this process. We will discuss our challenges and our obligations in this last step in ensuring compliance with project terms and conditions. We will spend some time sharing the new NIH subproject drawdown requirements and the increased demands on timely reporting to their agency. What is at risk if we fail to provide this information timely and accurately? Facilitators: Jenifer Gwaltney, Assistant Director, Financial Reporting & Cash Management Departmental Representative, TBN Effort Reporting Learn why effort reporting is necessary for individuals paid on sponsored agreements and some of the biggest issues associated with effort reporting. The presentation will introduce the updated ecrt effort certification software and will discuss updates to UNC-CH s effort reporting policies. Presenter: TBN Cost Share from Pre-Award to Closeout Cost shared dollars are real to the University! When should we commit the University s funds and when shouldn t we? Are there any impacts in not meeting cost share commitments? This session will speak to these questions and when a cost share commitment comes to be - pre-award to closeout. Discussions on how to proactively and properly manage, track and report these commitments will be included. We will also visit the UNC Policy Statement on Cost Share, spend some time in CC regarding the tracking of cost share, as well as define the criteria and types of costs that represent valid cost share dollars. Presenters: Brigid Poole, Senior Grant and Contract Specialist, Office of Sponsored Research Brian Collier, Lead Program Administrator, Office of Sponsored Research Grants Budgeting 101 (Concurrent Sessions C and D) see Concurrent Session C for description 4:00 pm - 5:00 pm Concurrent Sessions E E1 014 E2 E3 Research Service Centers (RSC) This session will assist in the understanding of the operational context of RSCs and key compliance issues related to RSC management. Attendees will gain an understanding of available references and guidance, establishing an RSC, developing RSC recharge rates, working with external customers, and the roles and responsibilities for RSC management. Presenter: Brian Bertlshofer, Director of Cost Analysis, Office of Sponsored Research NSF Overview This workshop will provide an overview of the NSF and Fastlane proposal submission process. Participants will learn how to prepare NSF proposals for submission in Fastlane from a departmental perspective. In addition, participants will be given tips concerning common compliance checks from the OSR Pre-Award review perspective. Presenters: Melissa (Missy) Wood, Mgr. Research Support & Communications, Department of Computer Science Marcia Margotta, Grant and Contract Specialist, Office of Sponsored Research Getting Out of the Gate: Finding Efficiencies in Project ID Set-Up Let's explore the steps in project set-up! We will visit the negotiation processes with special mention of the terms/ conditions and clauses that UNC cannot accept as a State agency. This session will also highlight research and animal subject protocols, financial conflicts of interest and subaward documentation steps. Numerous compliance topics are addressed in this stage of the project life cycle, so getting out of the gate toward Project ID set-up, will take all of us working together as efficiently as we can. Presenters: Sarah Van Heusen, Director of Research Administration, Department of Microbiology and Immunology Megan Tirpak, Program Administrator, Office of Sponsored Research
5 Annual Symposium July 29th 2015 To help you get oriented to campus building and bus routes please refer to the maps below. Arrow indicate building entrances Locations in Sitterson Hall () and Fred Brooks Building () Buses: RU route bus stop Cameron Avenue at Columbia NU route bus stop South Columbia at Sitterson Hall
6 Round Table Topics Please join a round table discussion group during the lunch hour. TRAINING GRANTS When it comes to training grants, the Gillings School of Global Public Health has close up and personal experience. The Department of Biostatistics currently manages four NIH training grants with 36 predoctoral slots and 7 postdoctoral slots while Epidemiology manages three NIH training grants with 12 predoctorals and 5 postdoctoral slots. Come talk with these professionals who combined have over 54 years experience in the grants arena. They welcome questions you may have regarding the administration of training grants. Facilitated by Vicki Moore with the Department of Epidemiology, Evie McKee and Debbie Quach from the Department of Biostatistics Location: COST ALLOCATION Direct cost allocations are a primary factor (as cited in the Uniform Guidance, 2 CFR Part 200) when charging sponsored projects. So what does it mean to properly allocate a direct cost? Is cost allocation different from cost allowability? Who is responsible for cost allocations and retaining the documentation to support these costing decisions? Join us over lunch for a quick chat about the concept of cost allocations to find out the answers to these questions! Facilitated by Sharon Brooks with the Office of Sponsored Research Location: 008 CLINICAL TRIALS Do you have questions about research administration for clinical trials and want a better understanding of how projects go through the Office of Clinical Trials (OCT) and also dove tail with RAMSeS and OSR submissions; how these two offices OCT and OSR interact; budget development for grants especially for those that involve clinical trials (incorporating all the relevant costs, hospital charges, and Medicare billing coverage analysis issues). Facilitated by Christine Nelson and Aylin Regulski with the Office of Clinical Trials Location: 011 Why Do Some Applications Require an Exception Letter? Using examples of program guidance language along with suggested language for inclusion in an exception letter, this luncheon session provides an overview of what to look for when working with proposals that will result in a contract or agreement between the sponsor and the University. For sponsors whose terms and conditions cannot be accepted by the University, an exception letter must be prepared. This may occur when working with Federal Contracts governed by the Federal Acquisition Regulations (FAR) clauses; or when working with another State contracts, non-profit organizations, or foreign entities. There may be requirements stipulating the agreement is subject to the laws of another state, or require the venue to occur in another state, or outside the US. Facilitated by Brigid Poole with the Office of Sponsored Research EXCEPTION LETTER Location: 020 COMMUNICATIONS After the Oct go-live of ConnectCarolina for Finance and HR/Payroll and the launch of the expanded InfoPorte reporting tool, there has been a lot of information about fixes and enhancements as well as business process changes. While we made every effort to communicate these changes, sometimes it was difficult to reach the right audiences with the right information. And, it was very, very difficult for people using the system to keep track of these changes which sometimes meant not knowing that something had been fixed or that there was a more efficient way to do the work. The communications and training team for ConnectCarolina and InfoPorte is always looking for ways to improve getting you what you need in terms of training and information. Join us to share your ideas and feedback on what went well and what we could do better to support you as you learn and use these new systems. Facilitated by CC and IFP Communications and Training Team Location:
Audits and Reviews: Who is Looking and How Can We be Prepared?
 Audits and Reviews: Who is Looking and How Can We be Prepared? 2015 Office of Sponsored Research Symposium Sharon Brooks Director, OSR Robin Cyr, AVC for Research, OSR Session Agenda What Should You Do
Audits and Reviews: Who is Looking and How Can We be Prepared? 2015 Office of Sponsored Research Symposium Sharon Brooks Director, OSR Robin Cyr, AVC for Research, OSR Session Agenda What Should You Do
Finance Reporting: Updates and Info
 Finance Reporting: Updates and Info ConnectCarolina User Group July 24, 2015 Welcome Anita Collins ConnectCarolina Change Management Lead Deep Dive Topic for Today Finance Reporting Reminders The Help
Finance Reporting: Updates and Info ConnectCarolina User Group July 24, 2015 Welcome Anita Collins ConnectCarolina Change Management Lead Deep Dive Topic for Today Finance Reporting Reminders The Help
SJSU Research Foundation
 SJSU Research Foundation The Nuts & Bolts of Proposal Preparation and Award Management Sponsored Programs Workshop February 16, 2017 Agenda Discussion of Common Issues and Solutions Terms and Conditions
SJSU Research Foundation The Nuts & Bolts of Proposal Preparation and Award Management Sponsored Programs Workshop February 16, 2017 Agenda Discussion of Common Issues and Solutions Terms and Conditions
OSR s Annual Symposium for Research Administrators: Conflict of Interest The Kitchen Sink Version
 OSR s Annual Symposium for Research Administrators: Conflict of Interest The Kitchen Sink Version Version 07.23.2015 Joy Bryde, MSW Conflict of Interest Officer Conflict of Interest Program Office of the
OSR s Annual Symposium for Research Administrators: Conflict of Interest The Kitchen Sink Version Version 07.23.2015 Joy Bryde, MSW Conflict of Interest Officer Conflict of Interest Program Office of the
East Carolina University Sponsored Projects Administration Roles and Responsibilities Matrix By Responsibility
 (See legend at end of matrix.) East Carolina University Sponsored Projects Administration Roles and Responsibilities Matrix By Responsibility This matrix is designed to provide general guidance on administrative
(See legend at end of matrix.) East Carolina University Sponsored Projects Administration Roles and Responsibilities Matrix By Responsibility This matrix is designed to provide general guidance on administrative
An Exercise in Effort
 3 rd Annual Symposium for Research Administrators An Exercise in Effort Brian Bertlshofer, Director, Cost Analysis and Compliance bertlsbj@email.unc.edu Aja Saylor, Central Effort Coordinator ajasaylor@unc.edu
3 rd Annual Symposium for Research Administrators An Exercise in Effort Brian Bertlshofer, Director, Cost Analysis and Compliance bertlsbj@email.unc.edu Aja Saylor, Central Effort Coordinator ajasaylor@unc.edu
Narration: Welcome to the Anatomy of an Administrative Shell mini course.
 Welcome to the Anatomy of an Administrative Shell mini course. 1 If you have previously joined us for other sponsored project mini courses, you will be familiar with the Sponsored Project Life Cycle. In
Welcome to the Anatomy of an Administrative Shell mini course. 1 If you have previously joined us for other sponsored project mini courses, you will be familiar with the Sponsored Project Life Cycle. In
3 rd Annual Symposium for Research Administrators
 3 rd Annual Symposium for Research Administrators Cost Transfer Policy Martha Martin, CRA, Grants Administration, School of Information and Library Science Sarah Van Heusen, MHA, Director of Research Administration,
3 rd Annual Symposium for Research Administrators Cost Transfer Policy Martha Martin, CRA, Grants Administration, School of Information and Library Science Sarah Van Heusen, MHA, Director of Research Administration,
University of Pittsburgh SPONSORED PROJECT FINANCIAL GUIDELINE Subject: SUBRECIPIENT MONITORING
 I. Scope Subrecipient monitoring is the process by which the University selects, monitors, controls, and accounts for University subcontractors or subrecipients utilized on sponsored projects in accordance
I. Scope Subrecipient monitoring is the process by which the University selects, monitors, controls, and accounts for University subcontractors or subrecipients utilized on sponsored projects in accordance
Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network of Clinical Research Professionals
 ORIENTATION FOR NEW CLINICAL RESEARCH COORDINATORS Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network of Clinical Research Professionals Overall Agenda for Orientation Module 1:
ORIENTATION FOR NEW CLINICAL RESEARCH COORDINATORS Presented by NC TraCS Institute UNC Office of Clinical Trials UNC Network of Clinical Research Professionals Overall Agenda for Orientation Module 1:
Tar Heel Certificate Program in Research Administration Level 1 Certification. Award Financial Management
 Tar Heel Certificate Program in Research Administration Level 1 Certification Award Financial Management Agenda Review the regulations from an award management perspective Federal Circulars Federal Demonstration
Tar Heel Certificate Program in Research Administration Level 1 Certification Award Financial Management Agenda Review the regulations from an award management perspective Federal Circulars Federal Demonstration
3 rd Annual Symposium for Research Administrators
 3 rd Annual Symposium for Research Administrators The UNC-CH Research IT Landscape An overview of the systems that support the research enterprise John Stephenson john@unc.edu July 29, 2016 Research Administration
3 rd Annual Symposium for Research Administrators The UNC-CH Research IT Landscape An overview of the systems that support the research enterprise John Stephenson john@unc.edu July 29, 2016 Research Administration
Roles & Responsibilities UR Office of Research and Project Administration
 Roles and Responsibilities - UR ORPA Roles & Responsibilities UR Office of Research and Project Administration The Office of Research and Projects Administration (ORPA) is the administrative office legally
Roles and Responsibilities - UR ORPA Roles & Responsibilities UR Office of Research and Project Administration The Office of Research and Projects Administration (ORPA) is the administrative office legally
Building Your Foundation: Administrative Requirements Boot Camp (Modules 1-8: Administrative Requirements for Non-Federal Entities)
 Course #400 Syllabus Grant Management Boot Camp (Covers 12 Grant Management Modules) This Boot Camp bundle includes 12 Grants Management training courses including 8 modules on the Administrative Requirements
Course #400 Syllabus Grant Management Boot Camp (Covers 12 Grant Management Modules) This Boot Camp bundle includes 12 Grants Management training courses including 8 modules on the Administrative Requirements
office of research administration newsletter
 july/august 2010 vol. 5 no. 1 office of research administration newsletter Yale has a clear obligation to comply with all regulations pertaining to the administration of federal grants, and we will spare
july/august 2010 vol. 5 no. 1 office of research administration newsletter Yale has a clear obligation to comply with all regulations pertaining to the administration of federal grants, and we will spare
FAQ S FOR UNIFORM GUIDANCE
 FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
FAQ S FOR UNIFORM GUIDANCE As Uniform Guidance (UG) and its implications continue to be defined, the Fred Hutch UG Team will release a series of Frequently Asked Questions (FAQs) to clarify current standings.
Principal Investigator
 Principal Investigator HEADER TEXT Award Management Roles & Responsibilities November 4, 2016 Susan Mundt, Chris Barnhill, & Jing Liu Agenda PI Definition and Eligibility PreAward Roles & Responsibilities
Principal Investigator HEADER TEXT Award Management Roles & Responsibilities November 4, 2016 Susan Mundt, Chris Barnhill, & Jing Liu Agenda PI Definition and Eligibility PreAward Roles & Responsibilities
ROLES AND RESPONSIBILITIES* FOR EXTERNAL AWARDS MADE TO DAVIDSON COLLEGE
 ROLES AND RESPONSIBILITIES* FOR EXTERNAL AWARDS MADE TO DAVIDSON COLLEGE Responsible (individual ultimately responsible for getting the work done) Accountable (signatory authority; expected to justify
ROLES AND RESPONSIBILITIES* FOR EXTERNAL AWARDS MADE TO DAVIDSON COLLEGE Responsible (individual ultimately responsible for getting the work done) Accountable (signatory authority; expected to justify
INTRODUCTION TO RESEARCH ADMINISTRATION. Office of Grants and Contracts Administration August 2015
 INTRODUCTION TO RESEARCH ADMINISTRATION Office of Grants and Contracts Administration August 2015 THE RESEARCH VESSEL SIKULIAQ FUNDED BY THE NATIONAL SCIENCE FOUNDATION Objectives To understand the UAF
INTRODUCTION TO RESEARCH ADMINISTRATION Office of Grants and Contracts Administration August 2015 THE RESEARCH VESSEL SIKULIAQ FUNDED BY THE NATIONAL SCIENCE FOUNDATION Objectives To understand the UAF
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates!
 FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
FDP Subaward Forms Frequently Asked Questions Check back frequently for updates! Categories of Questions (click hyperlink below): Invoicing & Final Statement of Cumulative Costs Uniform Guidance (UG) data
Rebecca Trahan. Office of Sponsored Programs December 9, ORED Limited Submission Update
 Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
Rebecca Trahan Office of Sponsored Programs December 9, 2015 1 NSF Updates NIH Updates Uniform Guidance Updates and Reminders ORED Limited Submission Update OSP Updates Q & A 2 1 3 4 2 NSF 16-1 effective
Research Administrators Forum September 13, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum September 13, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith OCGA Initiatives Patti Manheim, Kim Duiker Proposal Intake
Research Administrators Forum September 13, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith OCGA Initiatives Patti Manheim, Kim Duiker Proposal Intake
Trigger / Timing / Frequency: When a new award is received by the University and OSP determines that the award can be accepted.
 Kuali Research User Guide: Create a New Parent Award Version October 06 Purpose: To create a new parent award record in the system. Trigger / Timing / Frequency: When a new award is received by the University
Kuali Research User Guide: Create a New Parent Award Version October 06 Purpose: To create a new parent award record in the system. Trigger / Timing / Frequency: When a new award is received by the University
Subawards and Subrecipient Monitoring
 Subawards and Subrecipient Monitoring H O L LY S O M M E R S D I R E C T O R, O S P G R A N T S J O S H R O S E N B E R G A C T I N G D I R E C T O R, O G C A Objectives 2 Distinguish Between: Procurement
Subawards and Subrecipient Monitoring H O L LY S O M M E R S D I R E C T O R, O S P G R A N T S J O S H R O S E N B E R G A C T I N G D I R E C T O R, O G C A Objectives 2 Distinguish Between: Procurement
Please fill out the survey forms 2/9/2012. Upcoming ORA Training Opportunities. Agenda
 Agenda Welcome and Announcements Marcia Smith Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Data Management Tool Sharon Farb, Associate University Librarian,
Agenda Welcome and Announcements Marcia Smith Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Data Management Tool Sharon Farb, Associate University Librarian,
OMB Uniform Guidance ( UG ) Briefing. ASRSP & OSR Brown Bag Tuesday, January 27 th
 OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
OMB Uniform Guidance ( UG ) Briefing ASRSP & OSR Brown Bag Tuesday, January 27 th Background The UG is the single biggest regulatory change in the last fifty years in research administration Interesting
Policy on Cost Allocation, Cost Recovery, and Cost Sharing
 President Page 1 of 11 PURPOSE: Provide guidance and structure when allocating and documenting costs (direct and indirect) for extramurally funded awards. Serves to provide direction for budgeting, allocating
President Page 1 of 11 PURPOSE: Provide guidance and structure when allocating and documenting costs (direct and indirect) for extramurally funded awards. Serves to provide direction for budgeting, allocating
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Preparing campus for what is changing and what is not What is Grants Reform and when is it effective? What
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Preparing campus for what is changing and what is not What is Grants Reform and when is it effective? What
August 3, 2017 Clinical Trials at UNC. UNC Symposium for Research Administrators
 August 3, 2017 Clinical Trials at UNC UNC Symposium for Research Administrators UNC Symposium for Research Administrators Presenter Christine Nelson, RN, BSN, MBA, CCRC Director, Office of Clinical Trials
August 3, 2017 Clinical Trials at UNC UNC Symposium for Research Administrators UNC Symposium for Research Administrators Presenter Christine Nelson, RN, BSN, MBA, CCRC Director, Office of Clinical Trials
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018
 CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
CLOSEOUT - DON T SHUT DOWN SRA 2018 NORTHEAST/WESTERN SECTION MEETING MARCH 2018 PRESENTATION TEAM Christine Toalepai, University of Maryland, Baltimore, Office of Sponsored Programs Administration, Sponsored
Cost Sharing Administrative Guidelines
 Southern Illinois University Carbondale Cost Sharing Administrative Guidelines Summary Cost sharing refers to the resources contributed or allocated by the University to an externally sponsored project,
Southern Illinois University Carbondale Cost Sharing Administrative Guidelines Summary Cost sharing refers to the resources contributed or allocated by the University to an externally sponsored project,
Subrecipient Monitoring Procedures
 Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
Subrecipient Monitoring Procedures This procedure describes the proper management of subrecipient activity under Purdue sponsored program awards. Definitions Award: An award is a binding agreement between
OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015
 OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015 INTRODUCTION The University of Wisconsin-Milwaukee (UWM) is the prime recipient on a wide range of sponsored awards
OUTGOING SUBAWARD GUIDE: INFORMATION FOR UWM PRINCIPAL INVESTIGATORS VERSION 1, JULY 2015 INTRODUCTION The University of Wisconsin-Milwaukee (UWM) is the prime recipient on a wide range of sponsored awards
Sponsored Programs Roles & Responsibilities
 The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
AURA Meeting. July 23, 2014
 AURA Meeting July 23, 2014 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored
AURA Meeting July 23, 2014 Welcome William Mitchell, MPSA Supervisor, Systems & Reporting Sponsored Projects Administration Website Spotlight Cherrelle Duncan, MPSA Sponsored Projects Specialist Sponsored
Wake Forest University Financial Services: Grants Accounting and Compliance
 Wake Forest University Financial Services: Grants Accounting and Compliance 1 WFU Policies and Procedures a Policies b Fiscal Administration i Award Notification 1 The Office of Research and Sponsored
Wake Forest University Financial Services: Grants Accounting and Compliance 1 WFU Policies and Procedures a Policies b Fiscal Administration i Award Notification 1 The Office of Research and Sponsored
Sponsored Programs Roles & Responsibilities
 The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
The University of Florida is committed to acting with integrity in the management of sponsored programs. The goals of this document are to provide descriptions of key individuals or units and their responsibilities
SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients must complete this form when submitting a proposal to KSU, along with documents and certifications required by sponsors. This form must be endorsed by the
SUBRECIPIENT COMMITMENT FORM All subrecipients must complete this form when submitting a proposal to KSU, along with documents and certifications required by sponsors. This form must be endorsed by the
UNIFORM GUIDANCE IMPLEMENTATION
 UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
UNIFORM GUIDANCE IMPLEMENTATION Presented by Sara Judd, OSR Consultant October 2014 Uniform Guidance Implementation what we know and what we re guessing Ready, Set, Go 2014 Fred Hutchinson Cancer Research
OMB Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards
 OMB Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Research Advisory Board What PIs Need to Know January 2015 1 Background o When and Why? o Overview Important
OMB Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards Research Advisory Board What PIs Need to Know January 2015 1 Background o When and Why? o Overview Important
Uniform Guidance and Internal Controls: A Case Study
 Uniform Guidance and Internal Controls: A Case Study Rick Moyer, Stanford University Kim Ginn, Baker Tilly Ashley Deihr, Baker Tilly Expected outcomes from this session: Translate university expectations
Uniform Guidance and Internal Controls: A Case Study Rick Moyer, Stanford University Kim Ginn, Baker Tilly Ashley Deihr, Baker Tilly Expected outcomes from this session: Translate university expectations
AURA-Grants. Fundamentals
 AURA-Grants Fundamentals Agenda 2 Introductions What is AURA Grants? Why is AURA so Cool? Common Questions/Concerns Roles Workflows Websites System Overview Personal Workspace Funding Proposal Workspace
AURA-Grants Fundamentals Agenda 2 Introductions What is AURA Grants? Why is AURA so Cool? Common Questions/Concerns Roles Workflows Websites System Overview Personal Workspace Funding Proposal Workspace
NSF OIG Audit Update NORTHEAST CONFERENCE ON COLLEGE COST ACCOUNTING
 NSF OIG Audit Update 1 NORTHEAST CONFERENCE ON COLLEGE COST ACCOUNTING S e p t e m b e r 2 3, 2 0 1 4 Overview 2 Overview of NSF OIG Office of Audit Overview of Federal financial assistance in the U.S.
NSF OIG Audit Update 1 NORTHEAST CONFERENCE ON COLLEGE COST ACCOUNTING S e p t e m b e r 2 3, 2 0 1 4 Overview 2 Overview of NSF OIG Office of Audit Overview of Federal financial assistance in the U.S.
Ramp up and Wrap up projects. Riding the Wave to Close Out
 Ramp up and Wrap up projects Riding the Wave to Close Out To maximize our time together today: Please turn off or silence your cell phones Limit side chatter so everyone can hear Best of all. Stay for
Ramp up and Wrap up projects Riding the Wave to Close Out To maximize our time together today: Please turn off or silence your cell phones Limit side chatter so everyone can hear Best of all. Stay for
Office of Sponsored Projects 5/31/ Volume 2, Issue 5
 The OSP News & Updates, published by the Office of Sponsored Projects, is a bi-weekly subscription-based newsletter that provides OSP and sponsor updates and reminders, quick facts, guidance and training
The OSP News & Updates, published by the Office of Sponsored Projects, is a bi-weekly subscription-based newsletter that provides OSP and sponsor updates and reminders, quick facts, guidance and training
Sponsored Programs Administration Banner/FRAGRNT TABLE
 Sponsored Programs Administration Banner/FRAGRNT TABLE Concept of the FRAGRNT Table: The Grant Maintenance Form. FRAGRNT is used to enter or update grant information. Grant Field: This is a 6-Character
Sponsored Programs Administration Banner/FRAGRNT TABLE Concept of the FRAGRNT Table: The Grant Maintenance Form. FRAGRNT is used to enter or update grant information. Grant Field: This is a 6-Character
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards. AU SPAN Martha Taylor Larry Hankins
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards AU SPAN 6 23 2014 Martha Taylor Larry Hankins Grants Reform February 2011 President directed OMB to reduce
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards AU SPAN 6 23 2014 Martha Taylor Larry Hankins Grants Reform February 2011 President directed OMB to reduce
Proposal Preparation and Submission February 2018
 Proposal Preparation and Submission February 2018 Introductions We are. Chris Dye-Hixenbaugh, C&G Officer, Proposals Brooke Herevia, C&G Analyst, Proposals Shanna Nation Jose, C&G Analyst, Proposals Kassie
Proposal Preparation and Submission February 2018 Introductions We are. Chris Dye-Hixenbaugh, C&G Officer, Proposals Brooke Herevia, C&G Analyst, Proposals Shanna Nation Jose, C&G Analyst, Proposals Kassie
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards (New Uniform Guidance)
 Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards (New Uniform Guidance) Spring Research Administrators Series March 19, 2015 1 Agenda Background UW-Madison
Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards (New Uniform Guidance) Spring Research Administrators Series March 19, 2015 1 Agenda Background UW-Madison
Federal Grants and Financial Assistance 2017 Training Catalog
 1 P a g e Who are we? Meet Colleague Consulting Colleague Consulting LLC is a 19-year-old small business specializing in training, human resource development, and organizational development services for
1 P a g e Who are we? Meet Colleague Consulting Colleague Consulting LLC is a 19-year-old small business specializing in training, human resource development, and organizational development services for
Grants Financial Procedures (Post-Award) v. 2.0
 Grants Financial Procedures (Post-Award) v. 2.0 1 Grants Financial Procedures (Post Award) Version Number: 2.0 Procedures Identifier: Superseded Procedure(s): BU-PR0001 N/A Date Approved: 9/1/2013 Effective
Grants Financial Procedures (Post-Award) v. 2.0 1 Grants Financial Procedures (Post Award) Version Number: 2.0 Procedures Identifier: Superseded Procedure(s): BU-PR0001 N/A Date Approved: 9/1/2013 Effective
Office of Internal Audit
 Office of Internal Audit July 5, 2017 Dr. Kirk A. Calhoun, President UT Health Northeast 11937 U. S. Hwy 271 Tyler, TX 75708 Dr. Calhoun, We have completed the that was part of our Audit Plan. The objective
Office of Internal Audit July 5, 2017 Dr. Kirk A. Calhoun, President UT Health Northeast 11937 U. S. Hwy 271 Tyler, TX 75708 Dr. Calhoun, We have completed the that was part of our Audit Plan. The objective
UC San Diego Policy & Procedure Manual
 UC San Diego Policy & Procedure Manual Search A Z Index Numerical Index Classification Guide What s New CONTRACTS AND GRANTS (RESEARCH) Section: 150-45 Effective: 08/01/2001 Supersedes: 05/26/1999 Review
UC San Diego Policy & Procedure Manual Search A Z Index Numerical Index Classification Guide What s New CONTRACTS AND GRANTS (RESEARCH) Section: 150-45 Effective: 08/01/2001 Supersedes: 05/26/1999 Review
Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6
 Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6 KC-PRE28: Prepare Summary Proposal KC-AWD22: Research Gifts November 13, 2013 Goals and Outcomes Goals Obtain feedback on the proposed
Kuali Coeus Implementation Preaward/Award Blueprinting Workshop 6 KC-PRE28: Prepare Summary Proposal KC-AWD22: Research Gifts November 13, 2013 Goals and Outcomes Goals Obtain feedback on the proposed
Match and Leveraged Resources
 Match and Leveraged Resources Uniform Guidance vs. OMB Circulars Prior to the Uniform Guidance, Designed requirements for DOL-ETA governing direct cost recipients principles, and administrative their subrecipients
Match and Leveraged Resources Uniform Guidance vs. OMB Circulars Prior to the Uniform Guidance, Designed requirements for DOL-ETA governing direct cost recipients principles, and administrative their subrecipients
Uniform Guidance vs. OMB Circulars
 Program Income Uniform Guidance vs. OMB Circulars Prior to the Uniform Guidance, requirements governing cost Designed for DOL-ETA direct principles, administrative recipients and their requirements and
Program Income Uniform Guidance vs. OMB Circulars Prior to the Uniform Guidance, requirements governing cost Designed for DOL-ETA direct principles, administrative recipients and their requirements and
UNIVERSITY RESEARCH ADMINISTRATION FINANCIAL ROLES AND RESPONSIBILITIES MATRIX - WORK IN PROGRESS 10/03/2013 Roles.
 UNIVERSITY RESEARCH ADMINISTRATION Roles Business Internal Controller's Clinical Responsibilities PI Office Chair Dean Audit Office OCR GCFA GCA PROVOST Trials Office I. GENERAL RESEARCH ADMINISTRATION
UNIVERSITY RESEARCH ADMINISTRATION Roles Business Internal Controller's Clinical Responsibilities PI Office Chair Dean Audit Office OCR GCFA GCA PROVOST Trials Office I. GENERAL RESEARCH ADMINISTRATION
Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32)
 Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32) Overview: This document is intended to help you complete a KC System to System (S2S) proposal submission
Kuali Coeus Proposal Prep Guide Ruth L. Kirschstein National Research Service Awards (NRSA) (F31 and F32) Overview: This document is intended to help you complete a KC System to System (S2S) proposal submission
The Lifecycle of a Sponsored Project
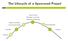 The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
The Lifecycle of a Sponsored Project Active Period Spending, Invoicing, Reporting, Monitoring Award Processing And Account Setup Final Reporting Proposal Closeout Funding Funding Sources Where is the Money?
Research Administration at Emory Foundation Program DAY TWO: The Financial Administration of Sponsored Programs
 Revised 05/2018 Research Administration at Emory Foundation Program DAY TWO: The Financial Administration of Sponsored Programs Presenter: Finance: Grants and Contracts (FGC) Jumionne Tiako AD Post Award
Revised 05/2018 Research Administration at Emory Foundation Program DAY TWO: The Financial Administration of Sponsored Programs Presenter: Finance: Grants and Contracts (FGC) Jumionne Tiako AD Post Award
Welcome to A Beginner s Guide to Sponsored Project Solicitations. This is one of the introductory mini courses in Northwestern s Sponsored Project
 Welcome to A Beginner s Guide to Sponsored Project Solicitations. This is one of the introductory mini courses in Northwestern s Sponsored Project Online Training series. 1 If you have previously joined
Welcome to A Beginner s Guide to Sponsored Project Solicitations. This is one of the introductory mini courses in Northwestern s Sponsored Project Online Training series. 1 If you have previously joined
Steve Relyea Executive Vice Chancellor and Chief Financial Officer. Audit Report 18-67, Sponsored Programs Post Award, Office of the Chancellor
 Date: May 4, 2018 To: From: Subject: Steve Relyea Executive Vice Chancellor and Chief Financial Officer Larry Mandel Vice Chancellor and Chief Audit Officer Audit Report 18-67, Sponsored Programs Post
Date: May 4, 2018 To: From: Subject: Steve Relyea Executive Vice Chancellor and Chief Financial Officer Larry Mandel Vice Chancellor and Chief Audit Officer Audit Report 18-67, Sponsored Programs Post
Michigan State University Contract and Grant Administration Post-Award Newsletter- Issue 4 December 2013 Table of Contents
 Michigan State University Contract and Grant Administration Post-Award Newsletter- Issue 4 December 2013 Table of Contents NIH Account Number Changes Page 1 Did you Know? Hardship Extensions.. Page 1 CGA
Michigan State University Contract and Grant Administration Post-Award Newsletter- Issue 4 December 2013 Table of Contents NIH Account Number Changes Page 1 Did you Know? Hardship Extensions.. Page 1 CGA
MAXIMUS Higher Education Practice
 Federal Compliance Webinar Series 2 CFR 200 Uniform Guidance Kris Rhodes, Director, MAXIMUS Jason Guilbeault, Senior Consultant, MAXIMUS 1 MAXIMUS Higher Education Practice Headquartered in Northbrook,
Federal Compliance Webinar Series 2 CFR 200 Uniform Guidance Kris Rhodes, Director, MAXIMUS Jason Guilbeault, Senior Consultant, MAXIMUS 1 MAXIMUS Higher Education Practice Headquartered in Northbrook,
Patti Manheim, Director, OCGA
 Research Administrators Forum Patti Manheim, Director, OCGA Agenda Welcome - Patti Manheim OCGA Updates Patti Manheim * Consortium/contractual arrangements versus purchase of services * Master Calendar
Research Administrators Forum Patti Manheim, Director, OCGA Agenda Welcome - Patti Manheim OCGA Updates Patti Manheim * Consortium/contractual arrangements versus purchase of services * Master Calendar
Sponsored Programs and Research Compliance SUBRECIPIENT COMMITMENT FORM
 SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a subaward proposal to University of North Carolina Wilmington (UNCW). Please complete this form and send all required
SUBRECIPIENT COMMITMENT FORM All subrecipients should complete this form when submitting a subaward proposal to University of North Carolina Wilmington (UNCW). Please complete this form and send all required
Research Administrators Forum May 8th, Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum May 8th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Office of Research Administration OHRPP Retention of Research
Research Administrators Forum May 8th, 2014 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith, Office of Research Administration OHRPP Retention of Research
Sponsored Projects: Curriculum Plan Johns Hopkins University Panel Members:
 Sponsored Projects: Curriculum Plan Johns Hopkins University Panel Members: Mike Alexander, University Administration Jim Aumiller, University Administration Holly Benze, Engineering Don Cornely, University
Sponsored Projects: Curriculum Plan Johns Hopkins University Panel Members: Mike Alexander, University Administration Jim Aumiller, University Administration Holly Benze, Engineering Don Cornely, University
Research Administration Forum March 9, Marcia Smith Associate Vice Chancellor for Research
 Research Administration Forum March 9, 2017 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements - Marcia Smith Immigration Update and Q & A Sam Nahidi, Assistant Director,
Research Administration Forum March 9, 2017 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements - Marcia Smith Immigration Update and Q & A Sam Nahidi, Assistant Director,
Research Administrators Round Table
 Research Administrators Round Table Recap Set framework for going forward Review new Uniform Guidance driven WVU policy changes Sponsored Agreement Closeouts (WVU-FS-23) http://adminfin.wvu.edu/policies/finance/financial-services/wvu-fs-23-policy
Research Administrators Round Table Recap Set framework for going forward Review new Uniform Guidance driven WVU policy changes Sponsored Agreement Closeouts (WVU-FS-23) http://adminfin.wvu.edu/policies/finance/financial-services/wvu-fs-23-policy
The Office for Sponsored Research (OSR): An Overview. Office for Sponsored Research
 The Office for Sponsored Research (OSR): An Overview Office for Sponsored Research OSR Mission The Office for Sponsored Research assists investigators in proposing and managing sponsored programs in support
The Office for Sponsored Research (OSR): An Overview Office for Sponsored Research OSR Mission The Office for Sponsored Research assists investigators in proposing and managing sponsored programs in support
Financial Oversight of Sponsored Projects
 Financial Oversight of Sponsored Projects Today s Agenda Introduction Reasons for Principal Investigator Oversight Sponsor and University Policies General Considerations Unallowable Costs Re-Budgeting
Financial Oversight of Sponsored Projects Today s Agenda Introduction Reasons for Principal Investigator Oversight Sponsor and University Policies General Considerations Unallowable Costs Re-Budgeting
Marcia Smith Associate Vice Chancellor for Research
 Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith Data Management Tool Sharon Farb, Associate University Librarian,
Research Administrators Forum February 9, 2012 Marcia Smith Associate Vice Chancellor for Research Agenda Welcome and Announcements Marcia Smith Data Management Tool Sharon Farb, Associate University Librarian,
How to Manage Externally Funded Grants PROJECTS - FUND CODE 501 or 502
 How to Manage Externally Funded Grants PROJECTS - FUND CODE 501 or 502 What is a Grant? Funding that is coming from a source external to Wesleyan Project conceived by the Investigator Response to a solicitation
How to Manage Externally Funded Grants PROJECTS - FUND CODE 501 or 502 What is a Grant? Funding that is coming from a source external to Wesleyan Project conceived by the Investigator Response to a solicitation
University of Colorado Denver
 University of Colorado Denver Campus Guidelines Title:, 4-13 Source: Prepared by: Approved by: Office of Grants and Contracts Director, Office of Grants and Contracts Vice Chancellor for Research Effective
University of Colorado Denver Campus Guidelines Title:, 4-13 Source: Prepared by: Approved by: Office of Grants and Contracts Director, Office of Grants and Contracts Vice Chancellor for Research Effective
University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures
 Summary 1. Subaward Definitions A. Subaward B. Subrecipient University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures C. Office of Contracts and Grants (OCG) 2. Distinguishing
Summary 1. Subaward Definitions A. Subaward B. Subrecipient University of San Francisco Office of Contracts and Grants Subaward Policy and Procedures C. Office of Contracts and Grants (OCG) 2. Distinguishing
Accelerated Translational Incubator Pilot (ATIP) Program. Frequently Asked Questions. ICTR Research Navigators January 19, 2017 Version 7.
 Accelerated Translational Incubator Pilot (ATIP) Program Frequently Asked Questions ICTR Research Navigators January 19, 2017 Version 7.0 TABLE OF CONTENTS Section # Title Page 1. ABOUT THE ATIP PROGRAM...
Accelerated Translational Incubator Pilot (ATIP) Program Frequently Asked Questions ICTR Research Navigators January 19, 2017 Version 7.0 TABLE OF CONTENTS Section # Title Page 1. ABOUT THE ATIP PROGRAM...
DFA Conversations Research Administration Forum
 DFA Conversations Research Administration Forum Fall 2017 10/23/2017 Agenda What is Financial Research Administration? Our Issues Recent Audits Your Issues Traveling for Cornell on Sponsored Awards Recent
DFA Conversations Research Administration Forum Fall 2017 10/23/2017 Agenda What is Financial Research Administration? Our Issues Recent Audits Your Issues Traveling for Cornell on Sponsored Awards Recent
Any observations not included in this report were discussed with your staff at the informal exit conference and may be subject to follow-up.
 Larry Mandel Vice Chancellor and Chief Audit Officer Office of Audit and Advisory Services 401 Golden Shore, 4th Floor Long Beach, CA 90802-4210 562-951-4430 562-951-4955 (Fax) lmandel@calstate.edu September
Larry Mandel Vice Chancellor and Chief Audit Officer Office of Audit and Advisory Services 401 Golden Shore, 4th Floor Long Beach, CA 90802-4210 562-951-4430 562-951-4955 (Fax) lmandel@calstate.edu September
Grant Closeout Process
 Grant Closeout Process Uniform Guidance vs. OMB Circulars Prior to the Uniform Guidance, requirements Designed for governing DOL-ETA cost direct principles, recipients administrative and their subrecipients
Grant Closeout Process Uniform Guidance vs. OMB Circulars Prior to the Uniform Guidance, requirements Designed for governing DOL-ETA cost direct principles, recipients administrative and their subrecipients
Financial Oversight of Sponsored Projects Principal Investigator and Department Administrator Responsibilities
 Principal Investigator and Department Administrator Responsibilities Boston College Office for Sponsored Programs Office for Research Compliance and Intellectual Property March 2004 Introduction This guide
Principal Investigator and Department Administrator Responsibilities Boston College Office for Sponsored Programs Office for Research Compliance and Intellectual Property March 2004 Introduction This guide
Emory Research A to Z ERAZ
 Emory Research A to Z ERAZ July 17, 2014 Whitehead Auditorium G01 Whitehead Building Agenda Controlled Substances Sub Award Processing RAS Updates SAM Kiosk Federal Updates OSP/OGCA Training Updates Christine
Emory Research A to Z ERAZ July 17, 2014 Whitehead Auditorium G01 Whitehead Building Agenda Controlled Substances Sub Award Processing RAS Updates SAM Kiosk Federal Updates OSP/OGCA Training Updates Christine
Ask the Experts Panel
 Ask the Experts Panel Compliance in Research Colleen Fritsche, Assistant Director of Office of Animal Care and Use Cassie Myers, Deputy Director of Office of Human Research Ethics Chris Nelson, Director
Ask the Experts Panel Compliance in Research Colleen Fritsche, Assistant Director of Office of Animal Care and Use Cassie Myers, Deputy Director of Office of Human Research Ethics Chris Nelson, Director
Navigating Grant Resources. 10/3/2018 Dr. Susan Fell
 Navigating Grant Resources 10/3/2018 Dr. Susan Fell sfell@fsu.edu 850-645-2172 Sponsored Research Administration https://www.research.fsu.edu/ Applying for a Grant Opportunity Does this require sign-off
Navigating Grant Resources 10/3/2018 Dr. Susan Fell sfell@fsu.edu 850-645-2172 Sponsored Research Administration https://www.research.fsu.edu/ Applying for a Grant Opportunity Does this require sign-off
The purpose of this survey
 The purpose of this survey Dear Colleague: As part of our ongoing effort to strengthen grants administration across Boston University, we have created this survey to gain a better understanding of your
The purpose of this survey Dear Colleague: As part of our ongoing effort to strengthen grants administration across Boston University, we have created this survey to gain a better understanding of your
Template D Plain-crimson-dark 1
 WSU Research Administration Series: Research Administration Update Dan Nordquist, Kim Small, Sponsored Programs Heather Lopez, Internal Audit September 12, 2014 Recording date of this workshop is September
WSU Research Administration Series: Research Administration Update Dan Nordquist, Kim Small, Sponsored Programs Heather Lopez, Internal Audit September 12, 2014 Recording date of this workshop is September
Grants Module. Create and Submit a Funding Proposal for Non System to System Submissions
 Grants Module Create and Submit a Funding Proposal for Non System to System Submissions Summary of Major Changes The following directions are a combination of all steps in the Funding Proposal submission
Grants Module Create and Submit a Funding Proposal for Non System to System Submissions Summary of Major Changes The following directions are a combination of all steps in the Funding Proposal submission
Office of Sponsored Programs Policies and Procedures. Sponsored Program Award Management Guidance
 Office of Sponsored Programs Policies and Procedures Contents Principal Investigator Eligibility... 2 Policy on Submission Deadlines for Applications Submitted by the Office of Sponsored Programs... 3
Office of Sponsored Programs Policies and Procedures Contents Principal Investigator Eligibility... 2 Policy on Submission Deadlines for Applications Submitted by the Office of Sponsored Programs... 3
University of North Carolina Finance Improvement & Transformation Contracts and Grants Standards. January 2015 Version 8
 University of North Carolina Finance Improvement & Transformation Contracts and Grants Standards January 2015 Version 8 Table of Contents Introduction.......3 Account Setup.....10 Time and Effort Reporting......14
University of North Carolina Finance Improvement & Transformation Contracts and Grants Standards January 2015 Version 8 Table of Contents Introduction.......3 Account Setup.....10 Time and Effort Reporting......14
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM. December 2017
 Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Office of Sponsored Programs RESEARCH ADMINISTRATORS FORUM December 2017 Agenda Feedback on Subawards Process What is FFATA and what does it mean to the Research Administration community at NYU? University
Uniform Guidance. Overview and Implementation Plan. November 21, 2014
 Uniform Guidance Overview and Implementation Plan November 21, 2014 Uniform Guidance. Change is coming! College or Department name here 1 Overarching goal of the reform is to: streamline the rules and
Uniform Guidance Overview and Implementation Plan November 21, 2014 Uniform Guidance. Change is coming! College or Department name here 1 Overarching goal of the reform is to: streamline the rules and
HUD-US DEPT OF HOUSING & URBAN DEVELOPMENT: Financial Grant Reporting. Ladies, and gentlemen, thank you for standing by and welcome to the
 Final Transcript HUD-US DEPT OF HOUSING : Financial Grant Reporting SPEAKERS Robin Booth PRESENTATION Moderator Ladies, and gentlemen, thank you for standing by and welcome to the Financial Grant Reporting
Final Transcript HUD-US DEPT OF HOUSING : Financial Grant Reporting SPEAKERS Robin Booth PRESENTATION Moderator Ladies, and gentlemen, thank you for standing by and welcome to the Financial Grant Reporting
ORA Quarterly Meeting April
 ORA Quarterly Meeting April 19 2017 Agenda 1. Workday Grants Implementation Items General Information Blackout Period Sub K s - Set-up and Invoicing Advance Accounts 2. Jackson/ UM Partnership Master Agreement
ORA Quarterly Meeting April 19 2017 Agenda 1. Workday Grants Implementation Items General Information Blackout Period Sub K s - Set-up and Invoicing Advance Accounts 2. Jackson/ UM Partnership Master Agreement
Sponsored by Office of Research Administration. Marcia Smith Associate Vice Chancellor Research Administration. July 9, 2009
 Research Administrators Forum Sponsored by Office of Research Administration Marcia Smith Associate Vice Chancellor Research Administration July 9, 2009 Agenda 2 Updates from the Animal Research Committee
Research Administrators Forum Sponsored by Office of Research Administration Marcia Smith Associate Vice Chancellor Research Administration July 9, 2009 Agenda 2 Updates from the Animal Research Committee
Financial Research Compliance. April 2013
 Financial Research Compliance April 2013 Overview I. What is a Sponsored Award? II. Sponsored Awards at WFU III. Compliance IV. Hot Topics V. Audits VI. WFU Updates VII. Questions VIII. Contacts What is
Financial Research Compliance April 2013 Overview I. What is a Sponsored Award? II. Sponsored Awards at WFU III. Compliance IV. Hot Topics V. Audits VI. WFU Updates VII. Questions VIII. Contacts What is
Current Issues in Sponsored Projects Administration. March 11 10:30-11:45 3:15 4:30
 Current Issues in Sponsored Projects Administration March 11 10:30-11:45 3:15 4:30 Workshop Presenters Name: Linda Learned Title: Associate Director/Interim Director, Office of Sponsored Programs and Research
Current Issues in Sponsored Projects Administration March 11 10:30-11:45 3:15 4:30 Workshop Presenters Name: Linda Learned Title: Associate Director/Interim Director, Office of Sponsored Programs and Research
USF HRPP Updates October 2017
 USF HRPP Updates October 2017 Julie Moore, J.D., M.S. PA, CIP Associate Director, Research Integrity & Compliance Research Integrity and Compliance Revised Common Rule 45 CFR 46 Effective date and compliance
USF HRPP Updates October 2017 Julie Moore, J.D., M.S. PA, CIP Associate Director, Research Integrity & Compliance Research Integrity and Compliance Revised Common Rule 45 CFR 46 Effective date and compliance
University of Kansas Medical Center Research Institute. What Every Administrator Ought to Know About Grant Management
 University of Kansas Medical Center Research Institute What Every Administrator Ought to Know About Grant Management Who is the RI? The University of Kansas Medical Center Research Institute, Inc. is a
University of Kansas Medical Center Research Institute What Every Administrator Ought to Know About Grant Management Who is the RI? The University of Kansas Medical Center Research Institute, Inc. is a
SPONSORED PROGRAMS AWARDS, EXPENDITURES, AND ALLOWABILTY APRIL 2015 Policy
 SPONSORED PROGRAMS AWARDS, EXPENDITURES, AND ALLOWABILTY APRIL 2015 Policy 2.2.01 Purpose: This policy was developed to ensure consistent compliance with 2 CFR part 200 Uniform Guidance and the Cost Accounting
SPONSORED PROGRAMS AWARDS, EXPENDITURES, AND ALLOWABILTY APRIL 2015 Policy 2.2.01 Purpose: This policy was developed to ensure consistent compliance with 2 CFR part 200 Uniform Guidance and the Cost Accounting
PEER (PAPERLESS ENVIRONMENT FOR ELECTRONIC REVIEW)
 PEER (PAPERLESS ENVIRONMENT FOR ELECTRONIC REVIEW) 1 What is PEER? PEER is a web portal maintained by Vanderbilt University which handles internal processing of grant related requests and contract submission
PEER (PAPERLESS ENVIRONMENT FOR ELECTRONIC REVIEW) 1 What is PEER? PEER is a web portal maintained by Vanderbilt University which handles internal processing of grant related requests and contract submission
